Neuropeptides and Depression Beyond Serotonin
Neuropeptides and Depression
30% non-response
Roughly 30% of patients with major depression do not respond to serotonin-based antidepressants, driving research into neuropeptide alternatives.
Kupcova et al., Behavioral Sciences, 2022
Kupcova et al., Behavioral Sciences, 2022
If you only read one thing
Antidepressants like Prozac and Zoloft work on serotonin, but they don't work for about 30% of people with depression. It turns out the brain has a whole separate system of signaling molecules called neuropeptides that control stress, resilience, social bonding, and motivation — and none of them are targeted by current antidepressants. Some of these peptides make depression worse (like CRF, the stress peptide), while others protect against it (like NPY, which is elevated in people who handle extreme stress well). No neuropeptide drug has been approved for depression yet, but the biology is strong and multiple targets are in development.
The dominant model of depression for the past four decades has centered on monoamine neurotransmitters: serotonin, norepinephrine, and dopamine. This model produced SSRIs, SNRIs, and other medications that remain first-line treatments. But roughly 30% of patients with major depressive disorder do not respond adequately to these drugs, and the monoamine hypothesis fails to explain why antidepressants take weeks to work despite altering neurotransmitter levels within hours.[1] Neuropeptides, a class of over 100 signaling molecules produced by neurons throughout the brain, offer a parallel biology of depression that operates through different mechanisms and different brain circuits.
This article maps the neuropeptides most implicated in depression research, from the stress-activating CRF system to the resilience-promoting NPY pathway to the social bonding effects of oxytocin. For deeper coverage of individual topics, see our dedicated articles on CRF and the stress hormone connection, substance P antagonists, the neuropeptide theory of depression, and VGF-derived peptide targets.
Key Takeaways
- About 30% of people with depression get no real help from SSRIs like Prozac or Zoloft.
- The brain has over 20 mood-regulating peptides — and current antidepressants target none of them.
- Serotonin is the brain's instant messaging. Neuropeptides are more like changing the thermostat for a whole region.
- Special Forces soldiers carry more NPY, the "resilience peptide" — combat veterans with PTSD carry less.
- In depression, the brain's stress alarm (CRF) gets stuck on — and SSRIs don't turn it off.
- A substance-P blocker once rivaled Prozac in a small trial. Bigger trials failed, and most programs got shelved.
- No neuropeptide drug is approved for depression yet — but multiple targets are in active development.
What Neuropeptides Are and How They Differ from Neurotransmitters
Neuropeptides are small protein molecules, typically 3 to 100 amino acids long, synthesized in neuronal cell bodies and transported to synaptic terminals. Unlike classical neurotransmitters (serotonin, dopamine, GABA, glutamate), which are synthesized locally at the synapse and act on millisecond timescales, neuropeptides are produced in the cell body, packaged into dense-core vesicles, and released under conditions of sustained or high-frequency neuronal firing. They diffuse over larger distances and act over seconds to minutes, modulating entire neural circuits rather than individual synapses.[2]
This makes neuropeptides ideal regulators of sustained emotional states like mood, anxiety, and stress responses. A single burst of serotonin release may transiently alter synaptic signaling, but neuropeptide release can shift the baseline excitability of entire brain regions for extended periods. Many neuropeptides co-localize with classical neurotransmitters in the same neurons, creating layered signaling systems where the fast neurotransmitter handles moment-to-moment communication and the neuropeptide sets the longer-term tone.
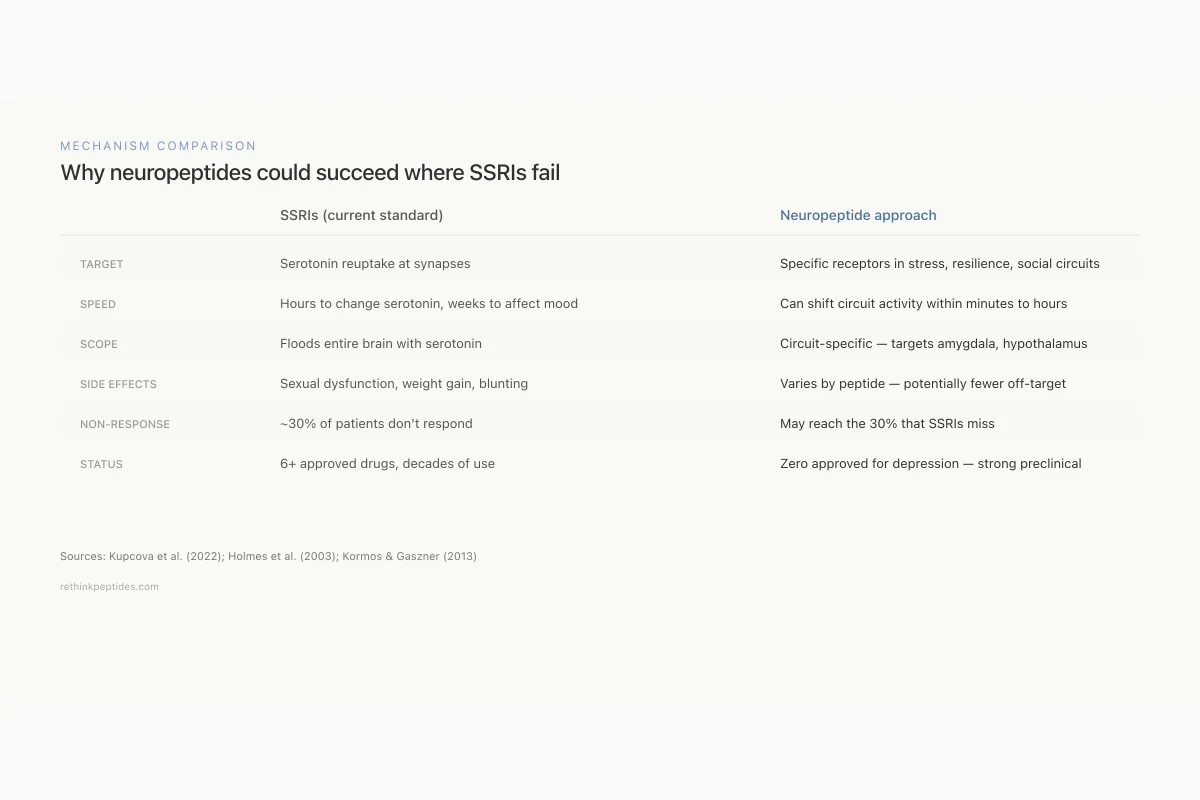
Mechanism Comparison
Why neuropeptides could succeed where SSRIs fail
SSRIs (current standard)
Neuropeptide approach
What they target
Serotonin reuptake at synapses
Specific receptors in stress, resilience, and social circuits
Speed of action
Hours to change serotonin, weeks to affect mood
Can shift circuit activity within minutes to hours
Scope
Floods entire brain with serotonin
Circuit-specific — targets amygdala, hypothalamus, or hippocampus
Side effects
Sexual dysfunction, weight gain, emotional blunting
Varies by peptide — potentially fewer off-target effects
Non-response rate
~30% of patients don't respond
May reach the 30% that SSRIs miss
Current status
6+ approved drugs, decades of clinical use
Zero approved for depression — strong preclinical data
What they target
SSRI: Serotonin reuptake at synapses
Neuropeptide: Specific receptors in stress, resilience, and social circuits
Speed of action
SSRI: Hours to change serotonin, weeks to affect mood
Neuropeptide: Can shift circuit activity within minutes to hours
Scope
SSRI: Floods entire brain with serotonin
Neuropeptide: Circuit-specific — targets amygdala, hypothalamus, or hippocampus
Side effects
SSRI: Sexual dysfunction, weight gain, emotional blunting
Neuropeptide: Varies by peptide — potentially fewer off-target effects
Non-response rate
SSRI: ~30% of patients don't respond
Neuropeptide: May reach the 30% that SSRIs miss
Current status
SSRI: 6+ approved drugs, decades of clinical use
Neuropeptide: Zero approved for depression — strong preclinical data
Sources: Kupcova et al. (2022); Holmes et al. (2003); Kormos & Gaszner (2013)
 View as image
View as imageThe brain expresses over 100 distinct neuropeptides, but depression research has concentrated on a smaller group: corticotropin-releasing factor (CRF), neuropeptide Y (NPY), substance P, oxytocin, vasopressin, galanin, orexin/hypocretin, and more recently, VGF-derived peptides and ProSAAS-derived peptides.[3]
CRF: The Stress Peptide Overactivated in Depression
Corticotropin-releasing factor (CRF, also called CRH) is a 41-amino acid neuropeptide that sits at the top of the hypothalamic-pituitary-adrenal (HPA) axis. CRF released from the hypothalamus triggers ACTH release from the pituitary, which drives cortisol production from the adrenal glands. But CRF also acts as a neurotransmitter in the amygdala, hippocampus, and cortex, where it modulates anxiety, fear, and mood independent of its hormonal effects.
CRF overactivity is one of the most replicated findings in depression neurobiology. Depressed patients show elevated CRF in cerebrospinal fluid, enlarged adrenal glands, and blunted ACTH responses to CRF challenge (indicating downregulated CRF receptors from chronic overstimulation). Reul and Holsboer (2002) documented that CRF acts through two receptor subtypes: CRF-1 receptors, which mediate anxiety-like behavior when activated, and CRF-2 receptors, which may have anxiolytic effects. This receptor duality complicates drug development but also creates opportunities for selective targeting.[4]
Fan et al. (2014) demonstrated that prenatal stress programs the CRH peptide family system, altering CRF receptor expression in offspring and increasing vulnerability to anxiety and depression in adulthood. This finding connects early-life stress to adult neuropeptide dysfunction through epigenetic mechanisms.[5]
Multiple pharmaceutical companies have developed CRF-1 receptor antagonists (pexacerfont, verucerfont, emicerfont), and while these showed antidepressant effects in animal models, clinical trials produced mixed results. The failure may reflect the complexity of CRF signaling rather than a flaw in the neuropeptide hypothesis itself. CRF acts differently in different brain regions: blocking CRF-1 in the amygdala may reduce anxiety, while blocking it in the prefrontal cortex may impair cognitive function needed for emotional regulation. Future approaches may need brain-region-specific delivery rather than systemic receptor antagonism.
Beyond the HPA axis, CRF peptides include urocortin I, II, and III, which preferentially activate CRF-2 receptors. Urocortin III produces anxiolytic effects in animal models, raising the question of whether CRF-2 agonism (not CRF-1 antagonism) might be the more productive therapeutic strategy. For the full clinical development history, see our article on CRF and the stress hormone connection.
Neuropeptide Y: The Resilience Peptide
Neuropeptide Y (NPY) is a 36-amino acid peptide and one of the most abundant neuropeptides in the mammalian brain. It is densely expressed in the amygdala, hippocampus, hypothalamus, and cortex. NPY consistently produces anxiolytic and antidepressant-like effects in animal models, and human studies associate higher NPY levels with stress resilience.
Cohen et al. (2012) used an animal model of PTSD to show that rats with higher NPY expression in the amygdala and hippocampus after traumatic stress showed fewer anxiety and depression-like behaviors. Crucially, this was not just a correlation: NPY expression patterns predicted which animals would be resilient versus vulnerable before behavioral testing. Exogenous NPY administration into the amygdala reduced anxiety behaviors, confirming a causal role.[6]
Enman et al. (2015) reviewed the therapeutic potential of the NPY system in stress-related psychiatric disorders, documenting that NPY acts through five receptor subtypes (Y1-Y5), each with distinct effects. Y1 receptor activation in the amygdala produces anxiolytic effects. Y2 receptor blockade (Y2 receptors are presynaptic autoreceptors that limit NPY release) also reduces anxiety by increasing NPY availability. The multiplicity of NPY receptors creates multiple drug development opportunities.[7]
Singanwad et al. (2025) extended NPY's role specifically to treatment-resistant depression (TRD), documenting that NPY modulates neuroplasticity through BDNF pathways, reduces neuroinflammation by suppressing microglial activation, and normalizes HPA axis dysregulation, three mechanisms that SSRIs address incompletely. The authors proposed NPY-based interventions as adjuncts for patients who fail conventional antidepressant therapy.[8]
Human data supports this preclinical picture. Military studies found that Special Forces soldiers, who are selected and trained for extreme stress resilience, have higher plasma NPY levels than general military personnel. Combat veterans with PTSD show lower NPY levels than combat-exposed veterans without PTSD. This human correlational data aligns with the animal causal data showing NPY administration reduces stress vulnerability, creating one of the strongest translational cases in neuropeptide depression research.
Substance P and the NK1 Receptor
Substance P is an 11-amino acid neuropeptide of the tachykinin family that acts primarily through NK1 (neurokinin-1) receptors. It is expressed in brain regions involved in emotional processing, including the amygdala, hypothalamus, and periaqueductal gray. Depressed patients show elevated substance P levels in cerebrospinal fluid, and NK1 receptor density is increased in the brains of suicide victims.
The substance P story in depression includes one of the most dramatic rise-and-fall narratives in psychopharmacology. Merck's aprepitant (an NK1 antagonist originally developed for nausea) showed antidepressant effects in an early clinical trial in 1998, rivaling paroxetine's efficacy without sexual side effects. This triggered a rush of NK1 antagonist development across the pharmaceutical industry. Subsequent larger trials failed to replicate the effect, and most programs were abandoned.
Rotzinger et al. (2010) reviewed the preclinical evidence and noted that substance P antagonists consistently show antidepressant and anxiolytic effects in animal models, but the doses and brain concentrations required may not have been achieved in human trials. The disconnect between robust preclinical data and inconsistent clinical results remains one of the central puzzles in neuropeptide drug development.[9] For the complete history, see our article on substance P antagonists for depression.
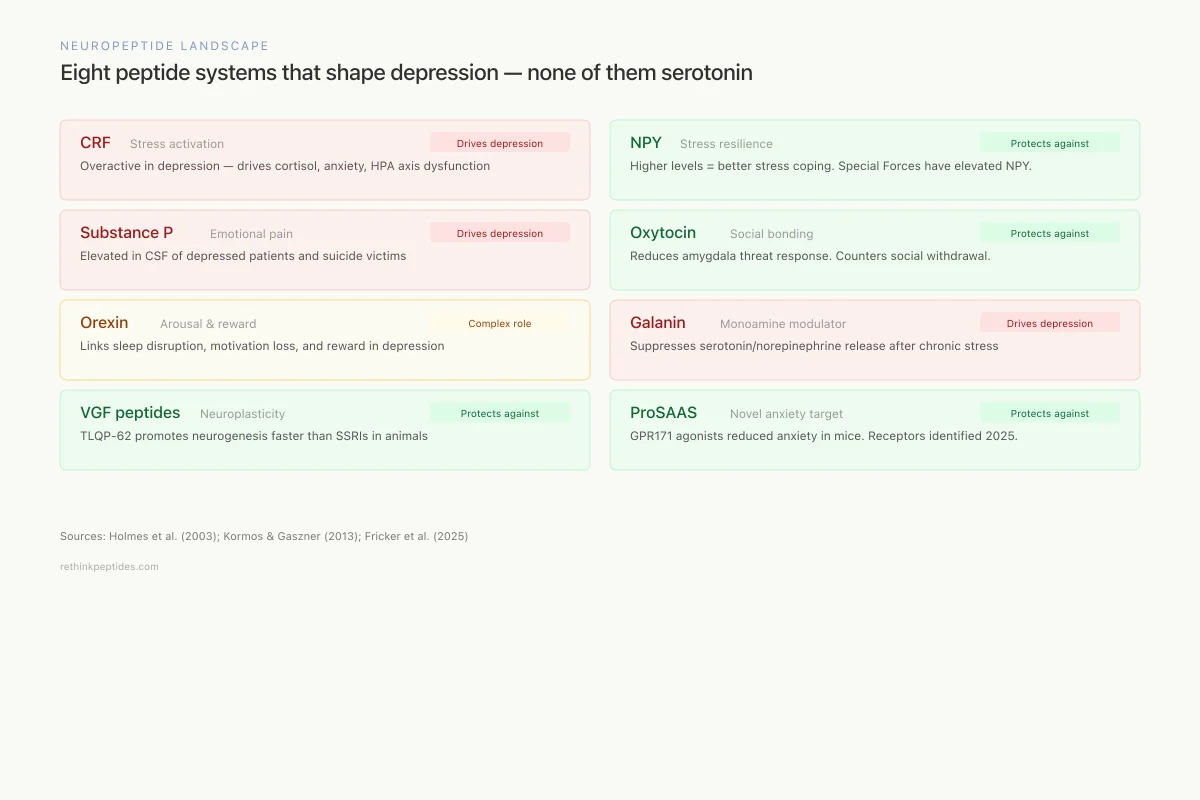
Neuropeptide Landscape
Eight peptide systems that shape depression — none of them serotonin
CRF
Corticotropin-Releasing Factor
Stress activation
Overactive in depression — drives cortisol, anxiety, HPA axis dysfunction
NPY
Neuropeptide Y
Stress resilience
Higher levels = better stress coping. Special Forces soldiers have elevated NPY.
Substance P
Substance P / NK1
Emotional pain signaling
Elevated in cerebrospinal fluid of depressed patients and suicide victims
Oxytocin
Oxytocin
Social bonding & trust
Reduces amygdala threat response. May counteract social withdrawal in depression.
Orexin
Orexin / Hypocretin
Arousal & reward
Links sleep disruption, motivation loss, and reward processing in depression
Galanin
Galanin
Monoamine modulation
Suppresses serotonin and norepinephrine release when overactive after chronic stress
VGF
VGF-derived peptides
Neuroplasticity
TLQP-62 promotes hippocampal neurogenesis — faster onset than SSRIs in animal models
ProSAAS
ProSAAS-derived peptides
Novel anxiety target
GPR171 agonists reduced anxiety in mice. Among the most abundant brain peptides.
Sources: Holmes et al. (2003); Kormos & Gaszner (2013); Fricker et al. (2025)
 View as image
View as imageOxytocin: The Social Bonding Peptide
Oxytocin is a 9-amino acid neuropeptide produced in the hypothalamus and released both into the bloodstream (endocrine function) and within the brain (neuromodulatory function). It is central to social bonding, trust, and attachment. Depression is fundamentally a disorder of social withdrawal, anhedonia, and disconnection, making oxytocin a logical candidate for investigation.
Kirsch et al. (2005) demonstrated that intranasal oxytocin administration in healthy humans reduced amygdala activation in response to fearful faces, showing that oxytocin can directly modulate the threat-processing circuitry that is overactive in depression and anxiety disorders. The amygdala dampening occurred without affecting amygdala responses to happy or neutral faces, suggesting a selective reduction in threat sensitivity rather than a blunting of all emotional processing.[10]
The oxytocin system interacts bidirectionally with the serotonin system. Oxytocin neurons in the hypothalamus express serotonin receptors (particularly 5-HT1A), and serotonin release in the hypothalamus stimulates oxytocin secretion. This creates a pathway through which SSRI antidepressants may exert some of their therapeutic effects indirectly through oxytocin, partially explaining why SSRIs improve social anxiety and interpersonal functioning beyond what pure serotonin elevation would predict.
However, oxytocin's effects are context-dependent and dose-dependent. In some social situations, oxytocin increases in-group trust but also out-group suspicion. At low doses it may reduce anxiety, while at higher doses it can increase salience of social threats. Individuals with insecure attachment styles may respond differently to oxytocin than those with secure attachment. This complexity has slowed its development as a standalone antidepressant, though it remains a promising adjunct for social anxiety and depression subtypes characterized by interpersonal withdrawal.
Vasopressin, oxytocin's structurally similar nine-amino-acid cousin (differing by just two amino acids), has complementary effects in social behavior. Where oxytocin generally promotes affiliation, vasopressin is more associated with vigilance and mate-guarding behaviors. The oxytocin-vasopressin balance may be relevant to depression subtypes characterized by anxiety-predominant versus withdrawal-predominant presentations.
Orexin/Hypocretin: Linking Arousal, Reward, and Mood
The orexin peptides (orexin-A and orexin-B, also called hypocretin-1 and hypocretin-2) are produced by a small cluster of neurons in the lateral hypothalamus. They are best known for regulating wakefulness (loss of orexin neurons causes narcolepsy), but they also modulate reward processing, stress responses, and mood through projections to the VTA, locus coeruleus, and amygdala.
Cohen et al. (2020) showed that the orexinergic system modulates stress-related behaviors through interactions with the serotonergic and noradrenergic systems. In a PTSD animal model, orexin-1 receptor antagonism reduced anxiety-like behaviors, while orexin-2 receptor modulation affected sleep architecture and arousal. The dual-receptor system creates the same kind of complexity seen with CRF: one receptor subtype may promote stress responses while the other may counteract them.[11]
Suvorexant (Belsomra) and lemborexant (Dayvigo), approved dual orexin receptor antagonists for insomnia, provide an opportunity to study orexin blockade effects on mood in clinical populations. Early observations suggest that orexin antagonism may improve sleep-related depression symptoms, though dedicated depression trials are lacking. For more on orexin biology, see our article on narcolepsy and orexin neurons.
Galanin: The Underexplored Modulator
Galanin is a 29-amino acid neuropeptide (30 in humans) with widespread expression in the brain, particularly in the locus coeruleus, dorsal raphe, and hypothalamus, regions central to noradrenergic and serotonergic function. Galanin co-localizes with both norepinephrine and serotonin in these nuclei, positioning it as a direct modulator of the monoamine systems targeted by conventional antidepressants.
Kask et al. (1997) described galanin's involvement in depression through its inhibitory effects on norepinephrine and serotonin release. Galanin acts through three receptor subtypes: GalR1 (inhibitory, linked to depression-like behavior), GalR2 (potentially antidepressant), and GalR3 (anxiogenic in some models). The pattern mirrors other neuropeptide systems: receptor subtype determines whether the peptide's effects are pro-depressive or antidepressive.[12]
Galanin levels rise in the locus coeruleus after chronic stress, potentially contributing to noradrenergic blunting and the emotional numbness characteristic of depression. This is particularly relevant because the locus coeruleus is the primary source of norepinephrine in the brain, and its dysfunction is central to the fatigue, concentration difficulties, and psychomotor retardation seen in depression. Galanin-mediated suppression of locus coeruleus firing may explain why some depressed patients respond poorly to norepinephrine-targeting drugs like SNRIs: the problem is not norepinephrine receptor sensitivity but upstream peptide-mediated suppression of norepinephrine release.
GalR2 agonists show antidepressant effects in animal models, and GalR3 antagonists reduce anxiety-like behavior, but no galanin-targeting drugs have reached clinical trials for depression. The galanin system exemplifies the receptor-subtype challenge: three receptors with different, sometimes opposing functions in the same brain circuits. For more on galanin, see our article on galanin as a sleep-promoting peptide.
Emerging Targets: ProSAAS and VGF-Derived Peptides
Two newer classes of neuropeptides are entering the depression research landscape. ProSAAS-derived peptides are among the most abundant peptides in the brain, with levels comparable to enkephalin and NPY, yet they remained functionally uncharacterized until recently. Fricker et al. (2025) identified two receptors for ProSAAS peptides, GPR171 and GPR83, and demonstrated that modulating these receptors affects anxiety-related behavior and body weight in animal models. GPR171 agonists reduced anxiety-like behavior in mice, positioning ProSAAS peptides as a genuinely novel target class.[13]
VGF (non-acronymic) is a neuropeptide precursor that produces multiple bioactive fragments, including TLQP-62 and TLQP-21. VGF expression is induced by BDNF and by antidepressant treatment, and VGF-derived peptides produce antidepressant-like effects in rodent models. TLQP-62 infusion into the hippocampus promotes neurogenesis and produces behavioral effects comparable to chronic SSRI treatment but with faster onset. The VGF pathway is notable because it sits downstream of the BDNF-TrkB signaling cascade that is increasingly recognized as central to antidepressant mechanism of action. This positions VGF peptides as potential mediators of the antidepressant effects that current drugs produce indirectly and slowly through monoamine modulation.
Both ProSAAS and VGF-derived peptides illustrate a recurring pattern: neuropeptides discovered through basic neuroscience (not psychiatry) turn out to have mood-regulatory functions that were invisible under the monoamine framework. The brain's peptide signaling system is far more complex than the three-neurotransmitter model that dominates clinical teaching. For a complete analysis of VGF, see our article on VGF-derived peptide targets.
Why Neuropeptide Drugs Have Been Difficult to Develop
Despite decades of compelling preclinical data, no neuropeptide-targeting drug has been approved specifically for depression. Several factors explain this gap. Neuropeptides do not cross the blood-brain barrier easily, requiring either intranasal delivery, intrathecal injection, or development of small-molecule receptor agonists/antagonists that mimic the peptide's effects. The receptor subtype complexity described above means that drugs must be highly selective, as activating one receptor subtype may produce therapeutic effects while activating another produces the opposite.
Satao and Doshi (2024) reviewed the current state of neuropeptide-based anxiety treatments and identified additional challenges: peptide degradation by brain enzymes, difficulty achieving consistent brain concentrations, and the absence of validated biomarkers to select patients most likely to benefit. These are solvable engineering problems, not fundamental biological obstacles. Advances in peptide stabilization (cyclization, D-amino acid substitution, PEGylation) and delivery (intranasal formulations, nanoparticle encapsulation, lipid-based carriers) are gradually closing the gap between preclinical promise and clinical viability.[14]
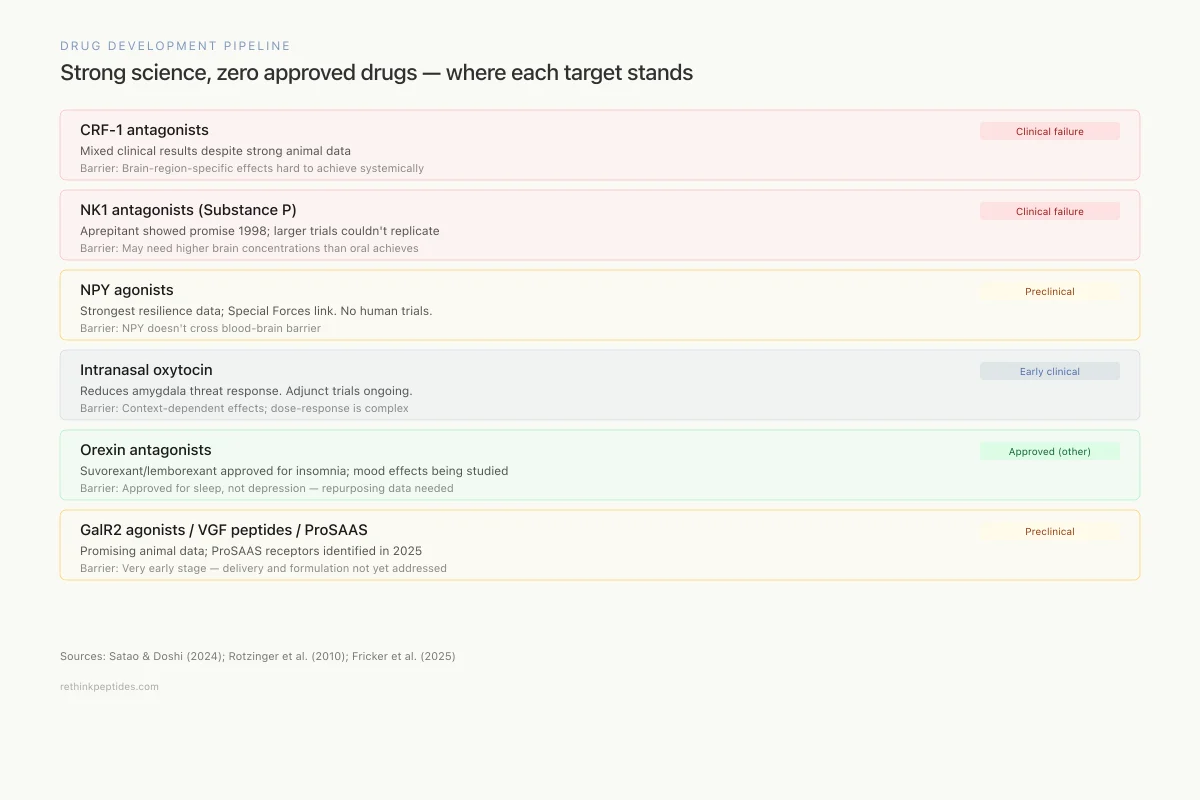
Drug Development Pipeline
Strong science, zero approved drugs — where each neuropeptide target stands
CRF-1 antagonists
Clinical failurePexacerfont, verucerfont — antidepressant effects in animals, mixed clinical results
Barrier: Brain-region-specific effects hard to achieve with systemic drugs
NK1 antagonists (Substance P)
Clinical failureAprepitant showed promise in 1998, but larger trials couldn't replicate
Barrier: May need higher brain concentrations than oral dosing achieves
NPY agonists
PreclinicalStrongest resilience data; Special Forces link. No drugs in human trials.
Barrier: NPY doesn't cross blood-brain barrier; delivery is the bottleneck
Intranasal oxytocin
Early clinicalReduces amygdala threat response. Adjunct trials ongoing.
Barrier: Context-dependent effects; dose-response is complex
Orexin antagonists
Approved (other)Suvorexant/lemborexant approved for insomnia; depression effects being studied
Barrier: Approved for sleep, not depression — repurposing data needed
GalR2 agonists
PreclinicalAntidepressant effects in animals; no compounds in human trials
Barrier: Three receptor subtypes with opposing effects
VGF-derived peptides
PreclinicalTLQP-62 promotes neurogenesis faster than SSRIs in animals
Barrier: Very early; delivery and formulation not yet addressed
ProSAAS / GPR171
PreclinicalNewly characterized receptors; anxiolytic effects in mice
Barrier: Discovery-stage — receptors identified in 2025
Sources: Satao & Doshi (2024); Rotzinger et al. (2010); Fricker et al. (2025)
 View as image
View as imageThe neuropeptide theory of depression does not replace the monoamine hypothesis. Instead, it expands it. Neuropeptides modulate the same brain circuits (amygdala, prefrontal cortex, hippocampus, hypothalamus) and interact directly with the same monoamine systems that current drugs target. The difference is resolution: neuropeptides offer finer-grained control over specific emotional and stress-response circuits, potentially enabling treatments tailored to specific depression subtypes rather than the one-size-fits-all approach of SSRIs. For the theoretical framework, see our article on the neuropeptide theory of depression.
Other neuropeptides relevant to depression biology include dynorphin and the kappa opioid receptor, which drives dysphoria, and beta-endorphin, which mediates natural reward and pleasure, both directly relevant to the anhedonia that defines depressive experience. The endogenous opioid peptide system represents yet another layer of neuropeptide regulation that intersects with depression biology at the level of reward, pain, and social connection.
The Bottom Line
Neuropeptides including CRF, NPY, substance P, oxytocin, orexin, galanin, and newer targets like ProSAAS and VGF-derived peptides operate through distinct mechanisms from serotonin-based antidepressants and regulate the stress, resilience, social bonding, and arousal circuits disrupted in depression. The 30% non-response rate to SSRIs and the weeks-long delay in their therapeutic effects suggest that monoamine modulation alone is insufficient for many patients. Neuropeptide-based treatments face pharmacological delivery challenges, but the biology is well-established, and multiple receptor targets remain available for drug development.
Sources & References
- 1RPEP-06277·Kupcova, Ida et al. (2022). “What We Know About Neuropeptides in Anxiety and Depression: A Comprehensive Review.” Behavioral sciences (Basel.Study breakdown →PubMed →↩
- 2RPEP-02212·Kormos, Viktória et al. (2013). “How Brain Neuropeptides Drive Anxiety, Stress, and Depression.” Neuropeptides.Study breakdown →PubMed →↩
- 3RPEP-00827·Holmes, Andrew et al. (2003). “Neuropeptide Systems: The Next Generation of Antidepressants and Anxiolytics.” Trends in pharmacological sciences.Study breakdown →PubMed →↩
- 4RPEP-00761·Reul, Johannes M H M et al. (2002). “CRF1 Drives Anxiety While CRF2 May Actually Help: Distinct Stress Receptor Roles in Mood Disorders.” Current opinion in pharmacology.Study breakdown →PubMed →↩
- 5RPEP-02380·Fan, Jun-Ming et al. (2014). “Prenatal stress, anxiety and depression: a mechanism involving CRH peptide family..” Neuro endocrinology letters.Study breakdown →PubMed →↩
- 6RPEP-01921·Cohen, Hagit et al. (2012). “Neuropeptide Y Linked to Stress Resilience in an Animal Model of PTSD.” Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology.Study breakdown →PubMed →↩
- 7RPEP-02622·Enman, Nicole M et al. (2015). “Targeting the Neuropeptide Y System in Stress-related Psychiatric Disorders..” Neurobiology of stress.Study breakdown →PubMed →↩
- 8RPEP-13611·Singanwad, Priyanka et al. (2025). “Neuropeptide Y offers a new biological target for treatment-resistant depression through stress and inflammation pathways.” Neuropeptides.Study breakdown →PubMed →↩
- 9RPEP-01682·Rotzinger, Susan et al. (2010). “Neuropeptides in Animal Models of Depression and Anxiety: CRF, NPY, and the Therapeutic Landscape.” Peptides.Study breakdown →PubMed →↩
- 10RPEP-01055·Kirsch, Peter et al. (2005). “Oxytocin Calms the Brain's Fear Center: Neural Imaging Reveals the Anti-Anxiety Mechanism.” The Journal of neuroscience : the official journal of the Society for Neuroscience.Study breakdown →PubMed →↩
- 11RPEP-04725·Cohen, Shlomi et al. (2020). “Significance of the orexinergic system in modulating stress-related responses in an animal model of post-traumatic stress disorder..” Translational psychiatry.Study breakdown →PubMed →↩
- 12RPEP-00414·Kask, K et al. (1997). “Galanin receptors: involvement in feeding, pain, depression and Alzheimer's disease..” Life sciences.Study breakdown →PubMed →↩
- 13RPEP-10982·Fricker, Lloyd D et al. (2025). “ProSAAS neuropeptides and receptors GPR171 and GPR83: Potential therapeutic applications for pain, anxiety, and body weight regulation..” The Journal of pharmacology and experimental therapeutics.Study breakdown →PubMed →↩
- 14RPEP-09212·Satao, Kiran S et al. (2024). “How Neuropeptides Shape Anxiety — and Could Lead to Better Treatments.” Pharmacology.Study breakdown →PubMed →↩