BPC-157 Legal Status in 2026: Where Things Stand
BPC-157 Regulation
Category 1 reclassified
On February 27, 2026, HHS announced BPC-157 would move from Category 2 (cannot compound) back to Category 1 (can compound with prescription).
HHS Secretary RFK Jr., February 27, 2026
HHS Secretary RFK Jr., February 27, 2026
If you only read one thing
BPC-157 is easier to get legally in 2026 than it was in 2025. In February 2026, the U.S. government said compounding pharmacies can start making it again with a doctor's prescription. But this is not the same as the FDA saying it works — no human clinical trial has ever finished. Athletes still can't use it. And much of what people buy online still comes from unregulated 'research chemical' websites where quality is hit or miss.
BPC-157's legal status has shifted three times in four years. WADA banned it in January 2022. The FDA classified it as Category 2 in September 2023, effectively blocking compounding pharmacies from manufacturing it. Then on February 27, 2026, HHS Secretary Robert F. Kennedy Jr. announced that BPC-157 would be among 14 peptides moving back to Category 1, restoring compounding pharmacy access with a physician's prescription.
Each of these changes carries specific implications for clinicians, patients, athletes, and researchers. This article maps the current regulatory landscape as of March 2026, what has changed, what has not, and what the remaining ambiguities are.
Key Takeaways
- BPC-157's legal status has flipped three times in four years — the latest change: it's coming back in 2026.
- A doctor can prescribe it again and a pharmacy can make it — but that's still not FDA approval.
- No human clinical trial for BPC-157 has ever been completed. Not one.
- Athletes still can't use it — the WADA ban runs on a separate rulebook from the FDA.
- Most BPC-157 being sold online still comes from unregulated "research chemical" websites.
- The 2023 ban pushed more demand onto the gray market, not less.
- As of early 2026 the formal FDA paperwork still isn't published — so the return isn't fully live.
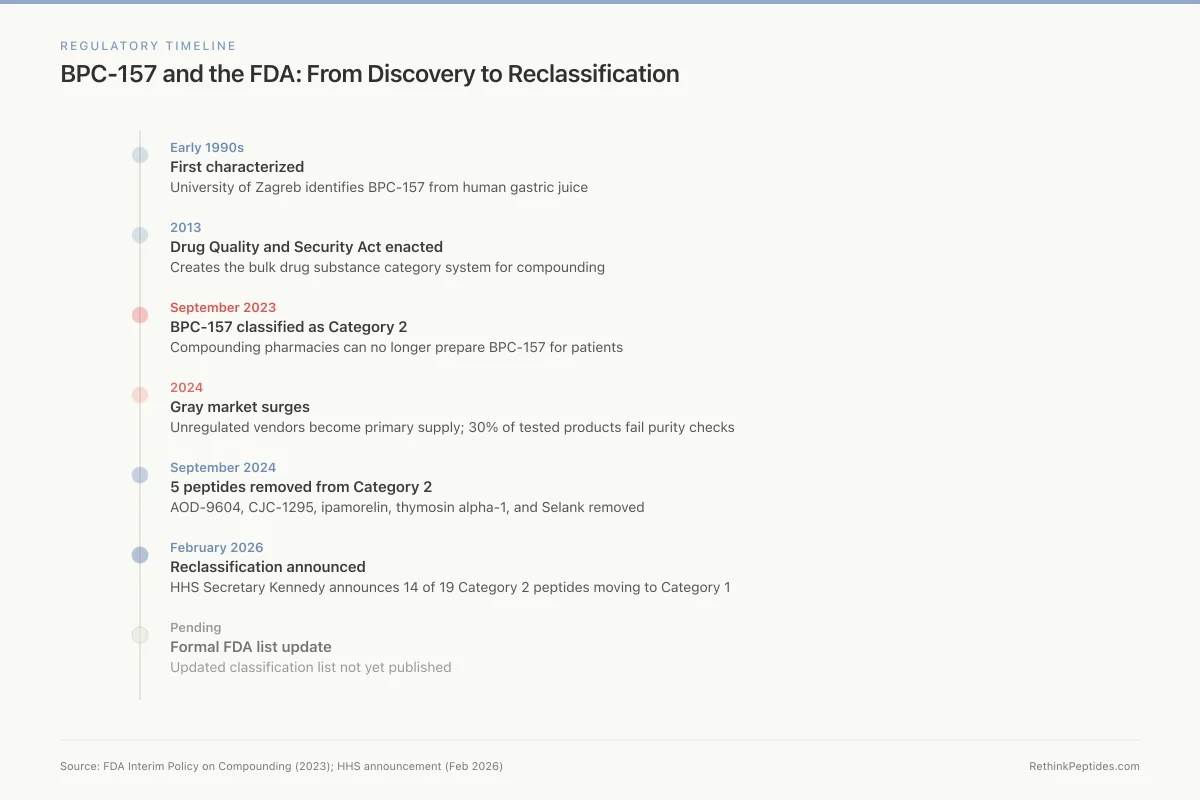
Regulatory Timeline
BPC-157 and the FDA: From Discovery to Reclassification
Early 1990s
First characterized
University of Zagreb identifies BPC-157 from human gastric juice
2013
DQSA enacted
Drug Quality and Security Act creates the bulk drug substance category system
Sept 2023
Category 2
FDA classifies BPC-157 as Category 2 — compounding pharmacies can no longer prepare it
2024
Gray market surges
Unregulated "research use only" vendors become primary supply; 30% of tested products fail purity checks
Sept 2024
5 peptides removed
AOD-9604, CJC-1295, ipamorelin, thymosin alpha-1, and Selank removed from Category 2
Feb 2026
Reclassification announced
HHS Secretary Kennedy announces 14 of 19 Category 2 peptides moving back to Category 1
Pending
Formal FDA list update
Updated classification list not yet published — compounding pharmacies waiting for formal guidance
Source: FDA Interim Policy on Compounding (2023); HHS announcement (Feb 2026)
 View as image
View as imageThe Category 2 classification: September 2023
In September 2023, the FDA added BPC-157 to its Category 2 bulk drug substance list under the Interim Policy on Compounding Using Bulk Drug Substances. This classification applied to both 503A pharmacies (traditional compounding pharmacies filling individual prescriptions) and 503B outsourcing facilities (which produce larger batches without patient-specific prescriptions).
Category 2 means the FDA evaluated the substance and determined it cannot be used in compounding. The agency's stated rationale cited three concerns: potential immune reactions, manufacturing impurities, and a lack of human safety data. The FDA noted it could take enforcement action against compounding pharmacies that continued to produce BPC-157.
This was a substantial change. Before Category 2 classification, compounding pharmacies across the United States routinely prepared BPC-157 for patients with physician prescriptions. The peptide had become one of the most commonly compounded peptides in functional medicine and sports medicine practices. Clinicians who had been prescribing it faced a choice between discontinuing a therapy they believed was working or accepting regulatory risk.
The Category 2 classification created legal uncertainty beyond just BPC-157. Eighteen other peptides were also placed in Category 2, including thymosin alpha-1, TB-500, CJC-1295, and ipamorelin. The breadth of the classification effectively shut down a substantial portion of the peptide compounding industry.
The WADA ban: January 2022
The World Anti-Doping Agency added BPC-157 to its Prohibited List effective January 1, 2022, classified under Section S0: Non-Approved Substances. Section S0 covers substances used for doping that are not approved by any governmental regulatory health authority for human therapeutic use, including drugs under pre-clinical or clinical development and discontinued drugs.
USADA (the U.S. Anti-Doping Agency) has specifically warned athletes about BPC-157, noting that it "is not a legitimate dietary supplement or approved medication" and that "there is no clinical basis for granting a TUE [Therapeutic Use Exemption] for this substance because it is not an approved therapeutic agent in any country."
The WADA ban operates independently from the FDA classification. Even with BPC-157's reclassification to Category 1 in 2026, the WADA prohibition remains in effect. Athletes subject to WADA testing who use BPC-157 risk a positive test and sanction regardless of whether they obtained it legally through a compounding pharmacy with a prescription. A 2025 systematic review noted that "despite lacking US Food and Drug Administration approval and its use being banned in professional sports, it is increasingly used by clinicians and athletes."[1]
The February 2026 reclassification
On February 27, 2026, HHS Secretary Robert F. Kennedy Jr. announced that approximately 14 of the 19 peptides on the Category 2 list would move back to Category 1. BPC-157 was named among the reclassified peptides. The announcement was made on the Joe Rogan Experience (Episode #2461).
The 14 peptides moving to Category 1 include:
- BPC-157
- Thymosin Alpha-1
- Thymosin Beta-4/TB-500
- PT-141/Bremelanotide
- Ipamorelin
- GHRP-2 and GHRP-6
- CJC-1295
- Selank and Semax
- Hexarelin
- DSIP
- Epitalon
- Pinealon
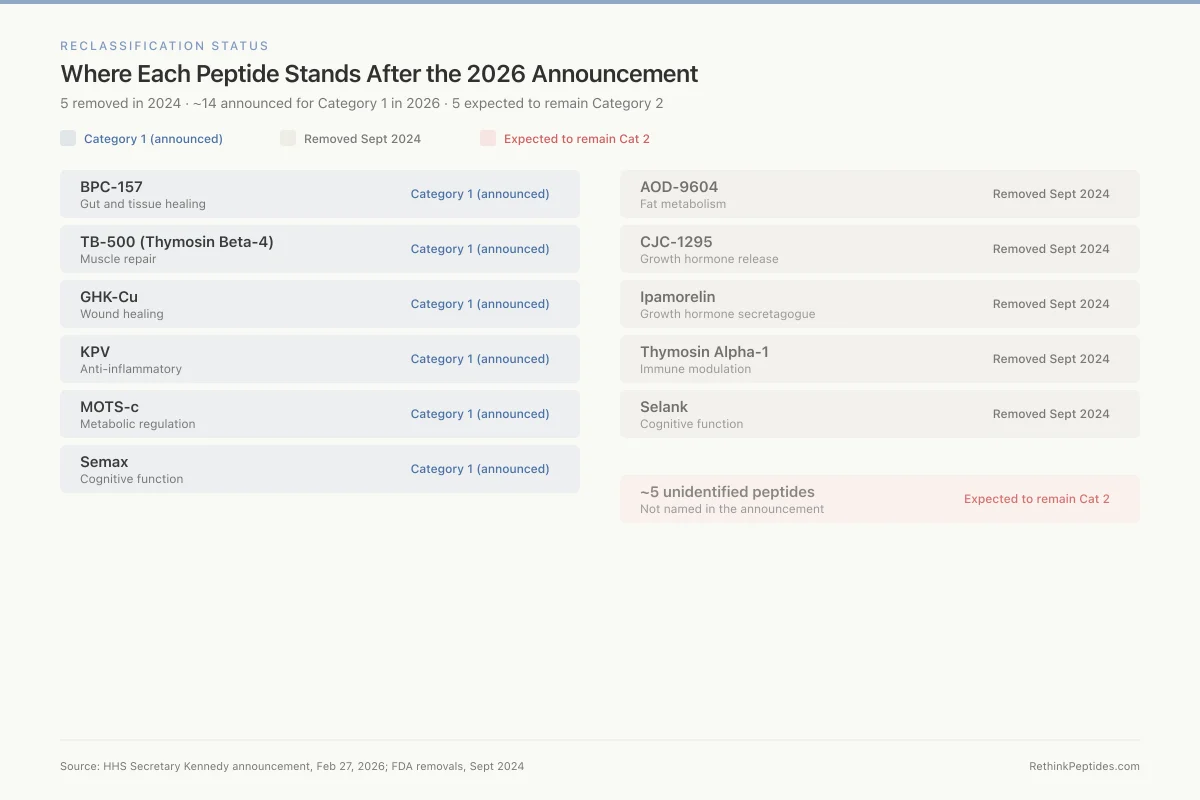
Five peptides remain in Category 2. The formal identification of which five remain restricted has not been published by the FDA as of March 2026.
Reclassification Status
Where Each Peptide Stands After the 2026 Announcement
5 removed in 2024 · ~14 announced for Category 1 in 2026 · 5 expected to remain Category 2
BPC-157
Gut and tissue healing
TB-500 (Thymosin Beta-4)
Muscle repair
GHK-Cu
Wound healing
KPV
Anti-inflammatory
MOTS-c
Metabolic regulation
Semax
Cognitive function
AOD-9604
Fat metabolism
CJC-1295
Growth hormone release
Ipamorelin
Growth hormone secretagogue
Thymosin Alpha-1
Immune modulation
Selank
Cognitive function
~5 unidentified peptides
Not named in the announcement
Source: HHS Secretary Kennedy announcement, Feb 27, 2026; FDA removals, Sept 2024
 View as image
View as imageImplementation timeline
The reclassification is not immediate. The announced timeline includes:
- February 27, 2026: Official announcement
- March-April 2026: 30-day stakeholder comment period
- May-June 2026: FDA finalizes technical requirements for compounding
- July 2026+: State pharmacy boards implement local regulations
Until the formal FDA updated list is published and state boards implement local rules, the legal status of compounding BPC-157 remains in a transitional period. Some compounding pharmacies have resumed preparation based on the announcement; others are waiting for formal guidance.
What Category 1 means in practice
Category 1 status allows licensed 503A and 503B compounding pharmacies to prepare BPC-157 under specific conditions:
- A physician must write a prescription for a specific patient (503A) or the facility must meet 503B outsourcing requirements
- The pharmacy must use pharmaceutical-grade raw materials meeting USP standards
- The compounded product must meet sterility and potency testing requirements
- The pharmacy must comply with state pharmacy board regulations, which vary by state
Reclassification is not FDA approval. BPC-157 has not undergone the Phase I, II, and III clinical trials required for FDA drug approval. No drug application (NDA or BLA) has been submitted for BPC-157 for any indication. Compounding pharmacies can prepare it, but no manufacturer can market it as an FDA-approved drug.
The legal challenge backdrop
The reclassification did not happen in a vacuum. Throughout 2025, the Outsourcing Facilities Association and other industry groups challenged FDA's peptide restrictions through federal litigation. While the most prominent cases involved GLP-1 agonists (semaglutide and tirzepatide compounding), the broader legal pressure on FDA's compounding authority created the political environment in which peptide reclassification became viable.
The legal arguments centered on whether the FDA's Category 2 process provided adequate notice and comment, whether the safety concerns cited were sufficient to justify the restriction, and whether the FDA overstepped its authority under the FD&C Act. These cases established that the peptide compounding regulatory framework was contested legal territory, not settled law.
The legal challenge created political pressure from multiple directions: compounding pharmacies losing revenue, clinicians losing prescribing options, patients losing access to therapies they valued, and advocacy organizations arguing the FDA had overstepped its statutory authority. The February 2026 reclassification can be understood partly as a response to this sustained pressure, combined with the new administration's broader skepticism of FDA regulatory restrictions on health products.
What buying BPC-157 looks like in 2026
BPC-157 has been available to purchase throughout the entire period of regulatory restriction. The Category 2 classification and WADA ban affected compounding pharmacies and athletes, respectively. They did not affect the research chemical market.
Buying Comparison
Research Chemical vs. Compounding Pharmacy: What You Actually Get
Research Chemical Vendor
Higher riskAvailability
Available throughout ban
Prescription
Not required
Quality control
Varies widely — some vendors provide CoAs, many do not
Purity
Unverified — studies find contamination, incorrect content, residual solvents
Typical cost
$30–$80 per vial (typical)
Oversight
None — labeled "for research use only"
Legal status
Legal to purchase (not a controlled substance)
Compounding Pharmacy (Category 1)
Lower riskAvailability
Resuming mid-2026 (pending state implementation)
Prescription
Required — physician must prescribe
Quality control
Pharmaceutical-grade raw materials, USP standards
Purity
Sterility and potency testing required
Typical cost
$150–$400+ per prescription
Oversight
FDA (503A/503B), state pharmacy boards
Legal status
Legal with prescription
Source: FDA compounding guidance; gray market vendor surveys
 View as image
View as imageResearch chemical vendors
BPC-157 is sold by dozens of online peptide vendors labeled "for research purposes only" or "not for human consumption." These products are manufactured by chemical synthesis companies without FDA oversight. Quality varies enormously. Some vendors provide third-party Certificates of Analysis (CoA) showing purity (>98% peptide content) and endotoxin testing. Others do not.
Purchasing BPC-157 as a research chemical is not illegal under federal law. BPC-157 is not a controlled substance under the Controlled Substances Act. It is not a Schedule I-V drug. The legal fiction of the "research use only" label allows vendors to sell and buyers to purchase without violating drug manufacturing or distribution laws. The FDA has generally not pursued enforcement against individual buyers of research peptides, focusing instead on manufacturers making therapeutic claims or selling products explicitly labeled for human use.
The quality concerns with research-grade peptides are not theoretical. Studies of gray-market peptide products have found issues including incorrect peptide content, bacterial contamination, heavy metals, and residual solvents from manufacturing. Without FDA oversight, the only quality assurance is the vendor's own testing and reputation. Third-party CoAs help, but verification of those certificates is rarely possible for individual buyers.
Safety
ModerateResearch chemical quality is not guaranteed
Concern
Research-grade BPC-157 is sold without FDA oversight. Independent testing of gray-market peptide products has found wrong peptide content, bacterial contamination, heavy metals, and leftover solvents from manufacturing. Because vendors label products 'for research use only,' no regulator is checking what's in the vial.
What the research says
Reputable vendors publish third-party Certificates of Analysis (CoAs). However, individual buyers rarely have a way to verify those certificates are real, or that the tested batch matches the vial they received.
Particularly relevant for: Anyone buying BPC-157 outside a compounding pharmacy
What to do
If you choose to purchase research-grade peptides, prefer vendors that publish batch-specific CoAs with mass spectrometry data and endotoxin testing. Understand that you are accepting unverified quality.
Independent gray-market peptide purity testing; FDA enforcement reports
Compounding pharmacies (post-reclassification)
With Category 1 status, licensed compounding pharmacies can prepare BPC-157 with a physician's prescription. This route offers several advantages over research chemicals: pharmaceutical-grade raw materials, sterility testing, accurate dosing, and a prescriber responsible for monitoring. It also costs more and requires a medical relationship.
The practical availability through compounding pharmacies will depend on how quickly individual pharmacies set up manufacturing, sourcing for pharmaceutical-grade BPC-157 bulk substance, and state pharmacy board timelines for implementing the federal guidance.
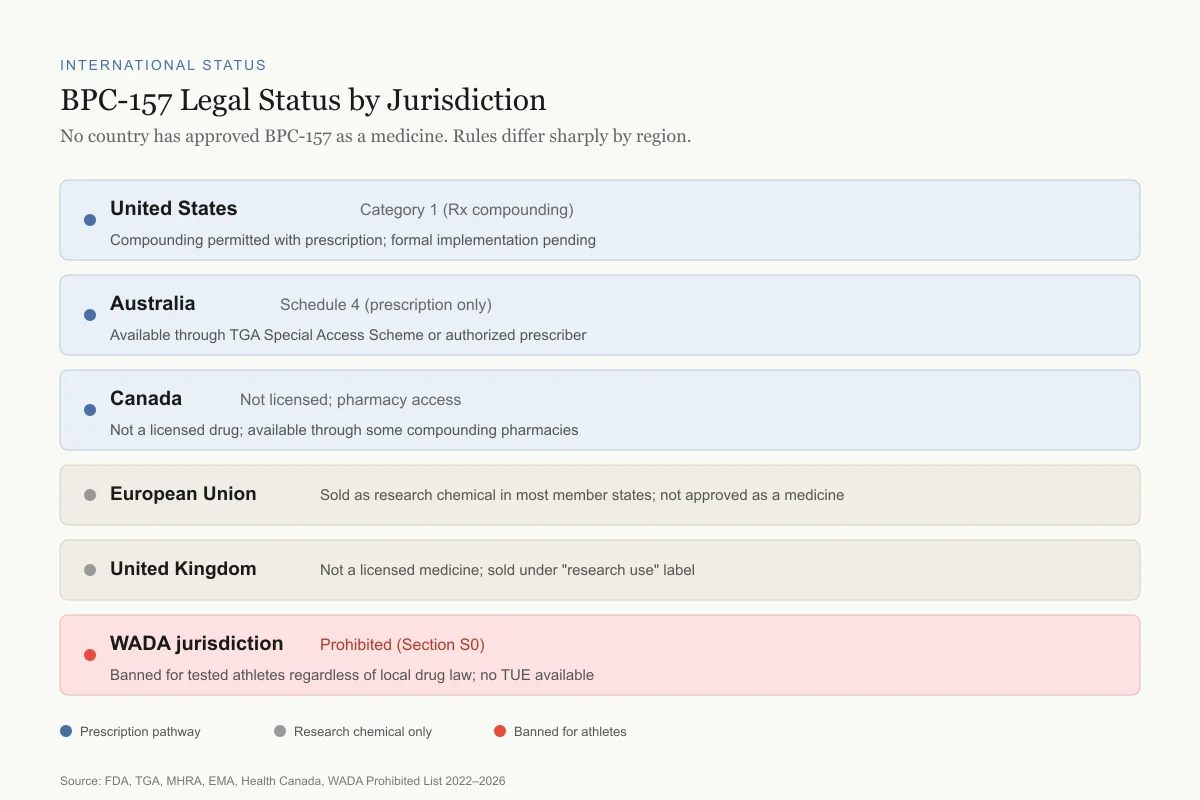
International status
International Status
BPC-157 Legal Status by Jurisdiction
No country has approved BPC-157 as a medicine. Rules differ sharply by region.
United States
Category 1 (Rx compounding)
Compounding permitted with prescription; formal implementation pending
Australia
Schedule 4 (prescription only)
Available through TGA Special Access Scheme or authorized prescriber
Canada
Not licensed; pharmacy access
Not a licensed drug; available through some compounding pharmacies
European Union
Research chemical
Not approved as a medicine; sold as research chemical in most member states
United Kingdom
Research chemical
Not a licensed medicine; sold under "research use" label
WADA jurisdiction
Prohibited (S0)
Banned for all tested athletes regardless of local drug law; no TUE available
Source: FDA, TGA, MHRA, EMA, Health Canada, WADA Prohibited List 2022–2026
 View as image
View as imageBPC-157's legal status varies by country:
- United States: Category 1 (compounding permitted with prescription as of the February 2026 announcement; formal implementation pending)
- European Union: Not approved as a medicine; available as a research chemical in most EU countries
- United Kingdom: Not a licensed medicine; sold as a research chemical
- Australia: Schedule 4 (prescription only) through the TGA Special Access Scheme or authorized prescriber pathway
- Canada: Not a licensed drug; available through some compounding pharmacies under physician authorization
- WADA: Prohibited under S0 in all member countries regardless of local drug classification
The lack of harmonized international classification reflects the unusual nature of BPC-157: a peptide with extensive preclinical research that has never completed the clinical trial process required for drug approval in any country.
In practice, people traveling internationally with BPC-157 face a patchwork of rules. A peptide obtained legally through a U.S. compounding pharmacy may have a different status in the destination country. Athletes competing internationally face WADA restrictions regardless of the domestic legality in their home country. Researchers ordering BPC-157 for laboratory use face different import regulations depending on whether the substance is classified as a pharmaceutical, a research chemical, or an unregulated compound in the receiving country.
What has not changed
The regulatory shifts of 2022-2026 changed who can sell BPC-157 and through what channels. They did not change the underlying evidence base.
BPC-157 remains a peptide with 35 preclinical studies and 1 retrospective clinical study meeting systematic review inclusion criteria.[1] It has no completed Phase II or Phase III human trials for any indication. The Phase II ulcerative colitis trials that were conducted never published results. The preclinical evidence for gut healing, liver protection, tendon repair, and bone healing is consistent and intriguing. It comes predominantly from one research group.[2]
Reclassification to Category 1 reflects a policy judgment that the safety concerns cited in the original Category 2 classification were insufficient to justify restricting compounding access. It does not constitute an endorsement of BPC-157's efficacy for any condition.
The core tension in BPC-157's regulatory story is the disconnect between its popularity and its evidence base. Tens of thousands of people use BPC-157 based on anecdotal reports, social media testimonials, and extrapolation from animal studies. The peptide generates substantial revenue for compounding pharmacies and research chemical vendors. Clinicians prescribe it based on clinical experience and preclinical data. Yet the controlled human trials that would definitively establish whether BPC-157 works for any specific condition in humans have never been completed.
The February 2026 reclassification does not resolve this tension. It makes BPC-157 easier to obtain through legitimate medical channels, which may improve product quality and patient safety. It does not generate the clinical evidence that would justify clinical confidence in any specific application. The peptide's journey from gastric juice to gray market to regulatory whiplash continues, and the most fundamental question, whether BPC-157 works in humans the way animal studies suggest, remains unanswered.
The Bottom Line
BPC-157's legal status as of March 2026: compounding pharmacies can prepare it with a physician's prescription following the February 2026 reclassification from Category 2 to Category 1. The formal FDA list has not been published and state implementation is pending. WADA's prohibition under S0 remains in effect for all athletes. BPC-157 is not FDA-approved for any indication and has no completed human clinical trials. Research chemical sales continue without restriction.
Sources & References
- 1RPEP-13892·Vasireddi, Nikhil et al. (2025). “BPC-157 for Sports Injuries: What a Systematic Review of the Evidence Actually Shows.” HSS journal : the musculoskeletal journal of Hospital for Special Surgery.Study breakdown →PubMed →↩
- 2RPEP-03561·Unknown (2018). “Seiwerth 2018 Bpc 157 And Standard.” .Study breakdown →↩