Selank vs Semax: How Two Neuropeptides Compare
Brain Peptide Bioregulators
84 Genes
Selank altered expression of 84 neurotransmission-related genes in rat frontal cortex in a single study, spanning GABA, serotonin, and dopamine systems.
Filatova et al., Frontiers in Pharmacology, 2017
Filatova et al., Frontiers in Pharmacology, 2017
If you only read one thing
Selank and Semax came out of the same Russian lab in the 1990s — but they do opposite jobs. Selank is built to calm anxiety without making you sleepy, like a benzodiazepine with fewer downsides. Semax is built to sharpen focus and protect brain cells by boosting a growth factor called BDNF. Think of them as siblings with different careers: Selank is the calm one, Semax is the quick-thinking one. Neither is FDA approved, and nearly all the research comes from Russia.
Selank and Semax are two synthetic peptides developed at the Institute of Molecular Genetics of the Russian Academy of Sciences during the 1990s. Both are registered medications in Russia. Both are administered intranasally. And both target the central nervous system. But they do fundamentally different things. Selank is classified as an anxiolytic. Semax is classified as a nootropic. Their parent molecules, receptor targets, and downstream effects diverge sharply, and the research base for each peptide tells a different story. This article is part of the brain peptide bioregulators cluster, which covers Russian-developed neuropeptides and their evidence base.
Key Takeaways
- Selank and Semax came out of the same Russian lab in the 1990s, but they do opposite jobs — one calms you down, one sharpens your focus.
- Selank works like a benzodiazepine without the downsides. In one trial, it matched Valium-class drugs for anxiety with no sedation, memory issues, or withdrawal.
- Semax tells your brain to produce 50 times more BDNF — a growth factor that keeps neurons alive and helps build new connections.
- Both are tiny 7-amino-acid peptides taken as a nasal spray. Neither is a pill and neither is swallowed.
- They are NOT interchangeable. A 2020 brain-imaging study showed they light up different networks and complement each other rather than substitute.
- Almost all research on both peptides comes from Russia. Outside that country, they are neither FDA approved nor widely studied.
- Both survive the blood-brain barrier easily and start working within 30 minutes — unusual for peptides, most of which are destroyed before they reach the brain.
Parent Molecules
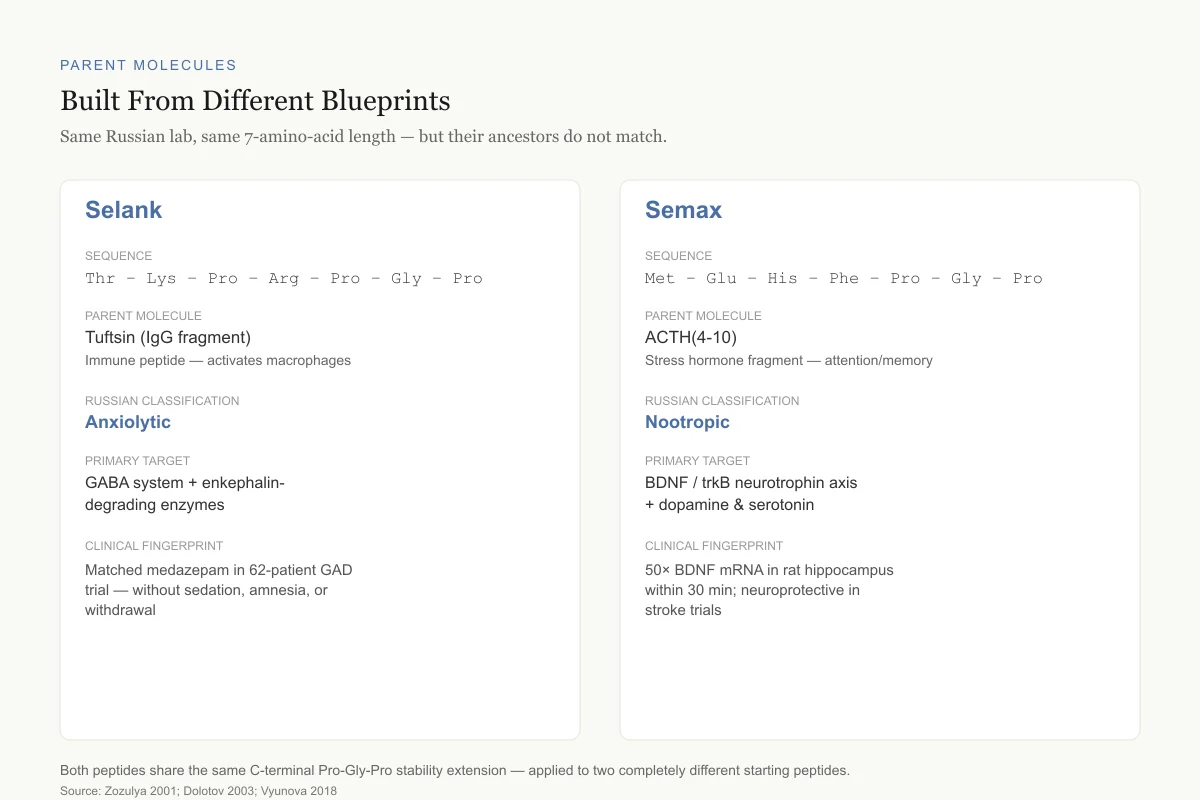
Built From Different Blueprints
Both peptides came out of the same Russian lab. Their ancestors did not.
Selank
Sequence
Thr – Lys – Pro – Arg – Pro – Gly – Pro
Parent molecule
Tuftsin (IgG fragment)
Immune peptide — activates macrophages and phagocytes
Russian classification
Anxiolytic
Primary target
GABA system + enkephalin-degrading enzymes
Clinical fingerprint
Matched medazepam in 62-patient GAD trial — without sedation, amnesia, or withdrawal
Semax
Sequence
Met – Glu – His – Phe – Pro – Gly – Pro
Parent molecule
ACTH(4-10)
Stress hormone fragment — attention and memory effects in animals
Russian classification
Nootropic
Primary target
BDNF / trkB neurotrophin axis + dopamine & serotonin
Clinical fingerprint
50× BDNF mRNA in rat hippocampus within 30 min; neuroprotective in stroke trials
Both peptides share the C-terminal Pro-Gly-Pro extension — the same stability trick, applied to two completely different starting peptides.
Source: Zozulya 2001; Dolotov 2003; Vyunova 2018
 View as image
View as imageWhere Selank and Semax Come From
Both peptides emerged from the same laboratory, but they were built from entirely different blueprints.
Selank (Thr-Lys-Pro-Arg-Pro-Gly-Pro) is a seven-amino-acid peptide derived from tuftsin, a naturally occurring tetrapeptide (Thr-Lys-Pro-Arg) cleaved from the heavy chain of immunoglobulin G.[1] The Russian team extended tuftsin's C-terminus with Pro-Gly-Pro to improve metabolic stability. Tuftsin itself is an immune modulator, not a neurotransmitter, which is why Selank's immunomodulatory effects were among the first properties documented.
Semax (Met-Glu-His-Phe-Pro-Gly-Pro) is also seven amino acids, but its core is ACTH(4-10), a fragment of adrenocorticotropic hormone. Like Selank, the C-terminal Pro-Gly-Pro extension prevents rapid enzymatic degradation.[2] ACTH fragments have long been associated with attention and memory in animal models, which is why Semax was developed as a nootropic rather than an anxiolytic.
The structural difference matters because it determines receptor binding. Selank's tuftsin backbone gives it affinity for systems involved in immune regulation and, unexpectedly, GABAergic signaling. Semax's ACTH backbone gives it access to melanocortin-adjacent pathways and neurotrophin regulation.
How Selank Works: GABA, Enkephalins, and Anxiety
Selank's primary mechanism centers on the GABAergic system. A 2017 study by Filatova et al. examined the expression of genes involved in GABAergic neurotransmission in human neuroblastoma cells (IMR-32) after Selank exposure. The peptide altered expression of genes encoding GABA-A receptor subunits, GABA transporters, and glutamic acid decarboxylase, the enzyme that synthesizes GABA.[3] The pattern resembled benzodiazepine-like modulation, but without direct receptor binding.
The enkephalin pathway adds a second layer. Zozulya et al. (2001) measured enkephalin-degrading enzyme activity in blood samples from 62 patients with generalized anxiety disorder and found that anxiety patients had accelerated enkephalin breakdown. Selank's anxiolytic effect correlated with its ability to inhibit these enzymes, preserving endogenous opioid-mediated anxiety relief.[1] The effect was comparable to medazepam in clinical assessment scales (Hamilton, Zung, CGI) but carried additional antiasthenic and psychostimulant properties that the benzodiazepine lacked.
In a 2017 animal study, Kasian et al. showed that Selank enhanced the anxiolytic effect of diazepam in rats subjected to unpredictable chronic mild stress. The combination produced greater anxiety reduction than either compound alone, suggesting Selank works through complementary rather than identical pathways to classical benzodiazepines.[4]
Selank also acts on serotonin and other neurotransmitter systems, which likely explains its broader spectrum compared to pure GABAergic drugs.
How Semax Works: BDNF, Dopamine, and Neurotrophin Cascades
Semax's signature effect is BDNF upregulation. Dolotov et al. (2003) administered Semax intranasally to rats and measured BDNF mRNA in multiple brain regions. Within 30 minutes, BDNF expression in the hippocampus increased roughly 50-fold. The effect was region-specific: basal forebrain and frontal cortex showed significant increases, while the hypothalamus did not respond.[5]
A follow-up study from the same group (Dolotov et al., 2006) confirmed that Semax not only increased BDNF mRNA but also raised BDNF protein levels and upregulated trkB, the receptor through which BDNF signals.[6] This is a critical distinction. Many compounds can transiently boost mRNA without producing functional protein. Semax appeared to do both, at least in rat hippocampus.
The dopamine and serotonin effects operate independently from the BDNF pathway. Eremin et al. (2005) measured monoamine metabolites in six brain regions of rats after Semax injection. HVA (a dopamine metabolite) increased by approximately 80% in the striatum, while 5-HIAA (a serotonin metabolite) rose by about 30% in the same region.[7] This dual monoaminergic activation likely underlies Semax's effects on attention and motivation.
Semax has also been studied as a neuroprotective agent. Romanova et al. (2006) tested Semax in rats with experimental ischemic infarction of the cerebral cortex and found both neuroprotective and antiamnesic effects: treated animals showed less tissue damage and better retention of learned behaviors compared to controls.[8] These neuroprotective properties connect to the broader field of neurotrophic peptides and brain health.
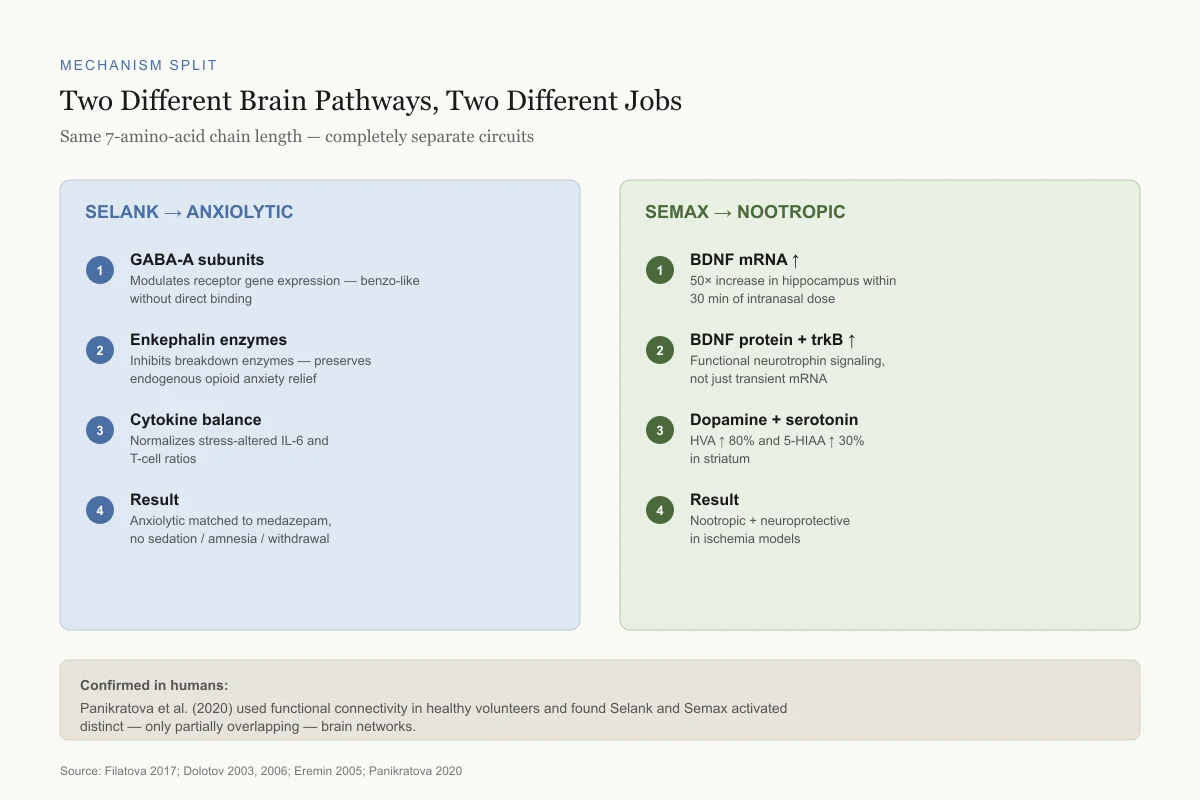
Mechanism Split
Two Different Brain Pathways, Two Different Jobs
The same 7-amino-acid chain length — completely separate circuits
Selank → Anxiolytic
GABA-A subunits
Modulates receptor gene expression — benzodiazepine-like effect without direct binding
Enkephalin enzymes
Inhibits enkephalin-degrading enzymes — preserves endogenous opioid anxiety relief
Cytokine balance
Normalizes stress-altered IL-6 and T-cell balance
Result
Anxiolytic matched to medazepam, no sedation/amnesia/withdrawal
Semax → Nootropic
BDNF mRNA ↑
50× increase in hippocampus within 30 min of intranasal dose
BDNF protein + trkB ↑
Functional neurotrophin signaling, not just transient mRNA
Dopamine + serotonin
HVA ↑ 80% and 5-HIAA ↑ 30% in striatum
Result
Nootropic + neuroprotective in ischemia models
Confirmed in humans: Panikratova et al. (2020) used functional connectivity in healthy volunteers and found Selank and Semax activated distinct — only partially overlapping — brain networks.
Source: Filatova 2017; Dolotov 2003, 2006; Eremin 2005; Panikratova 2020
 View as image
View as imageHead-to-Head: Where the Evidence Diverges
| Feature | Selank | Semax |
|---|---|---|
| Parent molecule | Tuftsin (IgG fragment) | ACTH(4-10) |
| Amino acid sequence | Thr-Lys-Pro-Arg-Pro-Gly-Pro | Met-Glu-His-Phe-Pro-Gly-Pro |
| Russian classification | Anxiolytic | Nootropic |
| Primary neural target | GABA system, enkephalins | BDNF/neurotrophin system |
| Monoamine effects | Serotonin modulation | Dopamine + serotonin activation |
| Human clinical data | GAD trial (62 patients) | Stroke trials (100+ patients) |
| Immune effects | Cytokine modulation (IL-6, T-cell balance) | Immune gene expression in ischemia |
| Administration | Intranasal | Intranasal |
| Side effect profile | Minimal reported | Minimal reported |
The comparison reveals a pattern: Selank is primarily anxiolytic with secondary cognitive effects, while Semax is primarily cognitive-enhancing with secondary mood effects. A 2024 study by Inozemtseva et al. tested Semax specifically for antidepressant and antistress properties in chronically stressed rats and found significant effects, but the mechanism involved dopaminergic and serotonergic activation rather than GABAergic modulation.[9] Semax can reduce stress, but it gets there through a fundamentally different route than Selank.
Brain Network Evidence: The Connectomic Study
One study directly compared both peptides in the same experimental framework. Panikratova et al. (2020) used functional connectivity analysis to examine how Selank and Semax altered brain network activity in healthy volunteers. The study found that each peptide activated distinct neural circuits, with only partial overlap.[10]
This is the closest thing to a direct head-to-head comparison in the literature. The results align with the pharmacological differences: Selank shifted activity in circuits associated with emotional regulation, while Semax shifted activity in circuits associated with attention and executive function. The partial overlap is consistent with both peptides affecting serotonergic signaling, just from different angles.
For context on how other peptides compare in cognitive enhancement, see the nootropic peptides overview and the Noopept profile.
Beyond Anxiety and Cognition: Other Research Directions
Both peptides show up in research areas beyond their primary classifications.
Alcohol-related research. Selank has been studied in ethanol models from multiple angles. Kolik et al. (2019) found that Selank protected against ethanol-induced memory impairment in rats by regulating BDNF content in the hippocampus and prefrontal cortex.[11] The BDNF connection is notable because it means Selank, typically characterized as a GABA modulator, can also influence neurotrophin pathways under certain conditions.
Neurodegeneration. Slominsky et al. (2017) tested both peptides in rats with 6-OHDA-induced Parkinson's-like symptoms. Both Semax and Selank improved behavioral outcomes, but gene expression analysis revealed different protective mechanisms: Semax upregulated neurotrophic factors while Selank modulated stress-response genes.[12]
Immune modulation. Leonidovna et al. (2021) examined Selank's effect on cytokine levels under social stress conditions and found that Selank normalized stress-altered cytokine profiles, connecting its anxiolytic effects to immune regulation.[13] Semax shows immune effects too, but primarily in the context of ischemic brain injury, where it upregulates chemokine and immunoglobulin genes as part of a broader neuroprotective response.
Limitations of the Current Evidence
The research base for both peptides shares several weaknesses worth noting.
Most studies come from Russian laboratories, many published in Russian-language journals with limited English-language peer review. This does not invalidate the science, but it does limit independent replication. The standard practice in Western drug development, where competing labs attempt to reproduce key findings before clinical adoption, has not been widely applied to either peptide. Few if any Western academic groups have published independent confirmation of the core mechanistic claims.
Human clinical data is thin. Selank's strongest human evidence comes from a single 62-patient anxiety trial from 2001 that compared it to only one benzodiazepine. Semax's clinical data comes primarily from Russian stroke studies conducted over two decades ago. Neither peptide has undergone the kind of large, multi-center, placebo-controlled trials that regulatory agencies in Europe or North America require for drug approval. Phase III trial data, the gold standard, does not exist for either compound in Western regulatory frameworks.
The animal studies, while internally consistent within individual research groups, use diverse models and dosing protocols that make cross-study comparison difficult. BDNF fold-changes, for instance, vary dramatically depending on the brain region sampled, the time point measured, the route of administration, and the strain of rat used. A 50-fold mRNA increase in one study does not necessarily translate to the same magnitude of protein-level change or functional outcome in a different experimental setup.
Dose-response data in humans is sparse. The optimal intranasal doses for cognitive or anxiolytic effects have not been established through systematic dose-ranging trials. Most human use outside of Russian clinical settings is based on extrapolation from animal data or anecdotal reports.
Neither Selank nor Semax is approved by the FDA, EMA, or most regulatory agencies outside of Russia and a few CIS countries. Both are available through research chemical suppliers, but quality control, purity verification, and peptide identity confirmation in this market are inconsistent.
Safety
ModerateGray-market quality is the biggest practical risk
Concern
Outside Russia, Selank and Semax are sold as 'research chemicals' without regulatory oversight. Published Russian trials used pharmaceutical preparations with verified purity. Retail research-chemical vials may contain underdosed, overdosed, or misidentified peptide, and no enforcement mechanism catches bad batches before they reach users.
What the research says
Clinical safety data for Selank and Semax used verified pharmaceutical-grade material. Those safety signals do not automatically transfer to anonymous vials from online vendors.
Particularly relevant for: Anyone buying Selank or Semax outside of Russia
What to do
If purchasing research-grade peptides, prefer vendors publishing batch-specific mass-spec and endotoxin CoAs. Recognize you are accepting unverified quality as part of the deal.
FDA gray-market peptide testing; research-chemical supply chain analyses
Who Studies What, and Why
The division between these two peptides maps to different clinical needs. Selank research clusters around anxiety, stress resilience, and conditions where emotional regulation is impaired. Semax research clusters around cognitive decline, neurodegeneration, and conditions where BDNF or neurotrophin deficiency plays a role.
This specialization reflects their origins. Tuftsin, Selank's parent, is an immune peptide that was repurposed for neuroscience. ACTH, Semax's parent, is a stress hormone fragment that was optimized for cognition. Each peptide inherited the biological neighborhood of its ancestor.
The two peptides are not competitors. They target different aspects of brain function through different mechanisms, and the limited evidence available suggests they may be complementary rather than redundant. Whether that complementarity translates into clinical benefit for any specific condition remains an open question, one that can only be resolved by trials designed to test it.
For comparison with other peptide-derived cognitive enhancers, see the profile on Dihexa, a peptide described as millions of times more potent than BDNF.
The Bottom Line
Selank and Semax are both seven-amino-acid peptides from the same Russian laboratory, but they diverge at every mechanistic level. Selank modulates GABA, enkephalins, and immune cytokines, producing anxiolytic effects comparable to benzodiazepines without their side effects in one 62-patient trial. Semax upregulates BDNF up to 50-fold, activates dopamine and serotonin pathways, and shows neuroprotective effects in ischemia models. Human data for both peptides remains limited and largely confined to Russian clinical settings.
Sources & References
- 1RPEP-00708·Zozulya, A A et al. (2001). “Selank Treats Anxiety by Protecting the Body's Natural Anxiety-Relieving Enkephalins.” Bulletin of experimental biology and medicine.Study breakdown →PubMed →↩
- 2RPEP-03968·Vyunova, Tatiana V et al. (2018). “How Selank Peptide Reduces Anxiety by Modulating GABA Receptors Differently Than Benzodiazepines.” Protein and peptide letters.Study breakdown →PubMed →↩
- 3RPEP-03286·Filatova, Elena et al. (2017). “GABA, Selank, and Olanzapine Affect the Expression of Genes Involved in GABAergic Neurotransmission in IMR-32 Cells..” Frontiers in pharmacology.Study breakdown →PubMed →↩
- 4RPEP-03340·Kasian, Anastasiya et al. (2017). “Peptide Selank Enhances the Effect of Diazepam in Reducing Anxiety in Unpredictable Chronic Mild Stress Conditions in Rats..” Behavioural neurology.Study breakdown →PubMed →↩
- 5RPEP-00812·Dolotov, O V et al. (2003). “The heptapeptide SEMAX stimulates BDNF expression in different areas of the rat brain in vivo..” Doklady biological sciences : proceedings of the Academy of Sciences of the USSR.Study breakdown →PubMed →↩
- 6RPEP-01129·Dolotov, Oleg V et al. (2006). “Semax Boosts Brain-Derived Neurotrophic Factor (BDNF) in the Hippocampus.” Brain research.Study breakdown →PubMed →↩
- 7RPEP-01031·Eremin, Kirill O et al. (2005). “Semax, an ACTH(4-10) analogue with nootropic properties, activates dopaminergic and serotoninergic brain systems in rodents..” Neurochemical research.Study breakdown →PubMed →↩
- 8RPEP-01182·Romanova, G A et al. (2006). “Neuroprotective and antiamnesic effects of Semax during experimental ischemic infarction of the cerebral cortex..” Bulletin of experimental biology and medicine.Study breakdown →PubMed →↩
- 9RPEP-08436·Inozemtseva, Ludmila S et al. (2024). “Antidepressant-like and antistress effects of the ACTH(4-10) synthetic analogs Semax and Melanotan II on male rats in a model of chronic unpredictable stress..” European journal of pharmacology.Study breakdown →PubMed →↩
- 10RPEP-05052·Panikratova, Ya R et al. (2020). “Selank and Semax Alter Brain Connectivity Between Amygdala and Temporal Cortex in Humans.” Doklady biological sciences : proceedings of the Academy of Sciences of the USSR.Study breakdown →PubMed →↩
- 11RPEP-04287·Kolik, L G et al. (2019). “Selank Peptide Prevented Alcohol-Induced Memory Loss by Regulating BDNF in Rat Brains.” Bulletin of experimental biology and medicine.Study breakdown →PubMed →↩
- 12RPEP-03474·Slominsky, P A et al. (2017). “Selank Peptide Reduced Anxiety in Rats with Parkinson's-like Brain Damage.” Doklady biological sciences : proceedings of the Academy of Sciences of the USSR.Study breakdown →PubMed →↩
- 13RPEP-05539·Leonidovna, Yasenyavskaya A et al. (2021). “The Influence of Selank on the Level of Cytokines Under the Conditions of "Social" Stress..” Current reviews in clinical and experimental pharmacology.Study breakdown →PubMed →↩