AOD-9604: The Growth Hormone Fragment for Fat Loss
AOD-9604
50% Less Weight Gain
In the first published AOD-9604 study, oral dosing at 500 micrograms per kilogram reduced body weight gain by over 50% in obese Zucker rats over 19 days, with increased adipose tissue lipolytic activity and no effect on IGF-1 levels.
Ng et al., Hormone Research, 2000
Ng et al., Hormone Research, 2000
If you only read one thing
AOD-9604 is a 16-amino-acid fragment of human growth hormone, engineered to keep the fat-burning part and drop the growth-promoting side effects. In mice, it worked beautifully — reducing weight gain by 50%. In humans, a small 12-week trial showed a modest 1.8 kg advantage over placebo, but the decisive 536-patient trial over 24 weeks failed. Its developer quit the drug in 2007. Today it's sold on the gray market as an injectable — a formulation that was never part of any of the six published clinical trials. The GRAS food-safety designation often cited in marketing is not a drug approval. The preclinical science is real; the human weight-loss claims do not match the clinical record.
AOD-9604 is a synthetic peptide consisting of 16 amino acids derived from the C-terminal region of human growth hormone (hGH), specifically residues 177 through 191, with an additional tyrosine substituted at the N-terminus.[1] Developed in the 1990s by Professor Frank Ng at Monash University in Australia, the peptide was designed to isolate the fat-metabolizing properties of growth hormone from its growth-promoting and diabetogenic effects. The logic was straightforward: if a small fragment of hGH could stimulate fat breakdown without triggering the side effects of the full hormone, it could become a safer obesity treatment. Preclinical results were promising. Clinical results were not. AOD-9604 completed six human trials involving over 900 participants before Metabolic Pharmaceuticals terminated development in 2007 after its largest trial failed to meet its primary endpoint. The peptide now circulates widely in the gray market and compounding pharmacy space, marketed with claims that go well beyond what the published evidence supports.
Key Takeaways
- AOD-9604 is a 16-amino-acid fragment of human growth hormone, engineered to keep the fat-burning part and drop the growth-promoting side effects.
- In obese rats, it cut weight gain in half. In mice, it burned fat without the blood sugar problems that full-length growth hormone causes.
- In humans, the story collapsed. A small 12-week trial showed a modest 2 kg advantage. The larger, decisive trial of 536 people failed entirely.
- The developer quit in 2007 after the Phase IIb failure. No pharmaceutical company has picked it up since.
- Unlike growth hormone, AOD-9604 doesn't bind the growth hormone receptor — meaning whatever it does in mice, it does through a different pathway nobody has fully mapped.
- It's not FDA-approved. The "GRAS" designation some marketers cite is a food-safety rating, not a drug approval.
- The injectable form sold through compounding pharmacies today was never part of any of the six published clinical trials.
What Is AOD-9604?
AOD stands for "Anti-Obesity Drug." The name reflects the peptide's original purpose: an obesity therapeutic derived from the portion of human growth hormone responsible for fat metabolism. Human growth hormone is a 191-amino-acid protein with multiple biological functions including growth promotion, glucose regulation, and lipid metabolism. AOD-9604 isolates residues 177 through 191 of this protein, the C-terminal fragment believed to carry the lipolytic signal, and adds a tyrosine residue at the N-terminus for stability.[1]
The development rationale emerged from earlier research showing that different regions of the growth hormone molecule mediate different biological functions. The N-terminal and central regions drive growth promotion and glucose regulation through the GH receptor/JAK2/STAT5 signaling cascade. The C-terminal region appeared to carry an independent lipolytic signal. By isolating this fragment, researchers hoped to create a peptide that burned fat without the problematic side effects of the full hormone.
The critical distinction between AOD-9604 and full-length growth hormone is what AOD-9604 does not do. Growth hormone raises IGF-1 levels (driving cell proliferation and potential cancer risk), causes insulin resistance and hyperglycemia, promotes fluid retention, and can trigger carpal tunnel syndrome and joint pain. AOD-9604 was specifically designed to avoid these effects by using only the fragment responsible for fat metabolism.
The Design Logic
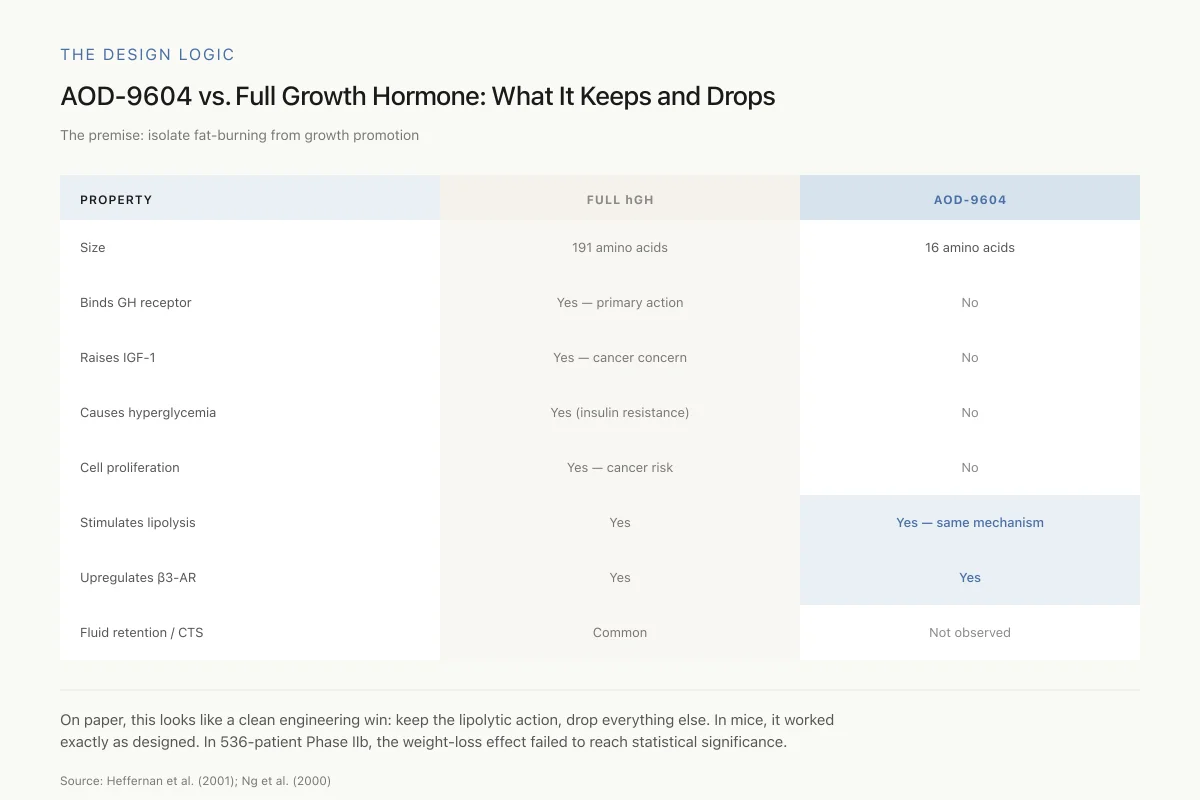
AOD-9604 vs. Full Growth Hormone: What It Keeps and What It Drops
The entire scientific premise: isolate fat-burning from growth promotion
On paper, this looks like a clean engineering win: keep the lipolytic action, drop everything else. In preclinical mice, it worked exactly as designed. In the largest human trial (536 patients, 24 weeks), the weight-loss effect failed to reach statistical significance.
Source: Heffernan et al. (2001); Ng et al. (2000)
 View as image
View as imageHeffernan et al. confirmed this distinction in 2001: AOD-9604 does not compete for the growth hormone receptor and does not induce cell proliferation in in vitro assays.[3] The peptide appears to act through a mechanism entirely independent of the classical GH-receptor/JAK2/STAT5 signaling pathway that mediates most of growth hormone's effects. This separation of lipolytic activity from growth-promoting activity was the entire scientific premise of AOD-9604's development. For a detailed analysis of how this selectivity works at the molecular level, see How AOD-9604 Targets Fat Cells Without Growth Hormone's Side Effects.
The Preclinical Evidence
The Ng Laboratory Studies (2000-2001)
The foundational AOD-9604 research came from Frank Ng's laboratory at Monash University. In the first published study, Ng et al. (2000) treated obese Zucker rats with oral AOD-9604 at 500 micrograms per kilogram body weight daily for 19 days.[1] The results were striking: treated animals gained only 15.8 grams compared to 35.6 grams in controls, a reduction of over 50%. Adipose tissue from treated animals showed increased lipolytic activity. The peptide did not affect blood glucose levels, insulin concentrations, or IGF-1 production. The study was notable for demonstrating oral bioavailability, a significant advantage for a peptide drug candidate since most peptides are destroyed by digestive enzymes and require injection.
Heffernan et al. followed with two studies in 2001 that expanded the evidence. The first, published in the International Journal of Obesity, treated obese (ob/ob) mice with AOD-9604 via mini-osmotic pumps for 14 days.[3] Both hGH and AOD-9604 significantly reduced body weight gain, and this reduction was associated with increased in vivo fat oxidation and elevated plasma glycerol levels (a marker of lipolysis). The key finding: unlike hGH, AOD-9604 did not induce hyperglycemia or reduce insulin secretion. In vitro assays confirmed that AOD-9604 did not bind the growth hormone receptor and did not stimulate cell proliferation.
The second Heffernan et al. study, published in Endocrinology, investigated the mechanism through which AOD-9604 stimulates fat breakdown.[2] Both hGH and AOD-9604 increased the expression of beta-3 adrenergic receptor (beta-3 AR) RNA in adipose tissue of obese mice, restoring levels comparable to lean mice. The beta-3 AR is the primary lipolytic receptor in fat cells. However, when the researchers repeated the experiments in beta-3 AR knockout mice, hGH and AOD-9604 failed to produce weight loss, suggesting beta-3 AR upregulation is necessary for the chronic weight-loss effect. In an acute (single-dose) experiment, AOD-9604 still increased energy expenditure and fat oxidation even in knockout mice, indicating the peptide has additional mechanisms beyond beta-3 AR signaling.
What the Preclinical Data Shows and Does Not Show
The mouse and rat studies established three things convincingly: AOD-9604 stimulates lipolysis, it increases fat oxidation, and it does this without the metabolic side effects of full-length growth hormone. These are real findings published in peer-reviewed endocrinology journals. The Ng 2000 study also demonstrated oral bioavailability, which was unusual for a peptide and made AOD-9604 attractive as a potential oral obesity drug.
The mechanism, however, remains incompletely understood. AOD-9604 upregulates beta-3 adrenergic receptors and is dependent on them for chronic weight loss effects, but the acute effects on energy expenditure and fat oxidation occur through a separate, unidentified pathway.[2] The actual molecular target through which AOD-9604 initiates its lipolytic signal has never been identified. This is a meaningful gap: without knowing the target, it is difficult to predict dose-response relationships, optimize the peptide sequence, or understand why clinical translation failed.
What the preclinical data does not establish is whether these effects translate to meaningful weight loss in humans. Mouse obesity models, particularly the ob/ob mouse (which lacks leptin entirely), are genetically extreme and do not represent typical human obesity. The 50% reduction in weight gain in Zucker rats is impressive in context but says little about what to expect in a 12-week human trial. The ob/ob mouse has no functional leptin signaling and gains weight relentlessly; any compound that slightly increases fat oxidation will show a measurable effect in this model. The bar for demonstrating clinical relevance in human obesity is substantially higher.
The Clinical Trials: Promise and Failure
The Clinical Arc
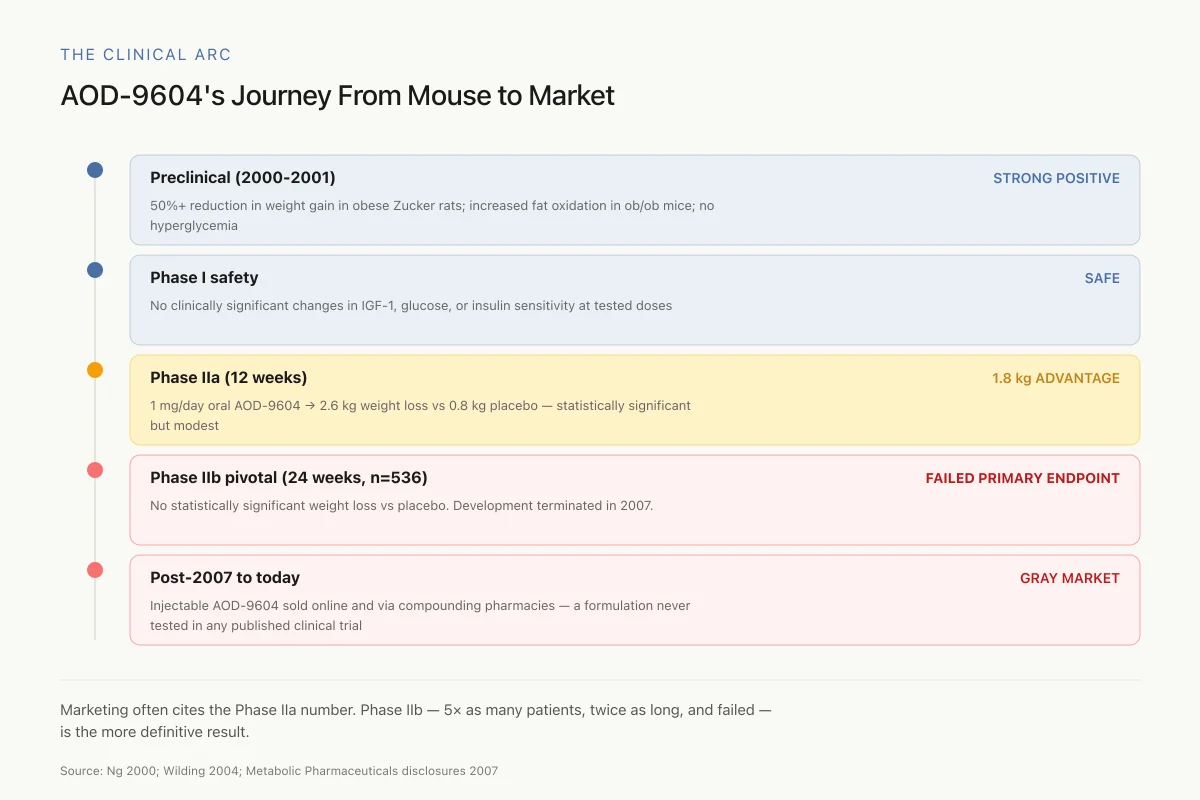
AOD-9604's Journey From Mouse to Market: A Cautionary Timeline
Marketing pages often cite the Phase IIa number as proof AOD-9604 works. They rarely mention Phase IIb — which enrolled 5× as many patients, ran twice as long, and failed. The larger, longer, better-powered trial is the more definitive one.
Source: Ng 2000; Wilding 2004 (Phase IIa); Metabolic Pharmaceuticals disclosures 2007
 View as image
View as imageMetabolic Pharmaceuticals Ltd., the Australian company that held the AOD-9604 patent, advanced the peptide through six human clinical trials involving over 900 participants total.[4]
Phase I and Phase IIa Results
Early-phase trials established safety and provided initial efficacy signals. In a 12-week Phase IIa randomized controlled trial, subjects receiving oral AOD-9604 at 1 mg per day lost an average of 2.6 kg compared to 0.8 kg in the placebo group, a statistically significant difference of 1.8 kg.[4] The safety profile was favorable: no clinically significant changes in IGF-1, glucose tolerance, or insulin sensitivity were observed. This was consistent with the preclinical finding that AOD-9604 acts independently of the growth hormone receptor.
The Phase IIb Failure
The pivotal Phase IIb trial enrolled 536 obese subjects and ran for 24 weeks. This was the trial that would determine whether AOD-9604 could produce clinically meaningful weight loss at scale. It did not. The trial failed to reach its primary endpoint of statistically significant weight loss versus placebo.[5]
Metabolic Pharmaceuticals terminated AOD-9604 development in 2007. The company's share price collapsed, and the obesity drug pipeline moved on. The reasons for the Phase IIb failure were never fully published in peer-reviewed form. Possible explanations include:
- Dose insufficiency: the oral doses used may have been too low for the duration tested
- Oral bioavailability limitations: despite the promising rat data, human gastrointestinal degradation of the peptide may have limited systemic exposure
- Endpoint sensitivity: 24-week weight loss trials require substantial effect sizes to reach significance in large, heterogeneous populations
- Biology does not scale: the ob/ob mouse model may have predicted efficacy that simply does not exist in human obesity
The honest assessment is that nobody knows exactly why AOD-9604 failed in Phase IIb because the detailed trial results were never published. This is a significant gap in the evidence base. Claims about AOD-9604's weight loss efficacy in humans must be evaluated against the reality that its largest clinical trial failed.
The Unexpected Cartilage Finding
In 2015, Kwon and Park published an unexpected result: intra-articular AOD-9604 injections enhanced cartilage regeneration in a collagenase-induced knee osteoarthritis rabbit model.[6]
The study randomized 32 rabbits into four groups receiving weekly knee injections for 4 to 7 weeks: saline (control), hyaluronic acid (HA) alone, AOD-9604 alone (0.25 mg), or AOD-9604 combined with HA. At 8 weeks, cartilage degeneration scores were significantly lower (better) in all treatment groups compared to saline controls. The combination group (AOD-9604 plus HA) scored significantly better than either treatment alone, and the lameness period was shortest in the combination group.
This study opened a secondary research avenue for AOD-9604 as a joint health compound, distinct from its original obesity indication. The mechanism by which a growth hormone fragment might promote cartilage repair is not established; speculation centers on stimulation of chondrocyte proliferation or extracellular matrix synthesis through pathways independent of IGF-1. This remains a single animal study with 8 animals per group and no human follow-up data. For a full analysis of this research direction, see AOD-9604 and Cartilage: An Unexpected Secondary Research Finding.
WADA, GRAS, and Regulatory Status
AOD-9604 occupies an unusual regulatory space. The World Anti-Doping Agency (WADA) banned it in 2013 under the category of peptide hormones, growth factors, and related substances. Cox et al. (2015) developed methods for detecting AOD-9604 and its metabolites in biological samples for anti-doping purposes, noting that the peptide was already available on multiple internet websites and had been identified in confiscated products.[7]
Safety
ModerateInjectable gray-market AOD-9604 has no human safety data
Concern
All six published clinical trials used oral AOD-9604. The injectable subcutaneous form — which is what compounding pharmacies and online vendors sell today — was never tested in any of those trials. Its pharmacokinetics, absorption, tissue distribution, and long-term safety in humans are unknown.
What the research says
The favorable safety profile from the oral trials does not automatically transfer to injection. Different route = different absorption, different peak concentrations, different metabolism. Product purity from unregulated sources is also variable.
Particularly relevant for: Anyone buying AOD-9604 injections from online vendors or compounding pharmacies
What to do
Don't assume 'the clinical trials showed it was safe' covers injection. If you're using it, use the lowest effective dose, source from a verified compounding pharmacy, and get baseline labs (glucose, lipid panel) to track changes.
Wilding (2004); Cox et al. (2015); WADA banned substances list
In the United States, AOD-9604 received GRAS (Generally Recognized as Safe) designation from the FDA, but this designation is frequently misunderstood. GRAS applies to AOD-9604 as a food ingredient, not as a drug. It means the FDA accepted evidence that the compound is safe for consumption at specified doses in food products. GRAS status does not constitute drug approval, does not validate therapeutic claims, and does not authorize marketing AOD-9604 for weight loss, fat metabolism, or any medical indication. For a complete breakdown of what this designation means in practice, see AOD-9604's FDA GRAS Status: What That Means and What It Doesn't.
AOD-9604 in Context: The Obesity Peptide Landscape
When AOD-9604 was in clinical development in the early 2000s, the obesity drug landscape looked very different. The options were limited to appetite suppressants (sibutramine, later withdrawn for cardiovascular safety) and lipase inhibitors (orlistat). Halford et al. (2006) reviewed the obesity drug pipeline and included AOD-9604 alongside other candidates, noting its novel mechanism as a growth hormone fragment acting on fat metabolism without affecting the GH receptor.[5]
The landscape has since been transformed by GLP-1 receptor agonists. Semaglutide (Wegovy) produces average weight loss of 15% of body weight; tirzepatide (Zepbound), a dual GIP/GLP-1 agonist, produces 20% or more. These are weight losses of 15 to 25 kg in clinical trials lasting 68 to 72 weeks. AOD-9604's best clinical result was 1.8 kg more than placebo over 12 weeks.
This context matters for evaluating current marketing of AOD-9604. The peptide is sold online and through compounding pharmacies with claims about fat loss, body composition improvement, and metabolic enhancement. These claims rest on preclinical mouse data and a small Phase IIa trial. The larger trial failed. No regulatory authority has approved AOD-9604 for obesity or any therapeutic indication. The evidence base is not comparable to approved GLP-1 therapies. For another peptide hormone involved in metabolic regulation, see Amylin (IAPP): The Third Pancreatic Peptide Hormone.
The Gray Market and Compounding Pharmacy Reality
AOD-9604 occupies a space familiar to anyone following the peptide market: a compound with genuine scientific origins, real but limited evidence, and marketing claims that far outstrip what the data supports. After Metabolic Pharmaceuticals abandoned development, AOD-9604 became widely available through research chemical suppliers, online peptide vendors, and compounding pharmacies.
The typical marketing narrative presents AOD-9604 as a proven fat-loss peptide backed by clinical trials. This framing is technically true in that clinical trials existed but misleading because it omits the most important trial result: failure. The Phase IIa data (1.8 kg advantage over placebo) is real but modest. The Phase IIb data (failure to reach significance in 536 subjects) is the larger and more definitive dataset.
Product quality is another concern. Cox et al. (2015) noted that AOD-9604 was already available on multiple internet websites and had been identified in confiscated pharmaceutical preparations seized by authorities.[7] The purity, concentration, and composition of gray-market peptides are variable. The clinical trial data, whatever its limitations, was generated with pharmaceutical-grade material under controlled conditions. There is no guarantee that commercially available AOD-9604 matches this standard.
The Route Mismatch
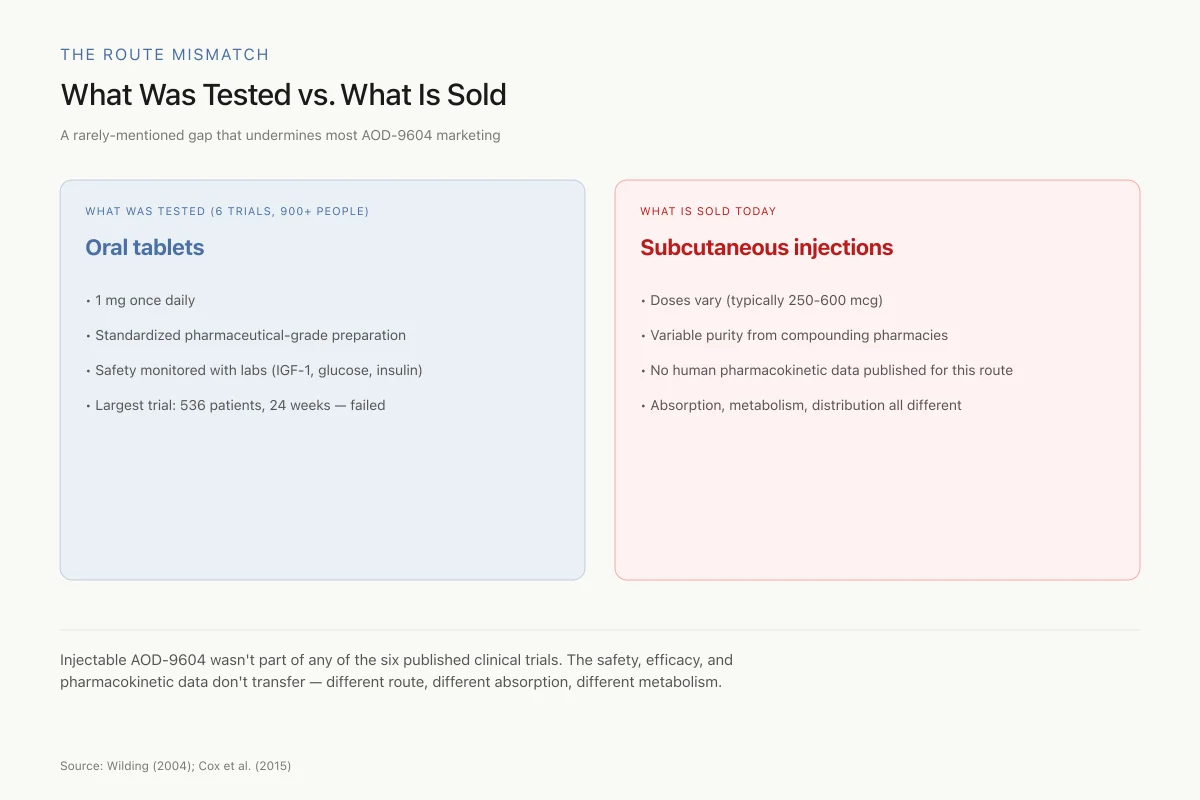
What Was Tested vs. What Is Sold
A rarely-mentioned gap that undermines most AOD-9604 marketing
- • 1 mg once daily
- • Standardized pharmaceutical-grade preparation
- • Safety monitored with labs (IGF-1, glucose, insulin)
- • Largest trial: 536 patients, 24 weeks — failed
- • Doses vary (typically 250-600 mcg)
- • Variable purity from compounding pharmacies and research-chemical vendors
- • No human pharmacokinetic data published for this route
- • Absorption, first-pass metabolism, and tissue distribution all different from oral
Injectable AOD-9604 wasn't part of any of the six published clinical trials. The safety, efficacy, and pharmacokinetic data don't transfer — different route, different absorption, different metabolism. The testimonials about injected AOD-9604 are not backed by the trial data they're often cited alongside.
Source: Wilding (2004); Cox et al. (2015); general peptide pharmacology
 View as image
View as imageInjectable AOD-9604, the most common form sold today, was never tested in any of the published clinical trials. The six human trials used oral formulations. The subcutaneous injection route, which is the standard in the compounding pharmacy and gray-market space, has a different pharmacokinetic profile. Whether the preclinical or clinical findings apply to injected AOD-9604 at the doses commonly used is an open question without published human data.
What Remains Unknown
Several important questions about AOD-9604 remain unanswered:
The mechanism of action is incomplete. The beta-3 AR pathway appears involved but is not the full story, as acute effects persist in knockout models. The actual molecular target through which AOD-9604 initiates lipolysis has not been identified.
The Phase IIb data is unpublished. The most important clinical dataset for AOD-9604 was never published in peer-reviewed form. Without access to the full trial data, including dose-response relationships, subgroup analyses, and reasons for failure, the clinical picture remains incomplete.
Long-term safety data is limited. The six clinical trials collectively provide safety data from over 900 participants, which is a reasonable database for common adverse events. Long-term effects of chronic use, particularly the gray-market pattern of months-long self-administration via injection, have not been studied.
The cartilage finding needs human validation. One rabbit study with 32 animals does not establish AOD-9604 as a joint health treatment. Controlled human trials are needed before any conclusions about cartilage regeneration can be drawn.
Compounding pharmacy products are not equivalent to clinical trial material. AOD-9604 sold through compounding pharmacies or online vendors varies in purity, concentration, and formulation. The clinical trial data, such as it is, applies to pharmaceutical-grade material under controlled conditions.
Injectable pharmacokinetics are unstudied. All six published clinical trials used oral AOD-9604. The subcutaneous injection route now standard in the gray market has different absorption kinetics, different first-pass metabolism characteristics, and potentially different tissue distribution. No published human data exists for injected AOD-9604.
The beta-3 AR dependence may limit human relevance. The chronic lipolytic effects of AOD-9604 in mice depended on beta-3 adrenergic receptors, which are abundantly expressed in rodent brown and white adipose tissue.[2] Humans express substantially less beta-3 AR in adipose tissue than rodents, and human beta-3 AR agonists have generally failed to produce meaningful weight loss in clinical trials. If AOD-9604's mechanism depends on a pathway that is less active in humans than in mice, this could partly explain the clinical translation failure.
The Bottom Line
AOD-9604 is a 16-amino-acid fragment of human growth hormone with documented lipolytic activity in preclinical models and a mechanism that avoids the metabolic side effects of full-length hGH. Its clinical history is defined by a pattern common in drug development: promising early results that did not survive a larger, more rigorous trial. The Phase IIb failure in 536 subjects effectively ended pharmaceutical development, and the detailed trial data was never published. The peptide's current circulation in the gray market and compounding pharmacy space is driven by preclinical findings and a small Phase IIa result, not by clinical proof of efficacy. The cartilage finding is preliminary. The GRAS designation is a food safety classification, not a drug approval. The evidence base does not support the marketing claims commonly made about AOD-9604.
Sources & References
- 1RPEP-00608·Ng, F M et al. (2000). “AOD9604: The Growth Hormone Fragment That Burned Fat in Rats Without Insulin Side Effects.” Hormone research.Study breakdown →PubMed →↩
- 2RPEP-00669·Heffernan, M et al. (2001). “The effects of human GH and its lipolytic fragment (AOD9604) on lipid metabolism following chronic treatment in obese mice and beta(3)-AR knock-out mice..” Endocrinology.Study breakdown →PubMed →↩
- 3RPEP-00670·Heffernan, M A et al. (2001). “Increase of fat oxidation and weight loss in obese mice caused by chronic treatment with human growth hormone or a modified C-terminal fragment..” International journal of obesity and related metabolic disorders : journal of the International Association for the Study of Obesity.Study breakdown →PubMed →↩
- 4RPEP-00993·Wilding, John (2004). “AOD-9604 Metabolic..” Current opinion in investigational drugs (London.Study breakdown →PubMed →↩
- 5RPEP-01143·Halford, Jason C G (2006). “Obesity drugs in clinical development..” Current opinion in investigational drugs (London.Study breakdown →PubMed →↩
- 6RPEP-02694·Kwon, Dong Rak et al. (2015). “Effect of Intra-articular Injection of AOD9604 with or without Hyaluronic Acid in Rabbit Osteoarthritis Model..” Annals of clinical and laboratory science.Study breakdown →PubMed →↩
- 7RPEP-02608·Cox, Holly D et al. (2015). “Detection and in vitro metabolism of AOD9604..” Drug testing and analysis.Study breakdown →PubMed →↩