Cortexin: The Brain Peptide Bioregulator
Nootropic Peptides & Russian Neuropeptide Drugs
25+ years in clinical use
Cortexin has been prescribed in Russia and CIS countries since 1999 for stroke, TBI, and cognitive disorders, yet a 2021 systematic review found only 1 eligible trial with 80 participants for meta-analysis.
Alsulaimani et al., Cerebral Circulation, 2021
Alsulaimani et al., Cerebral Circulation, 2021
If you only read one thing
Cortexin is a brain extract from cows or pigs that has been used as an injection drug in Russia and nearby countries for more than 25 years, mainly for stroke recovery and brain injury. It is not approved in the US or Europe and is not sold there. The laboratory work is genuinely interesting — rat stroke data look comparable to the better-known drug Cerebrolysin. The human evidence is the problem: when Western reviewers tried to pool the trials in 2021, only one trial with 80 patients met their standards. So it is used a lot by real doctors on real patients, but by modern evidence rules we still cannot say it works.
Cortexin occupies an unusual position in global pharmacology. In Russia and former Soviet states, it is a mainstream prescription drug administered by intramuscular injection for stroke recovery, traumatic brain injury, cognitive decline, and pediatric neurological conditions. Outside that region, it is virtually unknown. A systematic review and meta-analysis published in 2021 attempted to assess the evidence for animal-derived nootropics (Cortexin, Cerebrolysin, and Actovegin) in cognitive disorders and found the clinical trial data for Cortexin so limited that meta-analysis was impossible: only one eligible trial with 80 participants.[1] This article examines what Cortexin is, what the laboratory and clinical research actually shows, and why a drug used by millions of patients has so little Western-standard evidence behind it. For context on how Cortexin fits within the broader landscape of cognitive-enhancing peptides, see Nootropic Peptides: A Scientific Overview of Cognitive Enhancers.
Key Takeaways
- Cortexin is a lyophilized extract of animal cerebral cortex containing a mixture of low-molecular-weight polypeptides, amino acids, and trace elements (CAS 83270-84-2)
- In a head-to-head comparison with Cerebrolysin in rat stroke models, Cortexin at 1-3 mg/kg/day improved neurological function, reduced infarct volume, and preserved neurons comparably to Cerebrolysin at 538-1614 mg/kg/day (Kurkin et al., 2021)
- A 2021 systematic review found only 1 eligible Cortexin trial (80 participants) for cognitive disorder meta-analysis, precluding any pooled efficacy estimate (Alsulaimani et al., 2021)
- Cortexin stimulated expression of neurotrophic factors (BDNF, NGF) and anti-apoptotic proteins in neuronal cultures from aged rats (Umnov et al., 2014)
- In a 979-patient observational study on post-COVID neurological symptoms, Cortexin at 10-20 mg IM for 10 days improved cognitive and asthenic scores (Putilina et al., 2022)
- A 2025 rat study showed Cortexin modulated OPG/RANK/RANKL and TRPC1 expression in cerebral ischemia-reperfusion injury, identifying new molecular targets (Guven et al., 2025)
The Evidence Gap
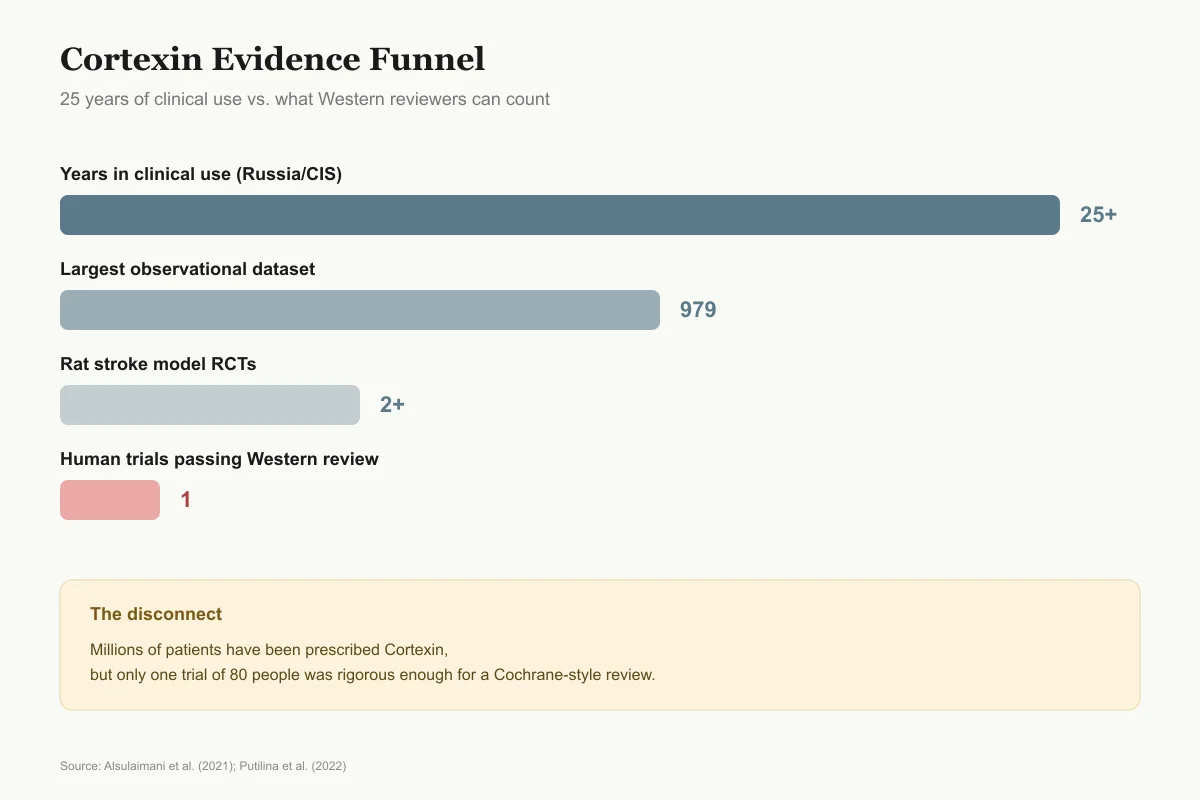
25 Years of Use vs What Western Reviewers Can Count
Years in clinical use (Russia/CIS)
25+
Approved by Russian authorities in 1999, prescribed routinely for stroke, TBI, cognitive decline
Largest observational dataset
979
CORTEX post-COVID program across Russia, Azerbaijan, Kyrgyzstan, Kazakhstan — no control group
Rat stroke model RCTs
2+
Kurkin 2021 and Zhang 2019 compared Cortexin vs Cerebrolysin in standardized models
Human trials passing Western review
1
80 participants; too few for any meta-analysis (Alsulaimani 2021 systematic review)
The disconnect: millions of patients have been prescribed Cortexin, but only one trial of 80 people was rigorous enough to include in a Cochrane-style review.
Source: Alsulaimani et al. (2021); Putilina et al. (2022)
 View as image
View as imageWhat Is Cortexin?
Cortexin is a multicomponent neuroprotective drug manufactured by Geropharm (Saint Petersburg, Russia). It is produced by lyophilizing an extract of the cerebral cortex of cattle or pigs, yielding a mixture of low-molecular-weight polypeptides (molecular weight below 10 kDa), L-amino acids (glycine being a significant component), and trace mineral elements. The resulting powder is reconstituted and administered by intramuscular injection.[2]
Unlike synthetic peptides with defined sequences (such as Semax or Selank), Cortexin is not a single molecule. It is a complex biological extract whose exact peptide composition varies between batches, though the manufacturing process aims for standardized biological activity. This distinction matters: the "active ingredient" is not one peptide but an ensemble of peptide fragments whose individual contributions to the overall effect are not fully characterized.
Cortexin received registration in Russia in 1999. It is classified as a nootropic and neuroprotective agent and is prescribed for ischemic stroke, traumatic brain injury, encephalopathy of various origins, cognitive disorders in children, and epilepsy-related cognitive decline. The standard adult dose is 10 mg intramuscularly daily for 10 days, with courses repeated at intervals. A pediatric formulation (5 mg) exists for children under 20 kg body weight.
The drug has no FDA approval, no European Medicines Agency authorization, and is not available in the United States, Canada, or Western Europe through regulated pharmacy channels. This creates a significant information asymmetry: millions of patients in Russia and CIS countries receive Cortexin as part of routine neurological care, while neurologists in the US and EU have typically never heard of it.
The composition question is fundamental to understanding Cortexin's evidence problem. When a drug's active ingredient is "a mixture of brain-derived peptides," reproducibility between studies becomes difficult. Two different batches of Cortexin may contain the same overall profile of peptide classes but different specific fragments, making it hard to attribute observed effects to particular molecular components. This contrasts with drugs like Semax (a synthetic heptapeptide with the defined sequence Met-Glu-His-Phe-Pro-Gly-Pro) where every dose contains identical molecules.
Proposed Mechanisms of Action
Because Cortexin is a multi-peptide mixture, it does not have a single mechanism of action. Research has identified several pathways through which its peptide components may exert neuroprotective effects.
Neurotrophic Factor Stimulation
Umnov et al. (2014) studied Cortexin's effects on organotypic brain cultures from young and old Wistar rats. Cortexin stimulated the expression of signal molecules involved in neuronal survival, including neurotrophic factors. The effective concentration of Cortexin for stimulating proliferative activity was lower than that of the synthetic peptide T-33 used for comparison, suggesting the multi-peptide mixture may have synergistic effects at lower concentrations.[3]
This neurotrophic activity connects to one of the best-characterized pathways in neuroprotection: brain-derived neurotrophic factor (BDNF) signaling. For a detailed examination of how BDNF supports neural connections, see BDNF: The Brain Peptide That Builds New Neural Connections.
Anti-apoptotic and Antioxidant Effects
Russian-language studies (PMID 30499504, PMID 28251948) have reported that Cortexin inhibits brain caspase-8 (an initiator of programmed cell death), restores the balance between pro-oxidative and antioxidant systems in aging rat brains, and demonstrates tissue-specific anti-inflammatory effects. Caspase-8 is an initiator caspase in the extrinsic apoptosis pathway, and its inhibition could theoretically prevent neurons from undergoing programmed cell death after ischemic or traumatic injury. The selectivity finding is relevant: Cortexin's effects on caspases-1, -3, and -9, cathepsin B, and calpain were much less pronounced or absent, suggesting a targeted rather than broadly anti-apoptotic action.
These studies also identified neuron-specific proteins (beta-5-tubulin, creatine kinase B, protein 14-3-3 alpha/beta) as molecular binding partners of Cortexin peptides in the brain. The binding partner identification provides a plausible molecular basis for Cortexin's tissue specificity: these proteins are concentrated in neurons, which could explain why a systemically administered drug has preferential effects on the nervous system. While these findings are published in peer-reviewed Russian journals, they have not been replicated by independent Western laboratories.
Ischemic Preconditioning Enhancement
Zarubina et al. (2016) investigated whether neurospecific peptide preparations could enhance ischemic preconditioning in rats. Their results showed that these preparations reduced neurological deficit severity, restored behavioral structure, and exerted antioxidant effects during chronic ischemic brain damage.[4]
OPG/RANK/RANKL Modulation
A 2025 study by Guven et al. identified a previously unrecognized molecular target for Cortexin. In a rat model of cerebral ischemia-reperfusion injury, Cortexin modulated the expression of osteoprotegerin (OPG), RANK, RANKL, and TRPC1 (Transient Receptor Potential Canonical 1). These biomarkers are implicated in the neuroprotective response to neuronal injury, and their modulation by Cortexin suggests the drug may engage bone morphogenetic protein-related signaling in the brain.[5]
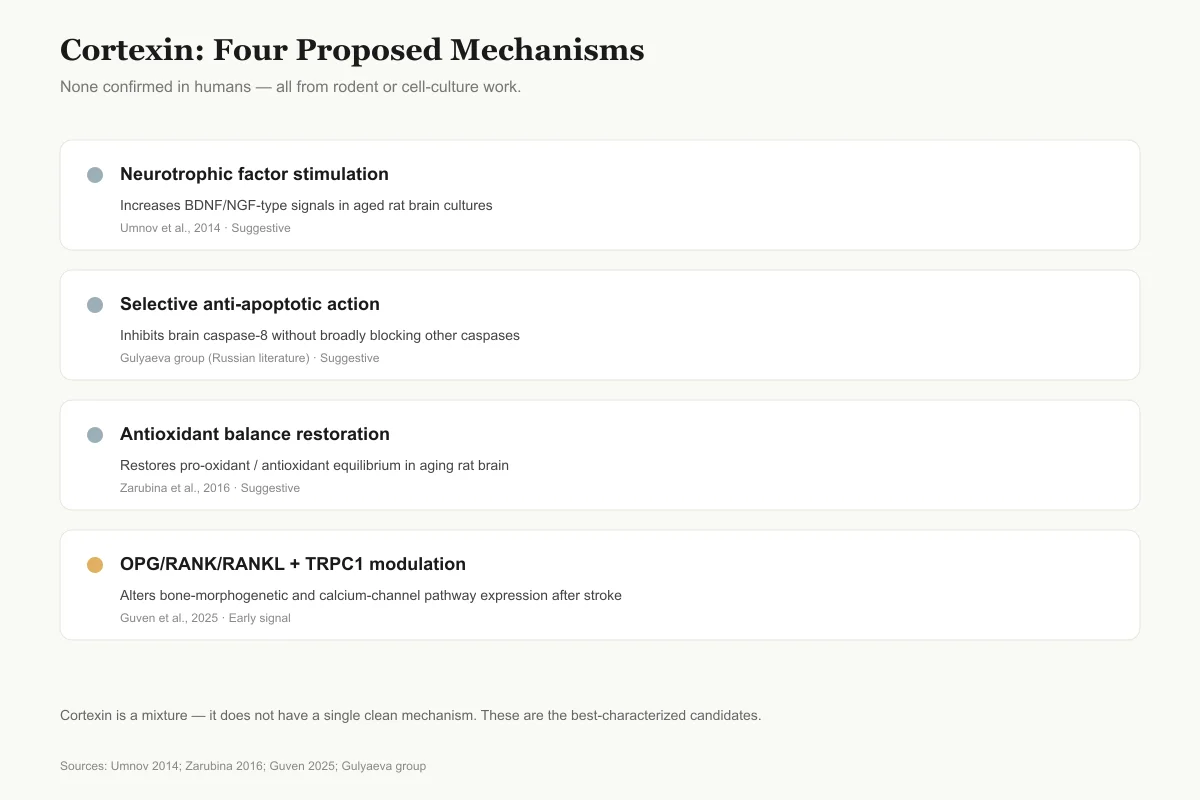
Proposed mechanisms
Four Pathways, None Fully Confirmed in Humans
Cortexin is a mixture, so it does not have one clean mechanism. Here are the four best-characterized candidate pathways from preclinical studies.
Neurotrophic factor stimulation
SuggestiveIncreases BDNF/NGF-type signals in aged rat brain cultures
Umnov et al., 2014
Selective anti-apoptotic action
SuggestiveInhibits brain caspase-8 without broadly blocking other caspases
Gulyaeva group (Russian literature)
Antioxidant balance restoration
SuggestiveRestores pro-oxidant / antioxidant equilibrium in aging rat brain
Zarubina et al., 2016
OPG/RANK/RANKL + TRPC1 modulation
Early signalAlters expression of bone-morphogenetic and calcium-channel pathways after stroke
Guven et al., 2025
Every pathway above comes from rodent or cell-culture work. No mechanism has been confirmed in humans using modern neuroimaging or biomarker studies.
Source: Umnov 2014; Zarubina 2016; Guven 2025; Gulyaeva group
 View as image
View as imageCortexin in Acute Brain Ischemia
The most rigorous preclinical data on Cortexin comes from a 2021 PLOS ONE study by Kurkin and colleagues, which was the first to directly compare Cortexin, Cerebrolysin, and Actovegin in standardized rodent stroke models.[2]
In the acute model (middle cerebral artery occlusion), rats received Cortexin at 1 or 3 mg/kg/day, Cerebrolysin at 538 or 1614 mg/kg/day, or Actovegin at 200 mg/kg/day for 10 days. Cortexin and Cerebrolysin both improved neurological function scores, reduced cerebral infarct volume, and preserved neuron counts in the ischemic penumbra. Cortexin achieved comparable neuroprotection at doses roughly 180 to 540 times lower than Cerebrolysin by weight, though the two drugs differ in composition and direct dose comparison has limited meaning.
In the chronic model (common carotid artery stenosis), both Cortexin and Cerebrolysin improved sensorimotor and cognitive outcomes. Actovegin showed weaker effects across both models.
This study is relevant for several reasons. First, it was published in an international peer-reviewed journal (PLOS ONE), used standardized stroke models recognized by Western neuroscience, and included blinded assessment. Most prior Cortexin evidence existed only in Russian-language journals with variable peer review standards. Second, the direct comparison with Cerebrolysin provides a clinical reference point: Cerebrolysin has accumulated enough international data that neurologists outside Russia recognize it, so demonstrating comparable efficacy positions Cortexin within a framework Western clinicians can evaluate. Third, the dose comparison is instructive. Cortexin's putatively greater potency per milligram could reflect the cortex-specific extraction process concentrating the most neurotrophically active peptide fractions, or it could reflect differences in how the two drugs are standardized. The study did not resolve which explanation is correct.
The chronic ischemia model adds clinical relevance because chronic cerebrovascular insufficiency (dyscirculatory encephalopathy in Russian neurology terminology) is one of the most common indications for which Cortexin is prescribed. The improvement in both sensorimotor and cognitive endpoints in the chronic model provides preclinical support for this clinical practice, though the translation from rat models to human chronic cerebral ischemia involves substantial uncertainty.
Cortexin vs. Cerebrolysin: The Head-to-Head Data
Head to head
Cortexin vs. Cerebrolysin at a Glance
The two most-used animal-brain peptide preparations compared on composition, dose, approval, and evidence.
| Attribute | Cortexin | Cerebrolysin |
|---|---|---|
| Tissue source | Cattle or pig cerebral cortex only | Porcine whole brain |
| Composition | Peptides <10 kDa + amino acids + trace minerals | Peptides + free amino acids |
| Typical rat neuroprotective dose | 1–3 mg/kg/day | 538–1614 mg/kg/day |
| Approved in Russia/CIS | Yes (since 1999) | Yes |
| Approved in US/EU | No | No US approval; used in some EU/Asia markets |
| Trials in Alsulaimani 2021 review | 1 trial / 80 patients | Enough for meta-analysis |
| Signal in vascular cognitive impairment | Not calculable | SMD -0.58 (95% CI -1.07 to -0.10) |
In rat models the two drugs look comparable. In human evidence they diverge sharply — Cerebrolysin has enough randomized data to pool; Cortexin has a single trial of 80 people.
Source: Kurkin et al. (2021); Alsulaimani et al. (2021); Zhang et al. (2019)
Zhang et al. (2019) conducted a prospective, randomized, double-blinded, placebo-controlled study comparing Cerebrolysin and Cortexin preparations in rat models of acute ischemic stroke and traumatic brain injury.[6]
Both neuropeptide preparations demonstrated neuroprotective effects compared to placebo, with improvements in neurological function, infarct volume reduction, and behavioral outcomes. The study noted that both drugs showed putative neurotrophic potential, though their molecular mechanisms may differ due to their distinct tissue sources (Cerebrolysin derives from porcine brain tissue generally, while Cortexin specifically targets cortical extracts).
The comparison matters because Cerebrolysin has a larger international evidence base, including randomized controlled trials in humans for stroke and Alzheimer's disease. If Cortexin shows comparable preclinical efficacy, the question becomes whether Cortexin can replicate Cerebrolysin's clinical results in properly designed human trials. To date, no such trial has been conducted. The Zhang study is also one of the few truly blinded comparisons between any neuropeptide preparations, addressing a common criticism that most studies in this drug class are open-label and susceptible to observer bias.
Clinical Evidence: What Exists and What Does Not
The Systematic Review Problem
Alsulaimani et al. (2021) conducted a systematic review and meta-analysis of Cerebrolysin, Cortexin, and Actovegin for cognitive disorders. For Cortexin, only one trial with 80 participants met inclusion criteria. This single trial was insufficient for meta-analysis, so no pooled efficacy estimate could be calculated.[1]
By contrast, Cerebrolysin had enough trials for pooled analysis, which showed a statistically non-significant trend toward improvement in all-cause dementia (standardized mean difference -0.44, 95% CI -0.93 to 0.04) and a statistically significant effect in the vascular cognitive impairment subgroup (SMD -0.58, 95% CI -1.07 to -0.10).
The gap between Cortexin's 25+ years of clinical use in Russia and its single eligible trial in Western-standard systematic review represents one of the sharpest disconnects in global pharmacology. Hundreds of clinical publications about Cortexin exist in Russian medical literature, but the vast majority lack the methodological features (randomization, blinding, placebo control, adequate sample size, pre-registered endpoints) required for inclusion in a Cochrane-style systematic review.
The Post-COVID Observational Study
The largest Cortexin clinical dataset comes from the CORTEX multicenter observational program, published by Putilina et al. (2022). This study enrolled 979 patients with post-COVID syndrome across Russia, Azerbaijan, Kyrgyzstan, and Kazakhstan. Patients received Cortexin at either 10 or 20 mg intramuscularly for 10 days.[7]
The study reported improvements in cognitive scores and reduction in asthenic symptoms. However, this was an observational study without a control group, without blinding, and without randomization. The natural trajectory of post-COVID symptom resolution over time cannot be separated from any drug effect without a placebo-controlled design.
Diabetic Neuropathy
Yazar et al. (2023) provided in vitro evidence that Cortexin may protect against high glucose-induced neuronal damage. Using primary cultured rat sensory neurons from dorsal root ganglia, the study demonstrated that Cortexin ameliorated the neurotoxic effects of high glucose exposure, preserving neuronal viability and function as measured by real-time cell impedance analysis.[8]
This is a single in vitro study and does not establish Cortexin as a treatment for diabetic neuropathy. It does, however, suggest that the peptide mixture's neuroprotective effects may extend beyond ischemic injury to metabolic neuronal stress, a finding that could be relevant given the growing recognition of diabetic neuropathy as a major clinical burden worldwide.
Otoprotection in Neonates
D'yakonova et al. (2020) investigated whether Cortexin could protect against aminoglycoside-induced hearing damage in immature rabbits. When administered simultaneously with netromycin (an aminoglycoside antibiotic known to cause hearing loss), Cortexin showed otoprotective effects as measured by distortion product otoacoustic emissions and auditory brainstem response tests.[9]
This finding extends Cortexin's proposed neuroprotective action to sensory neurons in the auditory system, consistent with its proposed broad-spectrum neurotrophic activity. It also represents one of the few studies testing Cortexin in a non-ischemic injury model, suggesting the neuroprotective effects may not be limited to stroke and TBI.
Cortexin in Pediatric Neurology
In Russia, Cortexin is widely prescribed for children with developmental delays, perinatal brain damage, and epilepsy-associated cognitive impairment. The pediatric formulation (5 mg for children under 20 kg) has been used since the early 2000s. Published data (PMID 33834730) from studies on children and adolescents with epilepsy reported improvement in electrophysiological parameters and cognitive function, with no seizure aggravation in 95% of cases.
However, the pediatric evidence shares the same structural limitations as the adult data: observational designs, lack of placebo controls, and publication primarily in Russian-language journals. No Western-standard randomized controlled trial has evaluated Cortexin in pediatric neurological conditions. The question of whether a multi-peptide animal brain extract is appropriate for developing nervous systems has not been addressed by the kind of rigorous safety pharmacology studies required by the FDA or EMA for pediatric drug approval.
Safety
ModerateUnregulated outside Russia and CIS
Concern
Cortexin is not approved by the FDA, EMA, Health Canada, or the UK MHRA. Any Cortexin obtained in the US, Canada, or Western Europe comes through unregulated channels. There is no guarantee of sterility, authenticity, or correct dosing, and no adverse-event reporting system to catch safety problems.
What the research says
Patients in Russia/CIS receive it through regulated pharmacies from a single registered manufacturer (Geropharm). Elsewhere, the product coming in an envelope is not the same thing — even if it carries the same name.
Particularly relevant for: Anyone considering self-importing Cortexin
What to do
Do not inject gray-market peptide products. If the intended indication is stroke recovery or cognitive decline, discuss evidence-based options with a neurologist rather than improvising with unapproved injectables.
FDA, EMA, Health Canada regulatory status databases; Geropharm product registration
The Regulatory Divide
Cortexin's regulatory status highlights a broader gap between Russian and Western pharmaceutical standards. In the Russian regulatory system (Roszdravnadzor), Cortexin completed the approval process based on clinical evidence from Russian centers. The drug's registration dossier includes pharmacological studies, clinical trials, and post-marketing surveillance data accumulated over more than two decades.
By FDA standards, however, several fundamental requirements remain unmet. The drug's composition is not fully characterized at the molecular level. The manufacturing process does not produce a chemically defined active ingredient. The clinical trials do not include Phase III studies with the design features (randomization, double-blinding, adequate sample sizes, pre-registered primary endpoints) required for FDA approval. And the drug's mechanism of action, while plausible based on preclinical data, has not been established with the precision expected for regulatory review.
This is not unique to Cortexin. The entire class of "bioregulator peptides" developed in Soviet and post-Soviet pharmacology operates under a different evidentiary paradigm than Western drug development. Cerebrolysin, the closest comparator to Cortexin, has bridged this gap partially by accumulating international trial data, but even its Western evidence base has been criticized for methodological limitations.
The Nootropic Family: Cortexin in Context
Cortexin belongs to a class of peptide-based nootropics developed in Soviet and post-Soviet neuropharmacology. Understanding its place in this family clarifies what makes it distinctive and where its evidence compares.
Cerebrolysin is the closest comparator: also an animal brain-derived peptide extract, but from whole brain rather than cortex specifically. It has the largest evidence base among this class, with multiple randomized controlled trials in stroke and dementia. Cortexin's preclinical data suggests comparable neuroprotection at lower doses, but the clinical evidence gap is wide.
Noopept (GVS-111) is a synthetic dipeptide derivative, not an extract. It was designed as an orally bioavailable compound inspired by the piracetam family. For a detailed comparison of its chemistry and evidence, see Noopept: The Peptide-Derived Nootropic (And Why It's Not Technically a Peptide).
Semax and Selank are synthetic peptides with defined amino acid sequences, making them fundamentally different from Cortexin's multi-component extract approach. Semax is an ACTH(4-10) analogue that modulates BDNF; Selank is a tuftsin analogue with anxiolytic properties. Their defined structures allow for more precise pharmacological characterization. See Selank vs Semax: How Two Russian Neuropeptides Compare.
The philosophical divide is between Cortexin's "many peptides working together" approach and the Western preference for single-molecule, well-characterized drugs. Both have trade-offs: the multi-peptide approach may capture synergies between peptide fragments but makes mechanistic study, quality control, and regulatory approval difficult; the single-molecule approach enables rigorous pharmacology and reproducible dosing but may miss combinatorial effects that exist when multiple neuroactive peptides are present simultaneously. This is not merely a philosophical distinction. It has practical consequences for which drugs get studied, which get approved, and which patients in which countries have access to which treatments. Cortexin's story is in large part a story about this divide.
Evidence Quality and Limitations
The Cortexin evidence base has several structural problems that are distinct from the question of whether the drug works.
Language barrier. The majority of Cortexin clinical studies are published in Russian-language journals that are not indexed in major Western databases or subject to the same peer review standards. The mechanistic studies from Gulyaeva and colleagues (PMID 30499504, 26356623) contain detailed molecular data but are accessible only through translated abstracts.
Study design. Most human studies are observational, open-label, or use historical controls. The 979-patient CORTEX post-COVID study had no control group. The open clinical trials use rating scales common in Russian neurology but not always validated against international standards.
Manufacturer involvement. Cortexin is manufactured by Geropharm, and much of the clinical research is conducted in institutions with existing relationships with the manufacturer. Independent replication by research groups without industry ties is sparse.
Batch variability. As a biological extract, Cortexin's exact peptide composition may vary between production lots. The manufacturing process is standardized for biological activity rather than chemical composition, which makes it difficult to attribute effects to specific peptide components.
Comparative effectiveness. No study has compared Cortexin to standard Western treatments for its approved indications (e.g., tissue plasminogen activator for stroke, cholinesterase inhibitors for dementia, or evidence-based rehabilitation protocols for TBI).
These limitations do not prove Cortexin is ineffective. They mean that by the evidentiary standards used by the FDA, EMA, and major Western clinical guideline organizations, the evidence is insufficient to draw conclusions about its efficacy for any indication. It is possible that Cortexin provides genuine neuroprotective benefit that has simply never been tested in a way that could confirm or deny it by Western standards. It is equally possible that the observed clinical improvements in uncontrolled studies reflect natural recovery, placebo effects, or regression to the mean. Without properly controlled data, both explanations remain viable.
The Bottom Line
Cortexin is a multi-peptide brain extract with 25+ years of clinical use in Russia for stroke, TBI, and cognitive disorders, with preclinical data showing neuroprotection through neurotrophic factor stimulation, caspase inhibition, and antioxidant effects. However, a 2021 systematic review found only a single eligible clinical trial (80 participants), and the largest human study (979 patients) was an uncontrolled observational program. The evidence base is concentrated in Russian-language journals, lacks independent replication, and does not meet Western regulatory standards for any indication.
Sources & References
- 1RPEP-05261·Alsulaimani, Rayan A et al. (2021). “The efficacy and safety of animal-derived nootropics in cognitive disorders: Systematic review and meta-analysis..” Cerebral circulation - cognition and behavior.Study breakdown →PubMed →↩
- 2RPEP-05518·Kurkin, Denis V et al. (2021). “Neuroprotective action of Cortexin, Cerebrolysin and Actovegin in acute or chronic brain ischemia in rats..” PloS one.Study breakdown →PubMed →↩
- 3RPEP-02526·Umnov, R S et al. (2014). “Peptides stimulate expression of signal molecules in neuronal cultures from animals of different age..” Bulletin of experimental biology and medicine.Study breakdown →PubMed →↩
- 4RPEP-03174·Zarubina, I V et al. (2016). “Neuroprotective Effects of Peptides during Ischemic Preconditioning..” Bulletin of experimental biology and medicine.Study breakdown →PubMed →↩
- 5RPEP-11237·Guven, Cengiz et al. (2025). “Cortexin Peptide Reduced Brain Damage After Stroke by Lowering Oxidative Stress in Rats.” Neurological research.Study breakdown →PubMed →↩
- 6RPEP-04591·Zhang, Li et al. (2019). “Prospective, double blinded, comparative assessment of the pharmacological activity of Cerebrolysin and distinct peptide preparations for the treatment of embolic stroke..” Journal of the neurological sciences.Study breakdown →PubMed →↩
- 7RPEP-06438·Putilina, M V et al. (2022). “Determination of the Prevalence of Postcovid Syndrome and Assessment of the Effectiveness of the Drug Cortexin in the Treatment of Neurological Disorders in Patients with Postcovid Syndrome. Results of the CORTEX Multicenter Clinical and Epidemiological Observational Program..” Neuroscience and behavioral physiology.Study breakdown →PubMed →↩
- 8RPEP-07589·Yazar, Uğur et al. (2023). “Cortexin® Ameliorates High Glucose-Induced Neuropathy in Cultured Rat Sensory Neurons..” Neuroendocrinology.Study breakdown →PubMed →↩
- 9RPEP-04744·D'yakonova, I N et al. (2020). “Otoprotective Effect of Cortexin, Cogitum, and Elkar Administered Simultaneously with Netromycin in the Experiment..” Bulletin of experimental biology and medicine.Study breakdown →PubMed →↩