Which 14 Peptides Are Moving Back to Category 1?
BPC-157 and the FDA
14 of 19 Peptides Returning
In February 2026, HHS Secretary Robert F. Kennedy Jr. announced that 14 of 19 peptides placed on the FDA's Category 2 restricted list in September 2023 will move back to Category 1, restoring legal compounding access.
RFK Jr., Joe Rogan Experience #2461, February 27, 2026
RFK Jr., Joe Rogan Experience #2461, February 27, 2026
If you only read one thing
In late 2023, the FDA blocked pharmacies from making 19 peptides including BPC-157, TB-500, and others. In February 2026, the government announced 14 of those 19 are coming back. Five stay restricted (Melanotan II, MK-677, Dihexa, PEG-MGF, LL-37) because of specific safety concerns. But 'coming back' means pharmacies can compound them with a doctor's prescription — it does NOT mean FDA-approved, over-the-counter, or covered by insurance. The formal paperwork isn't done yet.
On September 29, 2023, the FDA added 19 peptides to its Category 2 bulk drug substance list, blocking compounding pharmacies from preparing them for patients. On February 27, 2026, HHS Secretary Robert F. Kennedy Jr. announced on the Joe Rogan Experience that approximately 14 of those 19 peptides would move back to Category 1. For the full regulatory background on how the Category system works and why BPC-157 was restricted, see BPC-157 and the FDA: Category 2 Explained.
The announcement triggered immediate questions: which 14 peptides are coming back? Which 5 are staying restricted? And what does "Category 1" actually allow? This article covers every peptide on both lists, the research basis behind each one, and the regulatory steps that remain before compounding resumes.
Key Takeaways
- In February 2026, the government announced 14 of the 19 restricted peptides are coming back.
- "Coming back" means pharmacies can make them with a prescription — not that they're FDA-approved or over-the-counter.
- Not all 14 are equally researched — Thymosin Alpha-1 is used in 30+ countries, KPV has under five studies.
- The returning list includes BPC-157, TB-500, Ipamorelin, CJC-1295, Thymosin Alpha-1, Semax, Selank, and epitalon.
- Five peptides stay restricted: Melanotan II, MK-677, Dihexa, PEG-MGF, LL-37 — each flagged for safety concerns.
- The FDA hasn't published the formal reclassification paperwork yet, so the change isn't live.
- MK-677 technically isn't a peptide at all — it's grouped with them because it hits the same ghrelin receptor.
Regulatory Timeline
From Ban to Reclassification: Key Dates
FDA blocks compounding pharmacies from preparing these substances
AOD-9604, CJC-1295, Ipamorelin, Thymosin Alpha-1, Selank — nominators withdrew
Ipamorelin, Ibutamoren, and Kisspeptin-10 reviewed by advisory committee
AOD-9604, CJC-1295, and Thymosin Alpha-1 reviewed
14 of 19 peptides to return to Category 1 — announced on Joe Rogan Experience
Federal Register notice, public comment period, updated interim guidance — process ongoing
Source: FDA Federal Register; PCAC meeting records; RFK Jr. announcement
 View as image
View as imageReclassification Status
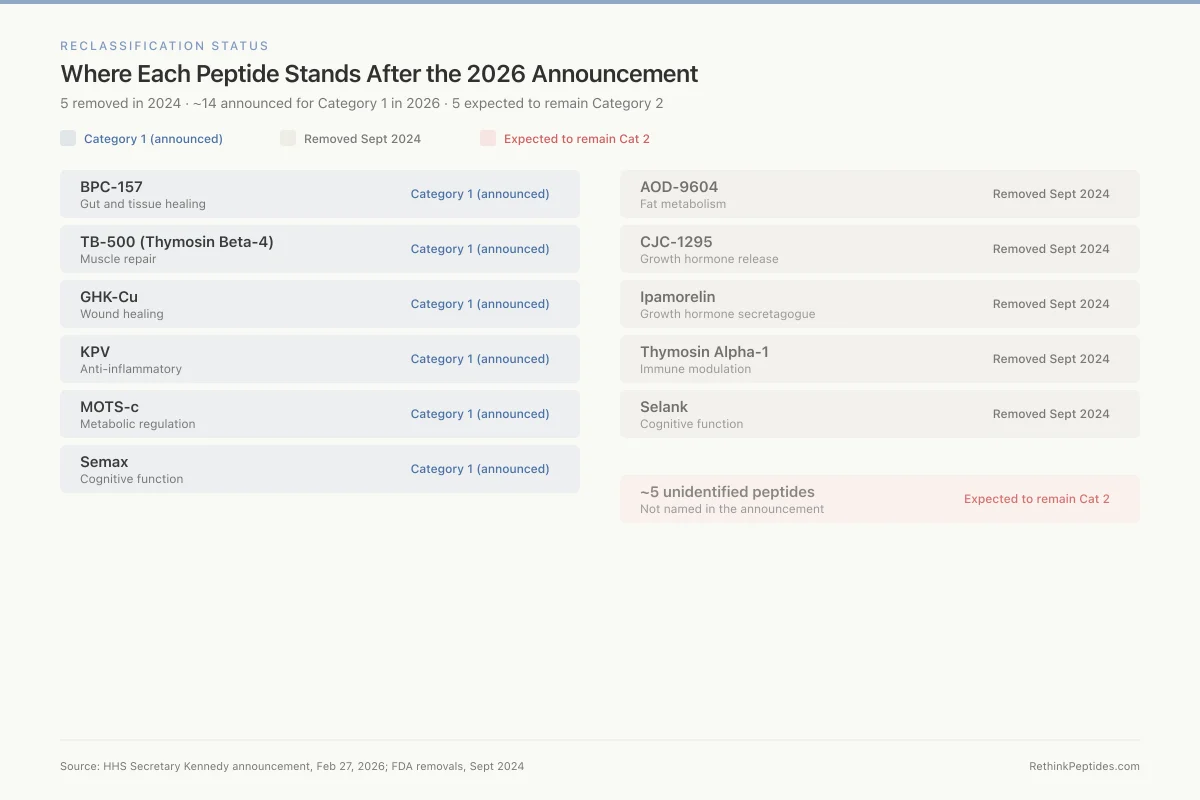
Where Each Peptide Stands After the 2026 Announcement
5 removed in 2024 · ~14 announced for Category 1 in 2026 · 5 expected to remain Category 2
BPC-157
Gut and tissue healing
TB-500 (Thymosin Beta-4)
Muscle repair
GHK-Cu
Wound healing
KPV
Anti-inflammatory
MOTS-c
Metabolic regulation
Semax
Cognitive function
AOD-9604
Fat metabolism
CJC-1295
Growth hormone release
Ipamorelin
Growth hormone secretagogue
Thymosin Alpha-1
Immune modulation
Selank
Cognitive function
~5 unidentified peptides
Not named in the announcement
Source: HHS Secretary Kennedy announcement, Feb 27, 2026; FDA removals, Sept 2024
 View as image
View as imageThe 14 Peptides Expected to Return
The following 14 peptides are widely expected to move from Category 2 back to Category 1 based on the February 2026 announcement. No official FDA list has been published. These identifications come from the announcement itself, subsequent reporting, and the September 2024 precedent where the FDA removed five of these peptides from Category 2 and referred them to the Pharmacy Compounding Advisory Committee (PCAC).
Tissue Repair and Recovery
BPC-157 (Body Protection Compound-157) is a synthetic 15-amino-acid peptide derived from human gastric juice. It is the most widely studied peptide on this list. Sikiric described its pharmacological profile in 1999, documenting effects on gastrointestinal mucosal integrity, wound healing, and interactions with the nitric oxide and dopamine systems.[1] Over 149 studies exist in the research database. The critical limitation: the overwhelming majority of data comes from animal models, with approximately 30 total humans studied in published trials. For a full analysis, see BPC-157 and the FDA: Category 2 Explained. For the current legal picture, see BPC-157 Legal Status in 2026.
TB-500 (Thymosin Beta-4 Fragment) is a synthetic fragment of Thymosin Beta-4, a 43-amino-acid protein involved in cell migration and wound healing. Maar et al. (2025) demonstrated that TB-500 modulates cardiac remodeling after myocardial infarction in mice through regulation of ROCK1 expression, with systemic injection increasing miR-139-5p expression in heart tissue.[2] Research spans 47 studies in the database, concentrated on wound healing, cardiac repair, and muscle recovery. Human clinical data remains sparse. For compounding status details, see TB-500, CJC-1295, Ipamorelin: Legal Compounding Status After Reclassification.
Growth Hormone Secretagogues
Ipamorelin is a pentapeptide that stimulates growth hormone (GH) release. Raun et al. (1998) characterized it as the first selective GH secretagogue, demonstrating that it released GH with potency similar to GHRP-6 but did not increase ACTH, cortisol, prolactin, or FSH even at doses 200-fold higher than effective GH-releasing doses.[3] This selectivity profile distinguishes it from older secretagogues. The FDA removed Ipamorelin from Category 2 in September 2024 and the PCAC reviewed it on October 29, 2024.
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH) with a drug affinity complex that extends its half-life. It is frequently combined with Ipamorelin in clinical protocols. Like Ipamorelin, CJC-1295 was removed from Category 2 in September 2024 and underwent PCAC review on December 4, 2024. Combined compounding status is covered in TB-500, CJC-1295, Ipamorelin: Legal Compounding Status After Reclassification.
AOD-9604 is a modified fragment (amino acids 177-191) of human growth hormone, originally developed as an anti-obesity agent. Wilding (2004) reviewed its metabolic profile, noting that phase IIa trials were underway by 2002.[4] AOD-9604 was designed to retain the lipolytic (fat-burning) properties of GH without the diabetogenic or growth-promoting effects. It was among the five peptides removed from Category 2 in September 2024 and reviewed by PCAC on December 4, 2024. The database contains 5 studies. For the broader context of AOD-9604 research, see AOD-9604: The Growth Hormone Fragment for Fat Metabolism.
Immune Modulation
Thymosin Alpha-1 is a 28-amino-acid peptide originally isolated from the thymus gland. It is the most clinically validated peptide on this list. Iino et al. (2005) published a randomized clinical trial in Japanese chronic hepatitis B patients showing HBeAg seroconversion and HBV DNA reduction with acceptable safety.[5] Thymosin Alpha-1 is approved in over 30 countries under the brand name Zadaxin for hepatitis B and as an immune adjuvant. The FDA removed it from Category 2 in September 2024 and the PCAC reviewed it December 4, 2024. For a dedicated analysis, see Thymosin Alpha-1 Reclassification: Restoring Access to an Immune Peptide.
KPV (Lys-Pro-Val) is a tripeptide fragment of alpha-melanocyte-stimulating hormone (alpha-MSH) with anti-inflammatory properties. Research is limited, with only 3 studies in the database. The evidence base for KPV is substantially thinner than for BPC-157 or Thymosin Alpha-1, and the strength of the reclassification case rests more on the peptide's structural simplicity and low safety signal than on robust clinical data.
Neuropeptides
Selank is a synthetic heptapeptide analog of the immunomodulatory peptide tuftsin. Vyunova et al. (2018) reviewed its molecular mechanisms as a peptide-based anxiolytic, describing its effects on GABA receptor expression and neurotrophic factor modulation.[6] Selank is approved in Russia as an anxiolytic but has no regulatory approval in the United States or European Union. It was among the five peptides removed from Category 2 in September 2024.
Semax is a synthetic analog of ACTH(4-10), a fragment of adrenocorticotropic hormone. Romanova et al. (2006) demonstrated neuroprotective and antiamnesic effects in a rat model of focal cerebral ischemia, with intranasal administration decreasing cortical infarction volume over 6 days.[7] Like Selank, Semax is approved in Russia for neurological applications but has no US or EU approval. The database contains 18 studies.
Anti-Aging and Longevity
Epitalon (Epithalon) is a synthetic tetrapeptide (Ala-Glu-Asp-Gly) modeled on the naturally occurring pineal peptide epithalamin. Khavinson et al. (2003) demonstrated that Epitalon induced telomerase catalytic subunit (hTERT) expression and telomere elongation in telomerase-negative human fetal fibroblasts.[8] The research is concentrated in a small number of studies, predominantly from Russian laboratories. For the pineal connection, see Epithalon and Melatonin: The Pineal Gland Connection.
GHK-Cu (Copper Tripeptide) is a naturally occurring tripeptide (Gly-His-Lys) that binds copper and is found in human plasma, saliva, and urine. Ogorek et al. (2025) reviewed GHK-Cu's skin permeation challenges, noting that the tripeptide's hydrophilic nature limits its ability to cross the lipophilic stratum corneum barrier, which has driven research into liposomal delivery systems.[9] GHK-Cu is primarily studied for wound healing and skin rejuvenation. Note that the Category 2 designation applied specifically to injectable routes of administration. For more detail, see GHK-Cu: The Copper Peptide That Modulates Over 4,000 Genes.
MOTS-C is a mitochondrial-derived peptide encoded by the 12S rRNA gene. Gudiksen et al. (2026) demonstrated that MOTS-C improved intrinsic muscle mitochondrial bioenergetic capacity and energy production efficiency through a PGC-1-alpha/AMPK-dependent mechanism, with potential therapeutic applications for aging and metabolic disease.[10] The database contains 14 studies. MOTS-C is among the newer peptides on the Category 2 list, with most published research dating from 2015 onward.
Other Returning Peptides
Emideltide (DSIP / Delta Sleep-Inducing Peptide) is a nonapeptide first isolated from rabbit cerebral venous blood during electrically induced sleep. Khvatova et al. (2003) studied its effects on mitochondrial respiration and stress protection, demonstrating protective effects against experimental hypoxia in rat brain mitochondria.[11] Research interest in DSIP has been intermittent since the 1970s, with the database containing 14 studies. For more detail, see DSIP for Insomnia: What the Limited Research Shows.
Kisspeptin-10 is a truncated form of the neuropeptide kisspeptin, which plays a central role in reproductive hormone regulation. Rackova (2025) reviewed how KNDy neurons (kisspeptin, neurokinin B, dynorphin) integrate hormonal feedback to control GnRH pulse frequency, positioning kisspeptin as a potential therapeutic target for reproductive disorders.[12] Kisspeptin-10 was reviewed by the PCAC on October 29, 2024. The database contains 28 studies.
The 5 Peptides Expected to Remain Restricted
Five of the original 19 Category 2 peptides are expected to remain restricted. The common thread: each one carries a specific safety concern that distinguishes it from the returning 14.
Melanotan II is a synthetic analog of alpha-MSH that stimulates melanogenesis (skin darkening) and has aphrodisiac effects. Case reports have documented cardiovascular events, priapism, and concern about melanoma promotion in predisposed individuals. It is the peptide most commonly cited as justifying continued restriction. For broader context on melanocortin peptide safety concerns, see The Peptide Craze Critique: What Eric Topol Got Right (and Wrong).
Ibutamoren Mesylate (MK-677) is technically not a peptide but a non-peptide ghrelin receptor agonist that stimulates GH release. It elevates cortisol and prolactin, an effect profile that distinguishes it from the more selective Ipamorelin. The PCAC reviewed Ibutamoren on October 29, 2024. It has 19 studies in the database.
Dihexa Acetate is a hexapeptide analog of angiotensin IV promoted for cognitive enhancement. The database contains only 1 study. Claims about its potency (sometimes described as "10 million times more potent than BDNF") derive from a single 2013 paper and have not been independently replicated. The limited evidence base and the outsized nature of the claims contributed to its restriction. For context on those claims, see Dihexa's Potency Claims: What the Original Research Actually Says.
PEG-MGF (Pegylated Mechano Growth Factor) is a PEGylated variant of a splice variant of IGF-1. PEGylation raises immunogenicity concerns, and the peptide occupies a regulatory gray area between traditional peptides and modified biologics. Research is limited.
Cathelicidin LL-37 is a 37-amino-acid antimicrobial peptide that is part of the innate immune system. Despite having the largest evidence base of any peptide on the restricted list (over 200 studies in the database), LL-37 presents unique safety challenges for compounding: it has complex immunomodulatory effects at higher concentrations, and there is limited human data on exogenous administration. The peptide's endogenous role in immune defense is well-documented, but that does not translate to a clear safety profile for compounded injectable use.
What Had Already Changed Before the Announcement
The February 2026 announcement did not arrive in a vacuum. The FDA had already taken action on five of the nineteen peptides in September 2024.
On September 20, 2024, the FDA announced that AOD-9604, CJC-1295, Ipamorelin Acetate, Thymosin Alpha-1, and Selank Acetate were being removed from Category 2. The mechanism: the original nominators who had petitioned the FDA to restrict these substances withdrew their nominations. The removal became effective September 27, 2024.
The FDA then referred these substances to the PCAC for formal review. Ipamorelin, Ibutamoren, and Kisspeptin-10 were reviewed at the PCAC meeting on October 29, 2024. AOD-9604, CJC-1295, and Thymosin Alpha-1 were reviewed on December 4, 2024. PCAC review is the first step toward formal Category 1 placement, but it does not automatically restore compounding access. Each substance still requires a notice-and-comment period before final determination.
For the legal and regulatory pressure that contributed to these changes, see The Legal Challenge That Forced FDA's Hand on Peptide Restrictions.
Safety
Moderate5 peptides staying restricted for specific safety reasons
Concern
Melanotan II (cardiovascular events, melanoma risk), MK-677 (cortisol/prolactin elevation), Dihexa (1 published study, unverified 'million times more potent' claims), PEG-MGF (immunogenicity from PEGylation), and LL-37 (complex immunomodulatory effects at injection doses). Each has a distinct safety profile that kept it restricted.
What the research says
The 14 returning peptides were judged to have lower safety risk profiles. But 'lower risk' is relative — Category 1 is not the same as safe. These are still unapproved substances without completed clinical trials.
Particularly relevant for: Anyone considering using compounded peptides
What to do
Even after reclassification, work with a physician who understands peptide pharmacology. Compounded products vary in quality between pharmacies. Category 1 is permission to compound, not a safety endorsement.
FDA Category 2 guidance; PCAC meeting records; published safety data
What Category 1 Does and Does Not Allow
Category 1 status means the FDA does not intend to take enforcement action against pharmacies that compound with the substance, provided the pharmacy meets all other conditions under Section 503A or 503B of the FD&C Act. In practice, this allows licensed compounding pharmacies to prepare patient-specific prescriptions.
Category 1 does not mean:
- FDA approval. None of these 14 peptides has completed a New Drug Application. FDA approval requires controlled clinical trials demonstrating safety and efficacy for a specific indication. Category 1 bypasses none of those requirements. For a deeper explanation, see What Reclassification Does NOT Mean.
- Over-the-counter availability. All compounded peptides require a physician prescription. The "research use only" gray market that expanded after the 2023 restrictions is a separate issue. See For Research Use Only: The Legal Fiction of Gray-Market Peptides.
- Insurance coverage. Compounded drugs are rarely covered by insurance. Patients typically pay out of pocket.
- Standardized quality. Compounding pharmacies are not held to the same manufacturing standards as FDA-approved drug manufacturers. Quality varies between pharmacies.
What Still Needs to Happen
As of March 2026, the formal regulatory process is incomplete. The Kennedy announcement signaled intent, but the FDA has not published updated Category lists in the Federal Register. Several administrative steps remain:
PCAC review completion. Not all 14 peptides have been through PCAC review. The peptides removed in September 2024 have been reviewed, but others (BPC-157, TB-500, MOTS-C, Epitalon, Emideltide, KPV, Semax, GHK-Cu) may still need advisory committee evaluation.
Notice-and-comment rulemaking. Formal reclassification requires a Federal Register notice and a public comment period. This process typically takes months.
Updated interim policy. The FDA will need to publish revised interim guidance documents reflecting the new Category 1 placements.
Until these steps are completed, the legal status of these peptides for compounding remains technically unchanged from the Category 2 designation. Pharmacies that resume compounding before formal reclassification take on regulatory risk.
For the political context behind the announcement, see RFK Jr. and Peptide Reclassification: What Was Announced and What It Means.
The Bottom Line
Fourteen of nineteen peptides placed on the FDA's Category 2 restricted list in September 2023 are expected to return to Category 1. The evidence base varies enormously across these peptides, from Thymosin Alpha-1 with randomized clinical trials and approval in 30+ countries, to KPV with fewer than 5 published studies. Category 1 restores legal compounding access but carries no FDA safety or efficacy endorsement. The formal administrative process remains incomplete as of March 2026.
Sources & References
- 1RPEP-00561·Sikiric, P (1999). “BPC-157: A Comprehensive Review of Its Healing Effects Across Multiple Organ Systems.” Inflammopharmacology.Study breakdown →PubMed →↩
- 2RPEP-12376·Maar, Klaudia et al. (2025). “Thymosin Beta-4 Peptide Reduces Heart Scarring After Heart Attack by Regulating the ROCK1 Protein.” International journal of molecular sciences.Study breakdown →PubMed →↩
- 3RPEP-00485·Raun, K et al. (1998). “Ipamorelin: The First Growth Hormone Peptide That Only Boosts GH Without Other Hormones.” European journal of endocrinology.Study breakdown →PubMed →↩
- 4RPEP-00993·Wilding, John (2004). “AOD-9604 Metabolic..” Current opinion in investigational drugs (London.Study breakdown →PubMed →↩
- 5RPEP-01050·Iino, S et al. (2005). “Thymosin Alpha-1 for Hepatitis B in Japanese Patients: Randomized Trial Shows Safety and Response.” Journal of viral hepatitis.Study breakdown →PubMed →↩
- 6RPEP-03968·Vyunova, Tatiana V et al. (2018). “How Selank Peptide Reduces Anxiety by Modulating GABA Receptors Differently Than Benzodiazepines.” Protein and peptide letters.Study breakdown →PubMed →↩
- 7RPEP-01182·Romanova, G A et al. (2006). “Neuroprotective and antiamnesic effects of Semax during experimental ischemic infarction of the cerebral cortex..” Bulletin of experimental biology and medicine.Study breakdown →PubMed →↩
- 8RPEP-00833·Khavinson, V Kh et al. (2003). “Epithalon Peptide Activates Telomerase and Lengthens Telomeres in Human Cells.” Bulletin of experimental biology and medicine.Study breakdown →PubMed →↩
- 9RPEP-12847·Ogórek, Karolina et al. (2025). “Can Liposomes Help the Anti-Aging Peptide GHK-Cu Penetrate Skin? Review Reveals a Major Research Gap.” Molecules (Basel.Study breakdown →PubMed →↩
- 10RPEP-15235·Gudiksen, Anders et al. (2026). “MOTS-c Peptide Improves Muscle Energy Production Efficiency.” Free radical biology & medicine.Study breakdown →PubMed →↩
- 11RPEP-00835·Khvatova, Elena M et al. (2003). “Delta sleep inducing peptide (DSIP): effect on respiration activity in rat brain mitochondria and stress protective potency under experimental hypoxia..” Peptides.Study breakdown →PubMed →↩
- 12RPEP-13160·Racková, Jana (2025). “How KNDy Neurons Control Reproduction Through Kisspeptin, Neurokinin B, and Dynorphin.” Ceska gynekologie.Study breakdown →PubMed →↩