MK-677 (Ibutamoren): Oral GH Secretagogue
MK-677 and Ibutamoren
97% GH increase
In a clinical study, 25 mg MK-677 daily increased mean 24-hour growth hormone concentration by 97% and IGF-1 levels by 55% in healthy older adults over 12 months.
Nass et al., Annals of Internal Medicine, 2008
Nass et al., Annals of Internal Medicine, 2008
If you only read one thing
MK-677 is the only growth hormone booster you can take as a pill instead of an injection. In clinical trials, it nearly doubled GH levels in older adults and improved deep sleep by 50%. But Merck — the pharmaceutical giant that invented it — abandoned it after trials showed it didn't actually improve the health outcomes they were testing. It raised blood sugar, may stress the heart, and despite making GH numbers look great, it didn't help Alzheimer's patients think better or hip fracture patients recover faster. It's now one of the most popular gray-market compounds, used mostly by young bodybuilders in ways no trial has ever tested.
In 1995, Arthur Patchett and colleagues at Merck Research Laboratories published the design and biological characterization of L-163,191, later designated MK-0677, in the Proceedings of the National Academy of Sciences. It was a small molecule that mimicked the action of growth hormone-releasing peptides (GHRPs) at a receptor that would later be identified as the growth hormone secretagogue receptor (GHSR-1a), the same receptor that binds the endogenous hunger hormone ghrelin.[1] The key innovation was that MK-677 could be taken orally, unlike every other growth hormone secretagogue, which required injection.
Over the following decade, Merck sponsored multiple clinical trials testing MK-677 in aging, obesity, hip fracture recovery, and Alzheimer's disease. None led to FDA approval. The compound was shelved, but it found a second life on the gray market as a bodybuilding and anti-aging supplement, where it remains one of the most widely used unapproved growth hormone-related compounds. This article covers the pharmacology, the clinical evidence, the safety profile, and what remains unknown. For how MK-677 compares to injectable secretagogues, see Hexarelin: The Most Potent Growth Hormone Releasing Peptide and Ipamorelin: The Selective Growth Hormone Secretagogue. For the underlying biology, see Ghrelin: The Hunger Hormone That Rises Before Meals. For why MK-677 technically is not a peptide, see Why MK-677 Isn't Actually a Peptide (and Why It's Discussed Alongside Them).
Key Takeaways
- MK-677 is the only growth hormone booster you swallow — every other one requires an injection.
- One pill a day nearly doubled growth hormone levels in older adults after twelve months.
- Merck ran real clinical trials and then walked away — the hormone numbers improved, the patients didn't.
- In an Alzheimer's trial, IGF-1 shot up 73% but cognitive decline didn't slow at all.
- That same trial was stopped early over a possible heart failure signal in the treated group.
- Fat-free mass gains were real but modest — about 2.4 pounds over a full year of daily dosing.
- The pill activates your hunger hormone receptor, which is why increased appetite is usually the first side effect.
Pharmacology: how MK-677 works
MK-677 is a non-peptidic, orally bioavailable agonist of the growth hormone secretagogue receptor type 1a (GHSR-1a). This is the same receptor that binds ghrelin, the 28-amino-acid peptide hormone released by the stomach before meals. When ghrelin binds GHSR-1a in the hypothalamus, it triggers growth hormone release from the anterior pituitary and stimulates appetite through activation of NPY/AgRP neurons in the arcuate nucleus.[1]
MK-677 mimics ghrelin at GHSR-1a but with important pharmacokinetic differences. Ghrelin has a half-life of approximately 30 minutes. MK-677, as a synthetic small molecule, has a half-life of approximately 6 hours, with active metabolites that extend its duration of action to allow once-daily oral dosing. This sustained receptor activation produces a distinct pharmacological profile: GH is released in amplified pulsatile bursts rather than as a single spike, and IGF-1 levels rise over days to weeks as the liver responds to the elevated GH signal.
Howard et al. (1996) identified the specific receptor, publishing their discovery of a new G-protein-linked receptor for growth hormone secretagogues in Science. This receptor, GHSR-1a, is expressed in the hypothalamus and pituitary but also in the heart, vasculature, immune cells, and gastrointestinal tract, explaining why MK-677 has effects beyond GH release.[2]
A critical distinction: MK-677 is not a growth hormone-releasing hormone (GHRH) analog. GHRH and ghrelin/GH secretagogues act through different receptors and different signaling pathways. GHRH acts at the GHRH receptor on pituitary somatotrophs to initiate GH synthesis and release. Ghrelin/MK-677 acts at GHSR-1a to amplify GH pulse amplitude. The two pathways are synergistic, which is why CJC-1295 (a GHRH analog) and IGF-1 elevation represents a complementary approach.
The clinical trial evidence
Chapman et al. (1996): early dose-finding
The first clinical trial of MK-677 was published by Chapman et al. in 1996 in the Journal of Clinical Endocrinology and Metabolism. In healthy older adults (ages 64-81), daily oral MK-677 at 25 mg for 28 days increased serum IGF-1 levels to those of young adults (ages 18-30), with dose-dependent effects observed at 2, 10, and 25 mg. At the 25 mg dose, MK-677 produced a peak GH response of approximately 56 micrograms per liter after a single dose and approximately 23 micrograms per liter after one week of dosing, compared to placebo peaks of approximately 9 and 7 micrograms per liter respectively. The treatment increased mean GH peak response and restored the GH secretory pattern toward that seen in younger individuals.[3]
This was the proof-of-concept: a pill could restore growth hormone levels in elderly adults to those of young adults. The magnitude of the effect was striking: both 10 and 25 mg doses increased mean 24-hour GH concentration by 57% and 97% respectively, and boosted pulsatile GH release by 1.7-fold over 24 hours. The GH release pattern maintained its normal pulsatile character rather than producing a flat, sustained elevation, which was considered pharmacologically important because pulsatile GH is the physiological pattern and is thought to have different tissue effects than continuous GH exposure. The question was whether this hormonal restoration would translate to clinical benefit.
Copinschi et al. (1997): sleep architecture
Copinschi et al. tested MK-677 in young healthy men and found effects beyond GH secretion. Seven days of treatment increased the duration of Stage IV (deep/slow-wave) sleep by 50% and REM sleep by 20%. GH secretion is naturally pulsatile and occurs primarily during deep sleep, and the improvement in sleep architecture was likely both a cause and consequence of the enhanced GH pulsatility.[4]
This finding has been one of the most persistent reasons for MK-677's popularity in the wellness community. The mechanism is plausible: GH secretion is naturally linked to slow-wave sleep through hypothalamic GHRH neurons that are active during early sleep stages. By stimulating the ghrelin receptor, MK-677 may amplify this endogenous sleep-GH coupling. However, the study was small (7 days, young men only), and whether the sleep benefits persist with chronic use or generalize to older adults or clinical populations with insomnia has not been studied. For the broader evidence on sleep and growth hormone peptides, see How Growth Hormone Peptides Affect Sleep Quality: The Evidence.
Murphy et al. (1998): reversing diet-induced catabolism
Murphy et al. showed that MK-677 could reverse the protein catabolism (muscle breakdown) induced by a calorie-restricted diet. In healthy volunteers placed on a calorie deficit, MK-677 restored nitrogen balance to baseline, indicating preservation of lean mass. Daily GH secretion increased, and the catabolic effects of the diet were attenuated.[5]
Svensson et al. (1998): body composition in obesity
Svensson et al. conducted a two-month trial in obese subjects. MK-677 at 25 mg daily increased GH and IGF-1 levels, increased fat-free mass, and was generally well tolerated. However, fasting glucose increased and insulin sensitivity decreased, foreshadowing the metabolic concerns that would later become the compound's primary safety issue.[6]
Nass et al. (2008): the definitive elderly trial
The largest and longest controlled trial was published by Nass et al. in the Annals of Internal Medicine. A total of 65 healthy older adults (ages 60-81) were randomized to MK-677 25 mg or placebo daily for 12 months. The results showed a 97% increase in mean 24-hour GH concentration and a 55% increase in IGF-1. Fat-free mass increased by 1.1 kg in the MK-677 group (versus a decrease in the placebo group). Fasting glucose increased by approximately 5 mg/dL, and insulin sensitivity decreased modestly.[7]
The study demonstrated sustained GH and IGF-1 elevation over 12 months without tachyphylaxis (the pituitary did not become desensitized to repeated GHSR-1a stimulation). This was a significant finding because many hormonal interventions show diminishing returns over time as receptor downregulation occurs. MK-677's ability to maintain GH elevation for a full year suggested that the ghrelin receptor pathway does not undergo the same desensitization that limits, for example, exogenous GnRH agonists.
The fat-free mass gain was statistically significant but modest at 1.1 kg. To put this in perspective, recombinant human growth hormone (rhGH) at replacement doses typically produces 2-3 kg of lean mass gain over 6-12 months. MK-677's more modest effect reflects the fact that it stimulates endogenous GH secretion rather than providing supraphysiological exogenous GH. The study was not powered to detect changes in functional outcomes like strength, mobility, or fall risk. For the implications of this finding in elderly populations, see MK-677 and Muscle Mass in Elderly Populations: Sarcopenia Research.
Bach et al. (2004): hip fracture
Bach et al. tested MK-677 in elderly patients with hip fracture, hypothesizing that the anabolic effects of GH elevation would improve recovery. MK-677 increased IGF-1 but did not significantly improve functional recovery outcomes in this population. The trial highlighted a recurring theme: MK-677 reliably elevates GH and IGF-1, but translating hormonal changes to clinical endpoints has proven difficult.[8]
Sevigny et al. (2008): Alzheimer's disease
The most sobering trial was the Alzheimer's disease study published by Sevigny et al. in Neurology. MK-677 at 25 mg daily raised IGF-1 by 72.9% at 12 months, confirming target engagement. But cognitive decline proceeded at the same rate in both groups over 12 months. The trial was stopped early because of a signal for congestive heart failure in the MK-677 group, though the numbers were small.[9]
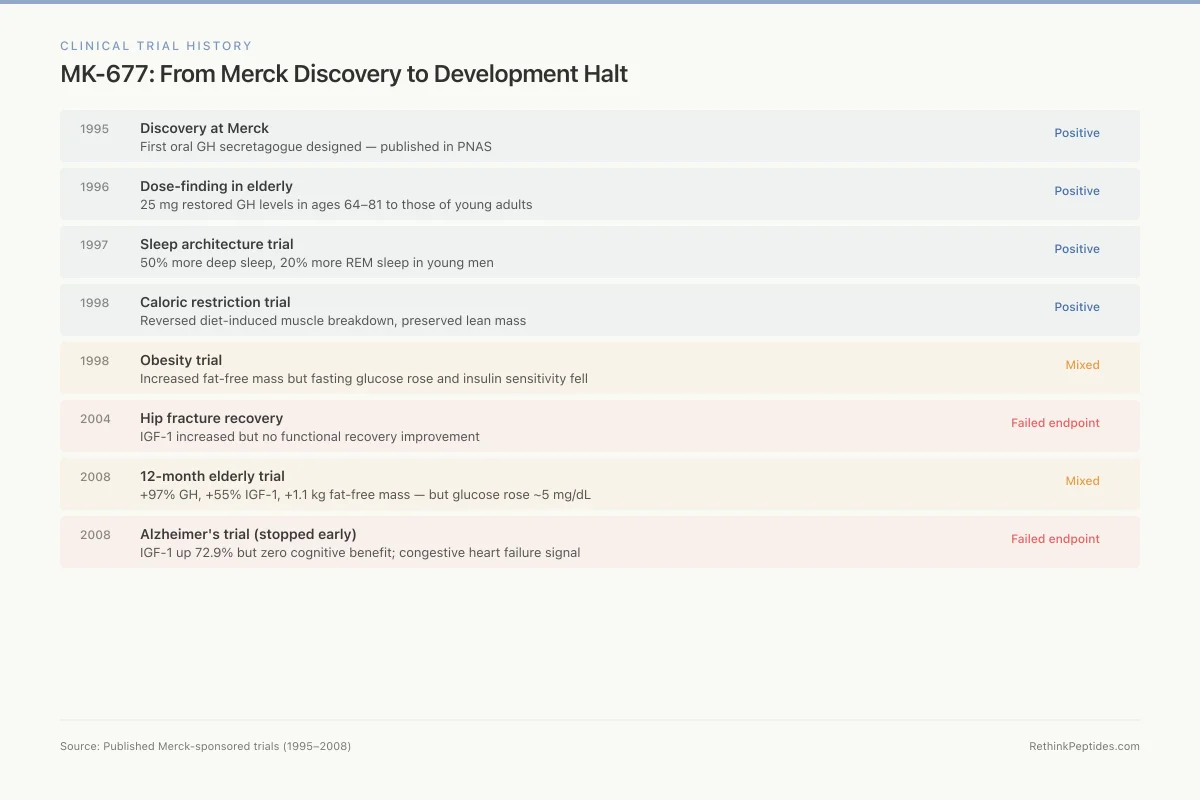
Clinical Trial History
MK-677: From Merck Discovery to Development Halt
Patchett et al. publish design of MK-0677, first oral GH secretagogue
PNAS
25 mg restored GH levels in 64–81 year olds to those of young adults
JCEM
50% more deep sleep, 20% more REM in young men at 25 mg
Neuroendocrinology
Reversed diet-induced muscle breakdown, preserved lean mass
JCEM
Increased fat-free mass but fasting glucose rose and insulin sensitivity fell
JCEM
IGF-1 increased but no functional recovery improvement
JCEM
+97% GH, +55% IGF-1, +1.1 kg fat-free mass, but glucose rose ~5 mg/dL
Ann Intern Med
IGF-1 up 72.9% but zero cognitive benefit; CHF signal emerged
Neurology
Source: Published Merck-sponsored trials (1995–2008)
 View as image
View as imageThe heart failure signal, combined with the metabolic effects (glucose elevation, insulin resistance), contributed to Merck's decision not to pursue MK-677 further. No Phase III trial was ever initiated. The Alzheimer's trial result also raised a broader question about whether GH/IGF-1 axis manipulation is a viable strategy for neurodegenerative disease: despite robust target engagement (IGF-1 increased by 72.9%), the downstream cognitive benefit was absent, suggesting either that IGF-1 elevation does not reach the central nervous system in sufficient quantity, or that IGF-1 is not a rate-limiting factor in Alzheimer's pathology.
Safety and side effects
The safety profile of MK-677 from controlled trials includes several consistent findings.
Appetite stimulation is the most immediate effect, occurring within hours of the first dose and persisting for several weeks before attenuating. This is expected from a ghrelin mimetic, as appetite stimulation is ghrelin's primary physiological function.
Fasting glucose elevation of approximately 5 mg/dL was observed across multiple trials. In the 12-month Nass trial, insulin sensitivity decreased. For individuals with pre-existing insulin resistance or prediabetes, chronic MK-677 use could theoretically accelerate progression to type 2 diabetes. For the full metabolic profile, see MK-677 Side Effects: Water Retention, Insulin Resistance, and Hunger.
Peripheral edema (fluid retention), particularly in the lower extremities, was reported in early trials and is a known effect of GH elevation.
The congestive heart failure signal from the Alzheimer's trial remains concerning. GH excess (as in acromegaly) is associated with cardiomyopathy. Whether chronic MK-677 use at standard doses carries a meaningful cardiac risk is unknown. For the theoretical risks of long-term IGF-1 elevation, see MK-677 Long-Term Safety: What Sustained IGF-1 Elevation Means.
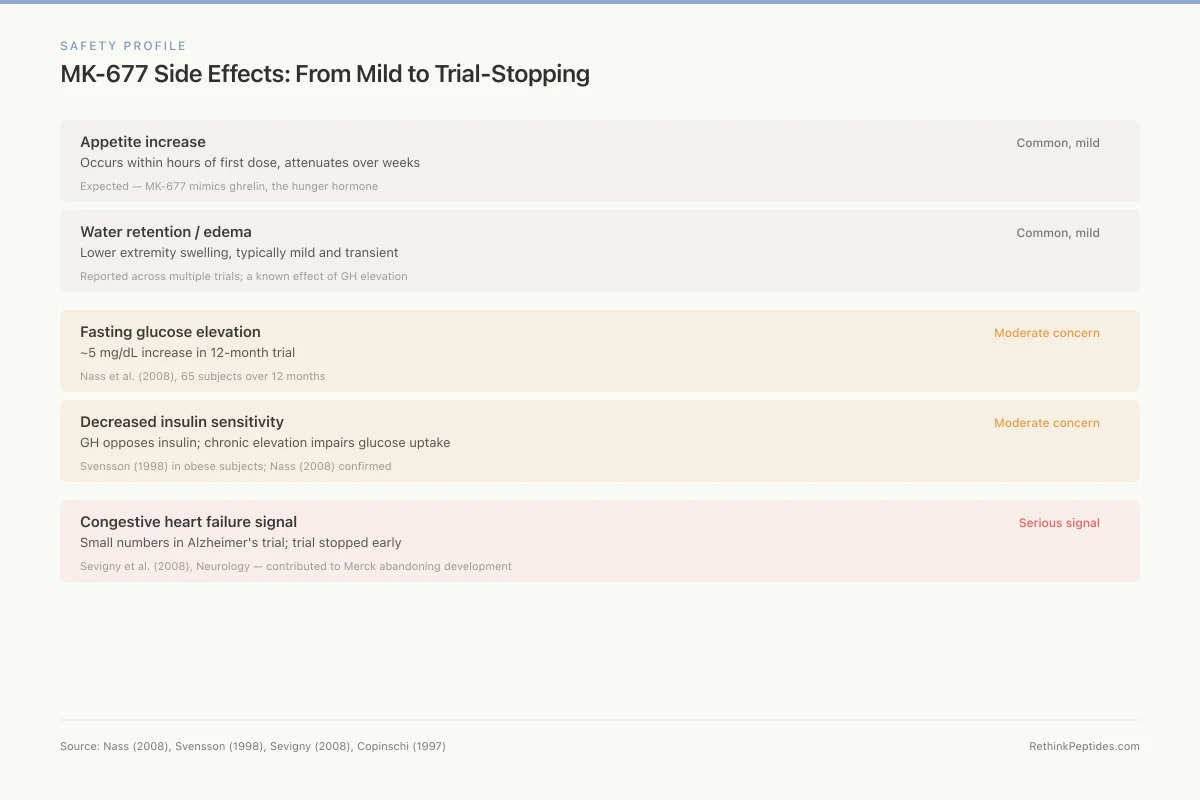
Safety Profile
MK-677 Side Effects: From Mild to Trial-Stopping
Occurs within hours of first dose, attenuates over weeks
Expected — MK-677 mimics ghrelin, the hunger hormone
Lower extremity swelling, typically mild and transient
Reported across multiple trials; a known effect of GH elevation
~5 mg/dL increase in 12-month trial
Nass et al. (2008), 65 subjects over 12 months
GH opposes insulin; chronic elevation impairs glucose uptake
Svensson (1998) in obese subjects; Nass (2008) confirmed
Small numbers in Alzheimer's trial; trial stopped early
Sevigny et al. (2008), Neurology — contributed to Merck abandoning development
Source: Nass (2008), Svensson (1998), Sevigny (2008), Copinschi (1997)
 View as image
View as imageSafety
ModerateInsulin resistance with chronic use
Concern
MK-677 raises fasting glucose by about 5 mg/dL and decreases insulin sensitivity. Growth hormone directly opposes insulin's effects on glucose metabolism. In the Svensson obesity trial, insulin sensitivity fell significantly.
What the research says
Clinical trials used 25 mg in selected healthy volunteers with monitoring. The real-world population — often with metabolic risk factors, using higher doses without monitoring — faces a different risk profile.
Particularly relevant for: Anyone with prediabetes, insulin resistance, obesity, or metabolic syndrome — populations most likely to seek MK-677 for body composition
What to do
If using MK-677, monitor fasting glucose and HbA1c regularly. The metabolic risk is higher in people who already have insulin resistance.
Nass et al. (2008), Annals of Internal Medicine; Svensson et al. (1998), JCEM
Cardaci et al. (2022) published the only study examining MK-677 use as it occurs in the real world (combined with the SARM LGD-4033). They found changes in body composition, circulating biomarkers, and skeletal muscle gene expression, but the study could not isolate MK-677's individual contribution from the SARM co-administration.[10]
The population using MK-677 today
The demographics of MK-677 users bear little resemblance to the clinical trial populations. Merck tested MK-677 primarily in elderly adults (ages 60-81) with age-related GH decline, hip fracture patients, and Alzheimer's disease patients. The gray-market user base is predominantly young men (ages 18-40) using MK-677 for bodybuilding, athletic performance, sleep improvement, or general anti-aging.
This mismatch matters for several reasons. Young men with intact GH-IGF-1 axes are elevating IGF-1 above already-normal levels, producing a state of pharmacological GH excess rather than restoration of a deficit. The risk-benefit calculus is fundamentally different: in elderly adults with demonstrably low GH, restoration carries plausible benefit. In young adults with normal GH, supraphysiological IGF-1 elevation carries the risks of excess (insulin resistance, potential cancer promotion, cardiac hypertrophy) without the clear rationale of correcting a deficiency.
The dosing regimens in online communities also frequently exceed trial protocols. Some users report 50 mg daily (double the maximum trial dose), extended cycles of 6-12 months or continuous use, and combination with other GH-elevating compounds that could produce additive or synergistic IGF-1 elevation. None of these protocols has safety data.
The bone metabolism data
MK-677 produced some of the most interesting findings in bone metabolism. Svensson et al. (1998) showed that treatment increased markers of bone formation (osteocalcin, type I procollagen) in a pattern suggesting enhanced bone turnover.[11]
The oral growth hormone secretagogue also increased markers of bone turnover when measured over longer time periods. Murphy et al. documented similar findings. The clinical significance of these changes, whether they translate to increased bone mineral density or reduced fracture risk, was never established because no trial was designed with bone density as a primary endpoint. The hip fracture trial (Bach 2004) did not show improved recovery, but that is a different question than whether MK-677 could prevent fractures in an at-risk population.
What the evidence does and does not show
MK-677's evidence base is unusually strong for a compound that never reached Phase III. Multiple randomized, placebo-controlled trials in hundreds of subjects demonstrate consistent, dose-dependent increases in GH secretion and IGF-1 levels. The compound reliably restores age-related decline in the GH-IGF-1 axis to levels seen in young adults.
The translation gap is equally clear. Despite 97% increases in GH and 55% increases in IGF-1, the clinical endpoints, functional improvement in the elderly, fracture recovery, cognitive preservation in Alzheimer's, were either absent or insufficient to justify further development. This raises a fundamental question about the GH-IGF-1 axis in aging: does the decline in GH with age cause functional decline, or is it an adaptive response? If it is adaptive (a protective downregulation to reduce cancer risk and metabolic demand), then restoring youthful GH levels in elderly bodies could be harmful.
The cancer question looms over all GH-elevating strategies. IGF-1 is a growth factor that promotes cell proliferation and inhibits apoptosis. Epidemiological studies link higher circulating IGF-1 with increased risk of several cancers, including prostate, breast, and colorectal cancer. The relationship is not simple: IGF-1 levels in the upper quartile of the normal range are associated with increased cancer risk, but whether pharmacologically elevating IGF-1 from the lower quartile (as MK-677 does in elderly adults) carries the same risk is unclear. No MK-677 trial has reported increased cancer incidence, but the longest trial was only 2 years, and cancer latency periods are typically measured in decades. The AOD-9604 approach to growth hormone fragments represents an attempt to capture metabolic benefits without IGF-1 elevation.
The insulin resistance findings deserve particular attention. Growth hormone is a counter-regulatory hormone that opposes insulin's effects on glucose metabolism. Chronically elevated GH, as produced by MK-677, increases hepatic glucose output and reduces peripheral glucose uptake. In clinical trials, the glucose elevation was modest (approximately 5 mg/dL fasting glucose increase), and no participant developed overt diabetes. However, the trial populations were selected healthy volunteers. In real-world use, where many users have risk factors for metabolic disease (obesity, sedentary lifestyle, concomitant use of other metabolically active compounds), the chronic insulin resistance could contribute to clinically meaningful metabolic deterioration. The 1998 Svensson trial in obese subjects specifically documented decreased insulin sensitivity, suggesting that MK-677 carries more metabolic risk in the populations most likely to seek it out for body composition purposes.
There is also the question of whether MK-677's effects on body composition are truly beneficial or partly cosmetic. The increased fat-free mass measured in trials includes both skeletal muscle and organ mass, and GH-driven fluid retention can inflate lean mass measurements without reflecting actual muscle protein accretion. Similarly, the appetite stimulation from ghrelin receptor activation can counteract the body composition benefits by driving increased caloric intake, particularly in users not following controlled diets.
MK-677 is not approved by the FDA, EMA, or any regulatory agency. It is sold through gray-market peptide vendors and online supplement retailers, often with minimal quality control. The FDA has specifically noted that MK-677 cannot legally be sold as a dietary supplement. Users are self-administering a compound that failed to reach Phase III development, at doses and durations that exceed any published trial, without medical supervision or monitoring.
The gap between the clinical evidence and actual use patterns is significant. Clinical trials typically administered 25 mg daily for periods of 2-24 months with regular monitoring of glucose, IGF-1, cardiac function, and adverse events. Gray-market use often involves similar or higher doses for years without monitoring. Some users combine MK-677 with SARMs (like LGD-4033, as documented by Cardaci et al.), exogenous testosterone, or other compounds, creating pharmacological interactions that have never been studied. The longest published trial was 2 years (the Alzheimer's study); many self-administering users report taking MK-677 continuously for longer periods.
The irony of MK-677's trajectory is that it was developed by one of the world's largest pharmaceutical companies with the full rigor of drug development, yet it found its largest market as an unregulated compound used outside of any medical framework. The clinical data from Merck's trials is freely available in peer-reviewed journals, giving MK-677 a veneer of evidence-based legitimacy that most gray-market compounds lack, while simultaneously revealing the safety signals that caused its developer to abandon it. The compound exists in an unusual space: better studied than most gray-market peptides, but studied enough to have demonstrated the very problems that ended its pharmaceutical development.
The Bottom Line
MK-677 (ibutamoren) is the only oral growth hormone secretagogue to reach Phase II clinical trials. It reliably increases GH and IGF-1 levels to those of young adults when given to elderly subjects, and it improves sleep architecture and preserves lean mass during caloric restriction. However, no trial demonstrated meaningful clinical endpoints (functional improvement, fracture prevention, cognitive preservation), and safety signals including glucose elevation, insulin resistance, edema, and a possible congestive heart failure risk halted development. It remains widely used through unregulated channels despite never receiving regulatory approval.
Sources & References
- 1RPEP-00339·Patchett, A A et al. (1995). “MK-677: The Oral Growth Hormone Pill That Changed the Field.” Proceedings of the National Academy of Sciences of the United States of America.Study breakdown →PubMed →↩
- 2RPEP-00380·Pong, S S et al. (1996). “Discovery of the Receptor That MK-677 and GHRPs Use to Release Growth Hormone.” Molecular endocrinology (Baltimore.Study breakdown →PubMed →↩
- 3RPEP-00357·Chapman, I M et al. (1996). “Oral MK-677 Restores Youthful Growth Hormone and IGF-1 Levels in Elderly Adults.” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 4RPEP-00402·Copinschi, G et al. (1997). “Oral MK-677 Significantly Improves Sleep Quality in Both Young and Older Adults.” Neuroendocrinology.Study breakdown →PubMed →↩
- 5RPEP-00480·Murphy, M G et al. (1998). “MK-677 Pill Reverses Muscle Breakdown From Calorie Restriction.” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 6RPEP-00497·Svensson, J et al. (1998). “Oral MK-677 Increases Growth Hormone, Lean Mass, and Calorie Burning in Obese Men.” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 7RPEP-01392·Nass, Ralf et al. (2008). “MK-677 for 1 Year in Healthy Elderly: Increased Lean Mass But Also Fat and Insulin.” Annals of internal medicine.Study breakdown →PubMed →↩
- 8RPEP-00882·Bach, Mark A et al. (2004). “MK-677 for Hip Fracture Recovery: Increased IGF-1 and GH But Mixed Functional Results.” Journal of the American Geriatrics Society.Study breakdown →PubMed →↩
- 9RPEP-01418·Sevigny, J J et al. (2008). “MK-677 Does NOT Slow Alzheimer's Disease Progression Despite Raising GH and IGF-1.” Neurology.Study breakdown →PubMed →↩
- 10RPEP-06030·Cardaci, Thomas D et al. (2022). “LGD-4033 and MK-677 use impacts body composition, circulating biomarkers, and skeletal muscle androgenic hormone and receptor content: A case report..” Experimental physiology.Study breakdown →PubMed →↩
- 11RPEP-00496·Svensson, J et al. (1998). “Oral MK-677 Activates Bone Remodeling in Obese Young Men.” Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research.Study breakdown →PubMed →↩