Gray-Market Peptides: The 'Research Use Only' Fiction
Gray-Market Peptide Regulation
30% Incorrect Sequences
Laboratory analysis of gray-market polypeptide products found that 30% contained incorrect amino acid sequences, with two-thirds of tested samples falling below 95% purity thresholds.
Janvier et al., Talanta, 2018
Janvier et al., Talanta, 2018
If you only read one thing
Those peptides labeled 'for research use only' are not research-grade products with a legal loophole — they're unregulated drugs with no quality control. Lab tests found that 30% had the wrong ingredient entirely, and some contained arsenic at 10 times the safety limit. People have ended up in comas from counterfeit products. The 'research only' label doesn't make them safe, legal for personal use, or tested in any way. If you're injecting something you bought online with that label, you genuinely don't know what's in the vial.
Gray-market peptides are sold worldwide under a four-word disclaimer: "for research use only." In 2025, a patient in Italy fell into a hypoglycemic coma after injecting what was sold as semaglutide for weight loss. The product was counterfeit.[1] In Belgium, laboratory analysis of the most commonly seized gray-market polypeptide products found that 30% contained incorrect amino acid sequences and two-thirds failed basic purity standards.[2] These are not outliers. They are the predictable consequences of a market built on a legal fiction.
Every year, thousands of peptide products ship under that label. The buyers are not researchers. The sellers know this. The FDA knows this. And an expanding body of analytical and clinical evidence documents what happens when unregulated peptide manufacturing meets human biology. This article examines the regulatory framework, the quality data, and the documented harms that define the gray-market peptide landscape. For a deeper look at vendor-specific liability, see Online Peptide Vendors: Legal Liability and Consumer Risk. For the legal line between research-grade and human-use products, see Research-Grade Peptides and Human Use: Where the Law Draws the Line.
Key Takeaways
- "For research use only" isn't a safety standard — it's a disclaimer that keeps the seller out of court.
- Lab tests of seized gray-market peptides found 30% contained the wrong ingredient entirely.
- Two-thirds of tested vials failed basic purity standards, and some contained arsenic at ten times the safety limit.
- A patient in Italy fell into a coma after injecting "semaglutide" that actually contained insulin.
- The FDA sent over 50 warning letters to peptide sellers in a single month of 2025.
- Most gray-market peptides are made in Chinese labs, relabeled through middlemen, and shipped with zero temperature control.
- Peptides that cost under $1 to make sell for $80, with nobody checking what's in the vial.
What "Research Use Only" Means Under Federal Law
The phrase "for research use only" (RUO) appears on virtually every peptide sold through gray-market channels. Its legal meaning is narrow: the product is intended for in vitro laboratory use and is not manufactured, tested, or approved for administration to humans or animals. Under the Federal Food, Drug, and Cosmetic Act (FD&C Act), any substance intended to diagnose, treat, cure, or prevent a disease in humans is classified as a drug. The moment a peptide is marketed, sold, or packaged in a way that implies human use, the RUO label becomes legally irrelevant.
The FDA has stated this position explicitly. In a December 2024 warning letter to Summit Research Peptides, the agency wrote that despite "research use only" labeling, evidence from the company's website established that the products "were intended to be drugs" for human use. The evidence included dosing instructions, inclusion of bacteriostatic water and syringes, and customer testimonials describing personal administration.
This is not a gray area. Under the FD&C Act, products intended for human use that have not completed a New Drug Application (NDA) or Biologics License Application (BLA) are unapproved new drugs. Their sale, distribution, and administration violate federal law regardless of what the label says.[3]
The legal framework extends internationally. The World Health Organization first flagged falsified medical products in 1985. By 2018, researchers estimated annual deaths from falsified medical products between 200,000 and one million globally.[4] Polypeptide drugs represent a growing segment of this problem because peptide synthesis technology has become inexpensive and widely accessible, while analytical verification remains complex and costly.
How the Gray Market Works
Gray-market peptide vendors operate through a consistent model. Manufacturing occurs predominantly in China, where contract synthesis laboratories produce peptides at scale without FDA oversight, Good Manufacturing Practice (GMP) compliance, or endotoxin testing. Products are shipped internationally with RUO labeling, often through intermediary countries to obscure origin.
Vendor websites employ a specific pattern. Product pages describe mechanisms of action, dosing protocols, and physiological effects while maintaining a legal disclaimer that the products are "not for human consumption." Some vendors sell peptides alongside bacteriostatic water, syringes, and alcohol swabs. Customer forums linked from vendor sites discuss personal administration, injection techniques, and subjective effects.
The FDA has identified this packaging and marketing pattern as sufficient evidence of intended human use. When a vendor sells a substance alongside the tools required for self-injection and provides information about physiological effects in humans, the RUO disclaimer does not provide legal protection.[3]
The economic incentive is substantial. Peptide synthesis costs have dropped dramatically over the past decade. A milligram of BPC-157 that costs under $1 to synthesize without quality controls can sell for $30 to $80 at retail, with no regulatory compliance costs, no clinical testing requirements, and no liability infrastructure. The global peptide therapeutics market was valued at over $50 billion in 2025, and the unregulated gray-market segment operates alongside it with no verifiable size estimate.
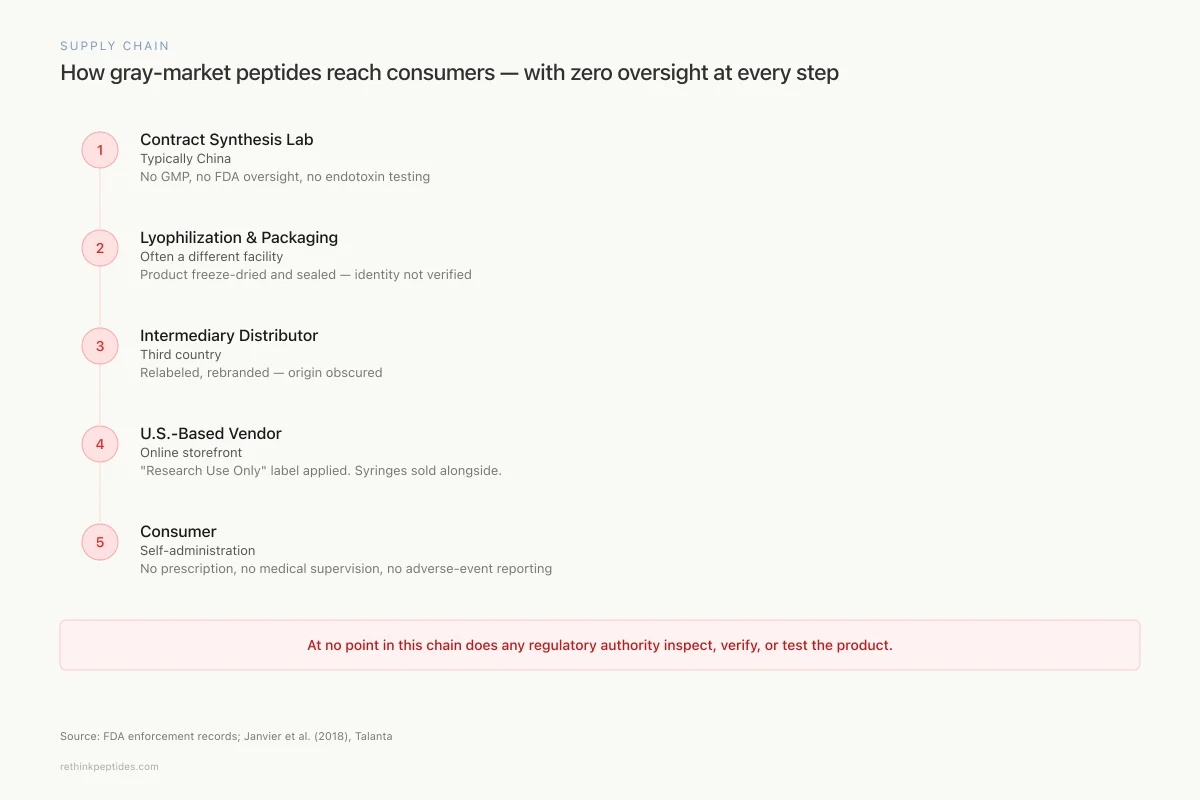
The supply chain problem
The lack of supply chain transparency compounds quality concerns. Gray-market peptides typically pass through multiple intermediaries between synthesis and delivery. A peptide synthesized in a Chinese contract laboratory may be lyophilized and packaged by a different facility, relabeled by a distributor in a third country, and shipped to a U.S.-based vendor who applies the final branding and RUO disclaimer. At no point in this chain does a regulatory authority inspect the product, verify its identity, or test for contaminants.
Supply Chain
How gray-market peptides reach consumers — with zero oversight at every step
Contract Synthesis Lab
Typically China
No GMP, no FDA oversight, no endotoxin testing
Lyophilization & Packaging
Often a different facility
Product freeze-dried and sealed — identity not verified
Intermediary Distributor
Third country
Relabeled, rebranded — origin obscured
U.S.-Based Vendor
Online storefront
"Research Use Only" label applied. Syringes and bacteriostatic water sold alongside.
Consumer
Self-administration
No prescription, no medical supervision, no adverse-event reporting
At no point in this chain does any regulatory authority inspect, verify, or test the product.
Source: FDA enforcement records; Janvier et al. (2018)
 View as image
View as imageTemperature control presents an additional risk. Peptides are sensitive to heat, light, and moisture. Pharmaceutical-grade peptide products are shipped under controlled cold-chain conditions with validated packaging. Gray-market products are shipped through standard postal and courier services with no temperature monitoring. Degradation during transit can alter the peptide's structure, reduce its potency, or generate breakdown products with unknown biological activity. The buyer has no way to assess whether the product arrived intact because the CoA, if one was provided, describes the product at the time of testing, not at the time of delivery.
What Independent Testing Reveals
The gap between what gray-market labels claim and what analytical chemistry finds is the central safety problem. Three decades of laboratory evidence document consistent quality failures in peptides manufactured outside regulated pharmaceutical frameworks.
Contamination identified early
In 1991, Castellino and colleagues reported that certain batches of synthetic peptides produced through azide coupling contained mutagenic contaminants detectable in the Ames test. The contaminant was identified as HOBt-azide, an explosive and mutagenic byproduct of the synthesis method. The researchers concluded that azide coupling methods introduce mutagenic risks that are not detectable through standard purity assays.[5]
This 1991 finding established a principle that remains central to the gray-market problem: purity testing and safety testing are not the same thing. A peptide can register as 99% pure by HPLC-UV while containing mutagenic, pyrogenic, or otherwise hazardous contaminants in the remaining 1%.
Falsified polypeptide drugs
Janvier and colleagues conducted the most systematic analysis of gray-market polypeptide quality to date, publishing two complementary studies in 2018. Their analysis of the most frequently seized falsified polypeptide drugs on the Belgian market revealed that 30% of samples contained incorrect amino acid sequences and two-thirds of samples fell below the 95% purity threshold considered minimum for research-grade applications.[2]
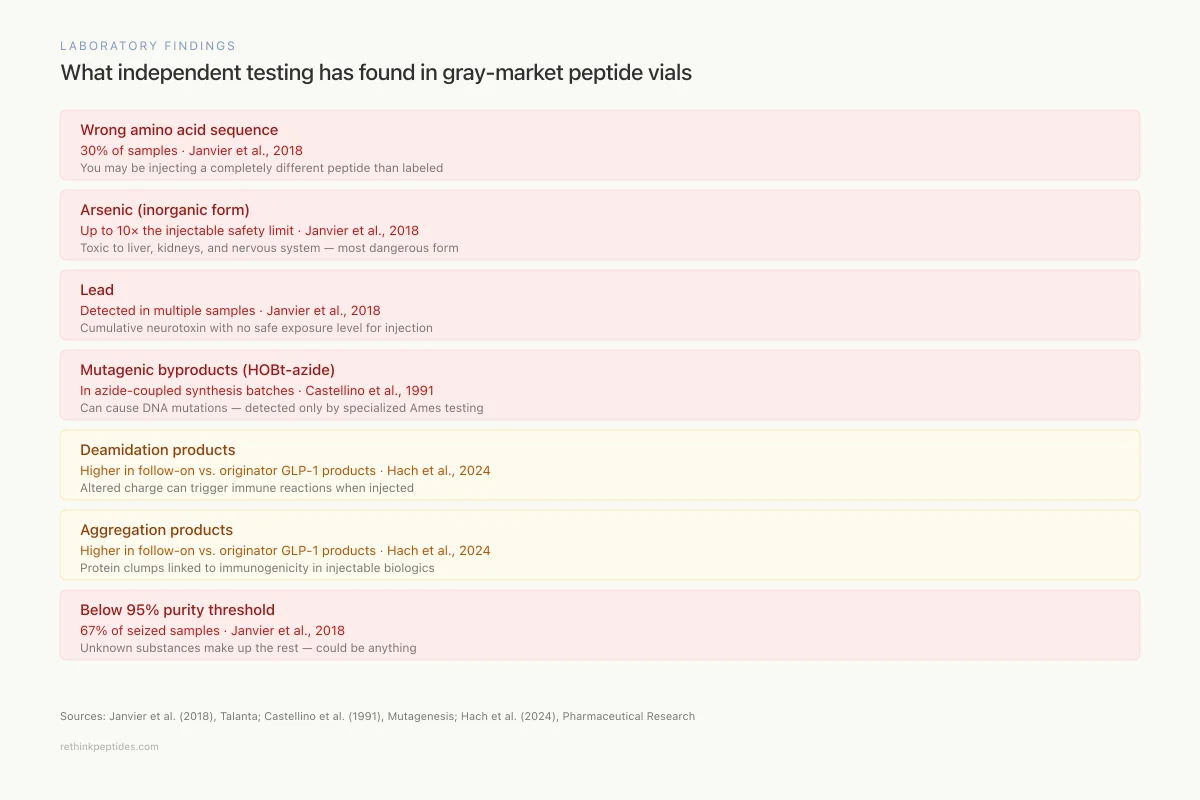
Laboratory Findings
What independent testing has found in gray-market peptide vials
Wrong amino acid sequence
Severe30% of samples
You may be injecting a completely different peptide than labeled
Janvier et al., 2018
Arsenic (inorganic form)
SevereUp to 10× the injectable safety limit
Toxic to liver, kidneys, and nervous system — the most dangerous form of arsenic
Janvier et al., 2018
Lead
SevereDetected in multiple samples
Cumulative neurotoxin with no safe exposure level for injection
Janvier et al., 2018
Mutagenic byproducts (HOBt-azide)
SevereIn azide-coupled synthesis batches
Can cause DNA mutations — detected only by specialized Ames testing
Castellino et al., 1991
Deamidation products
ModerateHigher in follow-on vs. originator GLP-1 products
Altered charge can trigger immune reactions when injected
Hach et al., 2024
Aggregation products
ModerateHigher in follow-on vs. originator GLP-1 products
Protein clumps linked to immunogenicity in injectable biologics
Hach et al., 2024
Below 95% purity
Severe67% of seized samples
Unknown substances make up the rest — could be anything
Janvier et al., 2018
Sources: Janvier et al. (2018); Castellino et al. (1991); Hach et al. (2024)
 View as image
View as imageIn their companion paper, Janvier and colleagues characterized the falsification of biotechnology drugs as a growing global threat. They documented cases where seized products contained entirely different peptides than labeled, incorrect concentrations, and undeclared excipients. The analytical methods required to detect these problems (LC-MS, peptide mapping, bioassays) are expensive and rarely performed by gray-market vendors or their customers.[4]
Follow-on GLP-1 products
In 2024, Hach and colleagues at Novo Nordisk published a direct comparison of originator versus follow-on and compounded GLP-1 polypeptide drug substances and products. The results documented measurable differences in purity, impurity profiles, and predicted stability. Follow-on semaglutide and liraglutide drug substances showed measurably higher levels of deamidation products, oxidation variants, and aggregates compared to originator products.[6]
The study identified specific impurity types with clinical relevance. Deamidation products alter peptide charge and can trigger immune responses. Aggregation products are associated with immunogenicity in injectable biologics. Oxidized variants may have reduced or altered biological activity. None of these impurities would be flagged by a basic HPLC-UV purity test of the type typically provided by gray-market vendors.
The purity-safety gap
Standard gray-market quality documentation consists of a Certificate of Analysis (CoA) showing HPLC-UV purity, typically claiming 98% or 99%. This metric is misleading for three reasons.
First, HPLC-UV confirms the presence of the target peptide but cannot identify what comprises the remaining percentage. The impurities could be truncated peptide sequences, racemized amino acids, residual solvents, heavy metals, or bacterial endotoxins. Second, HPLC-UV cannot detect endotoxin contamination. Bacterial endotoxins cause fever, inflammation, and in high doses, septic shock when injected. Pharmaceutical-grade injectables undergo specific endotoxin testing (Limulus Amebocyte Lysate assay) that gray-market products do not. Third, a CoA from an unaudited laboratory has no verification mechanism. There is no regulatory body inspecting these facilities, no consequence for falsified documentation, and no independent oversight.
Safety
CriticalA '99% pure' CoA doesn't mean safe to inject
Concern
Standard HPLC-UV purity tests — the kind gray-market vendors provide — cannot detect bacterial endotoxins, arsenic, lead, residual solvents, or mutagenic byproducts. A peptide can test as 99% pure while the remaining 1% contains substances that cause fever, septic shock, or long-term organ damage.
What the research says
Pharmaceutical-grade injectables undergo endotoxin testing (LAL assay), elemental impurity screening, residual solvent analysis, and sterility testing. These tests are expensive and are not performed by gray-market vendors.
Particularly relevant for: Anyone injecting peptides purchased from unregulated sources
What to do
A Certificate of Analysis showing high HPLC purity is not evidence of safety for injection. Only products from licensed pharmacies or FDA-approved manufacturers undergo the full testing required for injectable safety.
Janvier et al. (2018); Castellino et al. (1991); Hach et al. (2024)
Case Reports: When Counterfeit Peptides Cause Documented Harm
The clinical literature now includes case reports directly linking counterfeit peptide products to serious adverse events.
Counterfeit semaglutide: hypoglycemic coma
Antonacci and colleagues reported a 2025 case in which a patient purchased what was marketed as semaglutide (Ozempic) online for weight loss. After injection, the patient developed severe hypoglycemia resulting in coma. Genuine semaglutide, a GLP-1 receptor agonist, does not typically cause hypoglycemia in non-diabetic patients. The clinical presentation was consistent with insulin or an insulin analogue, suggesting the product contained an entirely different active substance than labeled.[1]
This case illustrates a worst-case scenario: the product did not merely fail a purity test or contain higher-than-expected impurity levels. It contained a completely different drug, one capable of causing life-threatening hypoglycemia in a person without diabetes.
Counterfeit semaglutide: euglycemic ketoacidosis
Sterckx and De Keyser reported a 2026 case of euglycemic ketoacidosis in a patient using counterfeit semaglutide for weight loss. The patient presented without hyperglycemia but with dangerous ketone accumulation, a metabolic emergency that can be fatal if unrecognized. The authors noted that the growing off-label use of GLP-1 agents for weight loss, particularly without medical supervision and through unregulated channels, creates conditions for adverse events that would not occur with authentic, properly supervised products.[7]
These cases are published precisely because the patients survived and sought medical care. The actual number of adverse events from counterfeit peptide products is unknown because there is no reporting infrastructure for gray-market products, no prescriber to file an adverse event report, and no systematic surveillance.
FDA Enforcement: Warning Letters, Seizures, and the Category System
The FDA's enforcement approach to gray-market peptides has escalated sharply since 2024. The agency's actions fall into three categories: warning letters, import alerts, and the peptide category system.
Warning letters
In late 2024, the FDA issued a series of warning letters to companies marketing peptides online with RUO labeling. The letters cited violations of the FD&C Act, specifically the sale of unapproved new drugs and misbranded products. The FDA stated that RUO disclaimers were "a ruse to avoid FDA scrutiny" when the products were clearly intended for human use.
In September 2025, the FDA issued more than 50 warning letters to companies compounding or manufacturing GLP-1 receptor agonists, specifically semaglutide and tirzepatide. This represented one of the largest coordinated enforcement actions in compounding pharmacy history and signaled that the agency was treating peptide regulation as a priority.[3]
Import alerts
The FDA expanded Import Alert 66-78 in 2025 to include 12 additional unapproved peptides. Import alerts authorize U.S. Customs and Border Protection to detain shipments without physical examination, effectively blocking specific products at the border. The FDA has also coordinated with the DEA in some seizure actions, not because peptides are controlled substances but because they qualify as unapproved drugs under the FD&C Act.
State-level enforcement
Federal enforcement is only part of the picture. State pharmacy boards and health departments have pursued their own actions against peptide distribution. The Ohio Board of Pharmacy issued summary suspension orders and settlement agreements involving clinics and distributors handling AOD-9604, BPC-157, and ipamorelin, characterizing these substances as unapproved drugs not permitted to be compounded under state law. Other states have taken similar approaches, with enforcement actions targeting both the sellers and the medical practitioners who administer gray-market products to patients.
State-level enforcement creates a patchwork of legal risk. A vendor operating legally in one state may face enforcement action in another. A practitioner who administers a gray-market peptide purchased with an RUO label faces potential discipline from their state medical board, their state pharmacy board, and federal regulators simultaneously. The legal exposure extends to clinics and wellness spas that administer these products, with some states treating administration of unapproved drugs as grounds for license suspension regardless of the practitioner's intent. For a detailed analysis of vendor and practitioner liability, see Online Peptide Vendors: Legal Liability and Consumer Risk.
The category system and 2026 regulatory shift
The FDA's Pharmacy Compounding Advisory Committee (PCAC) evaluates bulk drug substances for potential inclusion on the Section 503A list, which determines what compounding pharmacies can legally prepare. The FDA categorized peptides into two groups: Category 1 (potentially eligible for compounding) and Category 2 (not eligible without further evaluation).
In February 2026, HHS Secretary Robert F. Kennedy Jr. announced that approximately 14 of the 19 peptides on the Category 2 list would be reclassified to Category 1, allowing compounding pharmacies to legally prepare them under physician prescription. This reclassification changed the legal availability of several peptides through regulated channels but did not legalize gray-market sales.[3]
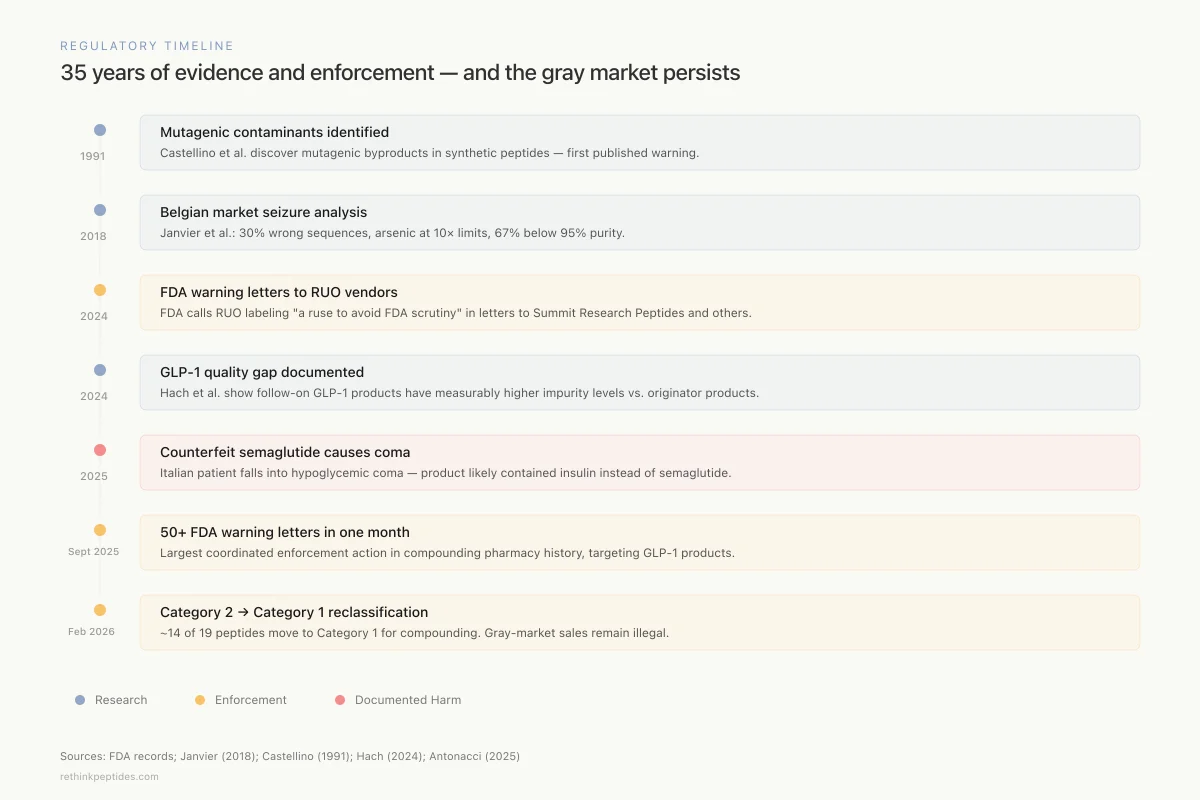
Regulatory Timeline
35 years of evidence and enforcement — and the gray market persists
1991
Mutagenic contaminants identified
Castellino et al. discover mutagenic byproducts in synthetic peptides — first published warning about non-GMP peptide safety.
2018
Belgian market seizure analysis
Janvier et al. publish systematic analysis of seized peptides: 30% wrong sequences, arsenic at 10× limits, two-thirds below 95% purity.
2024
FDA warning letters to RUO vendors
FDA issues warning letters to Summit Research Peptides and others, calling RUO labeling "a ruse to avoid FDA scrutiny."
2024
GLP-1 quality gap documented
Hach et al. at Novo Nordisk publish direct comparison showing follow-on GLP-1 products have measurably higher impurity levels vs. originator products.
2025
Counterfeit semaglutide causes coma
Italian patient falls into hypoglycemic coma after injecting falsified semaglutide — product likely contained insulin instead.
Sept 2025
50+ FDA warning letters in one month
Largest coordinated enforcement action in compounding pharmacy history, targeting GLP-1 compounders and manufacturers.
Feb 2026
Category 2 → Category 1 reclassification
HHS announces ~14 of 19 Category 2 peptides move to Category 1, allowing compounding pharmacies to legally prepare them. Gray-market sales remain illegal.
Sources: FDA records; Janvier (2018); Castellino (1991); Hach (2024); Antonacci (2025)
 View as image
View as imageThe distinction matters: a compounding pharmacy operating under Section 503A or 503B must follow GMP requirements, use pharmaceutical-grade ingredients, perform sterility and endotoxin testing, and operate under state pharmacy board oversight. A gray-market vendor selling the same peptide with an RUO label has none of these requirements. The 2026 reclassification expanded legal access to certain peptides but did nothing to address the quality and safety problems of unregulated products.
Compounding Pharmacies vs. Gray-Market Vendors
The quality gap between compounded and gray-market peptide products is measurable and documented. Hach and colleagues' 2024 analysis provides the clearest data. Their comparison of originator, follow-on, and compounded GLP-1 products revealed a quality hierarchy:[6]
Originator products (manufactured by the NDA/BLA holder under full GMP) showed the highest purity, lowest impurity levels, and best predicted stability. Follow-on drug substances (manufactured by third parties claiming equivalence) showed measurably higher impurity levels, particularly deamidation and aggregation products. Compounded drug products (prepared by 503A/503B pharmacies from bulk drug substances) showed variable quality depending on the source of the bulk ingredient and the compounding process.
Gray-market products were not included in this analysis because they fall entirely outside the regulatory framework. Based on the Janvier data showing 30% incorrect sequences and two-thirds failing basic purity standards, gray-market products represent a quality tier below the lowest category in the Hach analysis.
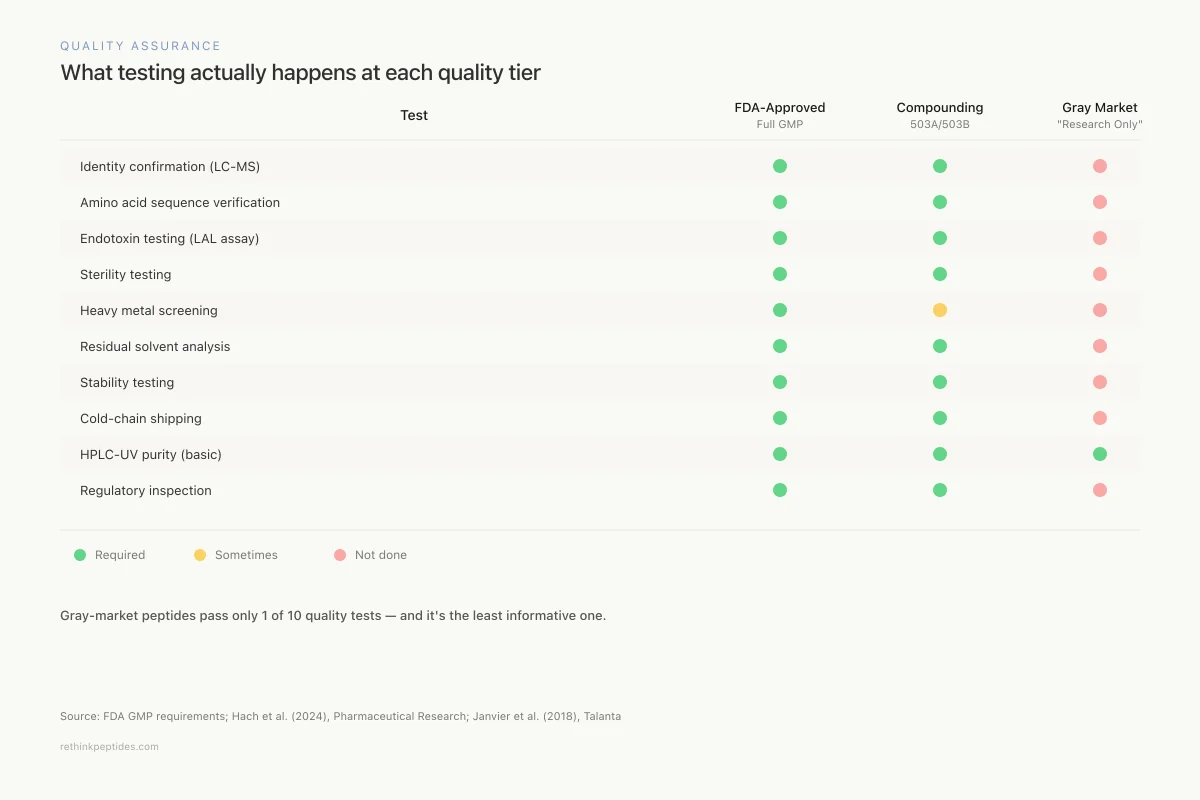
Quality Assurance
What testing actually happens at each quality tier
FDA-Approved
Full GMP
Compounding Pharmacy
503A/503B
Gray Market
"Research Only"
Identity confirmation (LC-MS)
Amino acid sequence verification
Endotoxin testing (LAL assay)
Sterility testing
Heavy metal screening
Residual solvent analysis
Stability testing
Cold-chain shipping
HPLC-UV purity (basic)
Regulatory inspection
Identity confirmation (LC-MS)
Amino acid sequence verification
Endotoxin testing (LAL assay)
Sterility testing
Heavy metal screening
Residual solvent analysis
Stability testing
Cold-chain shipping
HPLC-UV purity (basic)
Regulatory inspection
Source: FDA GMP requirements; Hach et al. (2024); Janvier et al. (2018)
 View as image
View as imageThe legal framework distinguishes these categories clearly. Compounding pharmacies operate under state and federal regulation, require a prescription, and face enforcement for quality failures. Gray-market vendors operate outside this framework entirely. For a detailed analysis of where the law draws this line, see Research-Grade Peptides and Human Use: Where the Law Draws the Line.
The Most Commonly Gray-Marketed Peptides
Several peptides appear consistently in gray-market seizures, FDA warning letters, and consumer forums. Their presence on the gray market reflects a pattern: strong preclinical data generates public interest, the absence of FDA approval creates unmet demand, and RUO labeling fills the gap.
BPC-157
Body Protection Compound-157 is a synthetic pentadecapeptide derived from a gastric protein sequence. It is among the most widely discussed peptides in online wellness communities and the most frequently purchased through gray-market channels. A 2025 systematic review by Vasireddi and colleagues screened 544 BPC-157 articles for orthopaedic sports medicine applications. Only one clinical study met inclusion criteria. The total published human evidence base consists of approximately 30 subjects across three pilot studies.[8] For a comprehensive analysis of the BPC-157 evidence base, see The Real BPC-157 Story: 544 Papers, 30 Human Subjects, 50 Million Views.
The Department of Justice prosecuted Tailor Made Compounding LLC for distributing unapproved peptides including BPC-157, resulting in a $1.79 million forfeiture. BPC-157 was placed on the FDA's Category 2 list, though the 2026 reclassification may move it to Category 1.
Thymosin Alpha 1
Thymosin alpha 1 (Ta1) has a more substantial clinical evidence base than most gray-marketed peptides. Dinetz and Lee's 2024 comprehensive review identified over 30 clinical trials involving more than 11,000 human subjects, examining applications in COVID-19, autoimmune conditions, and cancer treatment.[9] An FDA-approved version (Zadaxin) was marketed in several countries outside the United States. Despite this clinical track record, Ta1 remains unapproved in the U.S. and is frequently sold through gray-market channels with RUO labeling.
Growth hormone secretagogues
Compounds like ipamorelin, CJC-1295, and ibutamoren (MK-0677) are commonly sold as "research chemicals" despite widespread self-administration. Philip and colleagues' 2022 characterization of ibutamoren metabolites in thoroughbred horses was conducted specifically because the compound is used as a performance-enhancing agent in equine sport, illustrating how these substances circulate outside regulated channels across species.[10]
Semaglutide and tirzepatide
The explosive demand for GLP-1 receptor agonists for weight loss has created a large and dangerous counterfeit market. Unlike most gray-marketed peptides, semaglutide and tirzepatide are FDA-approved drugs with established manufacturers. The gray-market versions are explicitly counterfeit, not merely unapproved. The Antonacci and Sterckx case reports demonstrate that counterfeit GLP-1 products can contain substances that produce life-threatening effects entirely different from the expected pharmacology.[1][7]
What the Evidence Does and Does Not Establish
The available data on gray-market peptide quality is limited by a fundamental methodological problem: systematic studies can only analyze products that have been seized, purchased for research, or submitted voluntarily. The Janvier analyses represent government seizures in Belgium. The Hach study analyzed commercially available follow-on products, not gray-market sources. No peer-reviewed study has conducted a randomized, blinded analysis of products purchased from multiple gray-market vendors simultaneously.
This means the existing data likely underestimates the problem. Seized products may represent the worst offenders (those that triggered enforcement attention) or the most common ones (those with the highest volume). The quality distribution across the full gray-market is unknown.
What the evidence does establish: peptides manufactured outside GMP frameworks consistently show higher impurity levels, more frequent sequence errors, and variable potency compared to regulated products.[2][6] Counterfeit versions of approved peptide drugs can contain entirely different active substances.[1] And the RUO label provides no legal protection to sellers, no quality assurance to buyers, and no pharmacovigilance infrastructure for anyone.
The absence of a reporting system is itself a data problem. When a patient receives an FDA-approved drug and experiences an adverse event, there are multiple mechanisms for reporting: the prescribing physician, the pharmacy, the manufacturer, and the FDA's MedWatch system. When a person self-administers a gray-market peptide and experiences an adverse event, none of these mechanisms apply. The person may not seek medical care. If they do, the treating physician may not know what was injected. If the physician suspects a counterfeit product, there is no standardized process for reporting to the FDA. The result is a systematic undercount of harm that makes the gray market appear safer than it is.
The 2026 reclassification of certain peptides to Category 1 may reduce gray-market demand for those specific compounds by creating a legal alternative through compounding pharmacies. But the gray market is not limited to Category 2 peptides. It includes novel research peptides, foreign-manufactured versions of approved drugs, and compounds that have never been evaluated by any regulatory authority. The structural incentives that sustain it remain intact: low synthesis costs, high consumer demand, minimal enforcement risk for overseas manufacturers, and the persistent fiction that four words on a label create legal protection. For vendor-specific liability analysis, see Online Peptide Vendors: Legal Liability and Consumer Risk.
The Bottom Line
The "research use only" label on gray-market peptides is a legal fiction that the FDA has explicitly rejected. Laboratory analyses consistently find contamination, incorrect sequences, and substandard purity in unregulated peptide products. Published case reports document life-threatening harm from counterfeit peptides. The 2026 regulatory reclassification expanded legal access through compounding pharmacies but did not address the fundamental quality and safety problems of the gray market.
Sources & References
- 1RPEP-09953·Antonacci, Giulia et al. (2025). “Counterfeit Semaglutide Causes Hypoglycemic Coma: Dangerous Fake Drug Warning.” European journal of hospital pharmacy : science and practice.Study breakdown →PubMed →↩
- 2RPEP-03726·Janvier, Steven et al. (2018). “Counterfeit Peptide Drugs Sold Online Contained Arsenic, Lead, and as Little as 5% Purity.” Talanta.Study breakdown →PubMed →↩
- 3RPEP-10520·Combs, Blinn E et al. (2025). “The Legal Risks of Compounding Semaglutide and Tirzepatide for Weight Loss.” International journal of pharmaceutical compounding.Study breakdown →PubMed →↩
- 4RPEP-03725·Janvier, Steven et al. (2018). “Falsification of biotechnology drugs: current dangers and/or future disasters?.” Journal of pharmaceutical and biomedical analysis.Study breakdown →PubMed →↩
- 5RPEP-00187·Castellino, S et al. (1991). “Hidden Carcinogenic Contaminants in Synthetic Peptides That Standard Purity Tests Miss.” Mutagenesis.Study breakdown →PubMed →↩
- 6RPEP-08330·Hach, Morten et al. (2024). “Lab Tests Find Quality Problems in Generic and Compounded Semaglutide and Liraglutide Products.” Pharmaceutical research.Study breakdown →PubMed →↩
- 7RPEP-16173·Sterckx, Mira et al. (2026). “Euglycemic Ketoacidosis Following the Use of Counterfeit Semaglutide for Weight Loss..” Cureus.Study breakdown →PubMed →↩
- 8RPEP-13892·Vasireddi, Nikhil et al. (2025). “BPC-157 for Sports Injuries: What a Systematic Review of the Evidence Actually Shows.” HSS journal : the musculoskeletal journal of Hospital for Special Surgery.Study breakdown →PubMed →↩
- 9RPEP-08088·Dinetz, Elliot et al. (2024). “Review of Thymosin Alpha-1 Peptide Safety and Effectiveness Across 11,000 Patients.” Alternative therapies in health and medicine.Study breakdown →PubMed →↩
- 10RPEP-06432·Philip, Moses et al. (2022). “Characterization of growth hormone secretagogue small molecule ibutamoren (MK-0677) and its possible metabolites in thoroughbred horses for doping control..” Rapid communications in mass spectrometry : RCM.Study breakdown →PubMed →↩