Dihexa's Potency Claims: What the Research Says
Dihexa & HGF-Based Nootropics
10 millionx more potent claim
Dihexa was described as 10 million times more potent than BDNF at promoting synaptogenesis. In 2025, the key study establishing its mechanism was retracted due to image manipulation.
Benoist et al., 2014 (retracted April 2025)
Benoist et al., 2014 (retracted April 2025)
If you only read one thing
Dihexa is a research peptide that got famous online after one paper claimed it was ten million times more potent than BDNF — the brain's own growth factor — at forming new synapses. That claim came from one specific lab dish comparison, not any real-world cognitive outcome. The paper that supposedly explained how Dihexa works in the brain was retracted in April 2025 after investigators found its Western blot images had been faked. What is still standing is the behavioral side: Dihexa genuinely improved memory in two separate rodent studies, including an independent replication in a Chinese lab. But there is zero human data, no FDA approval, and the drug is sold through the gray market based on preclinical evidence whose mechanism story is in shambles.
Few peptides have generated as much online excitement with as little clinical data as Dihexa. The compound, formally known as N-hexanoic-Tyr-Ile-(6) aminohexanoic amide, was described in a 2013 paper as a metabolically stable angiotensin IV analog that improved cognitive performance in scopolamine-impaired rats and promoted synaptogenesis in hippocampal neurons.[1] A follow-up 2014 study claimed Dihexa was "seven orders of magnitude more potent than BDNF" at promoting new synaptic connections, and identified the hepatocyte growth factor (HGF)/c-Met receptor system as its molecular target. That 2014 paper was retracted in April 2025 following an investigation that found its lead author had systematically manipulated Western blot images.
This article examines what the original research actually demonstrated, what the retraction means for the evidence base, and what independent research exists to evaluate Dihexa's effects. For context on the BDNF comparison that drives most online discussion, see BDNF: The Brain Peptide That Builds New Neural Connections.
Key Takeaways
- Dihexa improved cognitive performance in scopolamine-impaired rats and increased hippocampal dendritic spine density by nearly 3-fold in cell culture (McCoy et al., 2013)
- The "10 million times more potent than BDNF" claim came from comparing synaptogenic activity in cell culture assays, not cognitive effects or clinical outcomes
- The 2014 Benoist et al. paper establishing Dihexa's HGF/c-Met mechanism was retracted in April 2025 after investigation found systematic image manipulation by lead author Leen Kawas
- An independent Chinese study (Sun et al., 2021) confirmed Dihexa rescues cognitive impairment in APP/PS1 Alzheimer's mice via PI3K/AKT signaling, providing partial independent replication
- A 2018 systematic review of angiotensin IV peptides found 32 eligible studies showing cognitive benefits, but noted most used the same research group's compounds (Ho et al., 2018)
- No human clinical trial of Dihexa has been published, and the compound has no FDA approval for any indication
The Original McCoy 2013 Study
Research & Commercialization Timeline
From Foundational Paper to Retraction: Dihexa's 12-Year Arc
Dihexa restores cognition in scopolamine-impaired rats at 2 mg/kg oral; picomolar synaptogenesis in cell culture
Proposes HGF/c-Met binding as molecular basis — becomes foundation for commercialization
Fosgonimeton (Dihexa prodrug) developer raises $238M on public markets
Four Kawas papers flagged for image manipulation; Kawas removed as CEO
Chinese group confirms cognitive rescue in APP/PS1 mice — via PI3K/AKT, not HGF/c-Met
Fosgonimeton misses primary and key secondary endpoints in 302 Alzheimer's patients
Wells et al. find Dihexa ineffective for motor/cognitive protection in HD rat model
Athira settles False Claims Act case for concealing research misconduct
Key mechanism paper formally retracted for systematic image manipulation
Sources: McCoy 2013; Benoist 2014 (retracted 2025); Sun 2021; Wells 2024; Athira LIFT-AD 2024; DOJ settlement 2025
 View as image
View as imageThe foundational Dihexa paper was published in the Journal of Pharmacology and Experimental Therapeutics by McCoy et al. from the Harding laboratory at Washington State University.[1]
The study's premise: angiotensin IV (AngIV), a six-amino-acid fragment of angiotensin II, had been known since the 1990s to enhance memory and learning in animal models. However, AngIV is rapidly degraded by peptidases and cannot cross the blood-brain barrier or survive oral administration. The Harding group synthesized a series of metabolically stabilized AngIV analogs, of which Dihexa emerged as the most potent.
In scopolamine-impaired rats (scopolamine blocks cholinergic signaling and mimics the cognitive deficits of Alzheimer's disease), Dihexa at 2 mg/kg per day orally restored performance in the Morris water maze to levels indistinguishable from healthy controls. Probe trials confirmed that treated animals retained spatial memory. Dihexa also outperformed donepezil (Aricept), a cholinesterase inhibitor that is one of the few FDA-approved Alzheimer's drugs, in the same model.
In cell culture, Dihexa increased hippocampal dendritic spine density by approximately 3-fold (41 spines per 50 micrometers of dendrite versus 15 in vehicle-treated controls). Dendritic spines are the physical structures where synapses form, so this increase represents new synaptic connections being built.
It was from this in vitro spine density data that the "10 million times more potent than BDNF" claim originated. At the concentrations tested, Dihexa produced similar synaptogenic effects to BDNF at concentrations seven orders of magnitude lower. This is a valid pharmacological comparison of relative potency at a specific endpoint in a specific assay. It is not a statement that Dihexa is "10 million times better" for the brain, for cognition, or for any clinical outcome.
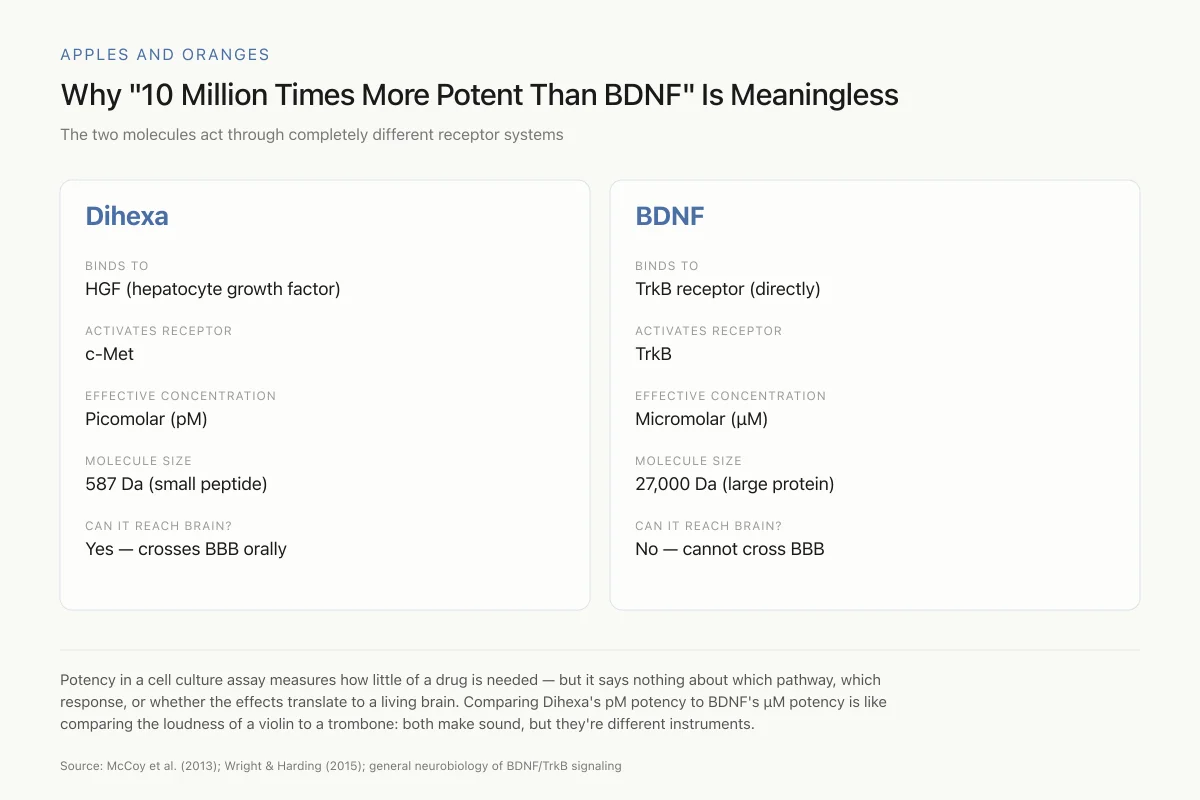
The comparison requires context that is almost always missing from online discussions. First, Dihexa and BDNF act through completely different receptor systems (Dihexa through HGF/c-Met; BDNF through TrkB). Comparing potencies across different receptor systems is like comparing the horsepower of a boat engine to the wattage of a lightbulb: the numbers are real but the comparison is not meaningful without shared units. Second, potency in cell culture does not predict potency in living organisms, where factors like absorption, distribution, metabolism, and excretion determine what reaches the brain. Third, BDNF is a large protein (27 kDa) that does not cross the blood-brain barrier, while Dihexa is a small molecule (587 Da) designed for oral bioavailability. The potency difference may partly reflect the fact that BDNF is being compared at a severe pharmacokinetic disadvantage in the in vitro assay context.
The study also showed that Dihexa had no cognitive effects in rats with normal cognition. It only improved performance in scopolamine-impaired animals. This is an important finding that contradicts the popular framing of Dihexa as a "cognitive enhancer" for healthy individuals. The data suggests it may be a cognitive rescuer for impaired states rather than a universal nootropic.
Apples and Oranges
Why "10 Million Times More Potent Than BDNF" Is a Meaningless Comparison
The two molecules act through completely different receptor systems
Potency in a cell culture assay measures how little of a drug is needed to produce a response — but it says nothing about which response, which pathway, or whether the effects translate to a living brain. Comparing Dihexa's pM potency to BDNF's µM potency is like comparing the loudness of a violin to a trombone: both make sound, but they're different instruments answering different questions.
Source: McCoy et al. (2013); Wright & Harding (2015); general neurobiology of BDNF/TrkB signaling
 View as image
View as imageThe Retracted 2014 Study: Benoist et al.
The 2014 study by Benoist, Bhatt, and Bhatt (with Leen Kawas as co-author) was published in the Journal of Pharmacology and Experimental Therapeutics. It was the study that established Dihexa's proposed mechanism of action: binding to hepatocyte growth factor (HGF) with high affinity and acting synergistically with HGF to promote c-Met receptor phosphorylation, hippocampal spinogenesis, and synaptogenesis.
In September 2021, four papers co-authored by Leen Kawas received expressions of concern from their respective journals, triggered by allegations of image manipulation in her 2011 doctoral dissertation at Washington State University. An investigation by the university's special committee found that Dr. Kawas had systematically altered images, including copying and pasting data between experiments, digitally modifying Western blot bands, and reusing identical images to represent different experimental conditions. Of 30 images examined from her dissertation, problems were identified in 19.
In April 2025, the Benoist et al. paper was formally retracted. This is the paper that provided the molecular basis for Dihexa's proposed HGF/c-Met mechanism. Without it, the mechanistic story for Dihexa loses its primary evidence.
The Broader Fraud Context
Kawas founded M3 Biotechnology (later renamed Athira Pharma) to commercialize HGF-targeting compounds derived from the Dihexa research. Athira went public in September 2020 at $17 per share, raising $238 million. After the image manipulation allegations surfaced in 2021, Kawas was removed as CEO. In January 2025, Athira paid $4,068,698 to settle False Claims Act allegations after a whistleblower exposed how the company had concealed research misconduct from federal agencies for five years.
This context matters for evaluating Dihexa because the retracted paper was the bridge between the behavioral data (McCoy 2013, which remains published) and the molecular mechanism (HGF/c-Met signaling). The behavioral finding that Dihexa improves cognition in rats has not been retracted. The proposed explanation for why it works has been.
The retraction does not prove that the HGF/c-Met mechanism is wrong. Image manipulation means the data supporting the claim is unreliable, not that the claim itself is false. It is possible that Dihexa genuinely binds HGF and activates c-Met, but the evidence presented in the retracted paper can no longer be used to support that conclusion. Independent replication of the binding and signaling data would be needed to reestablish this mechanism, and no such replication has been published.
What Evidence Remains?
After the retraction
What Dihexa Evidence Still Stands
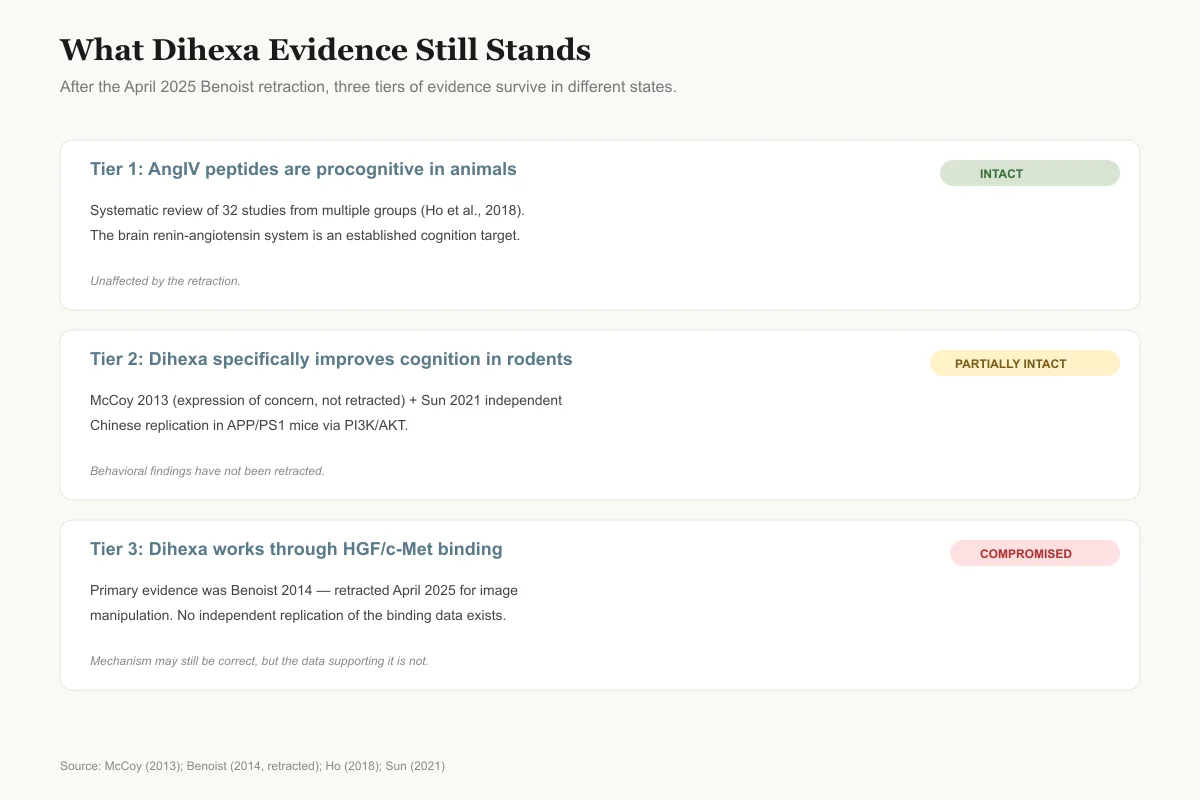
The retraction did not erase everything. It did knock out the mechanism story while leaving the behavioral findings partially intact.
Tier 1: Angiotensin IV–related peptides have procognitive effects in animal models
IntactSystematic review of 32 studies from multiple independent groups (Ho et al., 2018). Brain renin-angiotensin system is a legitimate cognition target.
Tier 2: Dihexa specifically improves cognition in rodent models
Partially intactMcCoy 2013 (published, expression of concern) + Sun 2021 (independent Chinese replication in APP/PS1 mice via PI3K/AKT). Behavioral findings not retracted.
Tier 3: Dihexa works through HGF/c-Met binding
CompromisedPrimary evidence was Benoist 2014 — retracted April 2025 for image manipulation. Mechanism may still be correct, but the data is not. No independent replication.
No human clinical trial of Dihexa has been published. No regulatory agency has approved it for any indication.
Source: McCoy (2013); Benoist (2014, retracted); Ho (2018); Sun (2021)
 View as image
View as imageThe McCoy 2013 Study (Published, Expression of Concern)
The original McCoy et al. behavioral study has an expression of concern but has not been retracted. It demonstrated that Dihexa improves cognitive performance in scopolamine-impaired rats and increases dendritic spine density in hippocampal cultures. These findings used different methodologies (behavioral testing, spine counting) than the Western blots that were the subject of the image manipulation investigation.[1]
However, the expression of concern means the data should be interpreted with caution. An expression of concern is not a retraction, it is a flag that the journal has identified concerns that have not yet been fully resolved.
Wright 2015: The HGF/c-Met Hypothesis (Different Authors)
Wright and Harding (2015) published a review article proposing the brain HGF/c-Met receptor system as a therapeutic target for Alzheimer's and Parkinson's diseases. This review paper, authored by the senior investigators in the Harding laboratory rather than by Kawas, laid out the theoretical framework for why HGF modulation could promote neuroprotection and synaptogenesis.[2] A companion review covered the development of small molecule angiotensin IV analogs for these diseases.[3]
These review papers are not affected by the retraction because they present theoretical arguments and review published literature rather than original experimental data. However, they rely heavily on the now-retracted Benoist 2014 data for their mechanistic claims about Dihexa specifically. The theoretical framework (HGF/c-Met as a neuroprotective target) is supported by broader HGF literature independent of the Dihexa research, but the specific claim that Dihexa activates this system rested primarily on the retracted paper.
Sun et al. 2021: Independent Chinese Replication
The most important evidence for Dihexa's cognitive effects comes from outside the Harding laboratory. Sun et al. (2021) at the Renmin Hospital of Wuhan University tested Dihexa in APP/PS1 transgenic mice, a model that develops Alzheimer's-like amyloid pathology. Dihexa rescued cognitive impairment and recovered memory in these mice, and the researchers attributed the effect to the PI3K/AKT signaling pathway rather than (or in addition to) the HGF/c-Met pathway proposed by the retracted Benoist paper.[4]
This is partial independent replication, and it is the single most important piece of evidence for Dihexa's cognitive effects. It confirms that Dihexa has procognitive effects in an Alzheimer's mouse model with genuine amyloid pathology (APP/PS1 mice develop plaques and tangles, unlike the scopolamine model which merely blocks cholinergic signaling). The cognitive improvements were assessed using the Morris water maze and novel object recognition tests, both standard behavioral assays.
The PI3K/AKT pathway finding is interesting because it is consistent with the HGF hypothesis (c-Met activates PI3K/AKT) but is also activated by many other receptor tyrosine kinases. The Sun study did not directly test whether Dihexa binds HGF or activates c-Met, so it neither confirms nor refutes the retracted mechanism specifically. It does confirm that Dihexa produces measurable cognitive improvement through identifiable molecular pathways in an independent laboratory, which is the minimal standard for taking a drug candidate seriously after a retraction of its proposed mechanism.
Ho et al. 2018: Systematic Review of AngIV Peptides
Ho et al. (2018) published a systematic review of experimental studies on the cognitive effects of angiotensin IV and angiotensin-(1-7). Of 450 articles identified, 32 met inclusion criteria. The review found consistent procognitive effects of AngIV-related peptides across multiple models and concluded that the brain renin-angiotensin system represents a legitimate target for cognitive enhancement research.[5]
This systematic review provides broader context: Dihexa is not the only angiotensin IV-derived compound with cognitive effects. The AngIV peptide family has a research history predating Dihexa by over a decade, and the procognitive effects of AngIV receptor activation are supported by multiple independent groups working with different AngIV analogs. Several mechanisms have been proposed for AngIV's cognitive effects, including activation of the AT4 receptor (also called insulin-regulated aminopeptidase, or IRAP), modulation of glucose uptake in the brain, and enhancement of long-term potentiation in the hippocampus. The Harding group's Dihexa specifically was proposed to work through HGF/c-Met rather than the AT4/IRAP pathway, which was a novel and controversial claim even before the retraction.
The question after the retraction is not whether AngIV signaling affects cognition (it does, supported by 32 studies) but whether Dihexa specifically works through the HGF/c-Met pathway that was proposed in the retracted paper, or through the more traditional AT4/IRAP mechanism shared by other AngIV analogs.
Wells et al. 2024: Huntington's Disease Model
Wells et al. (2024) tested an angiotensin IV analog in a rat model of Huntington's disease-like symptoms induced by 3-nitropropionic acid. The study assessed motor function, cognitive performance, and neuropathological markers, extending the AngIV analog research to a neurodegenerative disease beyond Alzheimer's.[6]
This study is from the same Harding laboratory group but uses different disease models and different experimental endpoints than the retracted Benoist paper. It adds to the body of evidence that AngIV analogs have neuroprotective effects across multiple neurodegenerative disease contexts, not just Alzheimer's disease. The extension to a Huntington's model is relevant because HD involves different pathology (huntingtin protein aggregation, striatal neuron loss) than Alzheimer's (amyloid plaques, tau tangles), suggesting that AngIV analog neuroprotection may operate through mechanisms that are not disease-specific. However, this study does not address the specific mechanism questions raised by the retraction of the Benoist paper.
How Dihexa Modulates the Brain: What We Know and Do Not Know
The proposed mechanism chain for Dihexa was: Dihexa binds HGF with high affinity, HGF activates the c-Met receptor, c-Met triggers downstream signaling including PI3K/AKT and ERK/MAPK, these pathways promote neuronal survival and synaptogenesis. For the full mechanistic analysis, see How Dihexa Modulates Hepatocyte Growth Factor in the Brain.
With the Benoist retraction, the first step of this chain (Dihexa binding to HGF specifically) has lost its primary evidence. The downstream effects (cognitive improvement, synaptogenesis) are supported by the McCoy 2013 study and the Sun 2021 replication. But these downstream effects could be mediated by a mechanism other than HGF/c-Met. The Sun study's finding that PI3K/AKT signaling is involved is consistent with the HGF hypothesis (c-Met activates PI3K/AKT) but is also consistent with numerous other upstream receptors.
For the specific synaptogenesis data and how it relates to Alzheimer's research, the spine density increase from McCoy 2013 remains the most cited finding. For a breakdown of how the "millions of times more potent than BDNF" claim originated and what it actually means, see Dihexa: The Peptide Described as Millions of Times More Potent Than BDNF.
The Amyloid-Beta Connection
Dihexa's relevance to Alzheimer's disease sits within a broader peptide landscape. The dominant Alzheimer's hypothesis targets amyloid-beta accumulation, while Dihexa targets a different pathway entirely (synaptogenesis via HGF or PI3K/AKT). The Sun 2021 study used APP/PS1 mice that develop amyloid pathology, showing Dihexa can improve cognition even in the presence of amyloid plaques. This positions Dihexa as a potential symptomatic rather than disease-modifying approach: it may improve cognitive function in the presence of amyloid pathology without addressing the underlying protein aggregation. This distinction is speculative given the absence of human data, but it is relevant to how Dihexa's potential role in Alzheimer's therapeutics should be framed.
Evaluating the Evidence After the Retraction
The evidence base for Dihexa can be assessed in three tiers:
Tier 1 (Intact): AngIV-related peptides have procognitive effects. This is supported by a systematic review of 32 studies from multiple independent groups (Ho et al., 2018). The brain renin-angiotensin system is a legitimate pharmacological target for cognitive enhancement.
Tier 2 (Partially intact): Dihexa specifically improves cognition in rodent models. The McCoy 2013 study remains published (with an expression of concern) and the Sun 2021 study provides independent replication in a different Alzheimer's model. These behavioral findings have not been retracted.
Tier 3 (Compromised): Dihexa works through HGF/c-Met binding. The primary evidence for this mechanism (Benoist 2014) has been retracted due to image manipulation. The mechanism may still be correct, but the data supporting it is no longer considered reliable by the scientific community. Independent confirmation of the binding data would be needed to rehabilitate this claim, and that confirmation does not exist.
The overall picture is of a compound with real but limited behavioral evidence, a compromised mechanistic story, zero human data, and enormous online hype driven by a potency comparison that was always narrower than how it was presented. The retraction does not erase Dihexa from the research landscape, but it removes the strongest argument that was being made for it and demands a more cautious evaluation of what remains.
No human clinical trial of Dihexa has been published. The compound has no FDA approval. It is not approved as a pharmaceutical in any country. It is available on the gray market as a "research chemical," and its use by individuals for cognitive enhancement is based entirely on preclinical animal data, of which the most mechanistically important study has been retracted.
What About Oral Bioavailability?
One of Dihexa's claimed advantages over other neuropeptides is oral bioavailability. The McCoy 2013 study demonstrated cognitive effects when Dihexa was administered orally to rats, which was a primary design goal (AngIV itself is destroyed by gut enzymes). The metabolic stabilization achieved by replacing natural amino acids with non-natural analogs is well-established peptide chemistry, and oral activity in rats is plausible based on Dihexa's molecular properties (molecular weight 587 Da, favorable calculated logP).
However, oral bioavailability in rats does not guarantee oral bioavailability in humans. The two species differ in gastric pH, intestinal transit time, first-pass liver metabolism, and drug transporter expression. No pharmacokinetic study of Dihexa in humans has been published. The effective dose in rats (2 mg/kg) would translate to roughly 140 mg for a 70 kg human by simple allometric scaling, but such scaling is unreliable without confirmed oral absorption data. Gray-market Dihexa products are typically sold at doses far below this, raising questions about whether the doses used by self-experimenters could achieve brain concentrations comparable to those in the animal studies.
The Gray Market Problem
Safety
ModerateDihexa is sold online without approval, trials, or purity testing
Concern
Dihexa has no FDA approval, no human clinical trial, and no standardized manufacturing. Gray-market vendors sell it in sublingual, nasal, and injectable forms with dosing based on unvalidated rat-to-human scaling. Product purity is not guaranteed. Effects on healthy human brain tissue are completely unstudied, and any compound that promotes synaptogenesis could in theory promote unwanted neural growth.
What the research says
The honest answer is that no one knows the right dose, safety profile, or long-term effect in humans. The key mechanistic paper being retracted in 2025 makes the situation worse, not better.
Particularly relevant for: Anyone considering self-experimentation with purchased Dihexa
What to do
Do not self-administer unapproved research chemicals. If cognitive decline is the concern, use evidence-based medical care. If you are a researcher, use only regulated supply chains with verified identity and purity.
FDA unapproved drug database; Benoist retraction (April 2025); Athira Pharma False Claims settlement (January 2025)
Dihexa is widely sold online as a "research peptide" or "nootropic compound." Multiple vendors offer it in sublingual, nasal spray, and injectable forms. The disconnect between the research base (small, preclinical, partially retracted, no human data) and the commercial availability (easily purchable online with implied cognitive enhancement benefits) is one of the sharpest in the peptide space.
The gray-market supply creates several risks beyond the absence of efficacy data. First, product purity is not guaranteed; without FDA oversight, purchased Dihexa may contain degradation products, synthetic impurities, or incorrect concentrations. Second, the appropriate human dose is unknown; vendors choose doses based on animal-to-human scaling calculations that have not been validated. Third, Dihexa's effects on healthy human brain tissue are completely unstudied. The McCoy 2013 study found no cognitive enhancement in unimpaired rats, which raises the question of what, if anything, Dihexa does in a healthy human brain. Fourth, any compound that promotes synaptogenesis could theoretically promote unwanted neural connections or cellular growth. This risk is speculative but cannot be assessed without human safety data.
The Bottom Line
Dihexa showed procognitive effects in multiple rodent models, including improved Morris water maze performance and increased hippocampal dendritic spine density. The "10 million times more potent than BDNF" claim reflected a narrow in vitro potency comparison, not overall cognitive superiority. The 2014 paper establishing Dihexa's HGF/c-Met mechanism was retracted in April 2025 due to systematic image manipulation by its lead author. An independent 2021 Chinese study partially replicated the behavioral findings in APP/PS1 Alzheimer's mice. No human trial exists.
Sources & References
- 1RPEP-02233·McCoy, Alene T et al. (2013). “Evaluation of metabolically stabilized angiotensin IV analogs as procognitive/antidementia agents..” The Journal of pharmacology and experimental therapeutics.Study breakdown →PubMed →↩
- 2RPEP-02836·Wright, John W et al. (2015). “The Brain Hepatocyte Growth Factor/c-Met Receptor System: A New Target for the Treatment of Alzheimer's Disease..” Journal of Alzheimer's disease : JAD.Study breakdown →PubMed →↩
- 3RPEP-02837·Wright, John W et al. (2015). “The development of small molecule angiotensin IV analogs to treat Alzheimer's and Parkinson's diseases..” Progress in neurobiology.Study breakdown →PubMed →↩
- 4RPEP-05800·Sun, Xiaojin et al. (2021). “AngIV-Analog Dihexa Rescues Cognitive Impairment and Recovers Memory in the APP/PS1 Mouse via the PI3K/AKT Signaling Pathway..” Brain sciences.Study breakdown →PubMed →↩
- 5RPEP-03709·Ho, Jean K et al. (2018). “Do Angiotensin Peptides Improve Memory? A Systematic Review of Animal Studies.” Neuroscience and biobehavioral reviews.Study breakdown →PubMed →↩
- 6RPEP-09517·Wells, Russell G et al. (2024). “Dihexa Failed to Protect Against Huntington's Disease Symptoms in Rats.” Journal of Huntington's disease.Study breakdown →PubMed →↩