Selank: The Anxiolytic Peptide
Selank Anxiolytic Peptide
7 amino acids
Selank (Thr-Lys-Pro-Arg-Pro-Gly-Pro) is a synthetic heptapeptide derived from the immunopeptide tuftsin, developed at the Russian Academy of Sciences as an anxiolytic with a fundamentally different mechanism than benzodiazepines.
Vyunova et al., Protein Pept Lett, 2018
Vyunova et al., Protein Pept Lett, 2018
If you only read one thing
Selank is a seven-amino-acid peptide nasal spray developed in Russia that reduces anxiety without making you drowsy — the opposite of how benzodiazepines (like Xanax or Valium) work. It does this by boosting three of your body's own calming systems at once: natural opioid peptides, GABA receptors, and brain growth factors. One clinical study of 62 patients found it worked as well as a benzodiazepine for anxiety. The catch: almost all the research comes from Russian labs, there's no FDA approval, and the one clinical study didn't include a placebo group.
Selank is a synthetic heptapeptide (Thr-Lys-Pro-Arg-Pro-Gly-Pro) developed at the Institute of Molecular Genetics of the Russian Academy of Sciences in cooperation with the V.V. Zakusov Research Institute of Pharmacology. It consists of the tetrapeptide tuftsin (Thr-Lys-Pro-Arg), a naturally occurring fragment of the immunoglobulin G heavy chain involved in immune regulation, extended with a Pro-Gly-Pro tripeptide tail that protects the molecule from enzymatic degradation and extends its biological half-life. Selank is approved in Russia as a nasal spray for generalized anxiety disorder and neurasthenia, but has not undergone clinical evaluation outside of Russian and former Soviet research institutions and holds no regulatory approval in the United States, European Union, or other Western regulatory jurisdictions. The research base, while containing over a dozen published studies, is concentrated in a small number of Russian laboratories, creating both a focused body of mechanistic evidence and a significant limitation in independent validation. This article examines the full evidence base for Selank's anxiolytic, nootropic, and immunomodulatory properties. For how Selank specifically modulates the GABAergic system, see How Selank Modulates GABA to Reduce Anxiety. For its effects on serotonin, see Selank and Serotonin: Effects on Multiple Neurotransmitter Systems.
Key Takeaways
- Selank is a Russian nasal spray that calms anxiety without making you drowsy — the opposite of Xanax.
- Most anxiety drugs flood your brain with a new signal. Selank just keeps your own calming molecules around longer.
- A single seven-amino-acid peptide appears to hit three anxiety systems at once — enkephalins, GABA, and BDNF.
- In a 62-patient study, it worked about as well as a benzodiazepine for generalized anxiety.
- That study had no placebo group, and almost all Selank research comes from the Russian labs that invented it.
- Selank isn't FDA-approved and has never been independently tested outside Russia.
- Because the effect scales with stress, Selank may calm you more when anxious and less when already relaxed.
From Tuftsin to Selank: The Design Logic
Selank's development began with tuftsin, a tetrapeptide (Thr-Lys-Pro-Arg) found naturally in the Fc region of immunoglobulin G. Tuftsin was first identified in the 1970s as a phagocytosis-stimulating peptide, meaning it activates macrophages and other immune cells. Researchers at the Russian Academy of Sciences observed that tuftsin also had effects on the central nervous system, modulating anxiety-related behaviors in animal models.
The problem with tuftsin as a drug was pharmacokinetic: as a four-amino-acid peptide, it was rapidly degraded by peptidases in the blood and tissues, giving it a biological half-life measured in seconds to minutes. The Pro-Gly-Pro extension was added to the C-terminus specifically to shield the molecule from carboxypeptidase-mediated degradation, the primary route of tuftsin clearance. The resulting heptapeptide, Selank, retained tuftsin's biological activities while achieving sufficient metabolic stability for intranasal administration.
Kozlovskaya et al.'s 2003 study in Neuroscience and Behavioral Physiology compared Selank directly to shorter tuftsin family peptides, demonstrating that Selank was more effective than the parent tetrapeptide in regulating adaptive behavior under stress conditions.[1] The study tested multiple tuftsin analogs and found that the Pro-Gly-Pro extension did not merely stabilize the molecule but actively contributed to its CNS activity, producing a qualitatively different pharmacological profile from tuftsin alone. This is an important distinction: Selank is not simply a stabilized tuftsin but a distinct molecule with broader biological activity than its parent peptide.
Mechanism 1: Enkephalin System Modulation
Selank's first characterized mechanism of action involved the endogenous opioid system. Zozulya et al.'s 2001 study in the Bulletin of Experimental Biology and Medicine demonstrated that Selank inhibited enzymes that degrade enkephalins, the body's endogenous opioid peptides.[2] Enkephalins (met-enkephalin and leu-enkephalin) bind to delta-opioid receptors and play a role in pain modulation, stress response, and anxiety regulation. By slowing their enzymatic degradation, Selank effectively increases the local concentration and duration of action of these endogenous anti-anxiety peptides.
Sokolov et al.'s 2002 follow-up study extended these findings to an animal model, showing that Selank altered both behavioral responses to stress and plasma enkephalin-degrading enzyme activities in mice with different anxiety phenotypes.[3] The effect was not uniform across all animals: mice with high baseline anxiety showed the most pronounced response to Selank, suggesting the peptide acts preferentially when the enkephalin system is under stress-related demand.
This mechanism is fundamentally different from both benzodiazepines (which potentiate GABA signaling) and SSRIs (which block serotonin reuptake). By preserving endogenous opioid peptides rather than introducing an exogenous pharmacological signal, Selank amplifies the body's own anti-anxiety system rather than overriding it with an external one. The distinction matters clinically: drugs that amplify endogenous signaling tend to produce effects proportional to the system's baseline activity, which means they may work more strongly when the system is under stress (high anxiety) and less strongly at baseline (low anxiety). This self-limiting pharmacology could explain why Selank reportedly produces anxiolysis without sedation, the system is amplified when needed but does not push beyond its natural operating range when not under stress.
The enkephalin pathway also connects Selank to pain modulation. Enkephalins are best known for their analgesic properties, and their role in anxiety regulation is mediated partly through effects on the amygdala, where delta-opioid receptors modulate fear conditioning and anxiety-related behaviors. This dual pain/anxiety action may explain why some patients using Selank report improvements in both anxiety and chronic pain conditions, though controlled data supporting this clinical observation is absent.
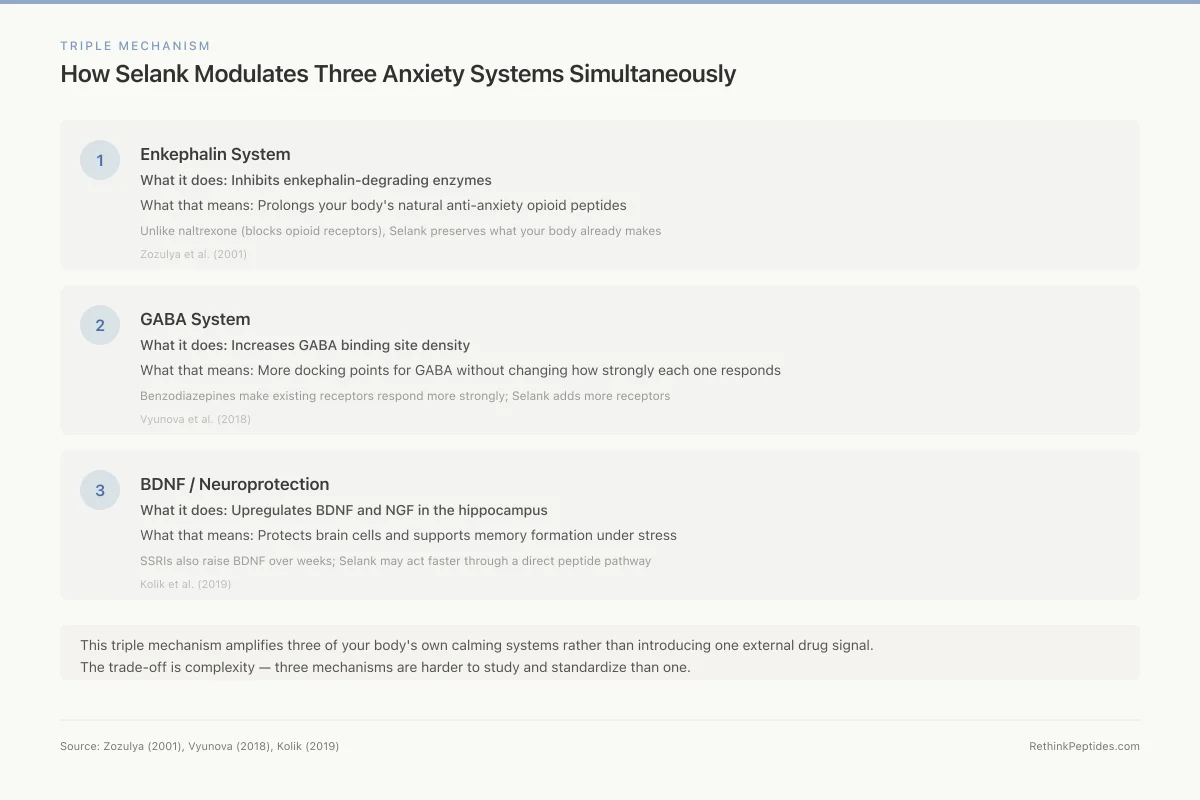
Triple Mechanism
How Selank Modulates Three Anxiety Systems Simultaneously
What it does: Inhibits enkephalin-degrading enzymes
What that means: Prolongs your body's natural anti-anxiety opioid peptides
Unlike naltrexone (blocks opioid receptors), Selank preserves what your body already makes
Zozulya et al. (2001)
What it does: Increases GABA binding site density
What that means: More docking points for GABA (the brain's calming signal) without changing how strongly each one responds
Benzodiazepines make existing receptors respond more strongly; Selank adds more receptors
Vyunova et al. (2018)
What it does: Upregulates BDNF and NGF in the hippocampus
What that means: Protects brain cells and supports memory formation under stress
SSRIs also raise BDNF over weeks; Selank may act faster through a direct peptide pathway
Kolik et al. (2019)
This triple mechanism is what makes Selank unusual: it amplifies three of your body's own calming systems rather than introducing one external drug signal. The trade-off is complexity — three mechanisms are harder to study and standardize than one.
Source: Zozulya (2001), Vyunova (2018), Kolik (2019)
 View as image
View as imageMechanism 2: GABAergic System Effects
Vyunova et al.'s 2018 comprehensive review in Protein and Peptide Letters described Selank's molecular-level interactions with the GABAergic system.[4] The review reported that Selank affected the expression of genes involved in GABAergic neurotransmission, altering the number of specific GABA binding sites on neurons without changing the affinity of individual GABA receptors. This means Selank modulates the capacity of GABAergic signaling (how many receptors are available) rather than the sensitivity of existing receptors (how strongly each receptor responds).
Kasian et al.'s 2017 study in Behavioural Neurology provided functional evidence for this GABAergic interaction.[5] In rats subjected to unpredictable chronic mild stress, a model that produces anxiety and depression-like behaviors, Selank enhanced the anxiolytic effect of diazepam (a benzodiazepine that acts on GABA-A receptors). Selank alone produced measurable anxiolytic effects, and the combination of Selank plus diazepam was more effective than either agent alone. The Selank-diazepam synergy is consistent with Selank increasing the number of available GABA binding sites while diazepam enhances the function of each individual receptor.
The clinical implication, if validated in humans, would be significant: Selank could potentially allow lower doses of benzodiazepines to achieve the same anxiolytic effect, reducing benzodiazepine side effects (sedation, cognitive impairment, dependence) while maintaining efficacy. For detailed mechanistic analysis, see How Selank Modulates GABA to Reduce Anxiety.
Mechanism 3: BDNF and Neuroprotection
Kolik et al.'s 2019 study in the Bulletin of Experimental Biology and Medicine demonstrated that Selank protected against ethanol-induced memory impairment in mice through regulation of brain-derived neurotrophic factor (BDNF) and nerve growth factor (NGF) expression in the hippocampus.[6] BDNF is the primary neurotrophin supporting learning, memory, and synaptic plasticity. Ethanol exposure reduces hippocampal BDNF levels, contributing to the cognitive impairment associated with alcohol use. Selank prevented this BDNF reduction, preserving memory function in treated animals.
This BDNF-mediated neuroprotective mechanism connects Selank to its molecular cousin Semax (a synthetic ACTH analog also developed at the Russian Academy of Sciences). Dolotov et al.'s 2003 study showed that Semax stimulated BDNF expression across multiple brain regions.[7] The shared BDNF upregulation between these two Russian-developed peptides, which target different neuropeptide systems (tuftsin for Selank, ACTH for Semax), suggests that BDNF elevation may be a common downstream effect of multiple neuropeptide signaling pathways. For how Semax specifically upregulates BDNF, see How Semax Upregulates BDNF: The Neuroplasticity Mechanism. For the broader role of BDNF as a brain peptide, see BDNF: The Brain Peptide That Builds New Neural Connections.
The Clinical Evidence: Generalized Anxiety Disorder
The most directly relevant clinical data for Selank comes from a study by Medvedev et al. (2014) comparing intranasal Selank to the benzodiazepine medazepam in 62 patients with generalized anxiety disorder (GAD) and neurasthenia. Thirty patients received Selank (administered intranasally at a dose of 0.15 mg three times daily) and thirty-two received medazepam (a benzodiazepine commonly used in Eastern European medicine, similar in class to diazepam) for a treatment course of several weeks. The study used the Hamilton Anxiety Rating Scale (HAM-A), the Clinical Global Impression scale, and the Spielberger State-Trait Anxiety Inventory to assess outcomes.
The study found that the anxiolytic effects of both drugs were comparable on all assessment scales. Selank additionally demonstrated antiasthenic (anti-fatigue) and psychostimulant properties that the benzodiazepine did not produce, meaning patients on Selank reported improved energy and mental clarity alongside anxiety reduction. This psychostimulant-anxiolytic combination is unusual: most anxiolytic drugs either sedate (benzodiazepines) or are neutral on energy (SSRIs). A drug that reduces anxiety while increasing alertness would fill a genuine unmet need, particularly for patients who find benzodiazepine sedation intolerable.
The absence of sedation, cognitive impairment, and dependence potential with Selank was the primary differentiating feature. Benzodiazepines produce their anxiolytic effects at the cost of drowsiness, impaired memory formation, psychomotor slowing, and physical dependence with chronic use. Withdrawal from benzodiazepines after prolonged use can produce rebound anxiety worse than the original condition, along with seizure risk. Selank produced anxiolysis without these trade-offs, and no withdrawal effects were reported upon discontinuation. For detailed analysis of the clinical trial data, see Selank Clinical Studies: Generalized Anxiety Evidence. For how this compares to benzodiazepine mechanisms, see Selank vs Benzodiazepines: A Fundamentally Different Approach.
The limitation of this clinical evidence must be stated plainly: a single comparative study in 62 patients conducted at Russian institutions, without placebo control, not registered on ClinicalTrials.gov, and not replicated independently by Western research groups, does not meet the evidence standard that the FDA, EMA, or other Western regulatory agencies require for drug approval. The study provides a signal of clinical activity but falls far short of the Phase 3, multicenter, randomized, placebo-controlled evidence base that established drugs like SSRIs have accumulated over thousands of patients across multiple countries.
Alcohol-Related Effects
A distinct line of Selank research has explored its effects on alcohol-related behaviors. Kolik et al.'s 2014 study showed efficacy of Selank during modeled alcohol withdrawal syndrome in rats with stable alcohol motivation, reducing withdrawal-associated anxiety and behavioral disturbances.[8] Their 2016 study demonstrated that Selank inhibited ethanol-induced hyperlocomotion and behavioral sensitization in DBA/2 mice, a strain genetically predisposed to high alcohol preference.[9]
These findings are consistent with Selank's multi-system mechanism: the enkephalin system, GABA system, and serotonin system are all disrupted by chronic alcohol exposure, and a drug that modulates all three simultaneously could theoretically address alcohol use disorder more comprehensively than drugs targeting a single pathway. Current approved medications for alcohol use disorder target individual systems: naltrexone blocks opioid receptors, acamprosate modulates glutamate/GABA balance, and disulfiram produces aversive reactions to alcohol. A peptide that simultaneously normalizes multiple disrupted systems is an appealing theoretical concept, though its practical value depends entirely on human clinical data that does not yet exist.
The alcohol research also demonstrates Selank's specificity: it reduced pathological alcohol-related behaviors without suppressing normal motor activity or feeding behavior at the tested doses, suggesting therapeutic selectivity rather than generalized CNS depression. This contrasts with benzodiazepines, which are used in alcohol withdrawal management but produce their own sedation, cognitive impairment, and cross-tolerance with alcohol.
Brain Imaging: Functional Connectomics
Panikratova et al.'s 2020 study in Doklady Biological Sciences used functional MRI to examine how Selank and Semax affected brain network connectivity in human subjects.[10] This study is notable because it represents one of the few pieces of human neuroimaging data available for either peptide. The analysis showed that Selank and Semax produced distinct patterns of brain network modulation: Selank preferentially affected connectivity in emotion-processing circuits (amygdala, prefrontal cortex, anterior cingulate), consistent with its anxiolytic profile, while Semax primarily modulated attention and executive function networks.
The functional connectomic approach provided objective, imaging-based confirmation that Selank reaches the CNS after intranasal administration and produces measurable changes in brain circuit activity, independent of subjective behavioral measures. This addresses a persistent skepticism about nasal peptide delivery: the blood-brain barrier normally excludes peptides, but the olfactory pathway provides a direct nose-to-brain route that bypasses the BBB. The fMRI data confirms that this delivery route achieves functional brain concentrations.
Selank in the Neuropeptide Anxiety Landscape
Holmes et al.'s 2003 review in Trends in Pharmacological Sciences mapped neuropeptide systems as novel therapeutic targets for depression and anxiety, identifying over a dozen endogenous peptide systems that modulate anxiety-related circuits.[11] Rotzinger et al.'s 2010 review in CNS & Neurological Disorders Drug Targets examined the behavioral effects of neuropeptides in rodent models of depression and anxiety, providing preclinical frameworks for evaluating peptide anxiolytics.[12]
Selank fits into this landscape as a peptide that modulates multiple anxiety-relevant systems simultaneously, rather than targeting a single receptor or neurotransmitter. This multi-target approach is increasingly recognized as potentially advantageous for psychiatric conditions, which are rarely driven by dysfunction in a single neurotransmitter system. The monoamine hypothesis of anxiety (that anxiety results primarily from serotonin or norepinephrine imbalance) has been progressively replaced by circuit-level models in which multiple neurotransmitter systems interact within specific brain networks. A drug that modulates GABA, serotonin, and enkephalin signaling simultaneously may normalize circuit-level dysfunction that single-target drugs cannot fully address.
The challenge is that multi-target mechanisms are harder to characterize, predict, and optimize than single-target drugs. When a molecule affects three systems, teasing apart which effect drives clinical benefit, which is irrelevant, and which might cause side effects requires extensive dose-response studies across each system independently. This pharmacological complexity is part of why Selank's mechanism has taken decades to map and remains incompletely understood. It also makes regulatory evaluation more difficult: demonstrating that a multi-target drug is safe and effective requires more extensive preclinical and clinical data than a drug with a single, well-characterized mechanism.
Other neuropeptides with anxiolytic potential include neuropeptide Y (NPY), which produces potent anxiolytic effects when administered to the amygdala; oxytocin, which reduces social anxiety through effects on the hypothalamic-pituitary-adrenal axis; and corticotropin-releasing hormone (CRH) receptor antagonists, which block the primary stress hormone initiator. None of these alternative peptide approaches have achieved clinical approval for anxiety, though several have reached early clinical trials. The field of peptide anxiolytics remains in its infancy compared to the established small molecule and antibody drug classes.
The Immune Connection
Selank's origin in the immunopeptide tuftsin means it retains immunomodulatory activity alongside its CNS effects. The peptide has been shown to modulate expression of interleukin-6 (IL-6) and affect the balance of T helper cell cytokines. This dual anxiolytic-immunomodulatory profile is relevant because anxiety disorders and immune dysregulation frequently co-occur, and inflammatory cytokines can directly promote anxiety-related behavior through effects on brain circuits. Whether Selank's immune effects contribute to its anxiolytic activity, or whether these are independent pharmacological actions, remains an open question. The bidirectional communication between the immune system and the brain (sometimes called the "immune-brain axis") is increasingly recognized as a factor in psychiatric disorders. Patients with elevated inflammatory markers have higher rates of anxiety and depression, and anti-inflammatory treatments can sometimes improve mood symptoms. If Selank reduces neuroinflammation through its tuftsin-derived immunomodulatory activity while simultaneously modulating neurotransmitter systems through its CNS actions, the combined effect could be greater than either mechanism alone. For detailed analysis, see Selank and Immune Function: The Unexpected Anxiety-Immunity Link.
Limitations of the Evidence Base
The central limitation of Selank research is geographic concentration. Nearly all published studies originate from a small number of Russian research groups, many affiliated with the same institutions that developed the peptide. Independent replication by Western laboratories is essentially absent. This does not invalidate the Russian findings, which are published in peer-reviewed journals and use standard pharmacological methods, but it limits confidence in the generalizability and reproducibility of the results.
The clinical evidence is sparse by Western regulatory standards. One comparative study with 62 patients does not establish efficacy to the level required for FDA approval, which typically demands two independent, adequately powered Phase 3 trials. The absence of placebo-controlled data (the comparator was medazepam, not placebo) means the absolute magnitude of Selank's anxiolytic effect is difficult to quantify.
Long-term safety data in humans is limited. Selank has been available as a prescription nasal spray in Russia since 2009, providing some post-marketing exposure data, but systematic pharmacovigilance reporting from Russian healthcare systems is less accessible to international researchers than FDA's FAERS database. Animal toxicology studies have not revealed serious safety concerns, and the peptide's rapid degradation to natural amino acids theoretically limits accumulation-related toxicity.
The intranasal delivery route introduces its own variables. Nasal absorption of peptides depends on nasal mucosa integrity, formulation pH, presence of permeation enhancers, and individual anatomical variation in the olfactory epithelium. Dose-to-CNS delivery is difficult to standardize compared to oral or injectable drugs. Different commercially available Selank nasal spray formulations may achieve different brain concentrations even at the same nominal dose. Patients with nasal congestion, rhinitis, or structural abnormalities may absorb substantially less peptide than healthy-nosed individuals.
Safety
ModerateGray-market Selank quality is unverifiable
Concern
Outside Russia, Selank is only available from gray-market peptide vendors as lyophilized powder. Purity, correct peptide sequence, and sterility cannot be verified by end users. The pharmaceutical-grade product used in published research may have a very different quality profile than commercially available gray-market preparations.
What the research says
The Russian pharmaceutical manufacturer produces a regulated nasal spray product. Users outside Russia have no access to this regulated product and must rely on unregulated sources.
Particularly relevant for: Anyone purchasing Selank from online peptide vendors outside the Russian pharmaceutical supply chain
What to do
If using gray-market Selank, understand that you are not using the same product studied in published research. Intranasal delivery of a non-sterile reconstituted peptide carries infection risk. Nasal mucosa absorption varies significantly between individuals.
General peptide quality literature; Russian pharmaceutical regulations
The quality of commercially available Selank is an additional concern for users outside Russia. Gray-market peptide suppliers offer Selank as a lyophilized powder for reconstitution, without the quality assurance of the Russian pharmaceutical manufacturer. Purity, sterility, and correct peptide sequence cannot be verified by end users, creating a gap between the pharmacological profile characterized in published research (using pharmaceutical-grade material) and the actual product that many users administer.
The Bottom Line
Selank is a seven-amino-acid synthetic peptide derived from the immunopeptide tuftsin, developed in Russia as an anxiolytic with a multi-system mechanism of action distinct from both benzodiazepines and SSRIs. Preclinical research has characterized three primary mechanisms: inhibition of enkephalin-degrading enzymes (prolonging endogenous opioid signaling), modulation of GABAergic neurotransmission (increasing GABA binding site density), and upregulation of BDNF (supporting neuroprotection and synaptic plasticity). A clinical study in 62 patients found anxiolytic effects comparable to medazepam without sedation, cognitive impairment, or dependence. The evidence base is concentrated in Russian research institutions with minimal independent Western validation. No FDA, EMA, or other Western regulatory approval exists. The peptide remains a subject of scientific interest for its novel multi-pathway anxiolytic mechanism, but the clinical evidence falls far short of the standard required for regulatory approval outside of Russia.
Sources & References
- 1RPEP-00837·Kozlovskaya, M M et al. (2003). “Selank and Tuftsin-Family Peptides Help Different Personality Types Handle Stress Differently.” Neuroscience and behavioral physiology.Study breakdown →PubMed →↩
- 2RPEP-00708·Zozulya, A A et al. (2001). “Selank Treats Anxiety by Protecting the Body's Natural Anxiety-Relieving Enkephalins.” Bulletin of experimental biology and medicine.Study breakdown →PubMed →↩
- 3RPEP-00774·Sokolov, O Yu et al. (2002). “Selank Reduces Anxiety in Mice by Protecting Their Natural Anti-Anxiety Enkephalins.” Bulletin of experimental biology and medicine.Study breakdown →PubMed →↩
- 4RPEP-03968·Vyunova, Tatiana V et al. (2018). “How Selank Peptide Reduces Anxiety by Modulating GABA Receptors Differently Than Benzodiazepines.” Protein and peptide letters.Study breakdown →PubMed →↩
- 5RPEP-03340·Kasian, Anastasiya et al. (2017). “Peptide Selank Enhances the Effect of Diazepam in Reducing Anxiety in Unpredictable Chronic Mild Stress Conditions in Rats..” Behavioural neurology.Study breakdown →PubMed →↩
- 6RPEP-04287·Kolik, L G et al. (2019). “Selank Peptide Prevented Alcohol-Induced Memory Loss by Regulating BDNF in Rat Brains.” Bulletin of experimental biology and medicine.Study breakdown →PubMed →↩
- 7RPEP-00812·Dolotov, O V et al. (2003). “The heptapeptide SEMAX stimulates BDNF expression in different areas of the rat brain in vivo..” Doklady biological sciences : proceedings of the Academy of Sciences of the USSR.Study breakdown →PubMed →↩
- 8RPEP-02421·Kolik, L G et al. (2014). “Selank Eliminated Anxiety and Pain Sensitivity During Alcohol Withdrawal in Rats.” Bulletin of experimental biology and medicine.Study breakdown →PubMed →↩
- 9RPEP-02996·Kolik, L G et al. (2016). “Selank Inhibits Ethanol-Induced Hyperlocomotion and Manifestation of Behavioral Sensitization in DBA/2 Mice..” Bulletin of experimental biology and medicine.Study breakdown →PubMed →↩
- 10RPEP-05052·Panikratova, Ya R et al. (2020). “Selank and Semax Alter Brain Connectivity Between Amygdala and Temporal Cortex in Humans.” Doklady biological sciences : proceedings of the Academy of Sciences of the USSR.Study breakdown →PubMed →↩
- 11RPEP-00827·Holmes, Andrew et al. (2003). “Neuropeptide Systems: The Next Generation of Antidepressants and Anxiolytics.” Trends in pharmacological sciences.Study breakdown →PubMed →↩
- 12RPEP-01682·Rotzinger, Susan et al. (2010). “Neuropeptides in Animal Models of Depression and Anxiety: CRF, NPY, and the Therapeutic Landscape.” Peptides.Study breakdown →PubMed →↩