BPC-157 and the FDA: Category 2 Explained
BPC-157 and the FDA
Category 2 Since Sept 2023
The FDA classified BPC-157 as a Category 2 bulk drug substance in September 2023, prohibiting licensed compounding pharmacies from preparing it for patients. In February 2026, HHS Secretary Kennedy announced plans to reverse the classification.
FDA Interim Policy on Compounding Using Bulk Drug Substances, 2023
FDA Interim Policy on Compounding Using Bulk Drug Substances, 2023
If you only read one thing
The FDA didn't ban BPC-157. It put it in a category that blocks compounding pharmacies from making it, mostly because almost no one has studied it in actual humans. About 30 people total, across three small studies. That restriction pushed buyers to unregulated gray-market vendors selling untested products. In February 2026, the government announced plans to reverse the restriction — but that still doesn't mean BPC-157 is FDA-approved or proven safe.
In September 2023, the FDA added BPC-157 to its Category 2 list of bulk drug substances that "may present significant safety risks." That single classification changed the peptide market overnight. Licensed compounding pharmacies could no longer legally prepare BPC-157 for patients. Clinics that had been prescribing it for tendon injuries, gut conditions, and recovery protocols lost access to their primary supply chain. Patient demand did not disappear. It moved underground. By early 2024, the gray market for BPC-157 was larger than the legitimate market had ever been. For the full picture of what BPC-157 is and what the preclinical data actually shows, see The Real BPC-157 Story: 544 Papers, 30 Human Subjects, 50 Million Views.
Key Takeaways
- The FDA never "banned" BPC-157 — it blocked compounding pharmacies from making it, which isn't the same thing.
- Regulators aren't saying BPC-157 is dangerous. They're saying no one has tested it on enough humans to know.
- Only about 30 people have ever been studied across three tiny pilot trials.
- In February 2026 the government announced plans to reverse the restriction for 14 of 19 peptides, BPC-157 included.
- Reversing the restriction isn't FDA approval — the evidence base didn't change, just the regulatory category.
- The 2023 restriction didn't reduce demand — it pushed buyers from licensed pharmacies to unregulated gray-market vendors.
- Athletes can still be sanctioned for BPC-157 even after reclassification — it remains banned under WADA rules.
What BPC-157 Is
BPC-157 is a synthetic 15-amino-acid peptide derived from a larger protein called Body Protection Compound (BPC) found in human gastric juice. It was first characterized in the early 1990s at the University of Zagreb by Predrag Sikiric and colleagues, who identified it as unusually stable in stomach acid.[1] The peptide's formal pharmacological profile was described in 1999, covering its effects on gastrointestinal mucosal integrity, wound healing, and interactions with the nitric oxide and dopamine systems.[2]
In animal models, BPC-157 has demonstrated effects across a wide range of tissues. It accelerated Achilles tendon healing in rats and stimulated tendocyte growth in vitro.[3] It modulated angiogenesis in muscle and tendon repair models.[4] It promoted healing of skin wounds, burns, and bone defects.[5] A 2021 review in Frontiers in Pharmacology compiled evidence across wound types, including incisional and excisional wounds, deep burns, and diabetic ulcers.[6] These results, spanning hundreds of animal studies, explain why BPC-157 became one of the most popular peptides in the biohacking and sports medicine communities. For a detailed analysis of the research, see BPC-157: The Body Protection Compound and What the Research Shows.
The critical caveat: over 80% of BPC-157 research originates from a single laboratory in Zagreb. And as of 2025, only three pilot studies totaling approximately 30 human subjects have been published. That evidence gap is central to the FDA's decision.
How the FDA Categories Work
The FDA's bulk drug substance categorization system was created under the Drug Quality and Security Act of 2013, which established two regulatory tracks for compounding. Section 503A governs traditional compounding pharmacies that fill patient-specific prescriptions. Section 503B governs outsourcing facilities that compound drugs in larger quantities without individual prescriptions but under enhanced FDA oversight.
Under the interim policy published in 2023, the FDA sorts nominated bulk drug substances into categories:
Category 1 substances have been evaluated and the FDA does not intend to take enforcement action against pharmacies compounding with them, provided they meet the conditions described in the guidance. In practical terms, Category 1 means a substance can be legally compounded.
Category 2 substances have been evaluated and the FDA has identified "significant safety risks" relating to their use in compounding. Pharmacies may not compound with Category 2 substances under the policies described for Category 1. In practice, Category 2 is a compounding prohibition.
Category 3 substances have not been nominated or do not have enough supporting information for FDA evaluation. (In January 2025, the FDA restructured the system, eliminating the interim categories for newly nominated substances.)
The distinction matters. Category 2 does not mean a substance is "banned" in the way the word is commonly used. BPC-157 is not a controlled substance. It is not a Schedule I drug. The classification specifically restricts licensed compounding pharmacies from preparing and distributing it. It does not make possession illegal. It does not apply to research use. And it does not apply in countries outside the United States.
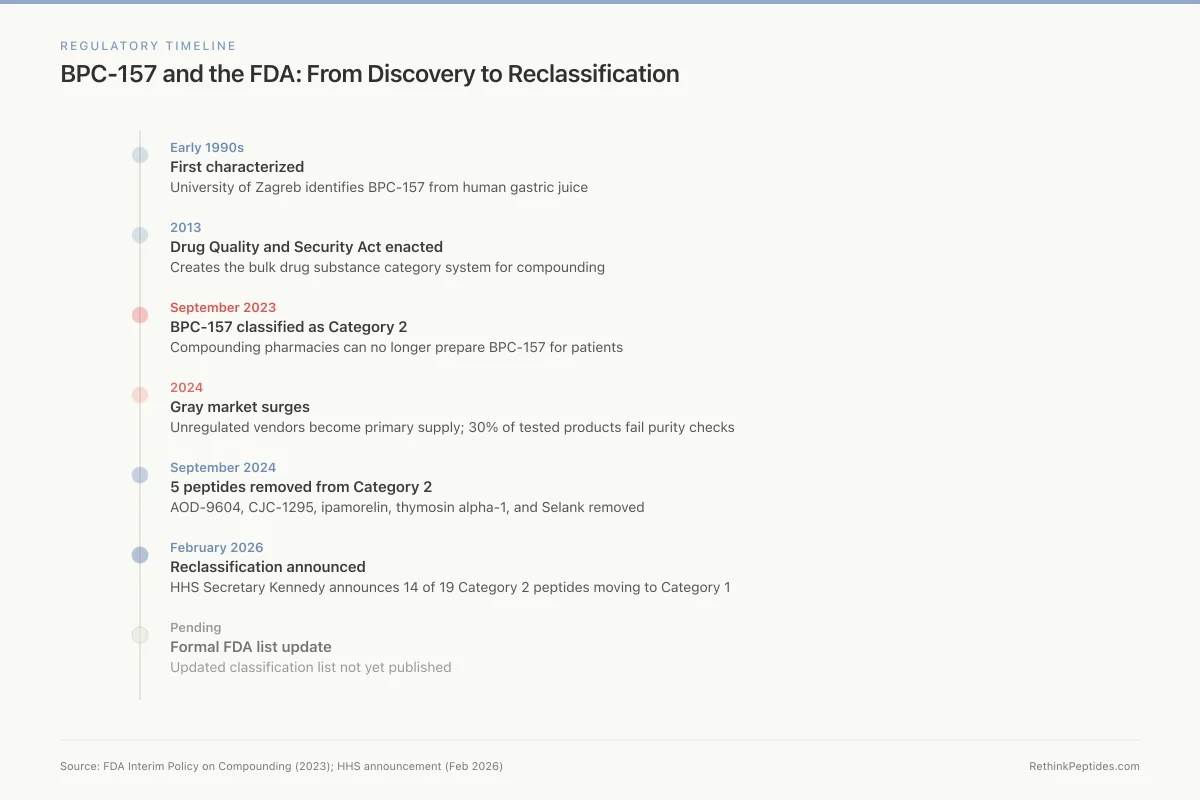
Regulatory Timeline
BPC-157 and the FDA: From Discovery to Reclassification
Early 1990s
First characterized
University of Zagreb identifies BPC-157 from human gastric juice
2013
DQSA enacted
Drug Quality and Security Act creates the bulk drug substance category system
Sept 2023
Category 2
FDA classifies BPC-157 as Category 2 — compounding pharmacies can no longer prepare it
2024
Gray market surges
Unregulated "research use only" vendors become primary supply; 30% of tested products fail purity checks
Sept 2024
5 peptides removed
AOD-9604, CJC-1295, ipamorelin, thymosin alpha-1, and Selank removed from Category 2
Feb 2026
Reclassification announced
HHS Secretary Kennedy announces 14 of 19 Category 2 peptides moving back to Category 1
Pending
Formal FDA list update
Updated classification list not yet published — compounding pharmacies waiting for formal guidance
Source: FDA Interim Policy on Compounding (2023); HHS announcement (Feb 2026)
 View as image
View as imageWhy the FDA Moved BPC-157 to Category 2
The FDA's stated justification for placing BPC-157 in Category 2 rests on three specific concerns.
Immunogenicity. All peptides carry some risk of triggering immune responses. The immune system can produce antibodies against injected peptides, particularly synthetic ones that differ from endogenous sequences. BPC-157 is a synthetic fragment. Although it is derived from a protein found in human gastric juice, the specific 15-amino-acid sequence does not exist in that exact form in the human body. The FDA cited the potential for immunogenic reactions as a safety concern, noting the absence of data characterizing the immune response to BPC-157 in humans.
Manufacturing impurities. Peptide synthesis involves chemical processes that can introduce contaminants, including truncated peptide sequences, racemized amino acids, and residual solvents. The FDA's concern was not specific to BPC-157; it applied broadly to peptides being compounded without the manufacturing controls required for FDA-approved drugs. A 2020 study examining BPC-157's modulatory effects on gastric lesions noted the peptide's protective properties but did not address purity standards for commercially produced formulations.[7]
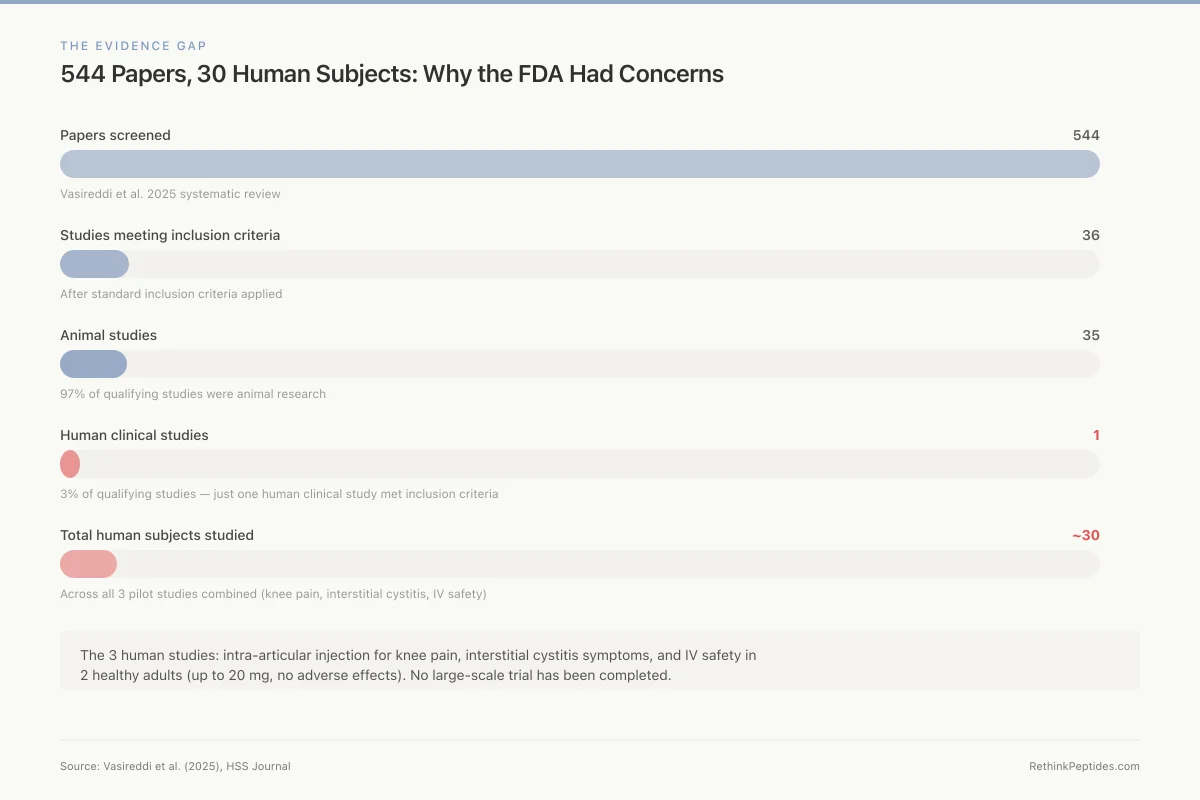
Absence of human safety data. This is the foundational issue. A 2025 systematic review screened 544 BPC-157 articles for orthopaedic applications. After applying standard inclusion criteria, 36 studies remained. Thirty-five were animal studies. One was a human clinical study.[8] The total published human evidence for BPC-157 consists of three pilot studies: one on intra-articular injection for knee pain, one on interstitial cystitis symptoms, and one on intravenous safety and pharmacokinetics.[9][10] The IV safety study administered up to 20 mg of BPC-157 to 2 healthy adults and reported no adverse effects on cardiac, hepatic, renal, or thyroid biomarkers.[10] Two subjects is not a safety profile. It is a starting point.
The FDA did not claim BPC-157 is dangerous. It claimed there is insufficient evidence to determine whether it is safe for compounding. That distinction is lost in most coverage of the decision.
The Evidence Gap
544 Papers, 30 Human Subjects: Why the FDA Had Concerns
Vasireddi et al. 2025 systematic review
After standard inclusion criteria
97% of qualifying studies
3% of qualifying studies
Across all 3 pilot studies combined
The 3 human studies: intra-articular injection for knee pain, interstitial cystitis symptoms, and IV safety in 2 healthy adults (up to 20 mg, no adverse effects). No large-scale trial has been completed.
Source: Vasireddi et al. (2025), HSS Journal
 View as image
View as imageThe Evidence That Does Exist
Dismissing BPC-157's evidence entirely would be inaccurate. The preclinical dataset is substantial, even if it comes predominantly from one research group.
In a 2019 review, Gwyer and colleagues summarized BPC-157's documented effects on musculoskeletal soft tissue healing, identifying consistent findings across tendon, ligament, muscle, and bone injury models.[11] The proposed mechanisms include upregulation of growth hormone receptors in tendon fibroblasts, activation of the VEGF pathway driving angiogenesis, modulation of nitric oxide synthesis through the Akt-eNOS axis, and engagement of ERK1/2 signaling in endothelial and muscle repair.
Sikiric's 2020 comprehensive review placed BPC-157 within the broader context of Robert's cytoprotection and Selye's stress coping response, documenting the peptide's organoprotective effects across gastrointestinal, cardiovascular, and neurological models.[12] In the gastrointestinal system, BPC-157 reduced lesion formation in models of ethanol, NSAID, and stress-induced damage. In cardiovascular models, it appeared to counteract arrhythmias and improve hemodynamic parameters. In neurological models, it showed anxiolytic and antidepressant effects.
These are real findings from published, peer-reviewed research. They are also almost entirely animal findings. The gap between "BPC-157 accelerated Achilles tendon healing in rats" and "BPC-157 is safe and effective for human tendon injuries" is not a small one. It is the gap that the FDA's Category 2 classification sits in.
For a closer look at BPC-157's gastrointestinal evidence, including the never-published Pliva Phase II ulcerative colitis trial, see The Real BPC-157 Story. For the liver-specific data, see BPC-157 and Liver Protection: What Animal Studies Suggest.
BPC-157 and Sports Anti-Doping
The FDA is not the only body that has restricted BPC-157. The World Anti-Doping Agency (WADA) prohibits BPC-157 under category S0: Unapproved Substances. Any pharmacological substance not addressed by any of the other prohibited categories that has no current approval by any governmental regulatory health authority for human therapeutic use is prohibited. BPC-157, which has never been approved for human use by any country's regulatory body, falls squarely within this definition.
The United States Anti-Doping Agency (USADA) has issued specific warnings about BPC-157, citing the lack of human safety data and the risk of contaminated gray-market products. Athletes who test positive for BPC-157 face sanctions regardless of how they obtained it or whether they intended to enhance performance. The peptide's popularity in sports medicine and recovery protocols creates a direct conflict between its off-label use and competitive eligibility. USADA has documented cases of BPC-157 appearing in supplements and products that did not disclose it on the label.
What Changed After the Classification
The immediate effect of the September 2023 classification was the closure of the legal compounding pathway. Compounding pharmacies that had been preparing BPC-157 for prescribing physicians stopped. The FDA signaled it would take enforcement action against pharmacies that continued to compound Category 2 substances.
What happened next was predictable. The demand for BPC-157 did not decrease. It was redirected.
Gray-market vendors, most based overseas, saw a surge in orders. Products labeled "for research use only" became the primary supply channel. These products are not manufactured under Good Manufacturing Practice (GMP) standards. They are not tested for purity, potency, or sterility by any regulatory body. Laboratory analyses of gray-market peptide products have found contamination rates that would be disqualifying in any regulated pharmaceutical setting. A 2018 analysis of seized gray-market polypeptide products found 30% contained incorrect amino acid sequences, and two-thirds failed basic purity thresholds. For the full data on gray-market peptide quality, contamination rates, and documented harms, see Gray-Market Peptides: The 'Research Use Only' Fiction.
Safety
ModerateGray-market peptide purity is a real problem
Concern
A 2018 analysis of seized gray-market polypeptide products found 30% contained incorrect amino acid sequences, and two-thirds failed basic purity thresholds. Products sold as 'research use only' are not manufactured under GMP standards and are not tested for sterility.
What the research says
If the reclassification to Category 1 is finalized, licensed compounding pharmacies would resume production under regulated conditions with quality controls. Until then, no regulated supply exists.
Particularly relevant for: Anyone purchasing BPC-157 from gray-market or 'research chemical' vendors
What to do
If you choose to use gray-market peptides, understand that purity and sterility are not guaranteed. Reconstituting a non-sterile lyophilized powder with bacteriostatic water does not sterilize it.
Analysis of seized gray-market polypeptide products (2018)
The irony of the Category 2 classification is that it may have increased, not decreased, the safety risk for people using BPC-157. Under the compounding system, patients received BPC-157 prepared by licensed pharmacists using verified raw materials, with some degree of quality control and physician oversight. Under the gray market, they receive products of unknown purity from unregulated suppliers, often self-administering based on internet dosing guides. The sterility concern alone is substantial: most gray-market peptides are sold as lyophilized powders without validated sterilization, and reconstituting a non-sterile powder with bacteriostatic water does not sterilize it.
HHS Secretary Kennedy acknowledged this dynamic directly. In his February 2026 announcement, he stated that the restrictions "created the gray market" by shutting down legitimate supply without addressing demand.
Other Peptides Affected by Category 2
BPC-157 was not the only peptide placed in Category 2. The classification affected multiple substances that had been widely compounded:
- TB-500 (Thymosin Beta-4): studied for muscle repair and recovery
- AOD-9604: a growth hormone fragment studied for fat metabolism
- CJC-1295: a growth hormone-releasing hormone analog
- Ipamorelin: a growth hormone secretagogue
- Thymosin Alpha-1: an immune-modulating peptide with published clinical data
- Selank and Semax: neuropeptides studied for cognitive function
- GHK-Cu: a copper peptide studied for wound healing
- KPV: an anti-inflammatory peptide derived from alpha-MSH
- MOTS-c: a mitochondrial peptide studied for metabolic regulation
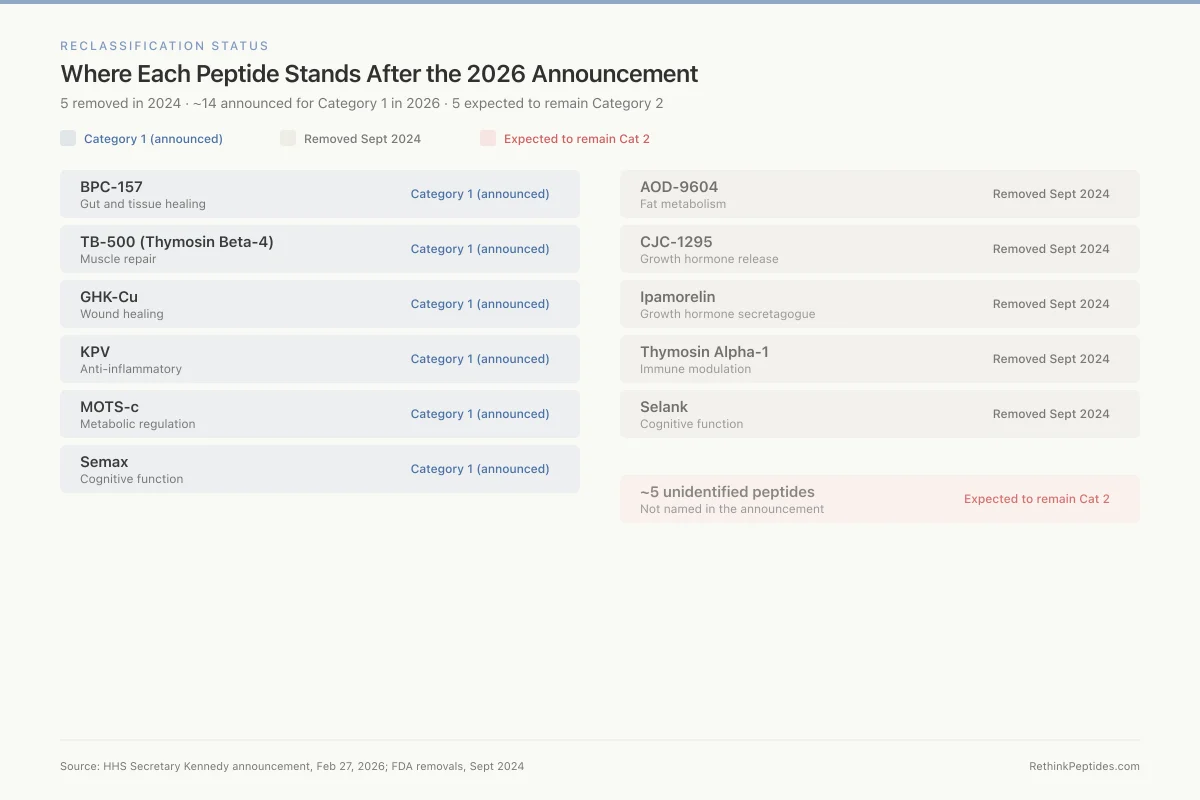
Reclassification Status
Where Each Peptide Stands After the 2026 Announcement
5 removed in 2024 · ~14 announced for Category 1 in 2026 · 5 expected to remain Category 2
BPC-157
Gut and tissue healing
TB-500 (Thymosin Beta-4)
Muscle repair
GHK-Cu
Wound healing
KPV
Anti-inflammatory
MOTS-c
Metabolic regulation
Semax
Cognitive function
AOD-9604
Fat metabolism
CJC-1295
Growth hormone release
Ipamorelin
Growth hormone secretagogue
Thymosin Alpha-1
Immune modulation
Selank
Cognitive function
~5 unidentified peptides
Not named in the announcement
Source: HHS Secretary Kennedy announcement, Feb 27, 2026; FDA removals, Sept 2024
 View as image
View as imageIn September 2024, the FDA removed five substances from Category 2: AOD-9604, CJC-1295, ipamorelin acetate, thymosin alpha-1, and Selank acetate. The removals were based on the nominators withdrawing their nominations, not on new safety data. For the regulatory details of each peptide's status after reclassification, see TB-500, CJC-1295, Ipamorelin: Legal Compounding Status After Reclassification. For the thymosin alpha-1 story specifically, see Thymosin Alpha-1 Reclassification: Restoring Access to an Immune Peptide.
The February 2026 Reclassification Announcement
On February 27, 2026, HHS Secretary Robert F. Kennedy Jr. announced that approximately 14 of the 19 peptides on the FDA's Category 2 list would be moved back to Category 1 status. The announcement was made on the Joe Rogan Experience (Episode #2461). BPC-157 was named among the peptides expected to return to Category 1.
Kennedy's stated rationale: the FDA lacked the required safety signal to justify placing these peptides on the restricted list. He framed the original Category 2 classifications as regulatory overreach that harmed patients by pushing them toward unregulated sources. The legal challenge that preceded the announcement argued the FDA had not demonstrated the "significant safety risks" required to justify the Category 2 designation, particularly for substances with long histories of compounding use and no documented adverse event patterns. For the full story of how this legal challenge unfolded, see The Legal Challenge That Forced FDA's Hand on Peptide Restrictions.
The announcement included several caveats. The FDA had not yet published its formal updated list at the time of the announcement. Kennedy stated he expected the formal reclassification "within a couple of weeks." The 5 remaining peptides that would not be reclassified were not identified. For a breakdown of all 14 peptides and what changes with each, see 14 Peptides Moving Back to Category 1: Which Ones and What Changes.
For a detailed analysis of what Kennedy specifically said and what it means for the compounding industry, see RFK Jr. and Peptide Reclassification: What Was Announced and What It Means.
What Reclassification Does Not Mean
Moving BPC-157 from Category 2 to Category 1 would restore the ability of licensed compounding pharmacies to prepare it under physician oversight. It would not make BPC-157 an FDA-approved drug.
The differences are substantial:
No approved indication. An FDA-approved drug has demonstrated safety and efficacy for a specific condition through controlled clinical trials. BPC-157 has no completed Phase III trial. It has no approved indication for any condition.
No standardized dosing. FDA-approved drugs come with established dose ranges, contraindications, and drug interaction profiles based on clinical trial data. BPC-157 has none of these. The dosing protocols used in clinical practice are extrapolated from animal studies and anecdotal reports.
No long-term safety data. The longest published human exposure to BPC-157 is a 2-day IV infusion protocol in 2 subjects.[10] No study has followed BPC-157 users for months or years. No study has examined interactions with other medications. No study has assessed effects in pregnant women, children, or immunocompromised individuals.
No manufacturing standard. Compounded drugs are not identical to manufactured drugs. They are prepared by pharmacies, not pharmaceutical companies, and are not subject to the same pre-market testing requirements.
Category 1 means the FDA will not take enforcement action against pharmacies that compound BPC-157 under the conditions described in its guidance. It does not mean the FDA endorses BPC-157 or considers it proven safe. For a detailed breakdown of this distinction, see What Reclassification Does NOT Mean: It's Not the Same as FDA Approval.
Where BPC-157 Stands in 2026
As of March 2026, the regulatory picture for BPC-157 is in transition.
The formal reclassification from Category 2 to Category 1 has been announced but the updated FDA list has not been published. Compounding pharmacies are in a holding pattern, waiting for the formal guidance before resuming production. The gray market continues to operate and will likely persist even after reclassification, because it offers lower prices and no prescription requirement.
The evidence base has not changed. BPC-157 still has approximately 30 published human subjects across three pilot studies. No Phase III clinical trial is underway. The preclinical data remains extensive in animal models and almost nonexistent in humans. The Pliva Phase II ulcerative colitis trial, registered in the early 2000s, still has no published results.
What has changed is the public conversation. The Category 2 classification forced a reckoning with the evidence gap. Publications like STAT News ran detailed examinations of BPC-157's evidence base. The 2025 Vasireddi systematic review provided the first rigorous accounting of the clinical data (or lack thereof). The reclassification announcement put peptide regulation into mainstream news.
For an analysis of the scientific and media debate, including Eric Topol's critique of the peptide ecosystem, see The Peptide Craze Critique: What Eric Topol Got Right (and Wrong). For the current legal status in plain terms, see BPC-157 Legal Status in 2026: Where Things Stand.
The question that the Category 2 classification raised has not been answered by the reclassification: is BPC-157 safe and effective in humans? The regulatory system moved the peptide into a restricted category and then moved it back out. Neither action was based on new clinical data. The animal evidence suggests biological activity across multiple tissue types. The human evidence consists of 30 subjects, no adverse events, and no controlled efficacy trials. That is where the science stands, regardless of what category the FDA assigns.
The Bottom Line
BPC-157's Category 2 classification in September 2023 blocked legal compounding and drove demand to unregulated gray-market sources. The February 2026 reclassification announcement would restore compounding access but does not change the evidence base: roughly 30 human subjects across three pilot studies, extensive animal data, and no completed Phase III clinical trial. The regulatory classification has moved. The science has not.
Sources & References
- 1RPEP-00279·Sikirić, P et al. (1993). “The Stomach May Protect the Entire Body During Stress Through Peptides Like BPC 157.” Journal of physiology.Study breakdown →PubMed →↩
- 2RPEP-00561·Sikiric, P (1999). “BPC-157: A Comprehensive Review of Its Healing Effects Across Multiple Organ Systems.” Inflammopharmacology.Study breakdown →PubMed →↩
- 3RPEP-00860·Staresinic, M et al. (2003). “BPC-157 Accelerates Achilles Tendon Healing and Directly Stimulates Tendon Cell Growth.” Journal of orthopaedic research : official publication of the Orthopaedic Research Society.Study breakdown →PubMed →↩
- 4RPEP-01458·Brcic, L et al. (2009). “BPC-157 Promotes Healing Through Angiogenesis in Both Muscle and Tendon.” Journal of physiology and pharmacology : an official journal of the Polish Physiological Society.Study breakdown →PubMed →↩
- 5RPEP-00555·Sebecić, B et al. (1999). “BPC-157 Heals Bone Defects in Rabbits as Effectively as Growth Factors.” Bone.Study breakdown →PubMed →↩
- 6RPEP-05748·Seiwerth, Sven et al. (2021). “BPC-157: A Gastric Peptide That Accelerates Wound Healing Across Nearly Every Tissue Type.” Frontiers in pharmacology.Study breakdown →PubMed →↩
- 7RPEP-04861·Hsieh, Ming-Jer et al. (2020). “BPC-157 Relaxes Blood Vessels Through a Mapped Molecular Pathway Involving Nitric Oxide.” Scientific reports.Study breakdown →PubMed →↩
- 8RPEP-13892·Vasireddi, Nikhil et al. (2025). “BPC-157 for Sports Injuries: What a Systematic Review of the Evidence Actually Shows.” HSS journal : the musculoskeletal journal of Hospital for Special Surgery.Study breakdown →PubMed →↩
- 9RPEP-08643·Lee, Edwin et al. (2024). “BPC-157 Injection Resolved Bladder Pain Symptoms in 10 of 12 Women: A Pilot Study.” Alternative therapies in health and medicine.Study breakdown →PubMed →↩
- 10RPEP-12011·Lee, Edwin et al. (2025). “First Study of Intravenous BPC-157 in Humans Shows No Side Effects in Two Patients.” Alternative therapies in health and medicine.Study breakdown →PubMed →↩
- 11RPEP-04213·Gwyer, Daniel et al. (2019). “BPC-157 for Tendon, Ligament, and Muscle Healing: What the Animal Research Shows.” Cell and tissue research.Study breakdown →PubMed →↩
- 12RPEP-05138·Sikiric, Predrag et al. (2020). “BPC-157 Review: From Stomach Protection to Full-Body Organ Defense.” Gut and liver.Study breakdown →PubMed →↩