MK-677 and Insulin Resistance: The Blood Sugar Trade-Off
GH Peptide Safety
26% glucose increase
Fasting glucose rose from 5.4 to 6.8 mmol/L after 4 weeks of MK-677 at 25 mg/day in elderly adults.
Chapman et al., J Clin Endocrinol Metab, 1996
Chapman et al., J Clin Endocrinol Metab, 1996
If you only read one thing
MK-677 is a pill that boosts your growth hormone levels. It works — IGF-1 goes up nearly 73% over a year. But there's a catch: every single clinical trial that checked blood sugar found it went up too. In one study of elderly adults, fasting glucose jumped 26% in just 4 weeks. This isn't a rare side effect. It's built into how the drug works, because growth hormone itself pushes blood sugar up. The benefits (more muscle, less fat, higher IGF-1) and the costs (higher blood sugar, insulin resistance) are inseparable.
MK-677 (ibutamoren) is one of the most studied oral growth hormone secretagogues, with clinical trial data spanning populations from young healthy men to elderly adults with Alzheimer's disease. It reliably raises growth hormone (GH) and insulin-like growth factor 1 (IGF-1) levels. It also reliably raises blood sugar. Every randomized controlled trial measuring glucose metabolism has documented some degree of impairment, from modest fasting glucose increases to clinically meaningful shifts in insulin sensitivity and HbA1c.[1][2] This is the central trade-off in MK-677 research, and it applies broadly to the growth hormone secretagogue risk profiles category.
This article examines exactly what happens to blood sugar during MK-677 use, why it happens, how large the effect is across different populations, and what the clinical trial data says about reversibility.
Key Takeaways
- Every MK-677 trial that measured blood sugar found it got worse — not sometimes, every time.
- In one study, fasting glucose jumped 26% in just four weeks — from normal into pre-diabetic territory.
- Your fasting glucose can look perfectly normal while a sugar load test reveals real damage.
- The blood sugar rise isn't a side effect — it's baked in, because growth hormone itself opposes insulin.
- Over two years, HbA1c drifted up 0.3% compared to placebo, and insulin sensitivity dropped.
- The same dose that made older adults' IGF-1 look young also pushed their glucose toward pre-diabetes.
- Most users only check fasting glucose — that's the wrong test to catch the real metabolic cost.
What happens to blood sugar during MK-677 use
Clinical Trial Summary
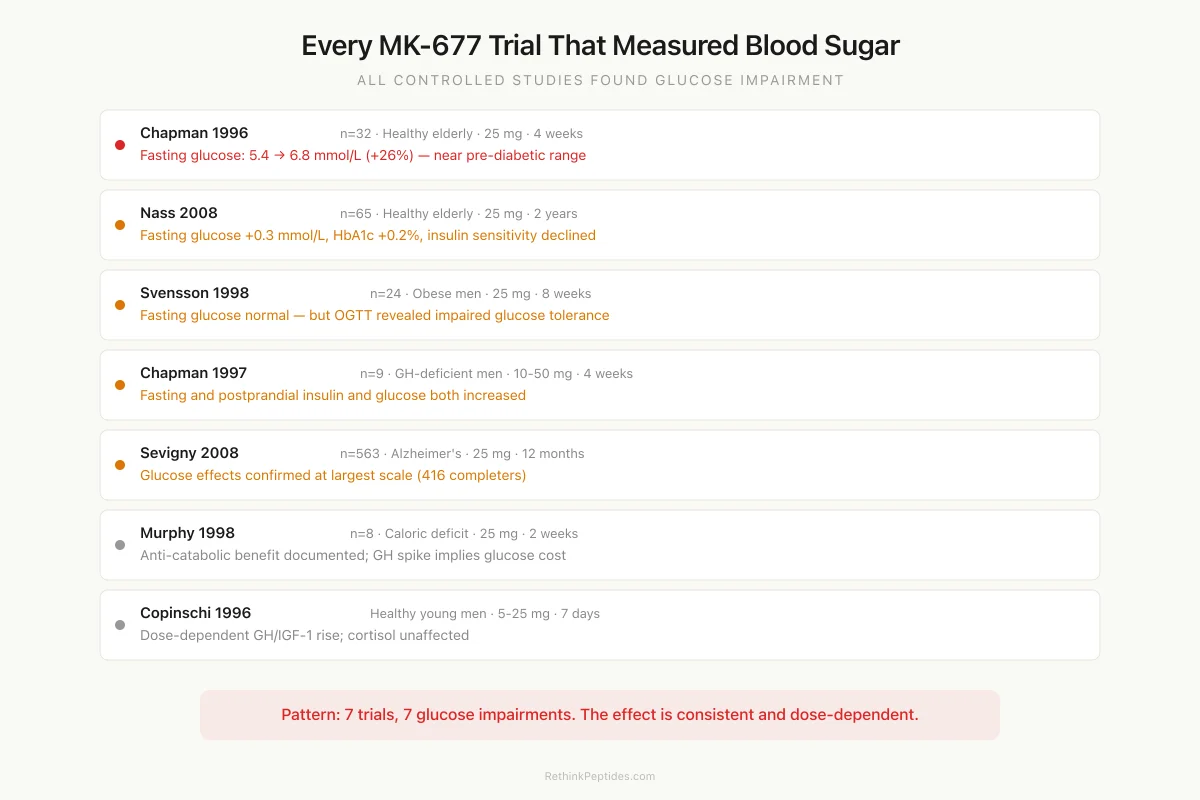
Every MK-677 Trial That Measured Blood Sugar
All controlled studies found some degree of glucose impairment
Fasting glucose: 5.4 → 6.8 mmol/L (+26%)
Fasting glucose +0.3 mmol/L, HbA1c +0.2%
Fasting glucose normal, but OGTT impaired
Fasting + postprandial insulin and glucose up
Glucose effects confirmed at scale (416 completers)
Anti-catabolic (+nitrogen balance) but GH spike implies glucose cost
Dose-dependent GH/IGF-1 rise; cortisol unaffected
Source: Chapman (1996, 1997); Nass (2008); Svensson (1998); Sevigny (2008); Murphy (1998); Copinschi (1996)
 View as image
View as imageThe glucose data from MK-677 trials is remarkably consistent. Every controlled study that measured blood sugar found some form of metabolic disruption, though the magnitude and clinical significance varied by population.
Chapman et al. (1996) conducted the first major MK-677 trial in 32 healthy elderly subjects (ages 64-81). At 25 mg/day, fasting glucose rose from 5.4 to 6.8 mmol/L within 4 weeks. That is a shift from normal to near the pre-diabetic range (6.1-6.9 mmol/L), and it was statistically significant (P < 0.01 vs. baseline).[1] The same dose that restored IGF-1 to youthful levels (from 141 to 265 micrograms/L at 4 weeks) simultaneously pushed glucose into territory that would prompt clinical monitoring.
The Nass et al. (2008) trial is the longest controlled MK-677 study: 65 healthy older adults randomized to 25 mg/day or placebo for up to 2 years. Fasting blood glucose increased an average of 0.3 mmol/L (5 mg/dL) in the MK-677 group (P = 0.015), and insulin sensitivity, estimated by the QUICKI index, declined.[2] HbA1c increased 0.2% in the MK-677 group while decreasing 0.1% in the placebo group (P = 0.002). At 12 months, 8 participants in the MK-677 arm had an HbA1c above 6%. The 2-year exploratory analyses confirmed these 1-year results persisted.
These numbers appear modest in isolation. A 5 mg/dL fasting glucose increase or a 0.2% HbA1c rise would not alarm most clinicians looking at a single lab report. But in healthy individuals with no prior metabolic risk, any sustained shift toward impaired glucose tolerance deserves attention, particularly over months or years of use. The significance is amplified by the fact that the Nass trial enrolled healthy volunteers with normal baseline glucose; a population with pre-existing metabolic risk would likely show larger shifts.
The OGTT reveals what fasting glucose hides
One of the most instructive findings comes from Svensson et al. (1998), who studied 24 obese men (BMI > 30) receiving MK-677 at 25 mg/day for 8 weeks. Fasting glucose and insulin concentrations did not change. On paper, the metabolic profile appeared clean.[3]
But an oral glucose tolerance test (OGTT) told a different story. Glucose homeostasis was impaired at both 2 and 8 weeks of treatment. The OGTT measures how efficiently the body clears a standardized glucose load over two hours, and it captures insulin resistance that fasting values miss entirely. In this study, the body's ability to process a glucose challenge deteriorated even though resting glucose levels held steady.
This distinction matters because fasting glucose is the most commonly reported metric in MK-677 discussions online, and it paints an incomplete picture. A person using MK-677 could check fasting glucose, see normal values, and assume their metabolism is unaffected. The Svensson data shows that assumption would be wrong. The metabolic cost was present but invisible to the most basic test.
The study also demonstrated that MK-677 increased fat-free mass (measured by both DEXA and a four-compartment model), increased IGF-1 by approximately 40%, and raised basal metabolic rate at 2 weeks (P = 0.01). These are meaningful anabolic benefits occurring alongside metabolic costs that only become visible with the right test. The trade-off is not hypothetical; it was measured in the same subjects, during the same treatment period.
Why MK-677 affects blood sugar: the growth hormone connection
Mechanism
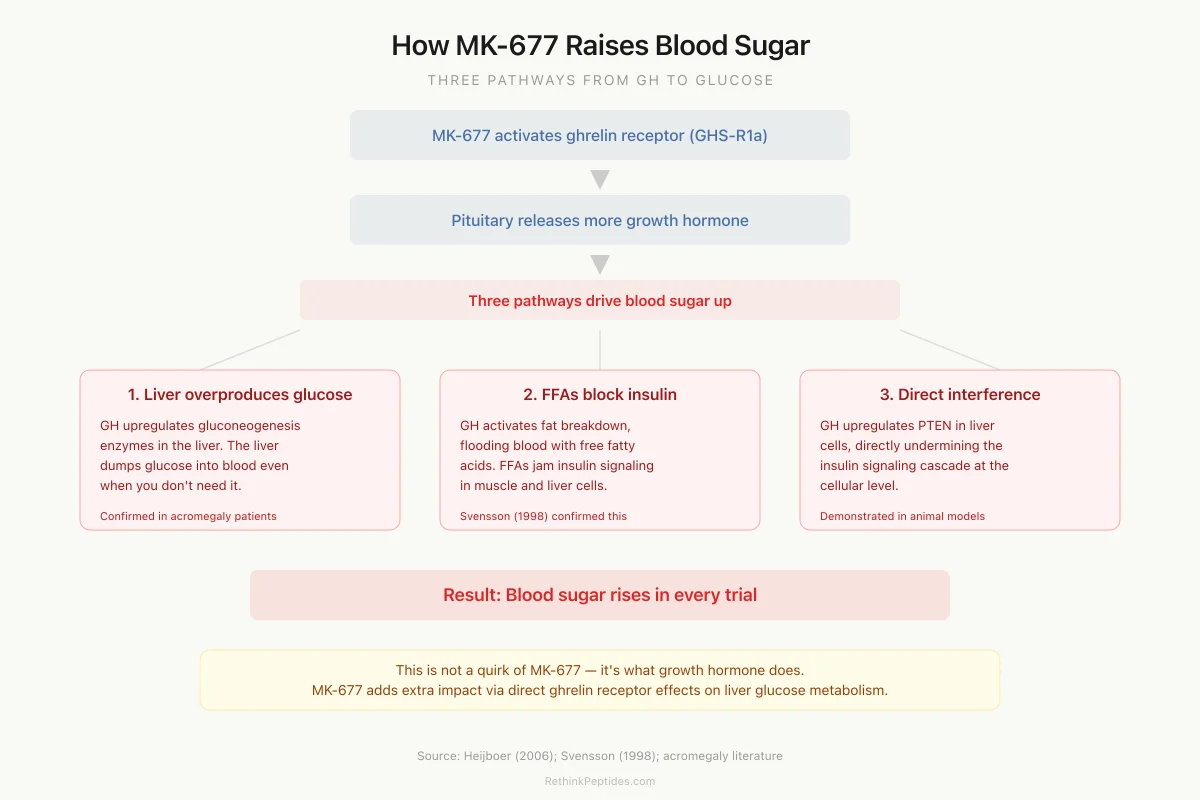
How MK-677 Raises Blood Sugar: Three Pathways
1. Liver overproduces glucose
GH upregulates gluconeogenesis enzymes — liver dumps glucose into blood even when you don't need it
2. Fat cells release free fatty acids
FFAs block insulin signaling in muscle and liver cells, making them "deaf" to insulin
3. Direct insulin pathway interference
GH upregulates PTEN in liver cells, directly undermining the insulin signaling cascade
Key point: This is not a quirk of MK-677. It's what growth hormone does. Any intervention that raises GH will push blood sugar up through these same pathways. MK-677 adds an extra layer because it also activates ghrelin receptors in the liver, which independently affect glucose metabolism.
Source: Heijboer (2006); Svensson (1998); acromegaly literature
 View as image
View as imageMK-677's glucose effects are not a quirk of the compound itself. They are a predictable consequence of raising growth hormone.
GH is classified as a counter-regulatory hormone, meaning it opposes insulin's actions. When GH levels rise, three interconnected processes drive blood sugar upward.
Increased hepatic glucose output. GH stimulates gluconeogenesis in the liver by upregulating key enzymes (phosphoenolpyruvate carboxykinase and glucose-6-phosphatase). The liver produces more glucose and releases it into the bloodstream, independent of whether the body needs it. In patients with acromegaly (chronic GH excess), hepatic glucose production is markedly elevated, confirming this pathway operates in humans at pharmacologically relevant GH concentrations.
Free fatty acid mobilization. GH activates hormone-sensitive lipase, driving lipolysis and increasing circulating free fatty acids (FFAs). Elevated FFAs interfere with insulin signaling in skeletal muscle and liver by inhibiting insulin receptor substrate-1 (IRS-1) and downstream PI3K activation. This creates a state where cells become less responsive to insulin even at normal or elevated insulin levels. The Svensson 1998 trial documented the lipolytic effects of MK-677 alongside the OGTT impairment, consistent with the FFA-mediated mechanism.[3]
Direct insulin signaling interference. Chronic GH elevation upregulates PTEN expression in hepatocytes, directly undermining insulin signaling pathways at the cellular level. This mechanism has been demonstrated in animal models and explains why GH-induced insulin resistance can persist even when FFA levels are experimentally controlled.
These mechanisms explain why the glucose effects of MK-677 are dose-dependent and population-dependent. Populations with already-elevated GH (young, lean individuals) may tolerate MK-677 differently than elderly subjects with low baseline GH, whose metabolic systems are recalibrated to operate at lower GH levels.
Ghrelin itself, independent of GH, also appears to modulate glucose metabolism. Heijboer et al. (2006) demonstrated in animal models that ghrelin differentially affects hepatic and peripheral insulin sensitivity, adding another layer to MK-677's metabolic effects beyond its GH-elevating properties.[4] Because MK-677 activates the ghrelin receptor (GHS-R1a), it engages both the GH-mediated and the direct ghrelin-mediated pathways affecting glucose metabolism. This dual mechanism distinguishes MK-677 from pure GH-releasing hormone (GHRH) analogs, which do not activate the ghrelin receptor.
This dual mechanism is relevant when comparing MK-677 to other GH secretagogues. Compounds like ipamorelin and hexarelin stimulate GH through different receptor profiles, and their metabolic side effect profiles may differ accordingly, though head-to-head glucose comparison data is limited.
The GH-deficient population: a different risk profile
Chapman et al. (1997) tested MK-677 in nine severely GH-deficient men (ages 17-34) at doses of 10 and 50 mg/day. This population had profoundly low GH levels (peak GH in response to insulin challenge: 1.2 +/- 1.5 micrograms/L). MK-677 successfully increased GH and IGF-1, with IGF-1 rising 52% at the 10 mg dose and 79% at the 50 mg dose.[5]
Fasting and postprandial insulin both increased after MK-677 treatment, as did postprandial glucose. The authors explicitly noted that "the clinical significance of these changes will need to be assessed in longer term studies."
This finding is particularly instructive. GH-deficient adults actually have increased insulin sensitivity at baseline (a well-documented paradox of GH deficiency). When MK-677 restored their GH levels toward normal, it also restored the normal degree of insulin resistance that accompanies adequate GH. The glucose shift was not necessarily pathological in this context; it was a reversion to physiological norms. But the practical implication remains: blood sugar goes up, and in patients who may have been managing metabolic conditions at their lower GH baseline, this shift could be clinically meaningful.
The GH response to MK-677 was positively correlated with baseline GH levels (r = 0.81, P = 0.009), meaning subjects with the most residual pituitary function responded best. This has implications for who might experience the greatest metabolic disruption: those with the strongest GH response may also see the largest glucose shifts.
Acromegaly: the natural experiment
The glucose effects of MK-677 are not theoretical concerns invented by cautious researchers. They are scaled-down versions of what occurs in acromegaly, a condition of chronic GH excess from pituitary tumors.
Patients with acromegaly develop insulin resistance at high rates. Up to 56% develop impaired glucose tolerance, and 15-38% develop frank diabetes. The same mechanisms are at work: excess GH drives hepatic gluconeogenesis, lipolysis, and direct insulin signaling impairment. Acromegaly effectively demonstrates what happens when GH stays elevated indefinitely without the self-limiting feedback that normal pulsatile secretion provides.
MK-677 does not produce acromegalic GH levels. The 25 mg dose increased mean 24-hour GH concentration by 97% in the Chapman 1996 trial, restoring elderly subjects' levels to the young-adult range rather than exceeding it.[1] But the direction of the effect is the same, and the question becomes whether sustained GH elevation at physiological-to-supraphysiological levels produces a clinically relevant fraction of acromegaly's metabolic consequences over years of use. The current evidence suggests it does, at least partially.
This is a core concern addressed in long-term GH peptide safety research, where the absence of multi-year data beyond the Nass trial leaves substantial uncertainty.
The 563-patient Alzheimer's trial: glucose data at scale
The largest MK-677 trial enrolled 563 patients with mild to moderate Alzheimer's disease, randomized to 25 mg/day or placebo for 12 months (Sevigny et al., 2008). The primary finding was negative: MK-677 did not slow AD progression despite successfully raising IGF-1 by 60.1% at 6 weeks and 72.9% at 12 months.[6]
The glucose data from this trial is less frequently discussed but is the largest dataset available. 416 patients completed treatment at 12 months. While the paper focused on cognitive endpoints (ADAS-Cog, CDR-sob, ADCS-ADL), the metabolic data confirmed the pattern seen in smaller trials: the same GH/IGF-1 elevation that failed to improve cognition came packaged with the same glucose-related effects.
This trial is important for the insulin resistance question because it provides two things the smaller studies cannot: statistical power and demographic diversity. With hundreds of participants spanning a wide age range and varying baseline metabolic profiles, the persistent glucose effect cannot be attributed to a few outliers or a narrow population selection. It persists at scale, across demographics, and at the standard 25 mg dose.
The anti-catabolic context: metabolic costs during caloric restriction
Murphy et al. (1998) demonstrated that MK-677 reverses diet-induced protein catabolism. Eight healthy volunteers under caloric restriction (18 kcal/kg/day) received MK-677 or placebo in a crossover design. Nitrogen balance shifted from -2.67 g/day to +0.31 g/day with MK-677, while placebo subjects remained catabolic at -1.48 g/day (P < 0.01). IGF-1 increased approximately 40%, and peak GH reached 55.9 micrograms/L after the first dose.[7]
This study did not specifically report adverse glucose findings, and the anti-catabolic effect was robust. But the GH and IGF-1 elevation documented in this trial would, based on every other MK-677 study, carry the same glucose implications. During caloric restriction, cells are typically in a state of enhanced insulin sensitivity (the body compensating for reduced energy intake). Introducing a GH secretagogue during this window pushes metabolism in two opposing directions simultaneously: preserving muscle protein while potentially impairing glucose disposal.
For the populations most likely to benefit from MK-677's anti-catabolic properties (post-surgical patients, critically ill individuals, elderly people losing muscle mass), metabolic fragility is often already present. The glucose trade-off is therefore most consequential precisely where the anabolic benefit is most needed.
Dose, duration, and reversibility
The glucose effects of MK-677 are dose-dependent. Copinschi et al. (1996) found that 5 mg produced lesser GH/IGF-1 responses than 25 mg in healthy young men over 7 days, while 25 mg significantly increased GH pulse frequency and IGF-1 levels in a dose-dependent manner.[8] Lower GH elevation logically implies lesser glucose disruption, though no trial has specifically compared glucose outcomes across MK-677 doses as a primary endpoint. The Copinschi study also confirmed that adrenocortical function (cortisol) was unaffected, meaning the glucose changes are specifically GH-mediated rather than stress-hormone-mediated.
The question of reversibility is partially answered by the available data. The Nass 2-year trial showed glucose effects persisting throughout treatment but did not report extended follow-up after discontinuation. The known pharmacology of GH-mediated insulin resistance supports reversibility: when GH levels normalize, hepatic glucose output decreases, FFA levels fall, and insulin sensitivity recovers. This is consistent with acromegaly data showing glucose metabolism improves after tumor removal normalizes GH levels.
Whether sustained use over many months creates lasting metabolic changes (beta-cell fatigue, progressive insulin resistance, or transition to frank diabetes) has not been tested in controlled trials. Case reports from clinical settings have documented new-onset diabetes in individuals using MK-677 in combination with other compounds, though isolating MK-677's contribution from confounders like concurrent SARM use, diet, and genetic predisposition is impossible from case data alone.
IGF-1 elevation: the other side of the metabolic equation
The relationship between IGF-1 elevation and glucose is more nuanced than the GH-glucose relationship. IGF-1 actually has insulin-sensitizing properties. It can enhance glucose uptake in skeletal muscle and suppress hepatic glucose output through mechanisms partially overlapping with insulin signaling.
This creates a paradox in MK-677 pharmacology: GH pushes glucose up while IGF-1 pushes it down. The net effect depends on the ratio and timing of these competing forces. In the early phase of MK-677 treatment, GH spikes acutely while IGF-1 takes days to weeks to rise (since it requires hepatic synthesis in response to GH). This temporal mismatch may explain why glucose disruption appears early in treatment.
Whether IGF-1's insulin-sensitizing effects partially offset GH's insulin-resistant effects during chronic MK-677 use is an open question. The Nass trial's persistent glucose elevation at 2 years suggests that if such compensation occurs, it is incomplete.
The theoretical risk of sustained IGF-1 elevation is a separate concern that adds another dimension to the benefit-risk analysis of long-term MK-677 use.
How MK-677 compares to GH injection
Exogenous GH injection also causes insulin resistance. The mechanism is identical. The difference is in the pattern of elevation: GH injections produce a supraphysiological spike followed by a return to baseline, while MK-677 enhances pulsatile GH secretion throughout the day, producing a more physiological pattern but a sustained elevation.
Whether the sustained-but-pulsatile pattern of MK-677 produces more or less insulin resistance than the spike-and-decline pattern of GH injection has not been directly compared. Both approaches elevate 24-hour mean GH levels, and both produce glucose effects. The theoretical advantage of MK-677's pulsatile pattern (preserving feedback regulation) may not translate to metabolic advantages.
AOD-9604, a modified GH fragment, was specifically designed to retain fat-metabolizing properties while eliminating the diabetogenic effects of full-length GH. Its existence as a research concept underscores that the glucose trade-off of GH-based interventions was recognized early enough to drive molecular engineering efforts to circumvent it.
Risk-Benefit
The MK-677 Trade-Off: What You Gain vs. What You Pay
Documented Benefits
IGF-1 restored to youthful levels (72.9% increase at 12 months)
Anti-catabolic: reversed diet-induced muscle loss
Increased fat-free mass (DEXA-confirmed)
Raised basal metabolic rate
Oral dosing (no injection needed)
Documented Metabolic Costs
Fasting glucose shifts toward pre-diabetic range
Impaired oral glucose tolerance (hidden by fasting tests)
HbA1c rises 0.2% over 12 months
Insulin sensitivity declines (QUICKI index)
Appetite increase (ghrelin receptor activation)
Both columns come from the same clinical trials, often measured in the same subjects during the same treatment period. The benefits and costs are inseparable — you cannot get the GH/IGF-1 elevation without the glucose effects.
Source: Nass (2008); Svensson (1998); Chapman (1996); Murphy (1998)
 View as image
View as imageSafety
CriticalConsistent blood sugar impairment across all trials
Concern
Every controlled MK-677 trial that measured glucose metabolism found impairment. Fasting glucose rises up to 26% (Chapman 1996), HbA1c increases 0.2% over 12 months (Nass 2008), and oral glucose tolerance tests show hidden impairment even when fasting glucose looks normal (Svensson 1998). This is not a rare side effect — it is a pharmacological certainty.
What the research says
The glucose effects are in the pre-diabetic range, not diabetic, for most healthy subjects studied. The impairment appears reversible based on GH pharmacology and acromegaly cure data. But no controlled trial has confirmed reversibility after prolonged MK-677 use.
Particularly relevant for: Highest risk: pre-diabetic individuals, family history of type 2 diabetes, metabolic syndrome, concurrent use of other insulin-resistant compounds
What to do
If considering MK-677, get a baseline OGTT (not just fasting glucose). Monitor HbA1c quarterly. Fasting glucose alone will miss the metabolic effects. Discuss with an endocrinologist if you have any metabolic risk factors.
Chapman (1996); Nass (2008); Svensson (1998); Sevigny (2008)
What the evidence does and does not show
The evidence clearly shows that MK-677 at 25 mg/day impairs glucose metabolism across multiple populations (healthy elderly, obese men, GH-deficient adults, Alzheimer's patients). The effect is consistent, dose-dependent, and mediated by the same mechanisms that drive GH-induced insulin resistance generally.
The evidence does not show that MK-677 at standard research doses causes diabetes in healthy individuals during the timeframes studied (up to 2 years). The glucose shifts documented are in the pre-diabetic range, not the diabetic range, for most participants. The distinction between "impaired glucose tolerance" and "diabetes" is meaningful, and conflating them misrepresents the data.
The evidence also cannot answer whether longer use, higher doses, or use in metabolically vulnerable populations (pre-diabetic, family history of type 2 diabetes, concurrent metabolic syndrome) tips the balance toward frank diabetes. The 2-year Nass trial is the longest controlled data available, and it enrolled healthy subjects. The real-world use pattern, which often involves longer durations in metabolically diverse populations, has no controlled data.
Sleep quality research on GH peptides including MK-677 adds another variable: sleep deprivation itself impairs insulin sensitivity, and MK-677's effects on sleep architecture may modulate its metabolic impact in ways not captured by the existing glucose-focused analyses. The ghrelin pathway that MK-677 activates also drives appetite increases, which in practice could compound the metabolic effects through increased caloric intake.
The Bottom Line
MK-677 reliably raises growth hormone and IGF-1, and it reliably raises blood sugar. Seven clinical trials across multiple populations show consistent glucose impairment: fasting glucose increases of 5-26%, impaired oral glucose tolerance, declining insulin sensitivity, and modest HbA1c rises. These effects are driven by the same counter-regulatory mechanisms that make all GH elevation insulin-resistant, compounded by direct ghrelin receptor effects on hepatic glucose metabolism. The changes are in the pre-diabetic range for most study participants and appear to reverse after discontinuation, but no controlled data exists beyond 2 years.
Sources & References
- 1RPEP-00357·Chapman, I M et al. (1996). “Oral MK-677 Restores Youthful Growth Hormone and IGF-1 Levels in Elderly Adults.” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 2RPEP-01392·Nass, Ralf et al. (2008). “MK-677 for 1 Year in Healthy Elderly: Increased Lean Mass But Also Fat and Insulin.” Annals of internal medicine.Study breakdown →PubMed →↩
- 3RPEP-00497·Svensson, J et al. (1998). “Oral MK-677 Increases Growth Hormone, Lean Mass, and Calorie Burning in Obese Men.” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 4RPEP-01146·Heijboer, A C et al. (2006). “Ghrelin Affects Insulin Sensitivity Differently in the Liver vs Muscles.” Diabetologia.Study breakdown →PubMed →↩
- 5RPEP-00400·Chapman, I M et al. (1997). “Oral MK-677 Boosts Growth Hormone in GH-Deficient Adults, Even Those Severely Deficient.” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 6RPEP-01418·Sevigny, J J et al. (2008). “MK-677 Does NOT Slow Alzheimer's Disease Progression Despite Raising GH and IGF-1.” Neurology.Study breakdown →PubMed →↩
- 7RPEP-00480·Murphy, M G et al. (1998). “MK-677 Pill Reverses Muscle Breakdown From Calorie Restriction.” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 8RPEP-00358·Copinschi, G et al. (1996). “Seven Days of Oral MK-677 Boosts 24-Hour Growth Hormone Profiles in Young Men.” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩