BPC-157 and Liver Protection: Animal Studies
BPC-157 and Liver Protection
7+ liver injury models
BPC-157 has been tested across more than seven distinct liver injury models in rats, including CCl4, ethanol, NSAIDs, bile duct ligation, ischemia-reperfusion, radiation, and restraint stress.
Sikiric et al., Life Sciences, 1993
Sikiric et al., Life Sciences, 1993
If you only read one thing
BPC-157 is a small peptide derived from a protein in your stomach. In rat studies going back to 1993, it's protected liver tissue from seven different types of damage — alcohol, ibuprofen, radiation, toxic chemicals, loss of blood flow, and more. The proposed mechanism involves boosting nitric oxide production, which improves blood flow to liver tissue. That's real preclinical evidence, consistent across three decades. But zero human clinical trials have tested BPC-157 for any liver condition, and most of the original studies came from one lab. The peptide sold online is 'research grade' with variable purity. So: genuinely interesting animal evidence, no human proof, gray-market supply.
BPC-157 (Body Protection Compound-157) is a synthetic pentadecapeptide derived from a protein found in human gastric juice. Among its many investigated properties, hepatoprotection is one of the earliest and most consistently documented. Since the first study in 1993, researchers have tested BPC-157 across more than seven distinct models of liver injury in rats: carbon tetrachloride (CCl4) poisoning, bile duct ligation, hepatic artery ligation, restraint stress, chronic alcohol administration, NSAID-induced liver damage, and radiation injury. In every model tested, BPC-157 reduced liver damage markers, prevented or reversed histological changes, and outperformed comparison drugs including ranitidine, somatostatin, and dopamine agonists.[1] This article reviews the full animal evidence for BPC-157's liver-protective effects, the proposed mechanisms, and the substantial gap between preclinical promise and clinical validation.
Key Takeaways
- The first hepatoprotective study (Sikiric et al., 1993) showed BPC-157 prevented liver necrosis and fatty changes from CCl4, bile duct ligation, hepatic artery ligation, and restraint stress in rats, outperforming somatostatin and dopamine agonists.[1]
- In chronically alcohol-drinking rats, BPC-157 prevented and reversed portal hypertension and liver lesions, effects that persisted after treatment cessation.[3]
- BPC-157 reduced radiation-induced liver injury by upregulating Kruppel-like factor 4 (KLF4), decreasing AST and ALT, and inhibiting cell apoptosis both in vivo and in vitro.[2]
- Multiple NSAID studies show BPC-157 counteracts diclofenac-, ibuprofen-, and celecoxib-induced liver lesions in rats through nitric oxide system modulation.[6]
- BPC-157 modulates vasomotor tone through the Src-Caveolin-1-eNOS pathway, which may explain its protective effects on hepatic blood flow.[10]
- All evidence comes from animal studies. No human clinical trial has evaluated BPC-157 for liver protection.[11]
The foundational study: Sikiric 1993
The hepatoprotective potential of BPC-157 was first documented by Sikiric et al. (1993) in a comprehensive study published in Life Sciences. They tested the peptide across four distinct liver injury models in rats:
Carbon tetrachloride (CCl4) administration produced severe liver necrosis. BPC-157, given either intragastrically or intraperitoneally, significantly reduced the extent of necrotic tissue compared to controls. CCl4 is a classic hepatotoxin that generates free radicals via cytochrome P450 metabolism, making this a test of BPC-157's ability to counteract oxidative liver damage.
Bile duct ligation produced obstructive cholestasis and secondary liver damage. BPC-157 reduced the histological signs of liver injury in this model, suggesting activity against bile-mediated hepatocyte damage.
Hepatic artery ligation created ischemic liver injury. BPC-157 reduced the severity of ischemic necrosis, indicating a protective effect on liver tissue deprived of arterial blood supply.
Restraint stress produced stress-induced liver lesions. BPC-157 prevented these lesions, extending its protective profile to neurally-mediated hepatic damage.
Across all four models, BPC-157 outperformed the comparison drugs: bromocriptine (a dopamine agonist), somatostatin, and domperidone. These reference drugs "had either little or no protective actions" in the same models where BPC-157 showed robust protection.[1]
This study established the foundational observation: BPC-157 protects rat liver tissue from chemically, mechanically, and neurally induced damage through mechanisms that do not overlap with conventional hepatoprotective drugs.
The breadth of models tested in a single study is worth noting. Most hepatoprotective candidates are evaluated against one or two injury types. Testing against four mechanistically distinct models in the same publication, and showing protection across all four, established a pattern of broad hepatoprotection that subsequent studies would reinforce and extend over the following three decades. The fact that BPC-157 worked when administered both intragastrically (simulating oral dosing) and intraperitoneally (systemic dosing) suggested that the peptide's liver effects do not require direct contact with hepatocytes from the gut lumen, supporting a systemic mechanism of action.
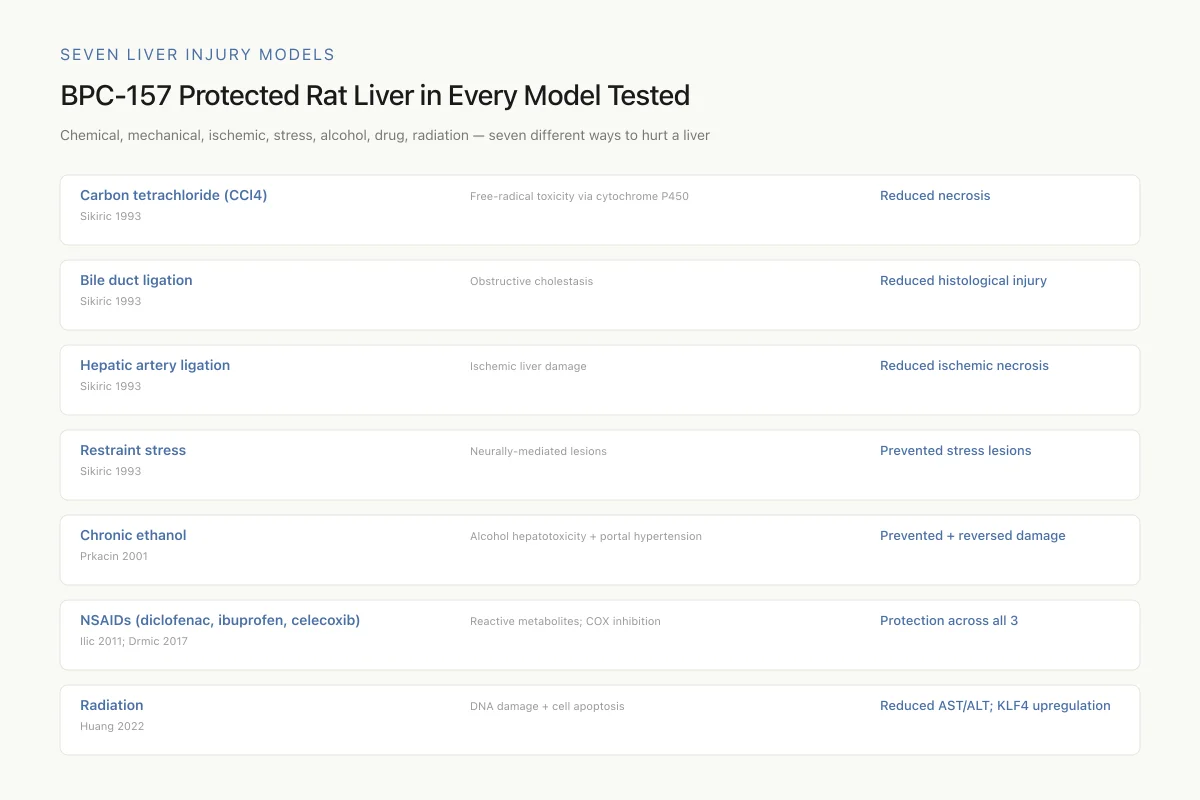
Seven Liver Injury Models
BPC-157 Protected Rat Liver in Every Model Tested
Chemical, mechanical, ischemic, stress, alcohol, drug, radiation — seven different ways to hurt a liver, one consistent result
Hepatoprotective drugs are usually validated against one or two injury models. BPC-157 has shown protection across seven mechanistically distinct ones. The consistency is genuinely unusual — it suggests a foundational protective pathway rather than a narrow drug effect.
Source: Sikiric 1993; Prkacin 2001; Ilic 2011; Drmic 2017; Huang 2022
 View as image
View as imageAlcohol-induced liver damage
Chronic alcohol consumption is one of the most clinically relevant causes of liver injury, and BPC-157 has been tested in several alcohol-induced liver damage models.
Prkacin et al. (2001) conducted two key studies. In the first, they demonstrated that chronically alcohol-drinking rats developed portal hypertension and progressive liver lesions, both of which were prevented and reversed by BPC-157 treatment. The peptide was administered as stable gastric pentadecapeptide in drinking water, a route that mimics the physiological exposure of the gastrointestinal tract to gastric peptides. Portal pressure measurements showed that BPC-157 normalized the elevated portal pressures that develop with chronic alcohol exposure.[3]
In their second study, Prkacin et al. (2001) compared BPC-157 with ranitidine and propranolol in a chronic cytoprotection model. All three agents were tested for their ability to prevent, attenuate, and reverse gastric and liver lesions in chronically alcohol-drinking rats. BPC-157 outperformed both comparison drugs. Ranitidine provided only partial protection, and propranolol was effective primarily against portal hypertension rather than hepatocyte damage. BPC-157 addressed both the vascular (portal pressure) and parenchymal (hepatocyte) components of alcohol-induced liver disease.[4]
Boban Blagaic et al. (2006) extended these findings by examining the influence of BPC-157 on both acute and chronic ethanol administration in mice, adding a second species to the evidence base. They also investigated the role of the nitric oxide (NO) system by co-administering L-NAME (an NO synthase inhibitor) and L-arginine (an NO precursor). When L-NAME was given alongside ethanol, liver damage worsened, indicating that NO depletion exacerbates alcohol hepatotoxicity. When BPC-157 was given with L-NAME, it partially overcame the NO blockade, suggesting that BPC-157 either restores NO production through an alternative pathway or provides hepatoprotection through additional mechanisms beyond NO signaling alone. L-arginine, the substrate for NO synthesis, provided partial but incomplete protection. The results positioned BPC-157 as a more effective hepatoprotectant than either NO pathway modulator alone.[5]
The alcohol studies are collectively the most clinically relevant subset of the BPC-157 liver data. Alcohol-related liver disease is the leading cause of liver transplantation in many countries, and current pharmacotherapy options are extremely limited. N-acetylcysteine is used for acute alcoholic hepatitis, and corticosteroids provide marginal benefit in severe cases, but no drug effectively prevents or reverses the progression from alcoholic fatty liver through steatohepatitis to cirrhosis. A peptide that demonstrates protection across this disease continuum in animal models, including reversal of established portal hypertension, addresses an unmet clinical need. The gap, as with all BPC-157 research, is the absence of human data.
NSAID-induced liver injury
Non-steroidal anti-inflammatory drugs (NSAIDs) can cause significant liver damage, particularly with chronic use. BPC-157 has been tested against liver injury from three different NSAIDs: diclofenac, ibuprofen, and celecoxib.
Ilic et al. (2011) demonstrated that BPC-157 counteracted diclofenac-induced gastrointestinal, liver, and encephalopathy lesions in rats. Diclofenac produced hepatomegaly, elevated liver enzymes, and histological liver damage. BPC-157 reduced all three markers. The same group showed similar results with ibuprofen, which induced hepatic encephalopathy, hepatomegaly, and gastric lesions that were all attenuated by BPC-157 treatment.[6][7]
Lojo et al. (2016) investigated the NO system dependence of BPC-157's hepatoprotective effects against diclofenac. By co-administering L-NAME and L-arginine, they demonstrated that BPC-157's protective effects involved modulation of nitric oxide synthesis. L-NAME aggravated the diclofenac-induced liver damage, while L-arginine partially attenuated it, and BPC-157 reversed the damage through a pathway that interacted with both NO system modulators.[9]
Drmic et al. (2017) extended the NSAID evidence to celecoxib, a selective COX-2 inhibitor. Celecoxib induced gastrointestinal, liver, and brain lesions in rats. BPC-157 and L-arginine both counteracted these lesions, while L-NAME aggravated them, reinforcing the consistent finding that BPC-157's hepatoprotective mechanism involves the NO system.[8]
The NSAID studies are collectively important because they demonstrate that BPC-157's hepatoprotective effects extend across different NSAID classes with distinct mechanisms of liver toxicity. Diclofenac causes idiosyncratic hepatotoxicity through reactive metabolite formation, while celecoxib and ibuprofen damage the liver through different pathways. BPC-157's protection against all three suggests a broad hepatoprotective mechanism rather than one specific to a single toxicity pathway.
Radiation-induced liver injury
Huang et al. (2022) investigated BPC-157's effects on radiation-induced liver disease, a clinically significant problem for cancer patients receiving abdominal radiation therapy. The study found that BPC-157 reduced radiation-induced liver injury through upregulation of Kruppel-like factor 4 (KLF4), a transcription factor with anti-apoptotic and tissue-protective functions.
Specifically, BPC-157 decreased plasma levels of AST and ALT (standard markers of hepatocyte damage), inhibited hydropic degeneration of liver tissue, reduced radiation-induced cell apoptosis, and decreased lipid accumulation in liver cells. The KLF4 mechanism was confirmed both in vivo (rat liver after radiation) and in vitro (irradiated hepatocyte cell cultures), providing a molecular pathway linking BPC-157 to transcriptional regulation of cell survival genes.[2]
This study is notable for providing a specific molecular target (KLF4) rather than relying solely on histological and biochemical endpoints. The identification of a transcription factor mechanism places BPC-157's hepatoprotection within a defined signaling framework, though whether KLF4 upregulation is the primary mechanism or one of several parallel pathways remains unclear.
The radiation injury model is also clinically relevant in a way that other models are not. Patients receiving radiation therapy for abdominal and thoracic cancers frequently develop radiation-induced liver disease (RILD), a dose-limiting toxicity that constrains treatment intensity. If BPC-157's hepatoprotective effects against radiation were confirmed in larger studies and eventually in human trials, it could potentially expand the therapeutic window for radiation oncology. This is speculative, but the preclinical rationale is stronger than for many radioprotective candidates that have failed in clinical development.
The nitric oxide system: a unifying mechanism
Across multiple liver injury models, BPC-157's hepatoprotective effects consistently involve the nitric oxide (NO) system. This is not unique to liver protection: NO system modulation appears to be a core mechanism of BPC-157 activity across many tissues.
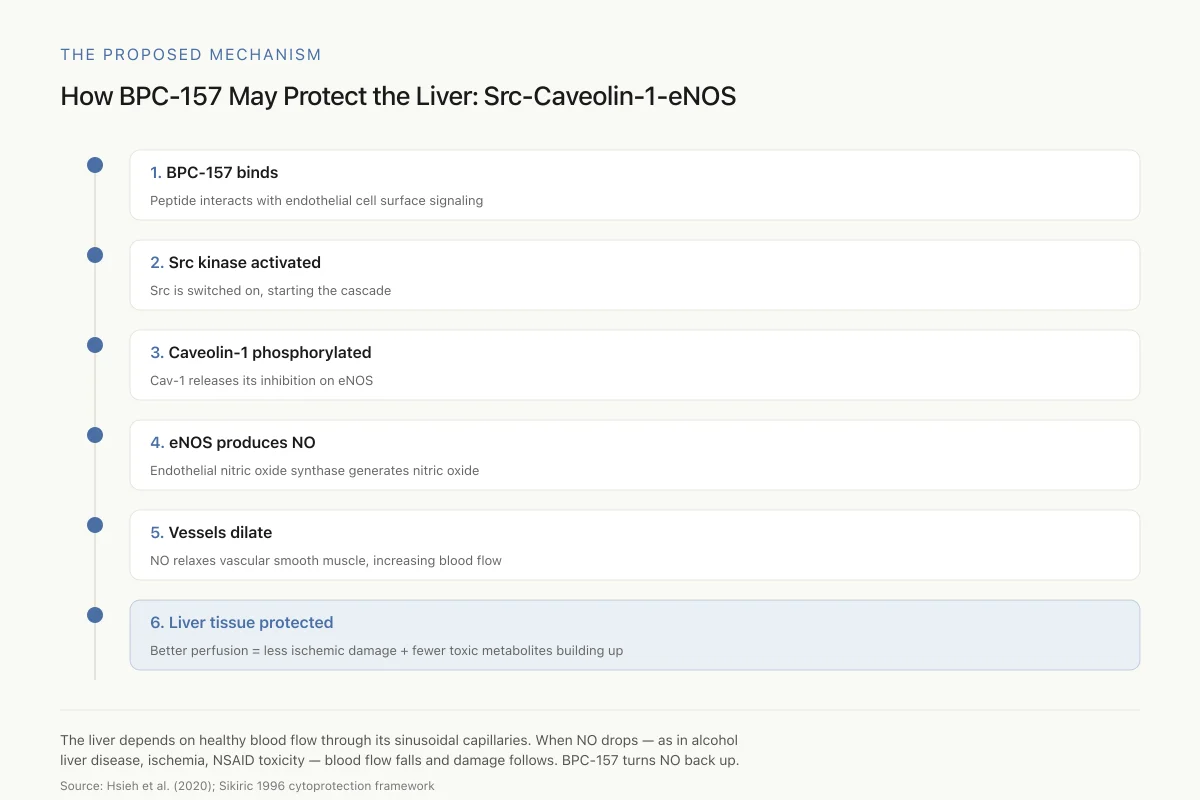
The Proposed Mechanism
How BPC-157 May Protect the Liver: The Src-Caveolin-1-eNOS Pathway
A signaling cascade that ends in more nitric oxide, better hepatic blood flow, and reduced liver damage
The liver depends on healthy blood flow through its sinusoidal capillaries. When NO production drops — as it does in alcohol-related liver disease, ischemia, and NSAID toxicity — blood flow falls and damage follows. BPC-157 seems to turn the NO signal back up.
Source: Hsieh et al. (2020); Sikiric 1996 cytoprotection framework
 View as image
View as imageHsieh et al. (2020) provided the most detailed mechanistic study, demonstrating that BPC-157 modulates vasomotor tone through the Src-Caveolin-1-endothelial nitric oxide synthase (eNOS) pathway. In their model, BPC-157 activated Src kinase, which phosphorylated caveolin-1, releasing the inhibition on eNOS and increasing NO production. This pathway directly regulates blood vessel dilation and blood flow, which in the liver context would improve hepatic perfusion and protect against ischemic damage.[10]
The NO system connection explains several observations across the BPC-157 liver literature:
- Portal hypertension reversal: NO is a potent vasodilator; increased NO production in hepatic vasculature would reduce portal pressure
- Ischemia protection: improved perfusion through NO-mediated vasodilation would reduce ischemic damage
- NSAID counteraction: NSAIDs inhibit prostaglandin synthesis and can reduce NO production; BPC-157's ability to restore NO signaling may directly counteract this mechanism
- Alcohol damage reversal: chronic alcohol exposure impairs endothelial NO synthase function; BPC-157's activation of the Src-Caveolin-1-eNOS pathway may restore this impaired function
The cytoprotective concept, which Sikiric's group has developed over three decades, frames BPC-157 as an endogenous protective factor that maintains tissue homeostasis through NO-mediated vascular and cellular mechanisms. Sikiric et al. (1996) proposed this framework early, showing that BPC-157's beneficial effects on gastric lesions extended to a broader protective physiology involving stress responses, ethanol, and indomethacin, all mediated partly through the NO system.[12]
The liver is a particularly appropriate organ for NO-mediated protection because of its unique dual blood supply (hepatic artery and portal vein) and its dependence on sinusoidal endothelial cell function for normal perfusion. The hepatic sinusoidal endothelium is one of the most active sites of NO production in the body, and impaired sinusoidal NO production is a hallmark of cirrhosis, portal hypertension, and ischemic liver disease. A peptide that restores or enhances endothelial NO signaling would be expected to protect against precisely the conditions tested in the BPC-157 literature.
The Src-Caveolin-1-eNOS pathway identified by Hsieh et al. (2020) is particularly relevant to liver biology because caveolin-1 is highly expressed in hepatic stellate cells and sinusoidal endothelial cells. Stellate cell activation drives liver fibrosis, the pathological scarring that underlies cirrhosis. If BPC-157's modulation of caveolin-1 affects stellate cell behavior in addition to endothelial function, it could theoretically influence fibrosis progression, though this has not been directly tested.[10]
Delivery innovation: BPC-157 in probiotics
Skrlec et al. (2018) took a different approach to BPC-157 liver research by engineering recombinant Lactococcus lactis bacteria to produce and deliver BPC-157. The engineered probiotic demonstrated antioxidant activity, and the researchers proposed oral delivery of the peptide via bacterial production in the gastrointestinal tract as a clinically feasible route. This approach would bypass the peptide stability issues that limit conventional oral delivery of small peptides and could provide sustained local BPC-157 release in the gut, where the peptide originates naturally.[13]
The delivery innovation is relevant to hepatoprotection specifically because of the gut-liver axis. Blood from the intestine drains directly into the liver via the portal vein, meaning that BPC-157 produced in the gut would reach the liver at relatively high concentrations before encountering systemic circulation. This first-pass effect, which usually limits the systemic bioavailability of oral drugs, would actually be advantageous for a hepatoprotective peptide: the liver would be exposed to higher concentrations than any other organ. Whether the engineered probiotic delivery system achieves therapeutic BPC-157 concentrations in the portal circulation has not been quantified, but the pharmacokinetic logic is sound.
How BPC-157 liver research fits the broader picture
Jozwiak et al. (2025) published a comprehensive literature and patent review of BPC-157's multifunctionality, documenting the peptide's effects across gastrointestinal, musculoskeletal, neurological, cardiovascular, and hepatic systems. The liver protection data represent one spoke of a broader cytoprotective profile that is remarkably consistent across tissue types. This consistency is both a strength (suggesting a fundamental protective mechanism) and a source of skepticism (single molecules rarely affect so many systems through one mechanism).[11]
The broader BPC-157 story is characterized by extensive animal data from a relatively small number of research groups, with the Sikiric laboratory in Zagreb, Croatia producing the majority of published studies. This concentration of research in one group is not inherently problematic, as specialized peptide research often develops in single labs, but it does mean that independent replication of the liver protection findings is limited.
Critical limitations
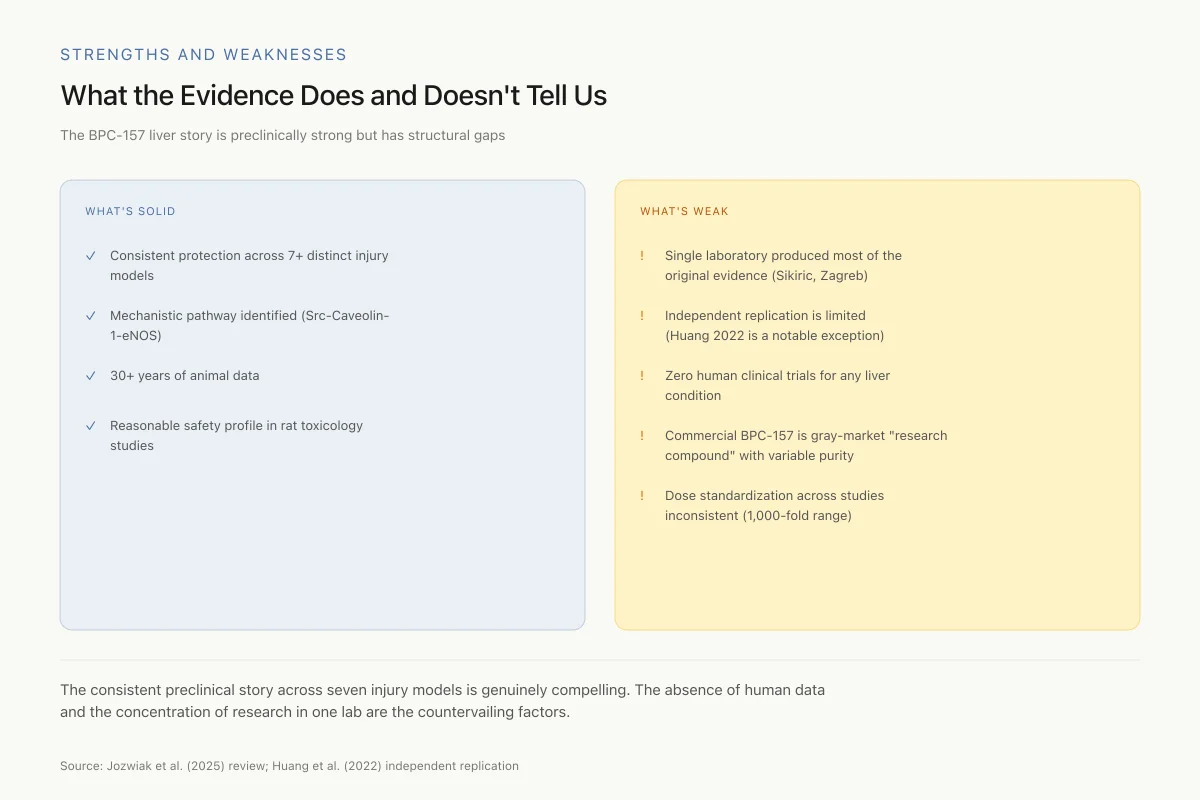
Strengths and Weaknesses
What the Evidence Does and Doesn't Tell Us
The BPC-157 liver story is preclinically strong but has structural gaps
- ✓Consistent protection across 7+ distinct injury models
- ✓Mechanistic pathway identified (Src-Caveolin-1-eNOS)
- ✓30+ years of animal data
- ✓Reasonable safety profile in rat toxicology studies
- !Single laboratory produced most of the original evidence (Sikiric group, Zagreb)
- !Independent replication is limited (Huang 2022 is a notable exception)
- !Zero human clinical trials for any liver condition
- !Commercial BPC-157 is gray-market "research compound" with variable purity
- !Dose standardization across studies inconsistent (1,000-fold range)
The consistent preclinical story across seven injury models is genuinely compelling. The absence of human data and the concentration of the original research in one lab are the countervailing factors. These don't cancel each other out — they define what we know vs. what we've assumed.
Source: Jozwiak et al. (2025) review; Huang et al. (2022) independent replication
 View as image
View as imageSafety
ModerateGray-market BPC-157 has no human data and variable purity
Concern
BPC-157 is commercially available as a 'research peptide' — a regulatory fiction that allows sale while technically prohibiting human consumption. Purity, concentration, and contamination vary by vendor. No human safety data exists for liver application. No human pharmacokinetic data exists to guide dosing.
What the research says
If you're exploring BPC-157 for a liver concern, the honest reality is that you'd be the experiment. The rat studies don't automatically transfer to humans, and gray-market peptide vials don't automatically match the pharmaceutical-grade peptide used in those studies.
Particularly relevant for: Anyone self-administering BPC-157 for liver conditions
What to do
Treat all self-administration as experimental. Don't use it to replace established liver treatments. Discuss with a hepatologist before combining with other liver-affecting drugs. Source matters — purity varies dramatically across vendors.
Jozwiak et al. (2025); commercial peptide market surveys
The entire BPC-157 liver protection evidence base rests on animal studies, primarily in rats with some mouse data. No human clinical trial has evaluated BPC-157 for any liver condition. The gap between "prevents CCl4-induced liver necrosis in rats" and "protects human liver from alcohol damage" is wide and filled with unknowns: human pharmacokinetics, dose-response relationships, long-term safety, and interaction with human liver metabolism are all uncharacterized.
The mechanistic picture, while increasingly detailed around the NO system and KLF4 pathways, remains incomplete. Whether these represent the primary mechanisms or downstream effects of a more fundamental action is uncertain. The peptide's effects across so many tissue types raise the question of whether the observed hepatoprotection reflects a specific liver-targeted mechanism or a general cytoprotective property that happens to benefit hepatocytes among many cell types.
Dose standardization across studies is inconsistent. Different research groups use different doses, routes, and durations of administration, making direct comparisons and dose-response modeling difficult. The most commonly used doses in rat studies range from 10 nanograms per kilogram to 10 micrograms per kilogram, spanning a 1,000-fold range. While BPC-157 appears to show activity across this range, the absence of formal dose-response curves in liver models makes it impossible to identify optimal dosing or predict human-equivalent doses with confidence.
The concentration of BPC-157 research within a single laboratory raises methodological questions that independent replication would address. The Sikiric group in Zagreb has produced the overwhelming majority of BPC-157 publications across all indications, not just liver protection. While their work is internally consistent and published in peer-reviewed journals, the lack of independent replication by unaffiliated laboratories is a weakness in the evidence base. The Huang et al. (2022) radiation study, conducted by a separate group in Taiwan, represents one of the few independent confirmations of hepatoprotective effects.
The commercial availability of BPC-157 as a "research peptide" and its growing off-label use create pressure to overstate the clinical implications of animal data. The preclinical evidence for hepatoprotection is genuine and consistent, but it does not support clinical claims about liver protection in humans. The peptide's regulatory status varies by jurisdiction, and its use outside of clinical trials carries inherent risks given the absence of human pharmacokinetic and safety data for liver applications specifically. Until human trials are conducted, the clinical relevance of BPC-157 hepatoprotection remains a hypothesis supported by consistent but exclusively animal evidence.
The Bottom Line
BPC-157 has demonstrated hepatoprotective effects across more than seven liver injury models in rats: CCl4 poisoning, bile duct ligation, hepatic artery ligation, restraint stress, chronic alcohol, NSAID toxicity, and radiation injury. The peptide consistently outperforms comparison drugs and operates through nitric oxide system modulation (via the Src-Caveolin-1-eNOS pathway) and transcriptional regulation (KLF4 upregulation). The evidence is remarkably consistent but comes exclusively from animal studies, primarily from one research group. No human clinical trial has evaluated BPC-157 for liver protection, and the translation from rat hepatoprotection to human clinical benefit remains entirely unvalidated.
Sources & References
- 1RPEP-00278·Sikiric, P et al. (1993). “BPC 157 Protects the Liver From Multiple Types of Damage — Even When Taken Orally.” Life sciences.Study breakdown →PubMed →↩
- 2RPEP-06206·Huang, Bing-Shen et al. (2022). “Oral BPC-157 Protects the Liver from Radiation Damage by Activating the KLF4 Pathway in Mice.” Life sciences.Study breakdown →PubMed →↩
- 3RPEP-00692·Prkacin, I et al. (2001). “BPC-157 Both Prevents and Reverses Alcohol-Induced Liver Damage and Portal Hypertension.” Journal of physiology.Study breakdown →PubMed →↩
- 4RPEP-00691·Prkacin, I et al. (2001). “BPC-157 Provides Long-Lasting Stomach Protection Against Chronic Alcohol Damage.” Journal of physiology.Study breakdown →PubMed →↩
- 5RPEP-01118·Boban-Blagaic, Alenka et al. (2006). “BPC-157's Anti-Alcohol Effects Involve the Nitric Oxide System.” Medical science monitor : international medical journal of experimental and clinical research.Study breakdown →PubMed →↩
- 6RPEP-01783·Ilic, Spomenko et al. (2011). “BPC-157 Protects Against Diclofenac-Induced Multi-Organ Damage: Gut, Liver, and Brain.” Life sciences.Study breakdown →PubMed →↩
- 7RPEP-01782·Ilic, Spomenko et al. (2011). “BPC-157 Protects Against Ibuprofen-Induced Liver Damage and Brain Swelling in Rats.” European journal of pharmacology.Study breakdown →PubMed →↩
- 8RPEP-03271·Drmic, Domagoj et al. (2017). “Celecoxib-induced gastrointestinal, liver and brain lesions in rats, counteraction by BPC 157 or L-arginine, aggravation by L-NAME..” World journal of gastroenterology.Study breakdown →PubMed →↩
- 9RPEP-03025·Lojo, Nermin et al. (2016). “BPC 157 Protects Against Organ Damage After Major Bowel Surgery in Rats.” PloS one.Study breakdown →PubMed →↩
- 10RPEP-04861·Hsieh, Ming-Jer et al. (2020). “BPC-157 Relaxes Blood Vessels Through a Mapped Molecular Pathway Involving Nitric Oxide.” Scientific reports.Study breakdown →PubMed →↩
- 11RPEP-11666·Józwiak, Michalina et al. (2025). “BPC 157: Comprehensive Review of the Body Protection Compound's Benefits, Safety, and Regulatory Status.” Pharmaceuticals (Basel.Study breakdown →PubMed →↩
- 12RPEP-00383·Sikirić, P et al. (1996). “BPC-157 Protects the Stomach Against Damage from Stress, Alcohol, NSAIDs, and Capsaicin.” Digestive diseases and sciences.Study breakdown →PubMed →↩
- 13RPEP-04022·Škrlec, Katja et al. (2018). “Engineering recombinant Lactococcus lactis as a delivery vehicle for BPC-157 peptide with antioxidant activities..” Applied microbiology and biotechnology.Study breakdown →PubMed →↩