Dihexa: Millions of Times More Potent Than BDNF?
Dihexa & HGF-Based Nootropics
0 human clinical trials
Despite widespread online promotion as a nootropic, Dihexa has zero published human trials. A key mechanistic study was retracted in 2025 for image manipulation, and its prodrug failed a Phase 2/3 Alzheimer's trial.
Benoist et al., 2014 (retracted April 2025); Athira LIFT-AD, 2024
Benoist et al., 2014 (retracted April 2025); Athira LIFT-AD, 2024
If you only read one thing
Dihexa is a small peptide that became internet-famous after a 2013 paper called it '10 million times more potent than BDNF' at building new brain connections — a comparison that sounds amazing but compares apples to oranges, since the two molecules work through totally different systems. The story has fallen apart since: the key mechanism paper was retracted in 2025 for fraud, the prodrug version failed a major Alzheimer's trial in 302 patients, and the company that commercialized it settled a $4 million fraud case. There are no human trials of Dihexa itself, and it activates a cancer-promoting pathway with no long-term safety data. Interesting molecule, but the gap between 'works in rat brain cells' and 'safe and useful for humans' has not been bridged.
Dihexa (N-hexanoic-Tyr-Ile-(6) aminohexanoic amide) is a small synthetic peptide that became one of the most hyped compounds in the nootropic community after a 2013 study described it as seven orders of magnitude more potent than BDNF (brain-derived neurotrophic factor) at promoting synaptogenesis in cell culture.[1] That comparison, often shortened to "10 million times more potent than BDNF," spread rapidly across forums, social media, and supplement vendor sites. It became the defining claim of Dihexa's online reputation. For a detailed breakdown of what that potency claim actually measured, see the pillar article in this cluster. This article covers the broader picture: what Dihexa is, what the full evidence base looks like, what went wrong with its commercialization, and what the compound's trajectory tells us about the gap between preclinical data and clinical reality.
Key Takeaways
- Dihexa became internet-famous after one 2013 paper called it "10 million times more potent than BDNF" at building new brain connections.
- That comparison is real but misleading — the two molecules work through totally different systems, like comparing a key for one lock to a key for another.
- The study establishing how Dihexa supposedly works was retracted in 2025 after investigators found the author had faked images across her research.
- The only human trial — of a Dihexa-based drug — failed in 302 Alzheimer's patients. No measurable benefit for thinking or function.
- In rats, Dihexa only helped brains that were already damaged. It did nothing in healthy rats — not the "smart pill for everyone" story the internet tells.
- The pathway it activates is the same one that drives cancer growth. There are FDA-approved cancer drugs that block it — and zero long-term safety studies on Dihexa.
- Nothing you can buy online as "Dihexa" has ever been tested in humans. The compound is a research chemical, not a proven therapy.
What Dihexa actually is
Dihexa is a peptidomimetic derived from angiotensin IV (AngIV), a six-amino-acid fragment of angiotensin II. AngIV had been known since the 1990s to enhance memory and learning in animal models, but it degrades within minutes in blood and cannot cross the blood-brain barrier. The Harding laboratory at Washington State University synthesized Dihexa as a metabolically stable AngIV analog: a small molecule (molecular weight 587 Da) designed for oral bioavailability and blood-brain barrier penetration.[4]
This is pharmacologically unusual for a peptide-derived compound. Most peptides are too large, too polar, or too easily degraded by proteases to survive oral administration and reach the brain. Dihexa's small size and chemical modifications (N-hexanoic acid cap, aminohexanoic amide tail) give it drug-like properties that native AngIV lacks.
The proposed mechanism: Dihexa binds to hepatocyte growth factor (HGF) at a site that prevents the dimerization that normally inhibits HGF activity. This increases the local concentration of active HGF available to bind the c-Met receptor, which triggers downstream signaling cascades that promote synaptogenesis (the formation of new synaptic connections between neurons). Wright and Harding (2015) positioned this HGF/c-Met system as "a new target for the treatment of Alzheimer's disease."[5]
Apples and Oranges
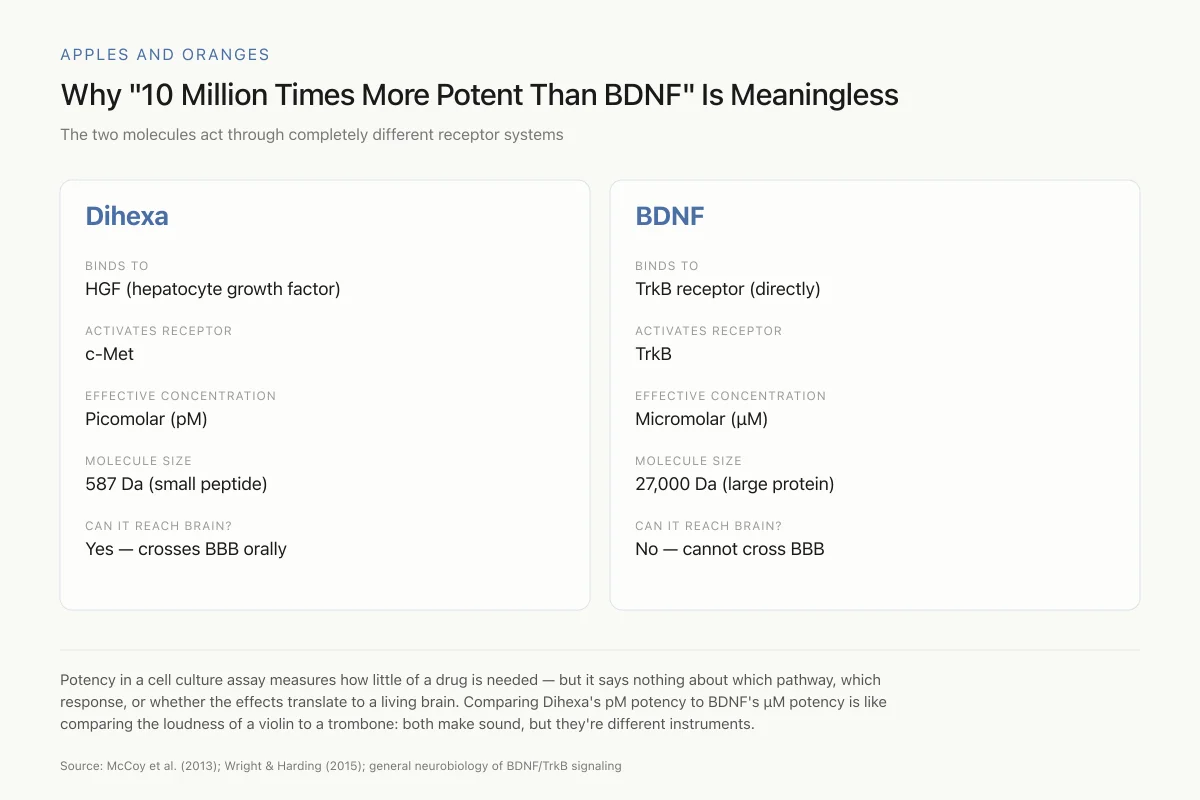
Why "10 Million Times More Potent Than BDNF" Is a Meaningless Comparison
The two molecules act through completely different receptor systems
Potency in a cell culture assay measures how little of a drug is needed to produce a response — but it says nothing about which response, which pathway, or whether the effects translate to a living brain. Comparing Dihexa's pM potency to BDNF's µM potency is like comparing the loudness of a violin to a trombone: both make sound, but they're different instruments answering different questions.
Source: McCoy et al. (2013); Wright & Harding (2015); general neurobiology of BDNF/TrkB signaling
 View as image
View as imageThe original animal data
McCoy et al. (2013) is the foundational study. In scopolamine-impaired rats (a model that mimics Alzheimer's-like cognitive deficits by blocking cholinergic signaling), Dihexa at 2 mg/kg per day orally restored performance in the Morris water maze to levels indistinguishable from healthy controls. It outperformed donepezil (Aricept), one of the few FDA-approved Alzheimer's drugs, in the same model.[1]
In hippocampal cell cultures, Dihexa increased dendritic spine density by approximately 3-fold (41 spines per 50 micrometers of dendrite versus 15 in controls). It was from this in vitro data that the potency comparison with BDNF originated. At the concentrations tested, Dihexa produced similar synaptogenic effects at concentrations seven orders of magnitude lower than BDNF. This is where the "10 million times" number comes from.
A critical finding often omitted from online discussions: Dihexa had no cognitive effects in rats with normal cognition. It only improved performance in animals with experimentally impaired memory. This contradicts the popular framing of Dihexa as a general-purpose "cognitive enhancer." The data points toward a cognitive rescuer for impaired states, not a universal nootropic for healthy brains.
Independent replication
Sun et al. (2021) from Nanjing Medical University provided the only published independent replication of Dihexa's cognitive effects. They administered Dihexa to APP/PS1 transgenic mice, which carry human mutations that cause familial Alzheimer's disease and naturally develop amyloid plaques and cognitive decline.[2]
Dihexa restored spatial learning and cognitive function in the Morris water maze. It increased neuronal cells and expression of synaptophysin (SYP) protein. It decreased activation of astrocytes and microglia, reduced pro-inflammatory cytokines IL-1-beta and TNF-alpha, and increased anti-inflammatory cytokine IL-10. The mechanism appeared to operate through PI3K/AKT signaling rather than the HGF/c-Met pathway proposed by the original group.
This is important for two reasons. First, it provides evidence from a lab with no connection to the original researchers, using a different and arguably more clinically relevant Alzheimer's mouse model. Second, the different signaling pathway raises the possibility that Dihexa's cognitive effects may not depend on HGF/c-Met at all, which matters because the evidence for the HGF/c-Met mechanism was compromised by the retraction.
The retraction and the fraud
The 2014 Benoist et al. paper, published in the Journal of Pharmacology and Experimental Therapeutics, was the study that provided the molecular basis for Dihexa's proposed HGF/c-Met mechanism. It claimed that Dihexa binds HGF with high affinity and acts synergistically with HGF to promote c-Met receptor phosphorylation and synaptogenesis.
In September 2021, four papers co-authored by Leen Kawas received expressions of concern from their journals, triggered by allegations of image manipulation in her doctoral dissertation at Washington State University. An investigation found systematic alteration of images, including copying and pasting data between experiments, digitally modifying Western blot bands, and reusing identical images to represent different conditions. Of 30 images examined from her dissertation, problems were identified in 19.
In April 2025, the Benoist et al. paper was formally retracted. The original behavioral study (McCoy 2013) received an expression of concern but was not retracted, likely because its primary data (behavioral testing, spine counting) used different methodologies than the manipulated Western blots.
Kawas had founded M3 Biotechnology (later Athira Pharma) to commercialize HGF-targeting compounds derived from this research. Athira went public in September 2020, raising $238 million. After the allegations surfaced, Kawas was removed as CEO. In January 2025, Athira paid $4,068,698 to settle False Claims Act allegations after a whistleblower revealed the company had concealed research misconduct from federal agencies for five years.
Research & Commercialization Timeline
From Foundational Paper to Retraction: Dihexa's 12-Year Arc
Dihexa restores cognition in scopolamine-impaired rats at 2 mg/kg oral; picomolar synaptogenesis in cell culture
Proposes HGF/c-Met binding as molecular basis — becomes foundation for commercialization
Fosgonimeton (Dihexa prodrug) developer raises $238M on public markets
Four Kawas papers flagged for image manipulation; Kawas removed as CEO
Chinese group confirms cognitive rescue in APP/PS1 mice — via PI3K/AKT, not HGF/c-Met
Fosgonimeton misses primary and key secondary endpoints in 302 Alzheimer's patients
Wells et al. find Dihexa ineffective for motor/cognitive protection in HD rat model
Athira settles False Claims Act case for concealing research misconduct
Key mechanism paper formally retracted for systematic image manipulation
Sources: McCoy 2013; Benoist 2014 (retracted 2025); Sun 2021; Wells 2024; Athira LIFT-AD 2024; DOJ settlement 2025
 View as image
View as imageFosgonimeton: the clinical failure
Athira Pharma developed fosgonimeton, a phosphate prodrug of Dihexa designed for subcutaneous injection, and advanced it into Phase 2/3 clinical trials for Alzheimer's disease. The LIFT-AD trial enrolled 302 patients with mild-to-moderate Alzheimer's and ran for 26 weeks.
The trial failed to meet its primary or key secondary endpoints. Fosgonimeton did not produce statistically significant improvement in cognition or function compared to placebo. Biomarker data showed reductions in phosphorylated tau217 (p < 0.01), suggesting some biological activity, but this did not translate into clinical benefit.
This result is consistent with a pattern in Alzheimer's research: compounds that promote synaptogenesis in cell culture and restore cognition in rodent models frequently fail in human trials. The complexity of human Alzheimer's disease, which involves amyloid plaques, tau tangles, neuroinflammation, vascular dysfunction, and progressive neuronal death, may overwhelm the capacity of any single synaptogenic mechanism. For context on how synaptogenesis studies translated to this clinical trial, see the dedicated cluster article.
Where Dihexa has not worked
Wells et al. (2024) tested Dihexa (designated PNB-0408) in a rat model of Huntington's disease. Rats received Dihexa alongside chronic exposure to 3-nitropropionic acid, a mitochondrial toxin that mimics Huntington's pathology. After five weeks of treatment, Dihexa did not protect rats from cognitive deficits, motor dysfunction, or weight loss induced by the toxin. The authors concluded that "PNB-0408 may not be an efficacious treatment strategy" for this indication.[3]
This negative result matters because it sets boundaries on Dihexa's therapeutic profile. The compound is not a universal neuroprotectant. Its cognitive effects appear specific to certain types of impairment (cholinergic blockade, amyloid-driven neurodegeneration) and absent in others (mitochondrial toxicity).
The systematic evidence landscape
Ho and Nation (2018) conducted a systematic review of experimental studies examining cognitive benefits of angiotensin IV and related peptides. Of 450 articles identified, 32 met inclusion criteria. Seven of 11 studies in normal animals found AngIV had beneficial effects on passive or conditioned avoidance and object recognition. In models of cognitive deficit, eight of nine studies found that AngIV and its analogs (including Dihexa) improved spatial working memory and passive avoidance performance.[6]
The review noted a significant limitation: most studies used compounds developed by the same research group (the Harding/Wright laboratory), meaning the evidence base was concentrated rather than independent. The review was published before the retraction, when the full scope of the data integrity issues was not yet known.
Safety: the c-Met oncogene concern
The HGF/c-Met signaling pathway is a well-characterized proto-oncogene system. In cancer biology, aberrant c-Met activation promotes tumor cell proliferation, invasion, and metastasis across multiple cancer types. Multiple c-Met inhibitors (capmatinib, tepotinib, crizotinib) are FDA-approved cancer drugs specifically because blocking c-Met activity slows tumor growth.
Dihexa does the pharmacological opposite: it potentiates HGF/c-Met signaling. While the proposed mechanism involves amplifying existing HGF signaling at subthreshold levels rather than constitutive receptor activation, no carcinogenicity studies have been conducted. No long-term safety data exists beyond approximately 3-month rodent studies. The theoretical oncogenic risk has not been systematically evaluated.
This is a substantive concern, not a hypothetical one. The c-Met pathway's role in cancer is supported by decades of research and multiple approved drugs targeting it. Activating this same pathway for cognitive benefits introduces a risk-benefit calculation that has not been characterized by any safety study.
Safety
CriticalActivating a known cancer pathway — with no long-term safety data
Concern
Dihexa potentiates the HGF/c-Met signaling pathway. c-Met is a proto-oncogene — its aberrant activation drives tumor growth and metastasis across multiple cancer types, and several FDA-approved cancer drugs (capmatinib, tepotinib, crizotinib) work by blocking exactly this pathway. Dihexa does the opposite direction of what those drugs do.
What the research says
No carcinogenicity studies have been conducted on Dihexa. The longest rodent safety studies are ~3 months. No human safety data exists at all. The theoretical risk is supported by decades of c-Met cancer biology, not speculation.
Particularly relevant for: Anyone using Dihexa from gray-market sources, especially long-term or at higher doses
What to do
Anyone considering Dihexa should weigh an unquantified but mechanistically plausible cancer risk against a cognitive benefit that has never been demonstrated in humans. The risk-benefit calculation on current evidence does not favor use.
Decades of c-Met cancer biology; absence of Dihexa-specific carcinogenicity studies
The state of the evidence
The Dihexa evidence base has a distinctive shape: extraordinary potency claims from a small number of preclinical studies, one formal retraction, one clinical trial failure, and zero published human data on the compound itself.
What has been established: Dihexa promotes synaptogenesis in hippocampal cell cultures at picomolar concentrations. It restores cognitive performance in at least two different rodent models of memory impairment (scopolamine-induced and APP/PS1 transgenic). It crosses the blood-brain barrier after oral administration. One independent replication exists.
What has been undermined: The molecular mechanism (HGF/c-Met binding and signaling) rests on a retracted paper. The clinical translation (fosgonimeton for Alzheimer's) failed. The development company settled fraud allegations.
What remains unknown: Whether Dihexa is safe for repeated use in humans. Whether the c-Met activation poses cancer risk. Whether the cognitive effects observed in rodents translate to any meaningful benefit in humans. Whether Dihexa works through HGF/c-Met at all, or through an alternative pathway like the PI3K/AKT axis identified by Sun et al.
For context on how other peptides approach cognitive enhancement through different neurotrophic pathways, see the article on how Semax upregulates BDNF. For the broader question of Alzheimer's peptide research, see cerebrolysin for Alzheimer's disease, which has a substantially larger clinical evidence base.
The Bottom Line
Dihexa produced striking preclinical results in cell culture and rodent models of cognitive impairment, but the path from laboratory to clinic has been marked by a key paper retraction, a failed Phase 2/3 clinical trial, and a fraud settlement. One independent replication of the cognitive effects exists. No human safety or efficacy data for the compound itself has been published. The c-Met activation mechanism raises unaddressed oncogenic concerns. The "10 million times more potent than BDNF" framing is a pharmacologically accurate but clinically meaningless comparison between two molecules acting through different receptor systems.
Sources & References
- 1RPEP-02233·McCoy, Alene T et al. (2013). “Evaluation of metabolically stabilized angiotensin IV analogs as procognitive/antidementia agents..” The Journal of pharmacology and experimental therapeutics.Study breakdown →PubMed →↩
- 2RPEP-05800·Sun, Xiaojin et al. (2021). “AngIV-Analog Dihexa Rescues Cognitive Impairment and Recovers Memory in the APP/PS1 Mouse via the PI3K/AKT Signaling Pathway..” Brain sciences.Study breakdown →PubMed →↩
- 3RPEP-09517·Wells, Russell G et al. (2024). “Dihexa Failed to Protect Against Huntington's Disease Symptoms in Rats.” Journal of Huntington's disease.Study breakdown →PubMed →↩
- 4RPEP-02837·Wright, John W et al. (2015). “The development of small molecule angiotensin IV analogs to treat Alzheimer's and Parkinson's diseases..” Progress in neurobiology.Study breakdown →PubMed →↩
- 5RPEP-02836·Wright, John W et al. (2015). “The Brain Hepatocyte Growth Factor/c-Met Receptor System: A New Target for the Treatment of Alzheimer's Disease..” Journal of Alzheimer's disease : JAD.Study breakdown →PubMed →↩
- 6RPEP-03709·Ho, Jean K et al. (2018). “Do Angiotensin Peptides Improve Memory? A Systematic Review of Animal Studies.” Neuroscience and biobehavioral reviews.Study breakdown →PubMed →↩