CJC-1295 DAC vs No DAC: Key Differences
CJC-1295 and IGF-1
8 days vs 30 min
The Drug Affinity Complex extends CJC-1295's half-life from approximately 30 minutes to 8 days by enabling albumin binding in the bloodstream.
Teichman et al., Journal of Clinical Endocrinology & Metabolism, 2006
Teichman et al., Journal of Clinical Endocrinology & Metabolism, 2006
If you only read one thing
CJC-1295 comes in two versions built around the same peptide. The 'with DAC' version has a tiny chemical attachment that grabs onto a blood protein called albumin. That attachment stretches how long it lasts in the body — from about 30 minutes to roughly 8 days. So one injection can keep growth hormone elevated for nearly a week, versus just a couple of hours for the no-DAC version. Neither is FDA approved, and both are banned in competitive sport.
CJC-1295 exists in two forms that share the same core peptide but behave very differently in the body. The version with DAC (Drug Affinity Complex) binds to albumin in the blood, extending its half-life to approximately 8 days and allowing once-weekly dosing. The version without DAC, commonly called Mod GRF 1-29 (Modified Growth Hormone Releasing Factor, amino acids 1-29), has a half-life of about 30 minutes and requires daily or twice-daily injections.[1] For the foundational data on what CJC-1295 does to growth hormone and IGF-1 levels, see the pillar article on CJC-1295 and IGF-1 elevation.
Both versions stimulate the pituitary gland to release growth hormone by mimicking GHRH (growth hormone releasing hormone). The difference in half-life creates fundamentally different pharmacological profiles: continuous versus pulsatile GH stimulation. This distinction matters for efficacy, side effects, and how closely the peptide mimics natural physiology.
Key Takeaways
- CJC-1295 comes in two versions built on the same core peptide — the only real difference is how long it stays active in your body.
- The "with DAC" version lasts about 8 days from one shot. The "without DAC" version is gone in about 30 minutes.
- DAC works by clipping onto a blood protein called albumin that rides around your bloodstream for weeks, taking the peptide along for the ride.
- One shot of the DAC version raises your growth hormone 2 to 10 times above normal — and keeps it elevated for nearly a week.
- Your body normally pulses growth hormone during deep sleep. DAC adds a steady background of GH on top of those pulses — not quite natural physiology.
- The two versions are not interchangeable. Switching from daily no-DAC to weekly DAC changes a short spike into a days-long exposure.
- Neither version is FDA-approved, and no long-term safety data exists for either — the research stops at a few weeks.
Side-by-Side
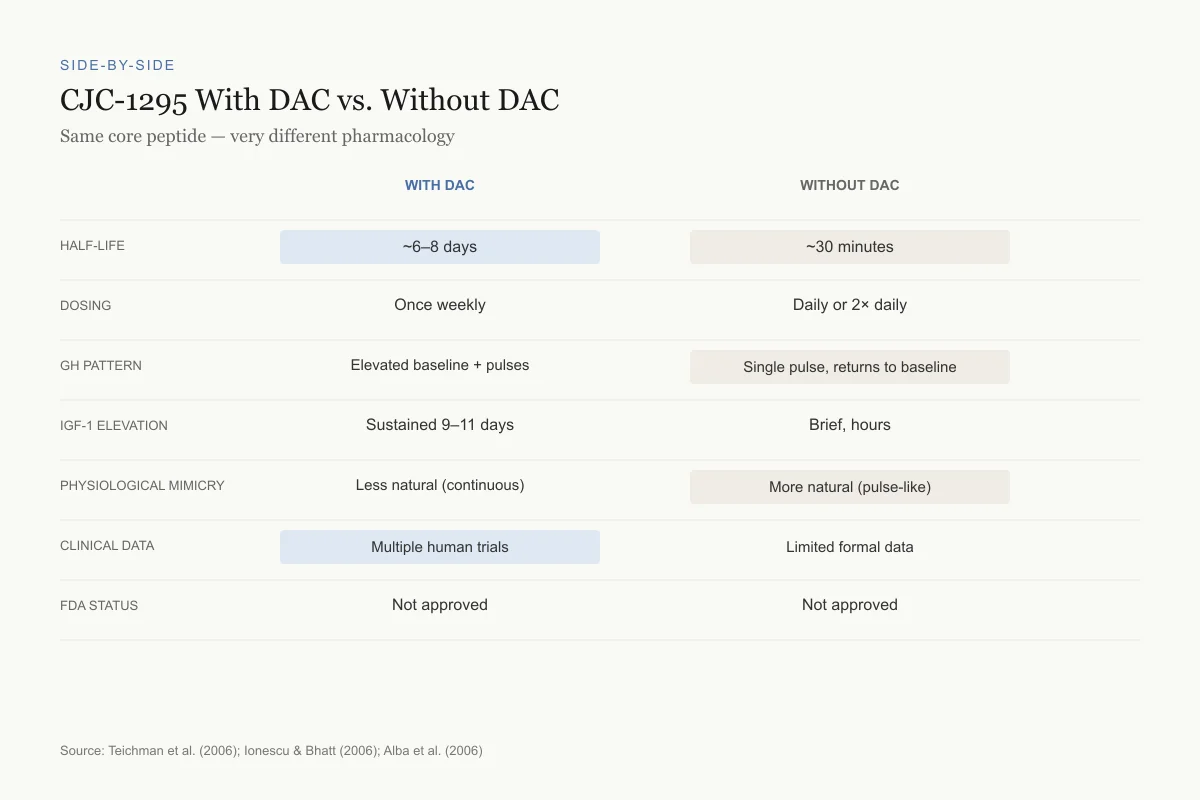
CJC-1295 With DAC vs. Without DAC
Same core peptide — very different pharmacology
Source: Teichman et al. (2006), Ionescu & Bhatt (2006), Alba et al. (2006)
 View as image
View as imageThe core peptide: what both versions share
Both CJC-1295 variants are based on a modified version of GHRH(1-29), the first 29 amino acids of the 44-amino acid growth hormone releasing hormone. Natural GHRH is rapidly degraded by dipeptidyl peptidase IV (DPP-IV) and other enzymes, giving it a plasma half-life of under 10 minutes. CJC-1295 incorporates four amino acid substitutions that protect it from DPP-IV cleavage, extending the unmodified peptide's half-life to approximately 30 minutes.[1]
The substitutions (at positions 2, 8, 15, and 27) were selected to resist enzymatic degradation while preserving receptor binding affinity. This core modified peptide is what is sold as "CJC-1295 without DAC" or "Mod GRF 1-29." It retains the basic GHRH receptor binding activity but with improved metabolic stability compared to native GHRH. For a detailed look at how CJC-1295 stimulates growth hormone at the molecular level, see the dedicated article. The Mod GRF 1-29 article covers the without-DAC version specifically.
What DAC does: the albumin binding strategy
The Drug Affinity Complex is a maleimidopropionic acid (MPA) linker attached to the peptide's lysine residue. After subcutaneous injection, the MPA group reacts covalently with albumin's cysteine-34 residue in plasma. Once bound to albumin (the most abundant protein in blood, with a half-life of approximately 19 days), the peptide is protected from renal clearance and enzymatic degradation, extending its functional half-life to 6-8 days.[1]
This albumin-binding approach is not unique to CJC-1295. It is a strategy now used in several approved drugs (semaglutide, for example, achieves its once-weekly dosing through fatty acid-mediated albumin binding). CJC-1295 with DAC was among the first peptides to use covalent albumin conjugation for half-life extension.
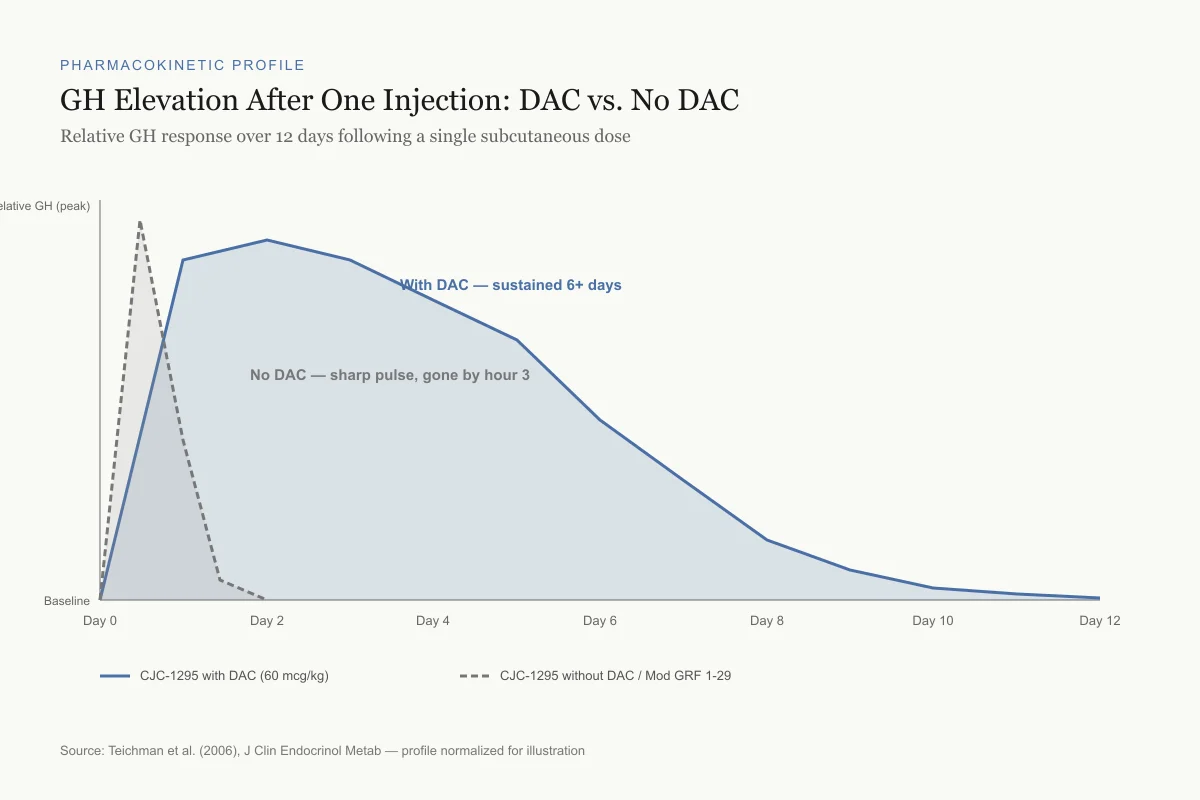
The pharmacological consequence is that a single injection of CJC-1295 with DAC produces sustained GH elevation for days rather than the hours-long pulse from the non-DAC version. Teichman et al. (2006) demonstrated this in healthy adults: after a single subcutaneous injection, mean GH levels rose 2-10 fold above baseline and remained elevated for up to 6 days. IGF-1 levels rose 1.5-3 fold and stayed elevated for 9-11 days at the highest doses.[1]
Mechanism
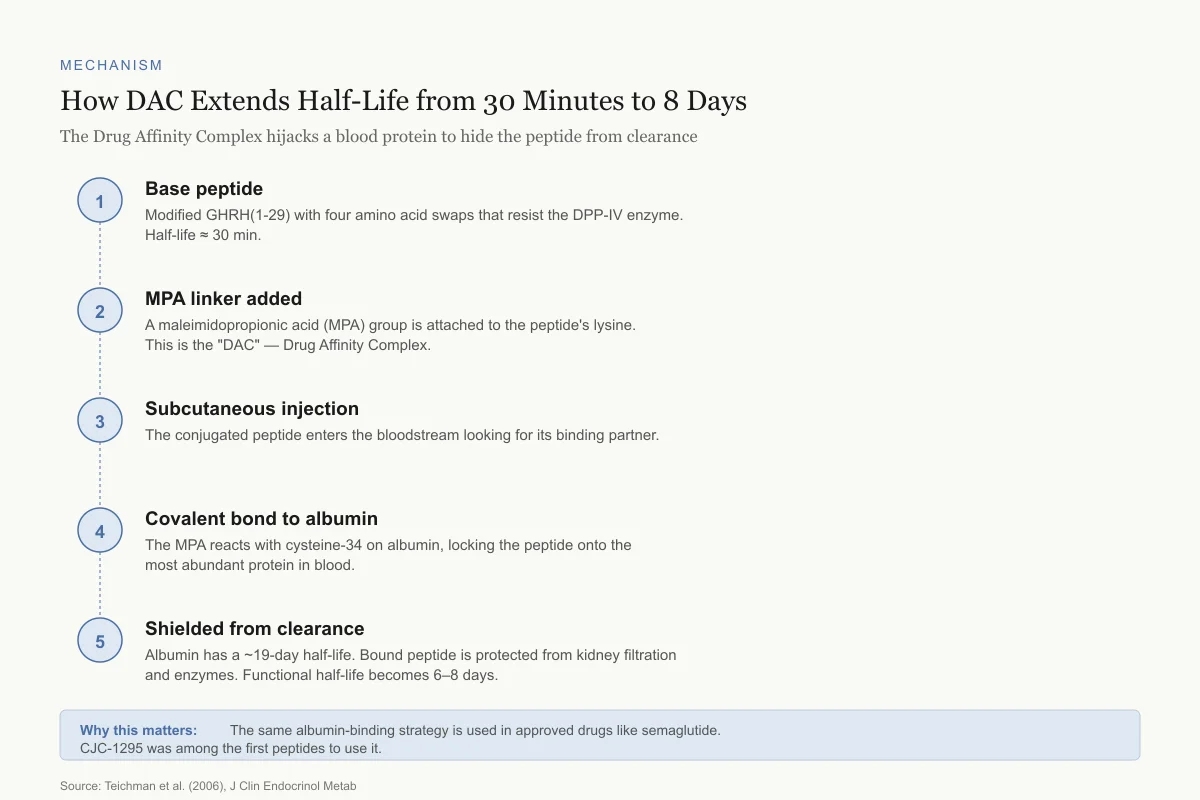
How DAC Extends Half-Life from 30 Minutes to 8 Days
The Drug Affinity Complex hijacks a blood protein to hide the peptide from clearance
Base peptide
Modified GHRH(1-29) with four amino acid swaps that resist the DPP-IV enzyme. Half-life ≈ 30 min.
MPA linker added
A maleimidopropionic acid (MPA) group is attached to the peptide's lysine. This is the "DAC" — Drug Affinity Complex.
Subcutaneous injection
The conjugated peptide enters the bloodstream looking for its binding partner.
Covalent bond to albumin
The MPA reacts with cysteine-34 on albumin, locking the peptide onto the most abundant protein in blood.
Shielded from clearance
Albumin has a ~19-day half-life. Bound peptide is protected from kidney filtration and enzymes. Functional half-life becomes 6–8 days.
Why this matters: The same albumin-binding strategy is used in approved drugs like semaglutide. CJC-1295 was among the first peptides to use it.
Source: Teichman et al. (2006), J Clin Endocrinol Metab
 View as image
View as imagePharmacokinetic Profile
GH Elevation After One Injection: DAC vs. No DAC
Relative GH response over 12 days following a single subcutaneous dose
Source: Teichman et al. (2006), J Clin Endocrinol Metab — profile normalized for illustration
 View as image
View as imagePulsatile vs continuous: why it matters
Growth hormone is normally secreted in pulses, with the largest surge occurring during the first phase of deep sleep. This pulsatile pattern is biologically significant: tissues respond differently to GH pulses than to sustained GH exposure. In animal models, pulsatile GH administration preferentially activates growth-promoting pathways, while continuous GH favors fat metabolism and can lead to receptor desensitization.
The concern with CJC-1295 with DAC was that its sustained activity might flatten the natural GH pulse pattern, creating a constant GH "drip" rather than physiological surges. Ionescu and Bhatt (2006) directly investigated this by measuring GH secretory dynamics during continuous CJC-1295 DAC exposure.[2] Their findings were reassuring: pulsatile GH secretion persisted even during sustained CJC-1295 stimulation. The amplitude and frequency of GH pulses were increased rather than abolished. This means the pituitary's intrinsic pulse-generating mechanism (the interplay between GHRH and somatostatin) was not overridden by the exogenous peptide.
This does not mean the pattern was identical to natural physiology. The baseline between pulses was elevated, and the overall 24-hour GH exposure was substantially higher than normal. Whether this elevated baseline carries long-term consequences (joint pain, insulin resistance, soft tissue edema, or carpal tunnel syndrome, all of which are known side effects of exogenous GH administration) has not been studied beyond the short-term trial data. The absence of long-term safety data is a significant limitation for both versions.
The without-DAC version, by contrast, produces a single GH pulse that resolves within 2-3 hours. This mimics a natural GHRH release more closely, though administered exogenously at fixed times rather than in response to physiological cues. The article on how growth hormone peptides affect sleep covers why timing of GH pulses matters for sleep architecture.
The clinical trial data
CJC-1295 with DAC
The primary published clinical data comes from ConjuChem Biotechnologies, the Canadian company that developed CJC-1295.
Teichman et al. (2006) conducted a dose-escalation study in healthy adults. Single subcutaneous doses of 30, 60, or 125 micrograms per kilogram produced dose-dependent increases in GH and IGF-1. The 60 mcg/kg dose produced mean GH increases of approximately 2-fold over baseline persisting for 6 days, with IGF-1 remaining elevated 1.5-fold for 9-11 days.[1] No serious adverse events were reported at any dose level.
Alba et al. (2006) tested once-daily CJC-1295 DAC in adults with growth hormone deficiency. Over 7 days, CJC-1295 DAC normalized GH and IGF-1 levels comparably to daily recombinant GH injections.[3] This was a proof-of-concept that a GHRH analog could replace GH replacement therapy, though longer-term studies were not completed.
Sackmann-Sala et al. (2009) examined the downstream effects of CJC-1295 DAC-induced GH/IGF-1 axis activation on the serum proteome, identifying changes in proteins related to lipid metabolism, complement activation, and coagulation pathways.[4] This systems-level analysis suggested that CJC-1295 DAC produced broad metabolic effects consistent with GH replacement.
CJC-1295 without DAC (Mod GRF 1-29)
Published clinical trial data specifically on the without-DAC version is sparse. The Mod GRF 1-29 formulation was not developed through a formal drug development program. Its pharmacological properties are inferred from the broader GHRH(1-29) analog literature and from the core peptide's behavior before DAC conjugation.
Tesamorelin, the only FDA-approved GHRH analog, provides the closest pharmacological reference. It is a modified GHRH(1-44) (not 1-29) with different amino acid substitutions and no DAC, approved for HIV-associated lipodystrophy. The Badran 2026 study extended tesamorelin's evidence base to body composition, hepatic fat, and metabolic outcomes.[5] Makimura et al. (2014) demonstrated that tesamorelin improved phosphocreatine recovery in obese subjects with reduced GH, providing functional evidence that GHRH stimulation translates to measurable metabolic improvements.[6]
The gray market reality
Neither CJC-1295 formulation is approved for human therapeutic use. Both are widely available through peptide suppliers as "research chemicals." Van et al. (2016) conducted a netnographic study of female users of synthetic CJC-1295, documenting use patterns, motivations, and self-reported experiences in online communities.[7] Users reported improvements in sleep quality, body composition, skin appearance, and recovery from exercise. Self-reported side effects included water retention, joint stiffness, and skin flushing.
Henninge et al. (2010) identified CJC-1295 in an unknown pharmaceutical preparation seized by authorities, highlighting the compound's presence in the unregulated supplement and performance-enhancement market.[8] Timms et al. (2019) developed analytical methods for detecting CJC-1295 in equine plasma, confirming its use in veterinary doping contexts.[9]
The distinction between DAC and no-DAC versions is particularly important in the gray market because the pharmacokinetic profiles are so different. A user who switches from daily Mod GRF 1-29 to weekly CJC-1295 DAC without understanding the difference in GH exposure pattern could experience unexpected side effects from the sustained elevation.
Product quality is also a concern. Unlike FDA-approved pharmaceuticals, research peptides are not subject to Good Manufacturing Practice standards. Purity, sterility, and actual peptide content vary between suppliers. The structural analysis work by Jeanne et al. (2018) on GHRH peptide analogs highlighted how subtle structural changes can alter receptor agonism versus antagonism, underscoring that manufacturing inconsistencies in complex peptides can produce unpredictable biological effects. Henninge et al. found that seized CJC-1295 preparations sometimes contained unexpected compounds, further emphasizing the risks of unregulated sourcing.
The anti-doping implications are also significant. CJC-1295 in both forms is prohibited by the World Anti-Doping Agency under the category of peptide hormones and growth factors. Timms et al. (2019) developed LC-MS/MS methods capable of detecting CJC-1295 in both equine and human biological matrices, and the long half-life of the DAC version makes it detectable for weeks after a single injection.[9]
Safety
ModerateSwitching between DAC and no-DAC is not a trivial change
Concern
A user who steps from daily Mod GRF 1-29 to weekly CJC-1295 DAC changes from short pulses of GH to a sustained, days-long elevation. That's a completely different exposure profile — not a simple dose conversion. Unexpected water retention, joint stiffness, or insulin resistance can follow.
What the research says
The two versions are not interchangeable. Short-term trial data covers weeks, not months, and no long-term safety data exists for either form.
Particularly relevant for: Anyone switching between DAC and no-DAC formulations
What to do
Do not treat the two versions as substitutes. If considering either, work with a clinician, and understand that the research behind them is limited to short-term trials from ~2006.
Teichman 2006 pharmacodynamics; general GH safety literature
Comparing the two versions directly
| Feature | CJC-1295 with DAC | CJC-1295 without DAC (Mod GRF 1-29) |
|---|---|---|
| Half-life | ~6-8 days | ~30 minutes |
| Dosing frequency | Once weekly | Daily or twice daily |
| GH pattern | Elevated baseline with preserved pulses | Single pulse, returns to baseline |
| IGF-1 elevation | Sustained 9-11 days | Brief, returns within hours |
| Physiological mimicry | Less natural (continuous stimulation) | More natural (pulse-like) |
| Published human data | Multiple clinical trials | Limited formal data |
| FDA status | Not approved | Not approved |
The choice between versions involves a trade-off: convenience versus physiological fidelity. The DAC version requires fewer injections and produces sustained GH/IGF-1 elevation, but the continuous stimulation pattern is less physiological. The no-DAC version requires more frequent dosing but produces discrete GH pulses that more closely resemble natural secretion.
For related peptides that stimulate GH through the ghrelin receptor rather than the GHRH receptor, see articles on ipamorelin, hexarelin, and MK-677. The sermorelin article covers the first-generation GHRH analog that preceded CJC-1295.
The Bottom Line
CJC-1295 with and without DAC are the same core GHRH analog peptide with radically different pharmacokinetics. The DAC version binds albumin and stays active for 6-8 days, producing sustained GH and IGF-1 elevation. The no-DAC version (Mod GRF 1-29) lasts about 30 minutes and produces a single GH pulse. Clinical trials of the DAC version showed significant GH and IGF-1 elevation with preserved pulsatile patterns. Neither version is FDA-approved; both are used off-label through gray market sources. The key difference is not potency but duration: continuous versus pulsatile GH stimulation, each with distinct physiological implications.
Sources & References
- 1RPEP-01190·Teichman, Sam L et al. (2006). “CJC-1295 in Humans: Single Injection Sustains GH and IGF-1 Elevation for Up to 2 Weeks.” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 2RPEP-01148·Ionescu, Madalina et al. (2006). “CJC-1295 Clinical Trial: Single-Dose Injection Maintains Pulsatile GH for Days in Humans.” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 3RPEP-01113·Alba, Maria et al. (2006). “CJC-1295: A Long-Acting GHRH Analog That Normalizes GH With Once-Daily Dosing.” American journal of physiology. Endocrinology and metabolism.Study breakdown →PubMed →↩
- 4RPEP-01542·Sackmann-Sala, Lucila et al. (2009). “CJC-1295 Changes Blood Protein Profiles: GH/IGF-1 Axis Activation Has Systemic Effects.” Growth hormone & IGF research : official journal of the Growth Hormone Research Society and the International IGF Research Society.Study breakdown →PubMed →↩
- 5RPEP-14810·Badran, Ahmed Samy et al. (2026). “Tesamorelin Reduces Belly Fat and Liver Fat in HIV Patients: Meta-Analysis of Trials.” Obesity research & clinical practice.Study breakdown →PubMed →↩
- 6RPEP-02435·Makimura, Hideo et al. (2014). “GHRH Peptide Tesamorelin Improves Mitochondrial Function in Obese Adults With Low Growth Hormone.” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 7RPEP-03144·Van Hout, Marie Claire et al. (2016). “What Women Say About Using CJC-1295: An Online Forum Study.” Substance use & misuse.Study breakdown →PubMed →↩
- 8RPEP-01626·Henninge, John et al. (2010). “CJC-1295 Identified in an Unknown Pharmaceutical Preparation: Forensic Analysis.” Drug testing and analysis.Study breakdown →PubMed →↩
- 9RPEP-04517·Timms, Mark et al. (2019). “New Test Detects the 'Invisible' Performance Peptide CJC-1295 in Horse Blood.” Drug testing and analysis.Study breakdown →PubMed →↩