GHK-Cu: The Copper Peptide That Modulates 4,000 Genes
GHK-Cu Copper Peptide Biology
4,000+ Genes Affected
GHK-Cu, a naturally occurring tripeptide-copper complex, up- and downregulates over 4,000 human genes involved in tissue repair, inflammation, antioxidant defense, and nervous system function.
Pickart et al., BioMed Research International, 2015
Pickart et al., BioMed Research International, 2015
If you only read one thing
GHK-Cu is a tiny, natural molecule in your blood — just three amino acids and a copper atom — that your body uses as a repair signal. It tells cells to make collagen, heal wounds, fight inflammation, and protect against damage. The remarkable part: it affects over 4,000 genes. The catch: your levels drop 60% as you age. Topical copper peptide creams do work for skin, but whether replacing GHK-Cu throughout your body could slow aging is still an unanswered question.
In 1973, a biochemist named Loren Pickart noticed something unexpected. When liver tissue from patients aged 60-80 was incubated in blood plasma from young donors (aged 20-25), the old cells started synthesizing proteins at rates resembling younger tissue. The active factor turned out to be a tiny tripeptide: glycyl-L-histidyl-L-lysine, bound to a copper ion. That molecule, now known as GHK-Cu, has since been shown to influence the expression of more than 4,000 human genes, touching pathways involved in tissue repair, inflammation, antioxidant defense, nerve regeneration, and cancer suppression.[1]
Three amino acids and one copper ion producing effects this broad raises an obvious question: how? This article traces GHK-Cu from Pickart's discovery through its molecular biology, clinical evidence, gene expression data, and the open questions that remain. For detailed coverage of specific aspects, see the cluster articles on GHK-Cu and DNA repair, stem cell regeneration, antioxidant effects, and age-related decline. For the skincare application of copper peptides more broadly, see Copper Peptides in Skincare.
Key Takeaways
- In 1973, old liver tissue bathed in young people's blood suddenly started acting young — GHK-Cu was the reason.
- A molecule of just three amino acids plus copper switches more than 4,000 human genes on or off.
- The amount of GHK-Cu circulating in your blood drops by roughly 60% between age 20 and 60.
- Copper is dangerous to cells on its own, but GHK carries it in safely and hands it off.
- In lab cultures, GHK-Cu boosts collagen output 70% and glycosaminoglycan production by over 200%.
- It's well studied for skin, but nobody has tested whether restoring GHK-Cu actually slows aging anywhere else.
- Scientists still don't fully understand how three amino acids can reprogram thousands of genes at once.
What Is GHK-Cu?
GHK-Cu is a tripeptide consisting of glycine, L-histidine, and L-lysine, complexed with a single copper(II) ion. The histidine residue provides the primary copper-binding site through its imidazole nitrogen, with additional coordination from the peptide backbone and the amino terminus. The resulting complex has a molecular weight of approximately 403 daltons, making it one of the smallest biologically active peptide complexes known.
GHK exists naturally in human plasma, saliva, and urine. In plasma, it is released through enzymatic degradation of larger proteins, particularly SPARC (secreted protein acidic and rich in cysteine, also called osteonectin) and collagen. The peptide binds copper(II) with high affinity (log K = 16.44 at physiological pH), and this copper binding is essential for many of its biological activities. When GHK complexes with copper, it silences copper's redox activity, preventing the generation of damaging hydroxyl radicals while still allowing copper to be delivered into cells for use as an enzymatic cofactor.
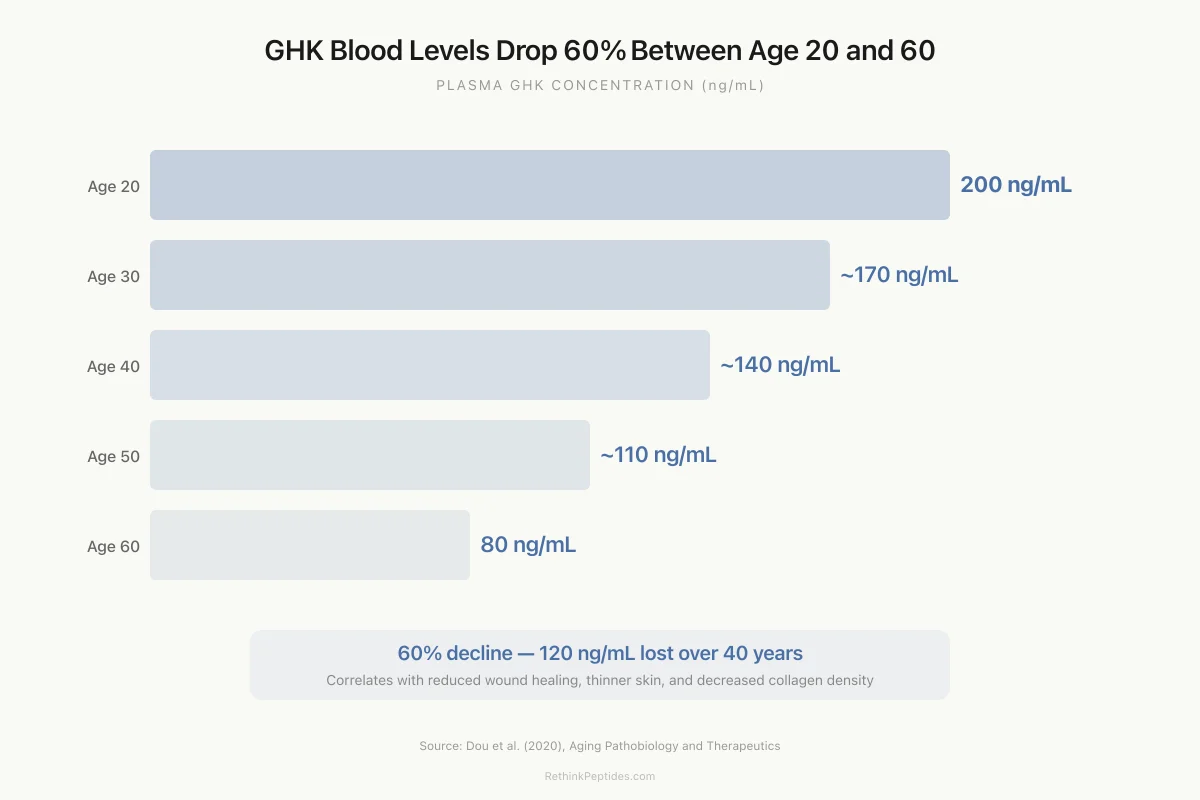
The decline in plasma GHK-Cu with age is well-documented. Levels average approximately 200 ng/mL in young adults (age 20) and fall to roughly 80 ng/mL by age 60.[2] This 60% decline parallels the age-related decrease in wound healing capacity, collagen production, and tissue repair that characterizes biological aging, though causation has not been established.
Discovery: Old Liver Meets Young Blood
Pickart's 1973 discovery began with a comparison of fibrinogen synthesis in human liver tissue. Liver samples from older patients (60-80 years) produced more fibrinogen than samples from younger donors (20-25 years), a pattern associated with chronic inflammation and age-related liver dysfunction. When older liver cells were cultured in plasma from the younger group, their protein synthesis patterns shifted toward the younger profile.
Pickart isolated the responsible factor and identified it as a small peptide with the sequence glycyl-L-histidyl-L-lysine. Subsequent work established that the peptide was naturally complexed with copper in plasma and that both the peptide and the copper were necessary for its biological effects.[3]
The discovery's implications went beyond liver biology. If a simple tripeptide could shift old tissue behavior toward a younger pattern, it suggested a broader role in tissue maintenance and repair that might operate across multiple organ systems. Over the following decades, Pickart (who passed away in 2023 at age 85) and collaborators documented GHK-Cu's effects on wound healing, collagen production, hair growth, nerve outgrowth, and inflammation, building a substantial body of evidence that the peptide functions as a systemic tissue repair signal rather than having effects limited to any single tissue type.
Collagen, Glycosaminoglycans, and Wound Healing
The earliest mechanistic work on GHK-Cu focused on extracellular matrix production, the structural scaffolding that holds tissues together.
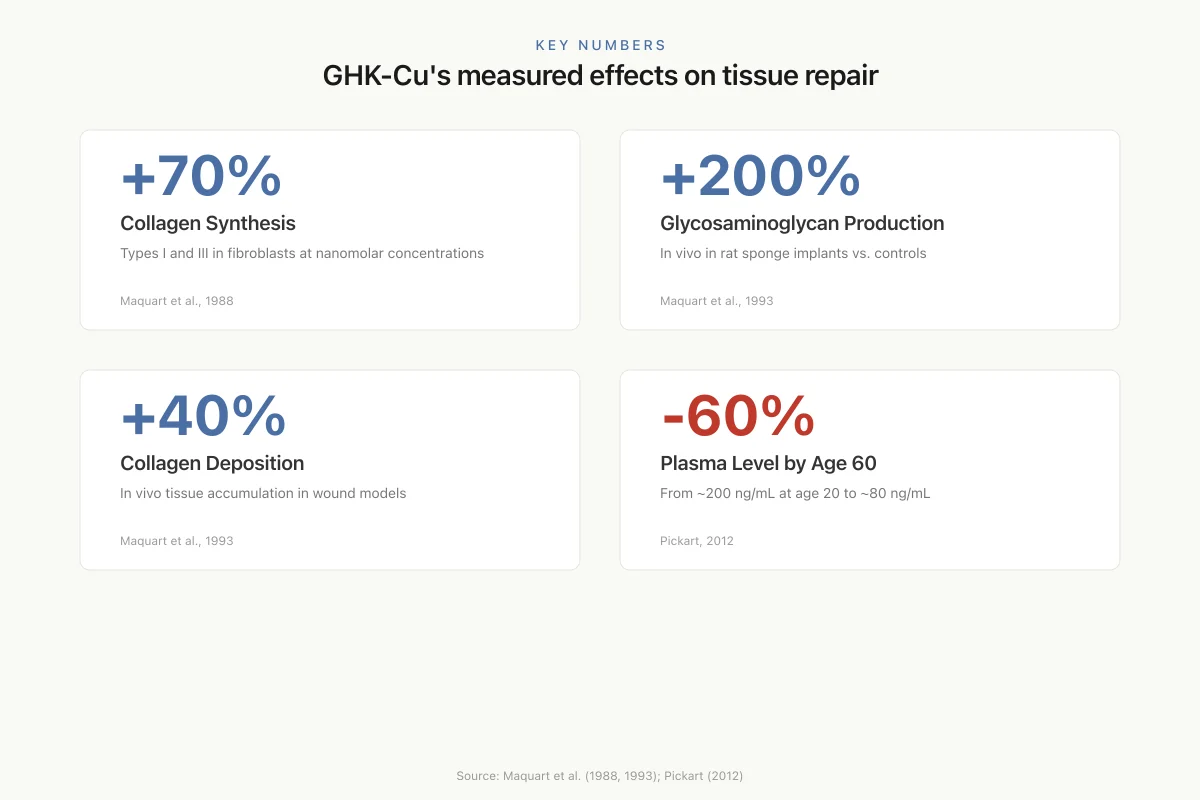
Collagen synthesis. Maquart et al. (1988) demonstrated that GHK-Cu stimulated collagen synthesis in human fibroblast cultures by approximately 70% at nanomolar concentrations, with effects on both type I and type III collagen.[4] The same group showed in 1993 that GHK-Cu stimulated connective tissue accumulation in vivo in rats, increasing collagen deposition in sponge implants by 40% and glycosaminoglycan synthesis by over 200% compared to controls.[5] These are substantial effects for a naturally occurring tripeptide at physiological concentrations.
Key Numbers
GHK-Cu's measured effects on tissue repair
+70%
Collagen Synthesis
Types I and III in fibroblasts at nanomolar concentrations
Maquart et al., 1988
+200%
Glycosaminoglycan Production
In vivo in rat sponge implants vs. controls
Maquart et al., 1993
+40%
Collagen Deposition
In vivo tissue accumulation in wound models
Maquart et al., 1993
−60%
Plasma Level by Age 60
From ~200 ng/mL at age 20 to ~80 ng/mL
Pickart, 2012
Source: Maquart et al. (1988, 1993); Pickart (2012)
 View as image
View as imageWound healing applications. Arul et al. (2005) incorporated biotinylated GHK peptide into collagenous matrices to create wound healing biomaterials, demonstrating enhanced fibroblast attachment and proliferation.[6] GHK-Cu has been shown to attract immune cells to wound sites (chemotaxis for macrophages and mast cells), stimulate blood vessel growth (angiogenesis), and promote nerve regeneration, addressing multiple phases of the wound healing process simultaneously.
Skin applications. Miller et al. (2006) tested topical copper tripeptide complex on skin healing after CO2 laser resurfacing in a controlled study comparing three treatment groups: copper peptide cream, tretinoin (retinoic acid) cream, and vitamin C cream. The copper peptide treatment produced significantly better outcomes in skin thickness, density, and overall clinical appearance than both comparators.[7] Patients treated with copper peptide showed faster re-epithelialization and reduced post-inflammatory erythema. This study remains one of the most clinically relevant demonstrations of GHK-Cu's dermatological effects and has driven the peptide's incorporation into numerous commercial skincare products.
Additional skin studies have examined GHK-Cu for photoaging, where UV-damaged skin shows characteristic collagen degradation and elastin abnormalities. The peptide's combined effect of stimulating new collagen production while suppressing matrix metalloproteinase expression (the enzymes that break down collagen) provides a mechanistically rational approach to photoaging that operates through different pathways than retinoids or sunscreen.
The Gene Expression Revolution: 4,000+ Genes
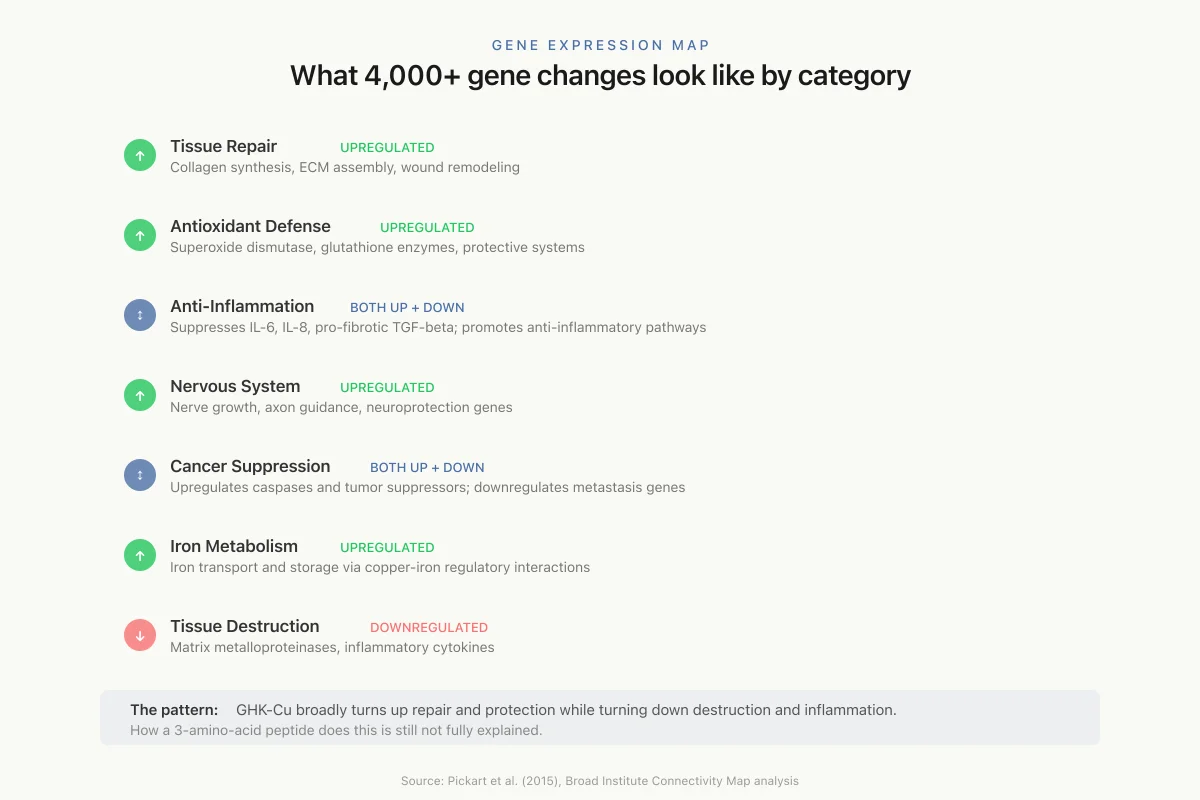
The most striking finding in GHK-Cu research came not from traditional biochemistry but from genomic analysis. Using the Broad Institute's Connectivity Map (cMap), a database that profiles gene expression changes produced by thousands of bioactive molecules, Pickart and Vasquez-Soltero identified that GHK-Cu influences the expression of more than 4,000 human genes.[1]
The affected genes cluster into several functional categories:
Tissue repair and remodeling. GHK-Cu upregulates genes involved in collagen synthesis, extracellular matrix assembly, and tissue remodeling while downregulating genes associated with tissue destruction (matrix metalloproteinases, inflammatory cytokines).
Antioxidant defense. Multiple antioxidant genes are upregulated, including those encoding superoxide dismutase, glutathione-related enzymes, and other protective systems. This genomic signature is consistent with the observed protective effects against oxidative stress in cell culture and animal studies. For deeper coverage, see GHK-Cu as an Antioxidant.
Anti-inflammatory effects. GHK-Cu suppresses expression of genes encoding pro-inflammatory cytokines (IL-6, IL-8, TGF-beta in its pro-fibrotic context) while promoting anti-inflammatory pathways. This dual action (repair promotion plus inflammation suppression) is unusual and may explain why GHK-Cu improves outcomes in chronic wounds where persistent inflammation blocks healing.
Nervous system. Genes involved in nerve growth, axon guidance, and neuroprotection are modulated, consistent with observed effects on nerve regeneration in wound models. For the DNA repair angle, see GHK-Cu and DNA Repair.
Cancer-related genes. GHK-Cu upregulates several tumor suppressor genes (caspases, growth arrest and DNA damage-inducible genes) and downregulates genes associated with metastasis, invasion, and angiogenesis. A study examining GHK's effects on human breast cancer MCF7 and prostate cancer PC3 cell lines found gene expression changes consistent with reduced proliferative and invasive capacity. Whether any of this translates to cancer-preventive or anti-metastatic effects in living organisms is unknown, and cancer biology is far too complex for cell culture gene expression changes to predict clinical outcomes.
Iron metabolism and hematopoiesis. GHK-Cu modulates several genes involved in iron transport and storage, likely through the intimate connection between copper and iron metabolism (copper-dependent ceruloplasmin is essential for iron release from cells). This connection to iron homeostasis may have broader implications for anemia and age-related changes in blood cell production.
Gene Expression Map
What 4,000+ gene changes look like by category
Tissue Repair
upregulatedCollagen synthesis, ECM assembly, wound remodeling
Antioxidant Defense
upregulatedSuperoxide dismutase, glutathione enzymes, protective systems
Anti-Inflammation
bothSuppresses IL-6, IL-8, pro-fibrotic TGF-beta; promotes anti-inflammatory pathways
Nervous System
upregulatedNerve growth, axon guidance, neuroprotection genes
Cancer Suppression
bothUpregulates caspases and tumor suppressors; downregulates metastasis genes
Iron Metabolism
upregulatedIron transport and storage via copper-iron regulatory interactions
Tissue Destruction
downregulatedMatrix metalloproteinases, inflammatory cytokines
The pattern: GHK-Cu broadly turns up repair and protection genes while turning down destruction and inflammation genes. How a 3-amino-acid peptide does this is still not fully explained.
Source: Pickart et al. (2015), Broad Institute Connectivity Map analysis
 View as image
View as imageThe breadth of gene modulation raises a fundamental question: how does a three-amino-acid peptide influence thousands of genes? The copper delivery hypothesis proposes that GHK-Cu regulates intracellular copper levels, which in turn affect copper-dependent transcription factors and enzymes. But this alone does not explain the specificity of the gene expression changes. Other proposed mechanisms include direct interaction with cell surface integrins (particularly alpha-2-beta-1), effects on iron metabolism through copper-iron regulatory interactions, and epigenetic modifications. The honest answer is that no single mechanism adequately explains the scope of GHK-Cu's gene modulation effects. This remains an active area of investigation.
Beyond Skin: Systemic Effects in Animal Models
While GHK-Cu is best known for dermatological applications, animal studies suggest effects that extend well beyond skin.
Pulmonary fibrosis. Ma et al. (2020) demonstrated that GHK-Cu protected against bleomycin-induced pulmonary fibrosis in mice via anti-inflammatory mechanisms, reducing fibrotic tissue accumulation and preserving lung function.[8] This is particularly relevant because pulmonary fibrosis involves the same extracellular matrix dysregulation and chronic inflammation that GHK-Cu modulates in skin wounds, suggesting the peptide's effects are not tissue-specific.
Cognitive health. Pickart's 2012 review examined GHK-Cu's potential role in preventing oxidative stress and degenerative conditions of aging, with a focus on cognitive health implications.[2] The decline in GHK-Cu levels correlates temporally with increased risk of neurodegenerative conditions, and the peptide's antioxidant and anti-inflammatory gene expression profile is theoretically relevant to brain aging. However, no human clinical studies have tested GHK-Cu for cognitive outcomes.
Copper homeostasis. Bossak-Ahmad et al. (2020) characterized the ternary copper complex formed between GHK, copper(II), and cis-urocanic acid (a UV-absorbing compound in skin), demonstrating a potential mechanism for GHK-Cu to function as a solar photoprotectant while maintaining its copper-delivery function.[9]
Stem cell effects. GHK-Cu has been reported to influence mesenchymal stem cell differentiation and proliferation, raising the possibility that some of its tissue repair effects operate through stem cell recruitment and activation rather than solely through direct effects on mature cells. If GHK-Cu can mobilize resident stem cell populations in wound beds and aging tissues, this would help explain the breadth of its regenerative effects across different tissue types. This is covered in detail in GHK-Cu and Stem Cells.
Age-Related Decline and the Anti-Aging Question
The 60% decline in plasma GHK-Cu from age 20 to 60 has prompted speculation that GHK-Cu supplementation could slow or partially reverse aspects of biological aging.[10] The logic: if the peptide declines with age and its gene expression profile favors tissue repair, restoration of youthful levels might restore youthful repair capacity. For the full analysis, see How GHK-Cu Declines with Age.
Age-Related Decline
GHK Blood Levels Drop 60% Between Age 20 and 60
Plasma GHK concentration (ng/mL) across the adult lifespan
60%
decline from age 20 to 60
120 ng/mL
lost over 40 years
Source: Dou et al. (2020), Aging Pathobiology and Therapeutics
 View as image
View as imageThe evidence supporting this hypothesis is suggestive but not conclusive. The gene expression data shows that GHK-Cu's genomic signature opposes many age-associated gene expression changes. Animal wound healing studies show improved outcomes. The topical skin data from Miller et al. shows measurable improvements in skin parameters.
What is missing: randomized controlled trials of systemic GHK-Cu supplementation measuring clinically relevant aging outcomes. The published human data is limited to topical skin applications and cosmetic endpoints. Whether injected, oral, or intranasal GHK-Cu would replicate the gene expression changes seen in cell culture studies in living humans is unproven. Bioavailability, half-life, and dose-response data in humans are sparse. The peptide is readily degraded by peptidases, creating delivery challenges similar to those faced by many therapeutic peptides.
The skincare industry has embraced GHK-Cu with considerable commercial enthusiasm, incorporating it into serums, creams, masks, and microneedling treatments at concentrations ranging from 0.01% to 1%. Some of these products have supporting evidence from controlled studies like Miller's laser resurfacing trial. Many do not. The gap between a skincare brand citing "copper peptide research" and the actual concentration, formulation stability, and skin penetration of their specific product is often large. GHK-Cu is susceptible to oxidation and degradation, and formulation chemistry matters for whether the active peptide reaches viable skin cells. Liposomal encapsulation and microneedle-mediated delivery have been explored as strategies to improve GHK-Cu skin penetration beyond what simple cream application achieves. The gap between the gene expression research (which suggests systemic biological effects) and the actual human evidence (which is limited to topical skin endpoints) is substantial. For related coverage of collagen peptides in skin and joints, see Collagen Peptides for Joint Health and Elastin Peptides.
Delivery Challenges and Formulation Science
GHK-Cu's therapeutic potential is constrained by the same delivery challenges that limit many bioactive peptides. The tripeptide is rapidly degraded by serum peptidases, with an estimated plasma half-life measured in minutes. Oral bioavailability is negligible due to gastrointestinal degradation. These pharmacokinetic limitations explain why the most successful applications have been topical (where the peptide is applied directly to the target tissue) rather than systemic.
Several engineering approaches aim to extend GHK-Cu's stability and reach. Liposomal encapsulation protects the peptide from enzymatic degradation and improves skin penetration. Dymek et al. (2023) developed GHK-Cu-loaded liposomes for cosmetic applications, demonstrating improved stability and sustained release profiles compared to free peptide. Li et al. (2015) explored microneedle-mediated delivery as a method to bypass the stratum corneum barrier that limits conventional topical absorption.
Peptide-polymer conjugates, nanoparticle encapsulation, and incorporation into hydrogel wound dressings represent additional delivery strategies under investigation. Each approach faces trade-offs between stability, bioavailability, manufacturing complexity, and cost. The challenge is not whether GHK-Cu has biological activity (that is well-established) but whether that activity can be delivered to tissues at concentrations and durations sufficient to produce clinical effects beyond the skin surface.
For systemic applications targeting pulmonary fibrosis, cognitive decline, or general anti-aging effects, the delivery problem becomes substantially harder. Injectable formulations with sustained-release carriers are one possibility, but no such product has entered clinical testing. The regulatory pathway for a systemic GHK-Cu therapeutic would require the full spectrum of preclinical toxicology, pharmacokinetics, and clinical trials that have not been conducted.
What We Know and What We Don't
GHK-Cu occupies an unusual position in peptide science. The basic biochemistry is well-established: it is a real endogenous peptide, it genuinely binds copper, it declines with age, and it produces measurable effects on collagen synthesis, wound healing, and gene expression. The Broad Institute gene expression data is particularly compelling because it was generated through an unbiased, genome-wide approach rather than hypothesis-driven experiments.
The limitations are equally real. Nearly all mechanistic work comes from cell culture and animal studies. Human data is limited primarily to topical dermatological applications. The mechanism linking three amino acids to 4,000+ gene changes is not understood. Whether restoring youthful GHK-Cu levels would have clinically meaningful effects on aging is speculative. And the peptide's instability and rapid degradation create practical challenges for any systemic application.
GHK-Cu is neither a miracle anti-aging compound nor a molecule without substance. It sits in the middle ground that characterizes many endogenous bioactive peptides: genuine biology, genuine effects, and genuine unanswered questions about how far those effects can be pushed therapeutically.
The field would benefit from three specific developments. First, a well-powered randomized controlled trial of topical GHK-Cu for a defined dermatological condition (photoaging, chronic wounds, or post-surgical healing) with objective endpoints including histological analysis and validated scoring systems. Second, pharmacokinetic studies characterizing systemic GHK-Cu levels after various routes of administration (topical, injectable, oral with encapsulation) to determine whether therapeutically relevant tissue concentrations are achievable beyond the skin. Third, mechanistic research explaining how a tripeptide produces gene expression changes across 4,000+ genes, which would both satisfy scientific curiosity and enable rational design of more potent or selective GHK-Cu derivatives.
Until those studies exist, GHK-Cu remains a fascinating molecule with strong preclinical credentials, proven topical skin benefits, and unresolved systemic therapeutic potential. The research base is genuine; the clinical translation is incomplete.
The Bottom Line
GHK-Cu is a naturally occurring copper tripeptide discovered in 1973 that declines 60% between ages 20 and 60. Gene expression analysis shows it modulates over 4,000 human genes, favoring tissue repair, antioxidant defense, and anti-inflammatory pathways. Human evidence is strongest for topical skin applications, where controlled studies show improved healing and skin quality. The mechanism linking three amino acids to thousands of gene changes remains incompletely understood. The gap between GHK-Cu's broad gene expression effects and its limited human clinical evidence is the central unanswered question in the field.
Sources & References
- 1RPEP-02768·Pickart, Loren et al. (2015). “GHK Peptide as a Natural Modulator of Multiple Cellular Pathways in Skin Regeneration..” BioMed research international.Study breakdown →PubMed →↩
- 2RPEP-02037·Pickart, Loren et al. (2012). “The human tripeptide GHK-Cu in prevention of oxidative stress and degenerative conditions of aging: implications for cognitive health..” Oxidative medicine and cellular longevity.Study breakdown →PubMed →↩
- 3RPEP-01402·Pickart, Loren (2008). “GHK-Cu: The Complete Review of This Tissue Remodeling Tripeptide's Biology and Applications.” Journal of biomaterials science. Polymer edition.Study breakdown →PubMed →↩
- 4RPEP-00080·Maquart, F X et al. (1988). “GHK-Cu Stimulated Collagen Production at Extremely Low Concentrations.” FEBS letters.Study breakdown →PubMed →↩
- 5RPEP-00269·Maquart, F X et al. (1993). “GHK-Cu Peptide Accelerates Wound Healing and Tissue Repair in Rats.” The Journal of clinical investigation.Study breakdown →PubMed →↩
- 6RPEP-01007·Arul, V et al. (2005). “GHK Peptide in a Collagen Scaffold Accelerates Wound Healing in Rats.” Journal of biomedical materials research. Part B.Study breakdown →PubMed →↩
- 7RPEP-01165·Miller, Timothy R et al. (2006). “Copper Peptide GHK-Cu Cream Improves Healing After Laser Skin Resurfacing in Humans.” Archives of facial plastic surgery.Study breakdown →PubMed →↩
- 8RPEP-04979·Ma, Wen-Hui et al. (2020). “GHK-Cu Peptide Reduces Lung Fibrosis and Inflammation in Mice by Blocking TGF-β Scarring Pathway.” Life sciences.Study breakdown →PubMed →↩
- 9RPEP-04672·Bossak-Ahmad, Karolina et al. (2020). “The GHK Copper Peptide May Form a Three-Way Complex With Urocanic Acid in Your Skin.” International journal of molecular sciences.Study breakdown →PubMed →↩
- 10RPEP-04782·Dou, Yan et al. (2020). “The potential of GHK as an anti-aging peptide..” Aging pathobiology and therapeutics.Study breakdown →PubMed →↩