How Growth Hormone Peptides Affect Sleep Quality
GH Peptides and Sleep
~50% more deep sleep
In a double-blind, placebo-controlled trial, high-dose MK-677 increased stage IV (deep) sleep duration by approximately 50% and REM sleep by over 20% in healthy young subjects.
Copinschi et al., Neuroendocrinology, 1997
Copinschi et al., Neuroendocrinology, 1997
If you only read one thing
Some GH peptides genuinely help you sleep deeper — but not for the reason most people think. MK-677 increased deep sleep by about 50% in a controlled trial. But here's the twist: blocking GH release by 93% didn't hurt sleep at all. The sleep benefit comes from the receptor in your brain, not from the growth hormone itself. So 'boosting GH' and 'sleeping better' are two separate effects that happen to come from the same pill.
Growth hormone and sleep are bidirectionally linked: the largest GH pulse of the day occurs during the first cycle of slow-wave sleep (SWS), and disrupting SWS reduces GH secretion. This relationship has led to widespread interest in whether GH-releasing peptides can improve sleep quality. The evidence is nuanced. MK-677, a non-peptide oral GH secretagogue, increased deep sleep by approximately 50% and REM sleep by over 20% in a double-blind, placebo-controlled trial.[1] Ghrelin, the endogenous ligand for the same receptor, also promotes slow-wave sleep when administered exogenously.[2] But the relationship is not what it first appears. Blocking endogenous GHRH receptors suppresses nocturnal GH by 93% without affecting slow-wave sleep at all, proving that GH secretion and SWS can be fully dissociated.[3] This article examines the evidence for every major peptide class that has been studied in relation to sleep, separating compounds that genuinely improve sleep architecture from those that simply coincide with sleep-associated GH release. For a deeper exploration of the underlying biology, see our article on the connection between deep sleep and growth hormone release.
Key Takeaways
- Shutting down 93% of nightly growth hormone didn't change deep sleep at all — the two just coincide.
- In one controlled trial, MK-677 increased deep sleep by about 50% and REM by over 20%.
- A lower dose raised growth hormone but did nothing for sleep — the two effects have different thresholds.
- The sleep benefit comes from a receptor in your brain, not from the growth hormone your body releases afterward.
- Ghrelin — the "hunger hormone" — also deepens slow-wave sleep when given at bedtime.
- How you deliver the peptide matters: the same dose in pulses works, while a steady drip does nothing.
- Most classic GHRPs like GHRP-6 and GHRP-2 haven't actually been shown to improve sleep in humans.
The GH-Sleep Connection: What Is Actually Linked?
The temporal association between slow-wave sleep and GH release has been observed for decades. Approximately 70% of daily GH secretion occurs during sleep, primarily in the first SWS cycle. This led to a natural hypothesis: the same signals that drive SWS also drive GH release, and therefore GH-releasing peptides should promote deeper sleep.
The 2004 study by Jessup et al. tested this hypothesis directly. Healthy men (20-33 years) received a 12-hour infusion of a GHRH receptor antagonist or saline overnight. The antagonist suppressed the GH response to a GHRH bolus by 93%, confirming effective receptor blockade. Despite this near-total GH suppression, polysomnography showed no difference in SWS percentage or any other sleep parameter between antagonist and placebo nights.[3]
This result is foundational: endogenous GHRH is indispensable for nocturnal GH secretion but does not generate slow-wave sleep. The GH pulse and SWS coincide in time because they share upstream regulators (circadian signals, sleep-wake transitions), but they are not causally linked through GHRH. This distinction matters because it means exogenous GHRH or GH secretagogues that improve sleep are doing so through a mechanism other than simply boosting GH.
MK-677: The Strongest Sleep Evidence in the GH Secretagogue Class
MK-677 (ibutamoren) is a non-peptide, orally active GH secretagogue that mimics ghrelin at the GHSR-1a receptor. The 1997 study by Copinschi et al. remains the most rigorous evidence for GH secretagogue-mediated sleep improvement.
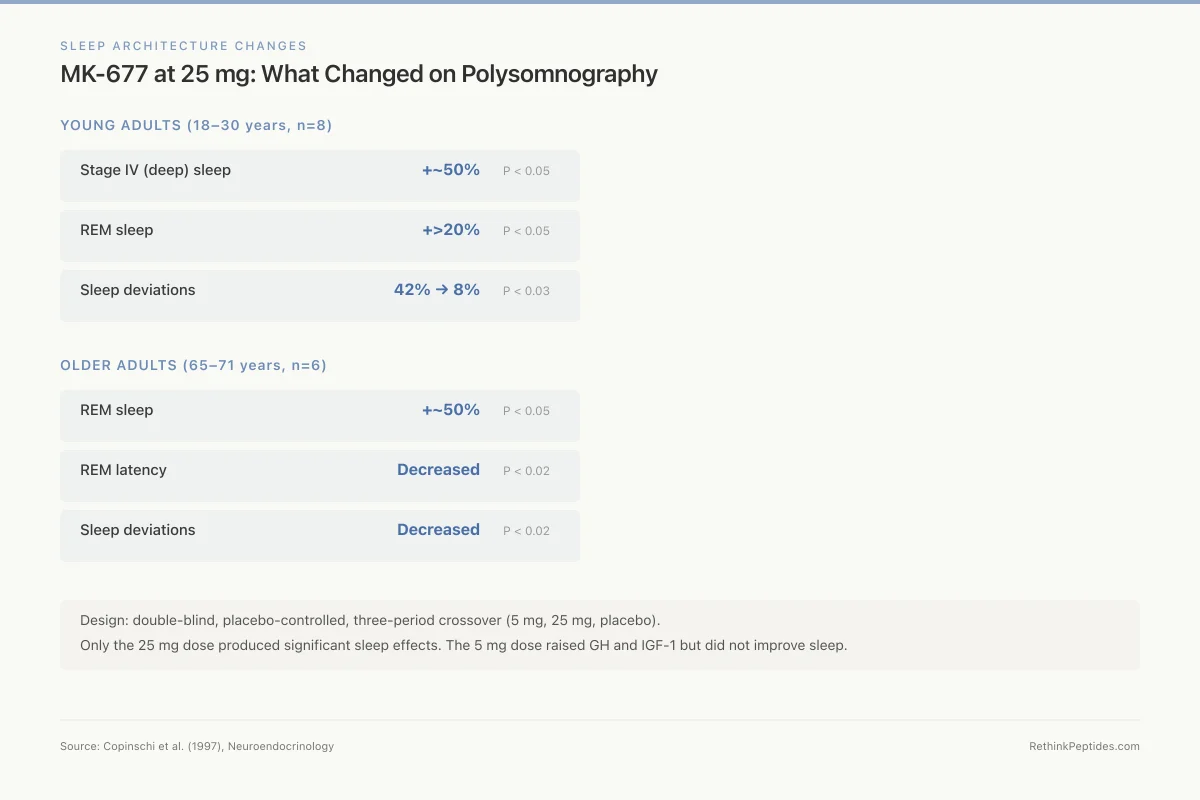
In the young adult cohort (8 subjects, 18-30 years), high-dose MK-677 (25 mg) produced three significant sleep changes compared to placebo: stage IV (deep) sleep duration increased by approximately 50%, REM sleep increased by more than 20%, and the frequency of deviations from normal sleep architecture dropped from 42% under placebo to 8% under MK-677 (P < 0.03).[1]
In the elderly cohort (6 subjects, 65-71 years), MK-677 at 25 mg produced a nearly 50% increase in REM sleep (P < 0.05), decreased REM latency (P < 0.02), and reduced sleep deviations (P < 0.02). The lower 2 mg dose did not produce these effects, establishing dose-dependency.
The mechanism likely involves GHSR-1a activation in brain regions that regulate sleep architecture. The ghrelin receptor is expressed in the hypothalamus, arcuate nucleus, ventromedial hypothalamus, and other areas involved in sleep-wake regulation. MK-677's sleep benefits may be partially independent of GH release, given the Jessup et al. finding that GHRH-mediated GH release does not drive SWS. The receptor's involvement in orexigenic and circadian signaling pathways may be more relevant to its sleep effects.
The study design is worth noting for its rigor. Young subjects followed a double-blind, placebo-controlled, three-period crossover design with Latin square ordering, receiving 5 mg, 25 mg, and placebo in random order during 7-day treatment periods separated by at least 14 days. Only the 25 mg dose produced significant sleep effects, establishing both dose-dependency and a therapeutic threshold. The lower 5 mg dose, while sufficient to raise GH and IGF-1 levels, did not improve sleep architecture, suggesting that the sleep-promoting effect requires higher receptor occupancy than the GH-releasing effect.
The practical implication is that MK-677 at bedtime may serve a dual purpose: augmenting the sleep-associated GH pulse (which peaks during the first SWS cycle) while simultaneously deepening the sleep architecture that facilitates that pulse. Whether this creates a true positive feedback loop or simply a pharmacological coincidence of two independent GHSR-mediated effects remains an open question.
Sleep Architecture Changes
MK-677 at 25 mg: What Changed on Polysomnography
Young adults (18–30 years, n=8)
Older adults (65–71 years, n=6)
Design: double-blind, placebo-controlled, three-period crossover (5 mg, 25 mg, placebo). Only the 25 mg dose produced significant sleep effects. The 5 mg dose raised GH and IGF-1 but did not improve sleep.
Source: Copinschi et al. (1997), Neuroendocrinology
 View as image
View as imageFor a comparison of GH secretagogues versus conventional sleep medications, see our article on GH secretagogues vs sleeping pills.
Ghrelin: The Endogenous Sleep-Promoting Peptide
Weikel et al. (2003) administered ghrelin (4 x 50 mcg IV boluses at hourly intervals from 2200 to 0100) to seven healthy men and measured polysomnographic sleep alongside plasma GH, ACTH, cortisol, prolactin, and leptin throughout the night.
Ghrelin increased slow-wave sleep during the total night and enhanced accumulated delta-wave activity (a quantitative measure of deep sleep intensity) during the second half of the night. REM sleep decreased during the second third of the night, while all other sleep EEG variables remained unchanged. GH and prolactin plasma levels were enhanced throughout the night, and cortisol levels increased during the first part of the night.[2]
The GH response to ghrelin showed tachyphylaxis: the response was strongest after the first injection and weakest after the fourth, suggesting rapid receptor desensitization. In contrast, the cortisol response showed the inverse pattern, increasing with successive injections. This divergent hormonal pattern indicates that ghrelin's effects on different neuroendocrine axes are mediated through partially independent pathways.
The authors concluded that ghrelin is an endogenous sleep-promoting factor with a role complementary to its regulation of energy balance. This positions ghrelin as an integrative hormone linking three fundamental processes: appetite (eat to acquire energy), sleep (rest to consolidate and repair), and growth hormone release (build and maintain tissue). The teleological logic is compelling: an organism that has eaten recently (high ghrelin from fasting would promote both food-seeking and subsequent sleep for digestion and repair) benefits from coordinating these processes.
The cortisol response to ghrelin is clinically relevant and often overlooked. While GH showed tachyphylaxis (weakening response with repeated injections), cortisol showed the opposite pattern: it increased with successive ghrelin doses. For individuals using ghrelin-mimicking compounds at bedtime to improve sleep, this rising cortisol trajectory could partially counteract the sleep benefit, since cortisol is an arousal-promoting hormone. This may explain why chronic use of GH secretagogues at night sometimes produces less sleep improvement than acute dosing studies predict.
For more on ghrelin's appetite-related functions, see our article on ghrelin: the hunger hormone, and for the connection between ghrelin receptor activation and hunger with GHRPs, see our article on GHRP-6 and hunger.
GHRH: Pulsatile Delivery Matters
While GHRH does not generate slow-wave sleep endogenously (Jessup et al.), exogenous GHRH administration can promote SWS under specific conditions. The key variable is the pattern of delivery.
Marshall et al. (1996) compared episodic and continuous GHRH administration in healthy volunteers. The total dose was identical (200 mcg): one group received it as four hourly 50 mcg IV boluses (at 2200, 2300, 0000, and 0100), while the other received a continuous infusion (57 mcg/h from 2130 to 0100).
Episodic GHRH significantly increased stage 4 SWS and REM sleep compared to placebo (P < 0.01 and P < 0.05 respectively) and diminished wakefulness and stage 1 sleep. Continuous infusion had no significant effects on any sleep parameter compared to placebo. Both conditions enhanced plasma GH, though the episodic administration produced slightly greater GH elevation.[4]
This finding has profound implications for how GH-related peptides interact with sleep. The body releases GHRH in pulses, not continuously, and the sleep-promoting effect appears to require this pulsatile pattern. The difference between episodic and continuous delivery is not simply a matter of dose timing; it reflects a fundamental feature of neuroendocrine signaling. Pulsatile GHRH allows receptor resensitization between pulses, maintaining responsiveness, while continuous exposure leads to receptor downregulation, analogous to how continuous GnRH agonist administration paradoxically suppresses gonadotropins. Continuous receptor stimulation, as occurs with long-acting GH secretagogues, may desensitize the sleep-regulating pathways more rapidly than the GH-releasing pathways. This is why GHRPs given as discrete doses (such as subcutaneous injections at bedtime) may be more sleep-relevant than continuous-infusion approaches.
GHRPs like hexarelin, GHRP-2, and GHRP-6 act through the ghrelin receptor rather than the GHRH receptor and thus work through a different pathway. A comprehensive 1996 review by Argente et al. noted that GHRPs' mechanism involves acting at both pituitary and hypothalamic levels through specific receptors distinct from GHRH receptors, with oral activity demonstrated in both animals and humans.[5] However, direct evidence for GHRPs (as opposed to MK-677 or ghrelin) improving sleep in humans is surprisingly limited. Single GHRP-6 injections around bedtime enhanced stage 2 sleep without affecting SWS or REM in one study, and single GHRP-2 injections in the latter part of the night had no sleep effect in another.
Beyond GH Secretagogues: Other Sleep-Active Peptides
Delta Sleep-Inducing Peptide (DSIP)
DSIP is a nonapeptide (Trp-Ala-Gly-Gly-Asp-Ala-Ser-Gly-Glu) isolated in 1977 from the blood of sleeping rabbits. It was shown to induce delta sleep (SWS) in rabbits, rats, mice, and humans, with a U-shaped dose-response curve. DSIP-like immunoreactivity was found in brain tissue and peripheral organs, and the peptide demonstrated effects on electrophysiology, neurotransmitter levels, circadian patterns, and hormone secretion beyond its sleep-inducing properties.[6]
However, clinical trials of DSIP for insomnia produced disappointing results. Monti et al. (1987) administered DSIP at 25 nmol/kg IV to chronic insomniacs in a double-blind crossover design over four nights. While total sleep time and NREM sleep time increased (related to stage 2 increases), slow-wave sleep and REM sleep were not modified. The authors concluded that sleep improvement under DSIP treatment was "of little clinical significance."[7]
The disconnect between DSIP's promising animal data and its weak clinical performance is a recurring theme in peptide sleep research. Several factors may explain this gap. DSIP has a U-shaped dose-response curve, meaning both too little and too much reduce its efficacy, making dosing difficult. Its half-life in human circulation is short, and the optimal timing of administration relative to sleep onset was never systematically established. DSIP also shows different effects across species: in cats, the primary effect was on REM sleep rather than delta sleep, while in rabbits and rats, delta sleep predominated.
The peptide's multiple biological activities beyond sleep induction, including effects on drug withdrawal symptoms, pain perception, circadian rhythms, and stress responses, suggest that "delta sleep-inducing peptide" is something of a misnomer for what is actually a broadly neuromodulatory compound. More recent research has focused on DSIP's potential in drug withdrawal and chronic pain rather than insomnia, applications where its broader neuromodulatory profile may be more therapeutically relevant than its modest sleep effects.
Neuropeptide Y: Sleep Through Stress Reduction
Neuropeptide Y (NPY) promotes sleep through an entirely different mechanism than GH secretagogues. Antonijevic et al. (2000) administered NPY intravenously (4 x 50 or 100 mcg between 2200 and 0100) to healthy young men and measured polysomnographic sleep and nocturnal hormones.
NPY enhanced sleep period time and stage 2 sleep, reduced sleep latency and time awake, and modulated REM sleep. Simultaneously, it reduced ACTH during the first half of the night and cortisol during the second half.[8]
The mechanism is functionally opposite to stress-related insomnia. Corticotropin-releasing hormone (CRH) promotes wakefulness, anxiety, and HPA axis activation. NPY opposes CRH at multiple levels. In depression and chronic stress, where CRH overactivity drives both insomnia and HPA dysregulation, NPY agonism could theoretically address both problems simultaneously. This makes NPY mechanistically distinct from GH secretagogues: it promotes sleep by reducing arousal rather than by stimulating sleep-promoting pathways directly.
Food-Derived Sleep Peptides
Emerging research has identified sleep-modulating peptides derived from dietary proteins. Bovine milk casein-derived sleep-enhancing peptides (CSEPs) act on the central nervous system through structure-activity compatibility mechanisms and also regulate gut microbiota composition through the gut-brain axis, indirectly mediating sleep effects.[9]
Thomas et al. (2024) tested glycine-rich collagen peptides (15 g daily before bedtime) in 13 athletic males with sleep complaints in a randomized crossover trial. Polysomnography showed significantly fewer awakenings with collagen peptides (21.3 vs 29.3, P = 0.028), and morning cognitive accuracy improved. Total sleep time, latency, and efficiency were unaffected.[10] The sleep-continuity improvement without changes in sleep architecture suggests a different mechanism from the GH secretagogue effects, likely involving glycine's role as an inhibitory neurotransmitter and its effects on core body temperature regulation.
What the Evidence Supports and What It Does Not
| Compound | Sleep Evidence | Mechanism | Strength of Evidence |
|---|---|---|---|
| MK-677 | Increased stage IV (+50%) and REM (+20%) | GHSR-1a agonism | Double-blind RCT, small sample |
| Ghrelin | Increased total-night SWS and delta activity | GHSR-1a agonism | Controlled, 7 subjects |
| GHRH (episodic) | Increased stage 4 SWS and REM sleep | GHRH receptor | Controlled, dose-pattern dependent |
| GHRH (continuous) | No significant sleep effects | GHRH receptor | Same study, same dose |
| GHRPs (GHRP-2, GHRP-6) | Minimal or no SWS/REM effects | GHSR-1a agonism | Limited data, no consistent benefit |
| NPY | Reduced latency, increased stage 2, reduced cortisol | CRH opposition | Controlled, hormonal + sleep measures |
| DSIP | Stage 2 increase only, no SWS/REM benefit | Unclear/neuromodulatory | Double-blind RCT, disappointing |
| Collagen peptides | Reduced awakenings, cognitive improvement | Glycine/thermoregulation | Crossover RCT, 13 subjects |
Evidence Matrix
Which Peptides Actually Improve Sleep? A Side-by-Side Comparison
| Compound | Deep Sleep | REM | Mechanism |
|---|---|---|---|
MK-677 (25 mg) Double-blind RCT, 14 subjects | Strong | Strong | GHSR-1a agonism |
Ghrelin (IV) Controlled study, 7 subjects | Moderate | — | GHSR-1a agonism |
GHRH (pulsatile) Controlled, dose-pattern dependent | Moderate | Moderate | GHRH receptor |
GHRH (continuous) Same study, same dose — no effect | — | — | GHRH receptor |
GHRP-6 (single dose) Stage 2 only, limited data | — | — | GHSR-1a agonism |
NPY (IV) Controlled, reduces cortisol | — | Weak | CRH opposition |
DSIP Double-blind RCT, disappointing | — | — | Neuromodulatory |
Collagen peptides Crossover RCT, fewer awakenings | — | — | Glycine / thermoregulation |
Source: Copinschi (1997), Weikel (2003), Marshall (1996), Jessup (2004), Monti (1987), Antonijevic (2000), Thomas (2024)
Aging, GH Decline, and Sleep Deterioration
Both GH secretion and sleep quality decline with age in parallel trajectories. By age 50, slow-wave sleep duration has typically decreased by 50-80% compared to young adulthood, and by the same age, daily GH secretion has declined by a similar proportion. This parallel decline has fueled the hypothesis that restoring GH secretion could restore sleep quality in older adults.
The MK-677 study partially supports this hypothesis: elderly subjects (65-71 years) showed a nearly 50% increase in REM sleep with MK-677 treatment. However, the Jessup et al. data complicates the picture by showing that the association between GH and SWS is not causal through GHRH. The age-related decline in both may reflect shared upstream deterioration (hypothalamic aging, reduced neurotransmitter signaling) rather than GH loss directly causing sleep deterioration.
This distinction matters for treatment strategy. If poor sleep in aging is driven by hypothalamic neuron loss or altered circadian signaling, then GH secretagogues may improve sleep by activating GHSR-1a in brain sleep-regulating areas rather than by restoring GH levels per se. The sleep improvement would be a direct neurological effect of receptor activation, with the GH increase being a parallel but independent benefit. This also means that monitoring IGF-1 levels alone (a common practice with GH secretagogue use) would not predict sleep quality improvement, since the two effects are mediated through different downstream pathways from the same receptor.
The evidence supports a clear hierarchy: MK-677 and ghrelin have the strongest evidence for improving deep sleep architecture through GHSR-1a activation. GHRH can promote SWS but only when delivered in pulsatile boluses that mimic physiological release patterns. Direct GHRPs (hexarelin, GHRP-2, GHRP-6) given as single injections have not consistently shown sleep benefits in the limited studies conducted. NPY offers a mechanistically distinct approach relevant to stress-related insomnia. DSIP's name promises more than its clinical data deliver. Food-derived peptides reduce sleep fragmentation through mechanisms unrelated to GH.
The absence of strong evidence for direct GHRPs (hexarelin, GHRP-2, GHRP-6) improving sleep despite their activation of the same receptor as ghrelin and MK-677 is puzzling. Several explanations are possible. First, the GHRP sleep studies used single injections rather than the sustained receptor activation achieved by MK-677's oral dosing. Second, the peptide GHRPs may have different binding kinetics or receptor engagement patterns at hypothalamic sleep-regulating neurons compared to the non-peptide MK-677. Third, the limited number of GHRP-sleep studies (only two published, both small) may simply be insufficient to detect a real effect. Until adequately powered, multi-dose GHRP-sleep studies are conducted, the question remains open.
What is clear is that the relationship between GH peptides and sleep is receptor-mediated rather than hormone-mediated. The peptides that most consistently improve sleep (MK-677, ghrelin) act through GHSR-1a in the brain. The GH they release is a parallel effect, not the mechanism of sleep improvement. This understanding should guide both clinical investigation and practical application: the goal is receptor activation in sleep-regulating neural circuits, not maximum GH secretion.
The Bottom Line
The relationship between growth hormone peptides and sleep is more complex than the simple "more GH = better sleep" narrative suggests. MK-677 and ghrelin genuinely improve sleep architecture through GHSR-1a activation, but this effect is likely mediated by the receptor's broader role in sleep-wake regulation rather than through GH release itself. The definitive Jessup et al. study showing that blocking GHRH suppresses GH by 93% without affecting slow-wave sleep at all should temper expectations that GH-boosting peptides will automatically improve sleep. The strongest sleep-promoting peptides may ultimately be those that target arousal regulation (NPY) or sleep continuity (glycine-rich peptides) rather than the somatotropic axis.
Sources & References
- 1RPEP-00402·Copinschi, G et al. (1997). “Oral MK-677 Significantly Improves Sleep Quality in Both Young and Older Adults.” Neuroendocrinology.Study breakdown →PubMed →↩
- 2RPEP-00873·Weikel, J C et al. (2003). “Ghrelin — The Hunger Hormone — Promotes Deep Sleep in Humans.” American journal of physiology. Endocrinology and metabolism.Study breakdown →PubMed →↩
- 3RPEP-00930·Jessup, Stacy K et al. (2004). “Blockade of endogenous growth hormone-releasing hormone receptors dissociates nocturnal growth hormone secretion and slow-wave sleep..” European journal of endocrinology.Study breakdown →PubMed →↩
- 4RPEP-00369·Marshall, L et al. (1996). “Greater efficacy of episodic than continuous growth hormone-releasing hormone (GHRH) administration in promoting slow-wave sleep (SWS)..” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 5RPEP-00353·Argente, J et al. (1996). “Growth Hormone-Releasing Peptides: A Comprehensive Review of How They Work.” Hormone research.Study breakdown →PubMed →↩
- 6RPEP-00021·Graf, M V et al. (1984). “Delta-Sleep-Inducing Peptide: The First Natural Sleep Molecule Discovered.” Neuroscience and biobehavioral reviews.Study breakdown →PubMed →↩
- 7RPEP-00055·Monti, J M et al. (1987). “Delta Sleep-Inducing Peptide Doesn't Meaningfully Improve Sleep in Insomniacs.” International journal of clinical pharmacology research.Study breakdown →PubMed →↩
- 8RPEP-00574·Antonijevic, I A et al. (2000). “Neuropeptide Y Promotes Sleep and Reduces Stress Hormones in Young Men.” Neuropharmacology.Study breakdown →PubMed →↩
- 9RPEP-12259·Liu, Yibo et al. (2025). “Bovine Milk Casein-Derived Sleep-Enhancing Peptides: Bioavailability, Functional Mechanisms, and Development Strategies..” Journal of agricultural and food chemistry.Study breakdown →PubMed →↩
- 10RPEP-09380·Thomas, Craig et al. (2024). “Collagen Peptides Before Bed Reduce Sleep Disruptions and Improve Next-Day Cognitive Function.” European journal of nutrition.Study breakdown →PubMed →↩