CJC-1295 and IGF-1: What the Research Shows
CJC-1295 and Growth Hormone
1.5-3x IGF-1 Elevation
A single subcutaneous injection of CJC-1295 elevated mean IGF-1 levels 1.5- to 3-fold above baseline for 9 to 11 days in healthy adults. Growth hormone levels increased 2- to 10-fold for 6 or more days.
Teichman et al., JCEM, 2006
Teichman et al., JCEM, 2006
If you only read one thing
CJC-1295 is a lab-made version of the hormone that tells your pituitary to release growth hormone. One injection can raise your GH and IGF-1 levels for over a week because the drug glues itself to a blood protein called albumin. Only one small human study — 2006 — actually measured those effects. The company halted the trial that same year after a patient death and never resumed. Every claim about muscle, fat, sleep, or injury recovery is based on the biology, not on outcome trials.
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH) that was developed by ConjuChem Biotechnologies to produce sustained elevation of growth hormone (GH) and insulin-like growth factor 1 (IGF-1) from a single injection. In a 2006 clinical trial in healthy adults, a single subcutaneous dose increased mean plasma GH concentrations 2- to 10-fold for 6 or more days and IGF-1 levels 1.5- to 3-fold for 9 to 11 days.[1] Those numbers made CJC-1295 one of the most discussed peptides in the growth hormone optimization community. They also raise questions the community does not always address: why was the clinical program discontinued, what do the numbers actually mean physiologically, and what are the risks of sustained GH/IGF-1 elevation that cannot be turned off?
Key Takeaways
- A single CJC-1295 injection produced dose-dependent GH increases of 2- to 10-fold and IGF-1 increases of 1.5- to 3-fold in healthy adults, with effects lasting 6+ and 9-11 days respectively (Teichman et al., JCEM, 2006)
- CJC-1295 preserved pulsatile GH secretion despite continuous GHRH receptor stimulation, with elevated trough GH levels driving the sustained IGF-1 increase (Ionescu and Bhatt, JCEM, 2006)
- After multiple doses, IGF-1 levels remained above baseline for up to 28 days (Teichman et al., JCEM, 2006)
- The phase II trial in HIV-associated lipodystrophy was halted in 2006 after a patient death, attributed by investigators to pre-existing coronary disease, but development was never resumed
- CJC-1295 achieves its long half-life (5.8-8.1 days) through a Drug Affinity Complex (DAC) that covalently binds serum albumin after injection
- No CJC-1295 product has received FDA approval; it was removed from the FDA's Category 2 compounding list in 2024 after nomination withdrawal
What CJC-1295 Is
CJC-1295 is a modified version of the first 29 amino acids of human GHRH (growth hormone-releasing hormone, also called GRF 1-29 or sermorelin). Native GHRH has a plasma half-life of approximately 7 minutes, making it impractical as a drug. CJC-1295 extends this through two strategies.
First, four amino acid substitutions (at positions 2, 8, 15, and 27) protect the peptide from enzymatic degradation by dipeptidyl peptidase IV (DPP-IV), which rapidly cleaves native GHRH at position 2. These substitutions increase the intrinsic half-life from minutes to approximately 30 minutes.
Second, and more significantly, CJC-1295 incorporates a Drug Affinity Complex (DAC): a maleimidopropionyl group attached to a lysine residue at position 30. After subcutaneous injection, this reactive group covalently and irreversibly binds to cysteine-34 on serum albumin. Because albumin has a circulating half-life of approximately 19 days, the CJC-1295-albumin conjugate persists in the bloodstream for days, continuously stimulating GHRH receptors on pituitary somatotroph cells.
The estimated half-life in humans is 5.8 to 8.1 days.[1] This represents a roughly 700-fold increase over the unmodified peptide and a roughly 1,000-fold increase over native GHRH. For context on how this albumin-binding strategy compares to other long-acting peptide modifications, the same pharmacokinetic principle underlies semaglutide (GLP-1) and cagrilintide (amylin). For a deeper explanation of CJC-1295's structure and design, see CJC-1295: The Growth Hormone Releasing Hormone Analog Explained.
The Clinical Evidence
The published human data for CJC-1295 comes from a small number of studies, all conducted before the clinical program was discontinued.
The Teichman 2006 Trial
The primary clinical evidence is a dose-escalation study published in the Journal of Clinical Endocrinology and Metabolism in 2006.[1] Healthy adults (ages 21-61) received single or multiple subcutaneous injections of CJC-1295 at doses of 30, 60, or 125 micrograms per kilogram.
After a single injection:
- GH levels: Increased 2- to 10-fold in a dose-dependent manner, remaining elevated for 6 or more days
- IGF-1 levels: Increased 1.5- to 3-fold, remaining elevated for 9 to 11 days
- Duration: At the 60 mcg/kg dose, mean GH remained above baseline for at least 6 days. Mean IGF-1 remained above baseline for at least 11 days.
After multiple weekly doses (2 to 3 injections):
- IGF-1 accumulation: Mean IGF-1 levels remained above baseline for up to 28 days after the last injection
- GH pulsatility: A companion study by Ionescu and Bhatt demonstrated that pulsatile GH secretion was preserved during continuous GHRH receptor stimulation by CJC-1295.[2] This was a significant finding because exogenous GH administration abolishes pulsatility, raising concerns about downstream effects. CJC-1295 elevated both trough and peak GH levels while maintaining the normal pulsatile pattern. The authors concluded that the sustained trough elevation, rather than peak enhancement, was the primary driver of IGF-1 increase.
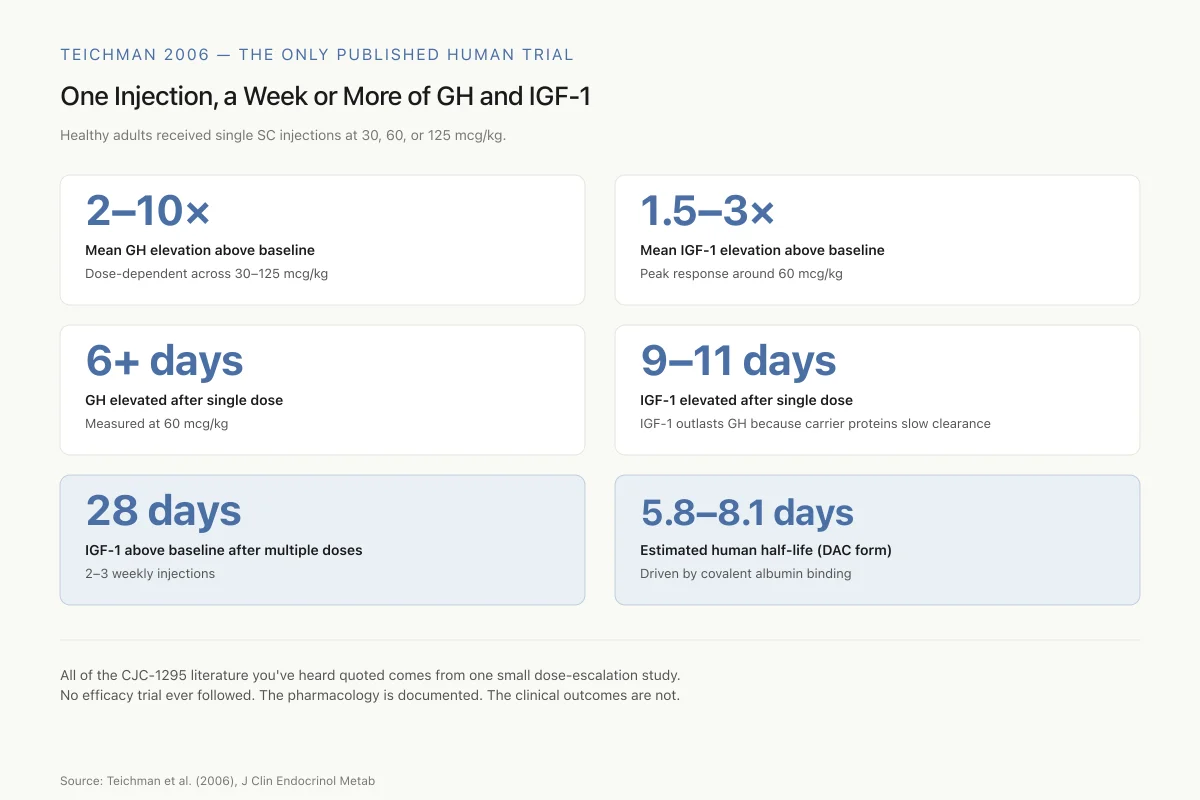
Teichman 2006 — The Only Published Human Trial
One Injection, a Week or More of GH and IGF-1
Healthy adults received single subcutaneous injections at 30, 60, or 125 mcg/kg. These are the numbers the entire CJC-1295 community cites.
2–10×
Mean GH elevation above baseline
Dose-dependent across 30–125 mcg/kg
1.5–3×
Mean IGF-1 elevation above baseline
Peak response around 60 mcg/kg
6+ days
Duration of elevated GH from single dose
Measured at 60 mcg/kg
9–11 days
Duration of elevated IGF-1 from single dose
IGF-1 outlasts GH because its carrier proteins slow clearance
28 days
IGF-1 still above baseline after multiple doses
2–3 weekly injections
5.8–8.1 days
Estimated human half-life (DAC form)
Driven by covalent albumin binding
All of the CJC-1295 literature you’ve heard quoted comes from one small dose-escalation study. No efficacy trial ever followed. The pharmacology is documented. The clinical outcomes are not.
Source: Teichman et al. (2006), J Clin Endocrinol Metab
 View as image
View as imageProteomic Analysis
A 2009 study used two-dimensional gel electrophoresis and mass spectrometry to examine serum protein changes in CJC-1295-treated subjects.[3] The analysis identified changes in 22 distinct proteins, spanning lipid metabolism (apolipoprotein changes), immune function (complement factors), and the acute-phase response (haptoglobin, alpha-1 antitrypsin). These changes were consistent with known downstream effects of GH/IGF-1 axis activation but provided the most detailed molecular fingerprint of what sustained GHRH stimulation does at a systemic level. The proteomic profile showed that CJC-1295 does not simply "boost GH." It triggers a cascade of metabolic changes across multiple organ systems, a point worth considering when evaluating the peptide's risk-benefit profile.
Animal Studies
In a GHRH-knockout mouse model, once-daily CJC-1295 administration normalized growth, confirming that the peptide's effects are mediated entirely through the GHRH receptor and not through alternative pathways.[4] The knockout mouse study was important for another reason: it demonstrated that CJC-1295 could functionally replace endogenous GHRH. This suggested potential therapeutic applications in conditions where GHRH production is impaired, such as age-related GH decline and certain forms of growth hormone deficiency.
CJC-1295 with DAC vs. without DAC
The peptide community commonly distinguishes between "CJC-1295 with DAC" and "CJC-1295 without DAC" (also called Mod GRF 1-29 or Modified GRF 1-29). This distinction is important but frequently misunderstood.
CJC-1295 with DAC is the original compound developed by ConjuChem and used in all published clinical trials. The DAC modification is what gives it the 5.8- to 8.1-day half-life. Without the DAC, you have Mod GRF 1-29: the same four amino acid substitutions for DPP-IV resistance, but no albumin-binding modification. Mod GRF 1-29 has a half-life of approximately 30 minutes.
The practical difference is profound. CJC-1295 with DAC produces continuous, sustained GHRH receptor stimulation for days. Mod GRF 1-29 produces a brief pulse of GHRH activity that more closely mimics the body's natural episodic GHRH release. Some practitioners prefer Mod GRF 1-29 precisely because the shorter duration allows for more physiological GH pulsatility, while CJC-1295 with DAC elevates trough GH levels continuously. For a detailed comparison, see CJC-1295 with DAC vs without DAC: What's the Difference?. For a dedicated analysis of the short-acting version, see Mod GRF 1-29: The Short-Acting Version of CJC-1295.
The GH/IGF-1 Axis: What Elevation Actually Means
Understanding what CJC-1295 does requires understanding the GH/IGF-1 axis. GHRH is released from the hypothalamus in pulses, typically concentrated during sleep and exercise. It stimulates pituitary somatotroph cells to release GH, which circulates to the liver and other tissues where it stimulates IGF-1 production.[5]
IGF-1 is the primary mediator of GH's anabolic effects: muscle protein synthesis, bone growth, cellular repair, and metabolism. When people discuss "growth hormone optimization," what they often mean functionally is IGF-1 optimization. GH itself is a relatively short-lived signal (half-life approximately 20 minutes). IGF-1, bound to carrier proteins in the bloodstream, has a much longer half-life and is the sustained effector molecule.
A 2013 review of the GHRH/GH/IGF-1 axis pathophysiology detailed the regulatory feedback loops: IGF-1 feeds back to both the hypothalamus and pituitary to suppress GHRH and GH release, while somatostatin provides additional inhibitory control.[5] CJC-1295 bypasses one layer of this regulation by providing continuous GHRH receptor stimulation, but the somatostatin and IGF-1 feedback loops remain intact, which is why GH pulsatility is preserved.
The age-related decline in GH/IGF-1 (somatopause) is well documented. GH secretion decreases approximately 14% per decade after age 30, driven by reduced GHRH output and increased somatostatin tone in the hypothalamus. By age 60, many adults have GH secretion rates comparable to patients with clinical GH deficiency, though they do not necessarily meet the diagnostic criteria for GH deficiency syndrome. A 2013 study examining the role of GHRH analogs in age-related GH decline found that GHRH stimulation could partially restore youthful GH secretion patterns, suggesting that the pituitary retains the capacity to respond even when hypothalamic drive diminishes.[6] This is the theoretical basis for CJC-1295's appeal in anti-aging contexts: restoring GH/IGF-1 to younger levels through receptor stimulation rather than direct GH replacement. The key word is "theoretical." No clinical trial has tested whether this restoration produces meaningful anti-aging outcomes. For a broader understanding of how CJC-1295 stimulates GH release at the receptor level, see How CJC-1295 Stimulates Growth Hormone: Mechanism of Action.
CJC-1295 and Ipamorelin: The Common Combination
CJC-1295 is frequently paired with ipamorelin, a selective growth hormone secretagogue (GHS) that acts through the ghrelin receptor (GHS-R1a) on pituitary cells. The rationale: GHRH (via CJC-1295) and GHS peptides (via ipamorelin) stimulate GH release through complementary but distinct pituitary mechanisms.
Ipamorelin was characterized in 1998 as the first GH secretagogue that selectively released GH without affecting cortisol, prolactin, or ACTH at GH-stimulating doses.[7] This selectivity profile makes it theoretically preferable to earlier GH secretagogues like GHRP-6 and GHRP-2, which also elevated cortisol and prolactin. Earlier work on growth hormone-releasing peptides had established the existence of the GHS receptor pathway as distinct from the GHRH receptor pathway.[8]
The theoretical basis for combining GHRH analogs with GHS peptides is well established. A 2005 study showed that GH-releasing peptide-2 (GHRP-2), acting through the GHS receptor, synergized with GHRH to produce GH release greater than either peptide alone.[10] The two receptor systems converge on pituitary somatotroph cells but through different intracellular signaling cascades: GHRH via cAMP/protein kinase A, and GHS peptides via phospholipase C/inositol triphosphate. This mechanistic independence predicts additive or synergistic GH release.
The CJC-1295/ipamorelin combination specifically has no published clinical trial data. Its popularity is based on the pharmacological logic of dual-pathway stimulation and anecdotal clinical reports. No controlled study has established optimal dosing, safety profile, or efficacy for this combination in humans. The absence of data is not evidence of harm, but it means that anyone using this combination is participating in an uncontrolled experiment on themselves.
Why the Clinical Program Stopped
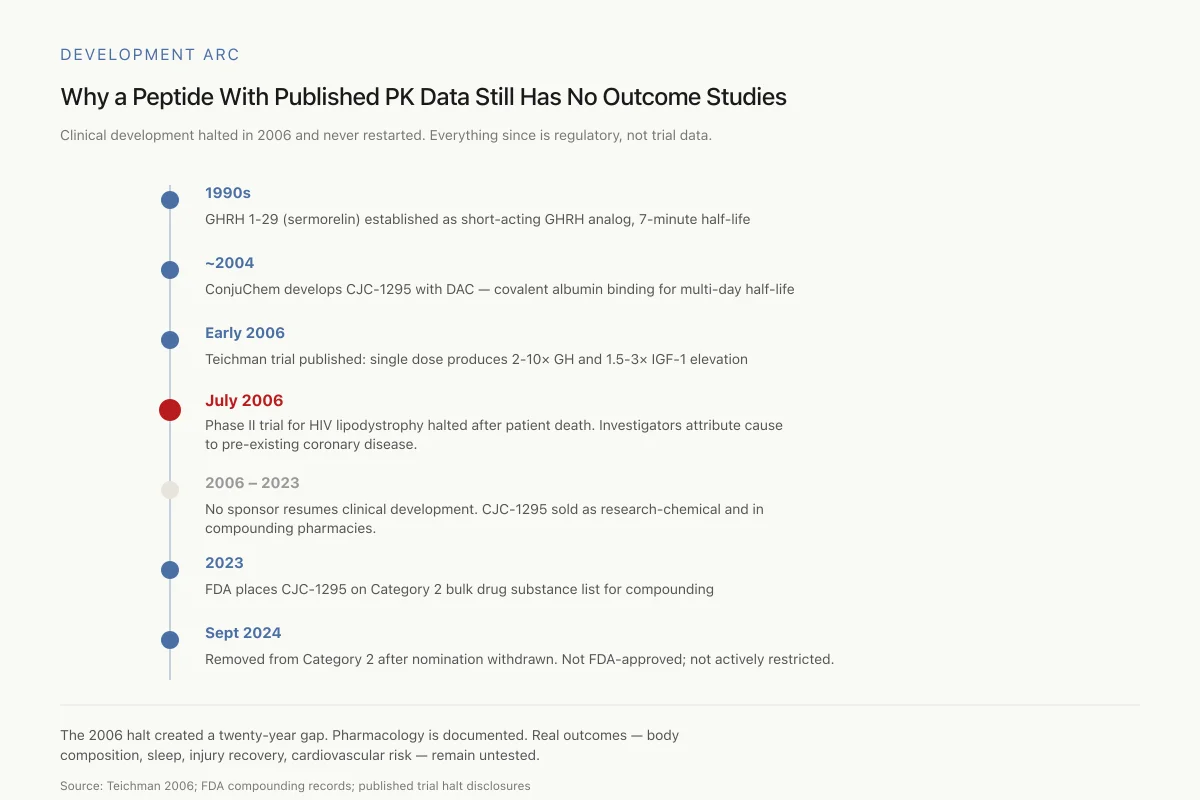
Development Arc
Why a Peptide With Published PK Data Still Has No Clinical Outcome Studies
Clinical development halted in 2006 and never restarted. Everything that followed is regulatory status, not trial data.
The 2006 halt created a twenty-year gap in the clinical record. What the peptide does to blood levels is well-documented. What it does to real outcomes — body composition, sleep, injury recovery, cardiovascular risk — remains untested.
Source: Teichman 2006; FDA compounding records; published trial halt disclosures
 View as image
View as imageIn July 2006, ConjuChem's Phase II trial of CJC-1295 for HIV-associated lipodystrophy was halted after a patient death at a study site in Argentina. The study had enrolled 192 participants with HIV-related visceral obesity across North and South American sites.
The attending physician attributed the death to pre-existing asymptomatic coronary artery disease with plaque rupture, concluding it was unrelated to CJC-1295 treatment. ConjuChem suspended the trial as a precaution. The company never resumed clinical development, and no subsequent sponsor has taken CJC-1295 (with DAC) into clinical trials.
The unresolved question is whether sustained GH/IGF-1 elevation carries cardiovascular risk. GH is known to have acute cardiovascular effects including increased heart rate and peripheral vasodilation. The FDA has cited these effects in safety assessments. Sustained elevation that cannot be "turned off" for 6 to 8 days, unlike short-acting GHRH analogs, raises theoretical concerns about cumulative cardiovascular stress. No study has conclusively established or ruled out this risk.
There is a separate concern about long-term IGF-1 elevation and cancer risk. Epidemiological data consistently associate high-normal and elevated IGF-1 levels with increased risk of several cancers, including prostate, breast, and colorectal. The relationship is correlational, not necessarily causal, and the magnitude of risk at the IGF-1 elevations produced by CJC-1295 (1.5- to 3-fold) is not established. This concern applies to all GH secretagogues and GH replacement therapies, not specifically to CJC-1295. However, the concern has particular relevance for CJC-1295 because of the sustained, multi-day IGF-1 elevation that cannot be interrupted once administered. With short-acting peptides like Mod GRF 1-29 or ipamorelin, a missed dose returns IGF-1 to baseline within hours. With CJC-1295 DAC, the committed duration of IGF-1 elevation is measured in days.
The insulin resistance question also merits attention. GH is a counter-regulatory hormone that opposes insulin's glucose-lowering effects. Sustained GH elevation raises fasting glucose and can reduce insulin sensitivity. In the Teichman trial, glucose and insulin parameters were monitored but detailed metabolic outcomes were not reported beyond the observation that no serious adverse events occurred. The absence of metabolic data from longer-term dosing is a meaningful gap.
Safety
ModerateMulti-day effects cannot be reversed
Concern
A single CJC-1295 injection produces 5–8 days of sustained GH/IGF-1 elevation. Unlike short-acting peptides (Mod GRF 1-29, ipamorelin) where a missed dose clears in hours, CJC-1295 DAC can't be 'turned off.' Long-term cardiovascular and cancer-risk implications of sustained IGF-1 elevation have never been tested.
What the research says
For people pursuing GH optimization, short-acting GHRH analogs allow dose interruption if adverse effects appear. DAC-modified CJC-1295 does not. Consider whether you're willing to commit to the full effect window of each injection.
Particularly relevant for: Anyone using compounded CJC-1295, especially with risk factors (family history of cancer, diabetes, cardiovascular disease)
What to do
Discuss with a physician familiar with GH secretagogue safety. Baseline and periodic IGF-1 monitoring is reasonable. Stop if IGF-1 exceeds age-adjusted reference range.
Teichman 2006; FDA GH secretagogue safety literature
Regulatory Status
CJC-1295 was placed on the FDA's Category 2 bulk drug substance list in 2023, alongside BPC-157 and other peptides. In September 2024, CJC-1295 was removed from Category 2 after its nominator withdrew the nomination. This removal does not constitute FDA approval or endorsement. It means the FDA no longer considers CJC-1295 among the substances requiring specific enforcement action against compounding pharmacies.
CJC-1295 is not FDA-approved for any indication. It has been identified as a substance of interest in anti-doping contexts. Henninge and colleagues published a 2010 method for identifying CJC-1295 in biological samples using liquid chromatography-mass spectrometry, establishing the analytical foundation for detection in competitive athletes.[9] WADA prohibits all growth hormone secretagogues under category S2 (Peptide Hormones, Growth Factors, Related Substances, and Mimetics). The prohibition covers CJC-1295 with and without DAC, Mod GRF 1-29, and all other GHRH analogs.
The regulatory landscape for GH secretagogues remains fragmented. CJC-1295 can be legally compounded by licensed pharmacies (since its Category 2 removal), but it is banned in competitive sports, unavailable as an approved drug, and sold through gray-market channels labeled "for research use only." This three-tier regulatory status creates confusion about what "legal" means in practice. For the broader regulatory context of peptide compounding, see AOD-9604: The Growth Hormone Fragment for Fat Metabolism.
What the Evidence Does and Does Not Support
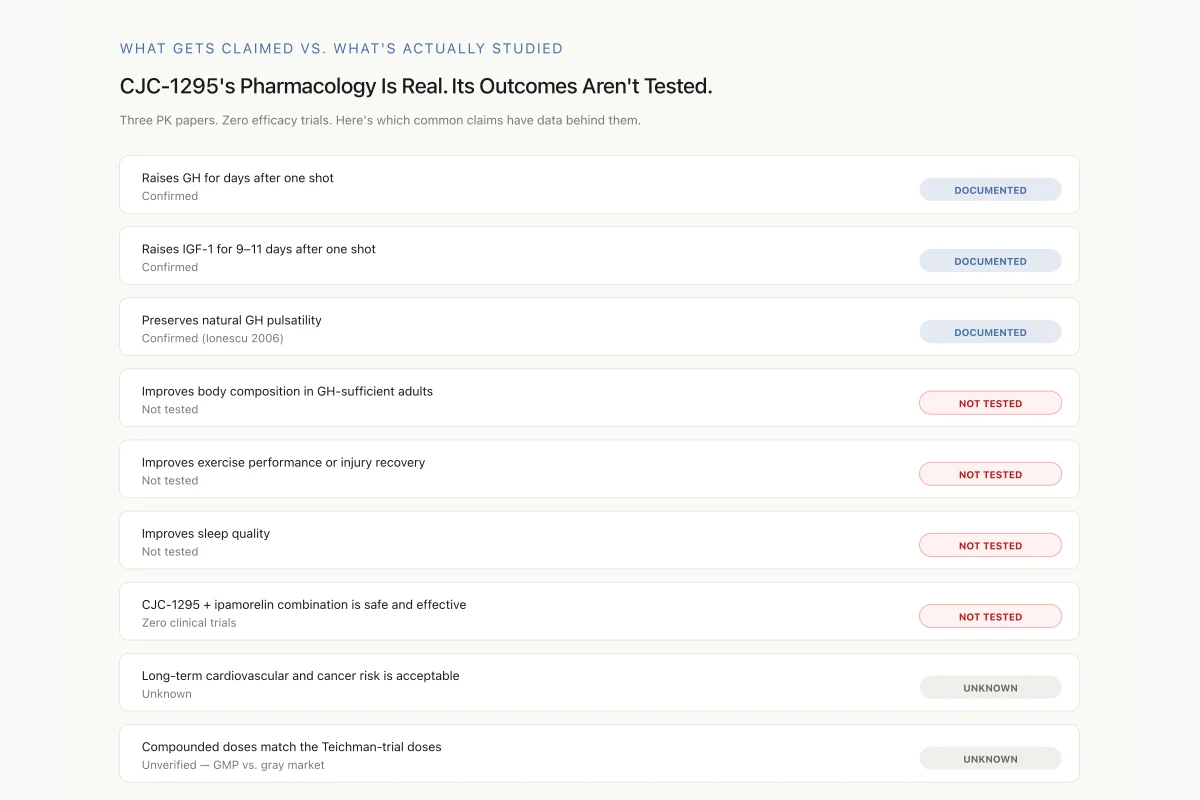
What Gets Claimed vs. What’s Actually Studied
CJC-1295’s Pharmacology Is Real. Its Outcomes Aren’t Tested.
Three pharmacokinetic papers. Zero efficacy trials. Here’s which common claims actually have data behind them.
Raises GH for days after one shot
Confirmed
Raises IGF-1 for 9–11 days after one shot
Confirmed
Preserves natural GH pulsatility
Confirmed (Ionescu 2006)
Improves body composition in GH-sufficient adults
Not tested
Improves exercise performance or injury recovery
Not tested
Improves sleep quality
Not tested
CJC-1295 + ipamorelin combination protocols are safe and effective
Zero clinical trials
Long-term cardiovascular and cancer risk is acceptable
Unknown
Compounded doses match the Teichman-trial doses
Unverified — GMP vs. gray market
Three documented pharmacology findings. Six claims ranging from “not tested” to “unknown.” That’s the actual evidence map — not what supplement marketing implies.
Source: Teichman 2006; Ionescu 2006; Sackmann-Sala 2009 — the complete CJC-1295 human literature
 View as image
View as imageThe published clinical data for CJC-1295 is small but methodologically sound. Three papers from the same clinical development program (Teichman 2006, Ionescu 2006, Sackmann-Sala 2009) provide consistent, dose-response evidence that CJC-1295 produces sustained GH and IGF-1 elevation in healthy adults. The preservation of GH pulsatility is a genuine pharmacological advantage over direct GH injection.
What the evidence does not support:
Specific clinical outcomes. No published study has shown that CJC-1295 improves body composition, exercise performance, injury recovery, sleep quality, or any other endpoint that drives its off-label popularity. The clinical program was halted before efficacy data could be generated. By contrast, recombinant human GH itself has been studied extensively in body composition trials, showing modest decreases in fat mass and increases in lean mass in GH-deficient populations. Whether CJC-1295-induced GH/IGF-1 elevation produces comparable body composition effects in GH-sufficient adults has never been tested.
Safety in chronic use. The longest published exposure is a few weeks of dosing in healthy volunteers. Long-term safety, particularly cardiovascular and oncological risk with sustained IGF-1 elevation, remains unknown. The 2009 proteomic analysis showed systemic metabolic changes that included acute-phase protein alterations, suggesting immune and inflammatory pathway engagement that could be relevant over months or years of use.[3]
Combination protocols. The CJC-1295/ipamorelin combination, the most commonly used protocol, has zero published clinical data. Its use is based on mechanistic reasoning, not empirical evidence. The dual-receptor stimulation concept is pharmacologically sound, as earlier work on GH-releasing substances documented the synergistic interaction between GHRH and GHS pathways.[10] But pharmacological plausibility is not clinical evidence. The doses, timing, duration, and risk profile of the combination in humans are entirely undocumented.
Dose equivalence with clinical trials. The doses used in the Teichman trial (30-125 mcg/kg) are sometimes cited to justify off-label dosing, but compounded CJC-1295 products are not manufactured to pharmaceutical standards. The actual dose delivered from a gray-market vial may differ from the labeled amount. The clinical trial used GMP-grade material with verified potency. Extrapolating trial results to unverified compounded products introduces a variable that the original researchers did not face.
The disconnect between CJC-1295's internet popularity and its evidence base is substantial. The clinical data shows it does what it claims pharmacologically: it elevates GH and IGF-1 for days from a single injection. Whether that elevation produces the outcomes people are using it for has never been tested in a controlled trial. The gap is not unique to CJC-1295; it is characteristic of the entire growth hormone secretagogue class, where pharmacokinetic validation has outpaced clinical outcome research.
The Bottom Line
CJC-1295 produces sustained, dose-dependent GH and IGF-1 elevation in humans, with effects lasting 6 to 11 days from a single injection. The clinical program was discontinued in 2006 after a patient death in a Phase II trial. Three published human studies provide pharmacokinetic and pharmacodynamic data, but no efficacy study for any clinical outcome has been completed. The peptide's use in anti-aging and body composition contexts is based on pharmacological plausibility, not clinical evidence.
Sources & References
- 1RPEP-01190·Teichman, Sam L et al. (2006). “CJC-1295 in Humans: Single Injection Sustains GH and IGF-1 Elevation for Up to 2 Weeks.” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 2RPEP-01148·Ionescu, Madalina et al. (2006). “CJC-1295 Clinical Trial: Single-Dose Injection Maintains Pulsatile GH for Days in Humans.” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 3RPEP-01542·Sackmann-Sala, Lucila et al. (2009). “CJC-1295 Changes Blood Protein Profiles: GH/IGF-1 Axis Activation Has Systemic Effects.” Growth hormone & IGF research : official journal of the Growth Hormone Research Society and the International IGF Research Society.Study breakdown →PubMed →↩
- 4RPEP-01113·Alba, Maria et al. (2006). “CJC-1295: A Long-Acting GHRH Analog That Normalizes GH With Once-Daily Dosing.” American journal of physiology. Endocrinology and metabolism.Study breakdown →PubMed →↩
- 5RPEP-02197·Jain, Shobhit et al. (2013). “How HIV and Its Treatment Disrupt the Growth Hormone Axis — And Why Tesamorelin Was Approved.” Reviews in endocrine & metabolic disorders.Study breakdown →PubMed →↩
- 6RPEP-02243·Nair, Deepti et al. (2013). “Growth hormone releasing hormone (GHRH) signaling modulates intermittent hypoxia-induced oxidative stress and cognitive deficits in mouse..” Journal of neurochemistry.Study breakdown →PubMed →↩
- 7RPEP-00485·Raun, K et al. (1998). “Ipamorelin: The First Growth Hormone Peptide That Only Boosts GH Without Other Hormones.” European journal of endocrinology.Study breakdown →PubMed →↩
- 8RPEP-00401·Cheng, J et al. (1997). “GHRP-2 and GHRP-6 Work Synergistically with GHRH But Not with Each Other.” Life sciences.Study breakdown →PubMed →↩
- 9RPEP-01626·Henninge, John et al. (2010). “CJC-1295 Identified in an Unknown Pharmaceutical Preparation: Forensic Analysis.” Drug testing and analysis.Study breakdown →PubMed →↩
- 10RPEP-00563·Smith, R G et al. (1999). “The Complete Landscape of Growth Hormone Releasing Compounds and Their Receptors.” Hormone research.Study breakdown →PubMed →↩