Beta-Endorphin: The Runner's High Peptide
Beta-Endorphin and Endogenous Opioids
31 amino acids
Beta-endorphin is a 31-residue opioid peptide cleaved from proopiomelanocortin (POMC) that binds mu-opioid receptors with high affinity and produces analgesia comparable to morphine.
Abrimian et al., International Journal of Molecular Sciences, 2021
Abrimian et al., International Journal of Molecular Sciences, 2021
If you only read one thing
Beta-endorphin is a 31-amino-acid opioid peptide your body makes from a precursor protein called POMC — the same precursor that makes your stress hormone ACTH. It binds the same brain receptor as morphine, but your body produces it naturally during hard exercise, acute stress, and certain pleasurable activities. It's behind the 'runner's high' feeling, although endocannabinoids do more of the work than the popular story admits. Beyond the high, beta-endorphin helps control pain, boosts your immune system, and affects feeding and metabolism. The runner's high is real — but 'flood of endorphins' is an oversimplification of a much richer system.
Beta-endorphin is the most potent endogenous opioid peptide in the human body. This 31-amino acid molecule, cleaved from the precursor protein proopiomelanocortin (POMC), binds mu-opioid receptors with high affinity and produces analgesia, euphoria, and reward signaling that rivals exogenous opioids like morphine. It is the molecule most commonly associated with the "runner's high," that sense of euphoria and reduced pain perception that follows sustained aerobic exercise. But beta-endorphin does far more than make running feel good. It modulates pain perception, participates in stress responses, influences feeding behavior, regulates immune function, and interacts with other endogenous opioid systems in ways that are still being mapped. This article covers the full biology of beta-endorphin: how it is made, where it acts, what it does, and what remains unknown.
Key Takeaways
- Beta-endorphin is your body's own morphine. It binds the same brain receptor as opioids and can dull pain almost as strongly.
- Your body releases it during hard exercise, stress, and some pleasurable activities — part of what you feel as a "runner's high."
- But the runner's high story isn't just endorphins. Another system (endocannabinoids) does more of the work at moderate intensities.
- People with chronic abdominal pain have measurably lower beta-endorphin in their spinal fluid — and feel more pain as a result.
- Beta-endorphin isn't only a mood chemical. It also activates immune cells and influences appetite and metabolism.
- It comes from the same parent protein as your stress hormone ACTH. When your body produces one, the other often rises too.
- You can't take beta-endorphin as a pill. It breaks down in digestion and can't cross the blood-brain barrier — which is why all attempts to use it as a drug have failed.
How beta-endorphin is made: the POMC precursor
Beta-endorphin is not synthesized directly. It is cleaved from proopiomelanocortin (POMC), a 241-amino acid precursor protein that also gives rise to adrenocorticotropic hormone (ACTH), alpha-melanocyte-stimulating hormone (alpha-MSH), and several other bioactive peptides. This shared precursor means that beta-endorphin production is tightly linked to the stress response: the same hypothalamic-pituitary-adrenal (HPA) axis activation that releases cortisol also releases beta-endorphin.
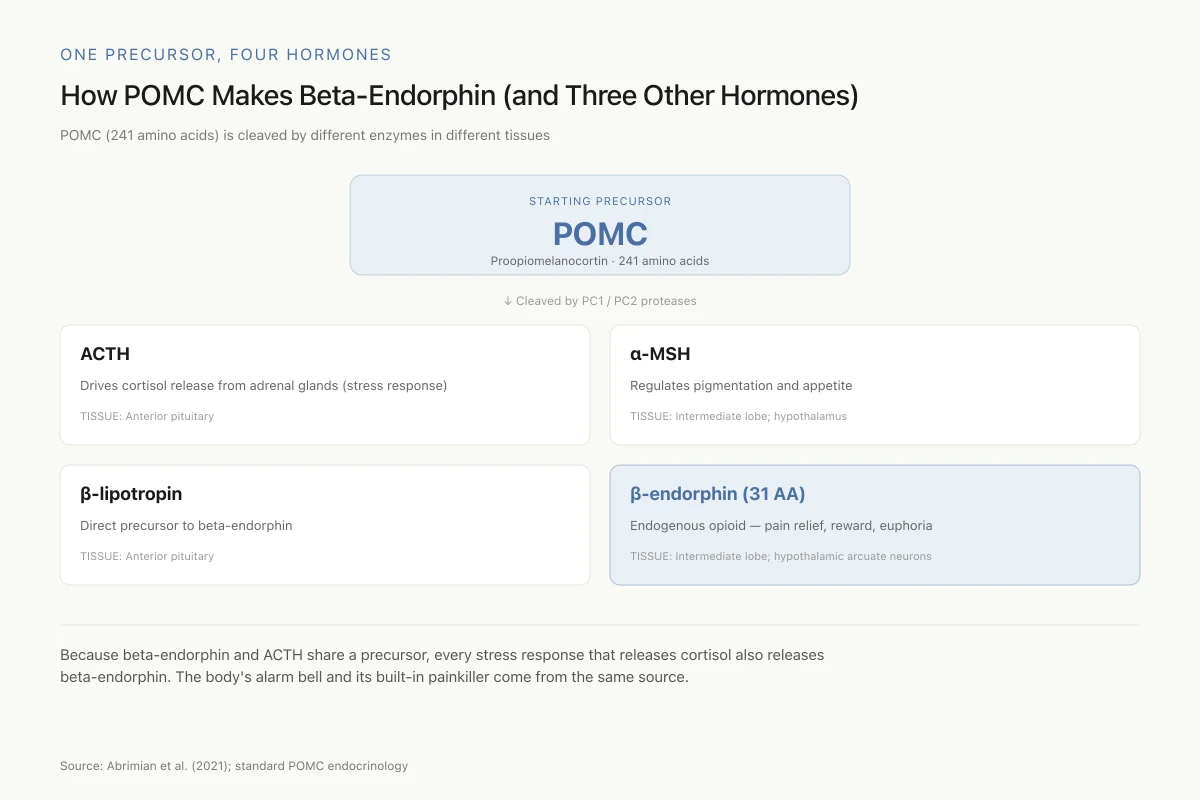
One Precursor, Four Hormones
How POMC Makes Beta-Endorphin (and Three Other Hormones)
POMC (241 amino acids) is cleaved by different enzymes in different tissues to produce different products
Because beta-endorphin and ACTH share a precursor, every stress response that releases cortisol also releases beta-endorphin — which is why acute stress can temporarily reduce pain perception. The body's alarm bell and its built-in painkiller come from the same source.
Source: Abrimian et al. (2021); standard POMC endocrinology
 View as image
View as imagePOMC is expressed primarily in the arcuate nucleus of the hypothalamus and in the anterior pituitary gland. In the pituitary, POMC is cleaved by prohormone convertase 1 (PC1) to generate ACTH, which is then further processed by prohormone convertase 2 (PC2) to yield beta-endorphin. The processing is tissue-specific: different cell types cleave POMC at different sites, producing different peptide products from the same precursor. In the anterior pituitary, the dominant products are ACTH and beta-lipotropin (the direct precursor of beta-endorphin). In the intermediate lobe and hypothalamic neurons, PC2-mediated processing predominates, producing alpha-MSH and beta-endorphin as the primary active peptides.
This shared precursor system has a critical functional consequence: any stimulus that increases ACTH release, primarily stress, also increases beta-endorphin release. The simultaneous release of a stress hormone (ACTH, which drives cortisol production) and an analgesic, euphorigenic peptide (beta-endorphin) provides the molecular basis for stress-induced analgesia, the well-documented phenomenon that acute stress temporarily reduces pain perception.
Yamamoto et al. (2025) revealed an additional layer of complexity. They found that POMC can be processed extracellularly, generating short beta-endorphin fragments outside the cell that have distinct biological activities from the full-length 31-amino acid peptide. In rat keratinocytes, these short fragments regulated cellular function through pathways that differed from canonical mu-opioid receptor signaling. This finding suggests that the biological repertoire of POMC-derived peptides is broader than previously appreciated, with extracellular processing creating a parallel set of signaling molecules.[6]
Where beta-endorphin binds: the mu-opioid receptor system
Beta-endorphin is the prototypical endogenous ligand for the mu-opioid receptor (MOR), the same receptor targeted by morphine, fentanyl, and other exogenous opioids. It binds MOR with higher affinity than met-enkephalin or leu-enkephalin, the other major endogenous opioid peptides, and produces more sustained signaling due to its larger size and slower dissociation kinetics.
Abrimian et al. (2021) characterized the relationship between endogenous opioid peptides and alternatively spliced mu-opioid receptor variants. The MOR gene produces multiple splice variants with different carboxyl-terminal sequences, and these variants show different signaling profiles when activated by beta-endorphin versus other endogenous opioids. This means the cellular response to beta-endorphin depends not just on receptor density but on which splice variants are expressed in a given brain region or cell type.[1]
Gupta et al. (2021) extended this picture by examining how endogenous opioid peptides regulate opioid receptor expression itself. Beta-endorphin binding to MOR triggers receptor internalization and recycling, a process that modulates receptor availability on the cell surface. Chronic exposure to high levels of beta-endorphin, as might occur during prolonged stress or sustained exercise, can alter the baseline receptor density and sensitivity of the opioid system.[2]
Lavigne et al. (2020) demonstrated that different endogenous opioid peptides produce "biased agonism" at the mu-opioid receptor. Endomorphin-1, endomorphin-2, and dynorphin-B each activated different intracellular signaling cascades through the same receptor. This biased signaling means that the biological effects of beta-endorphin at MOR are not identical to those produced by other endogenous opioids, even though they share the same receptor. Beta-endorphin's unique signaling profile likely contributes to its distinct behavioral effects, including the sustained euphoria associated with exercise.[11]
The runner's high: exercise and beta-endorphin release
The association between exercise and beta-endorphin is one of the most widely known facts in exercise science, though the underlying biology is more nuanced than popular accounts suggest. Vigorous aerobic exercise, typically sustained for 30 minutes or longer at moderate to high intensity, triggers the release of beta-endorphin from the anterior pituitary into the bloodstream. PET imaging studies using radiolabeled opioid receptor ligands have demonstrated that extended running also increases central opioid release in frontolimbic brain regions, with the magnitude of opioid release correlating with subjective euphoria ratings.
The precise threshold for exercise-induced beta-endorphin release varies between individuals and depends on fitness level, exercise intensity, and duration. Generally, exercise must exceed approximately 70% of maximal oxygen uptake (VO2max) to produce measurable increases in plasma beta-endorphin. Below this threshold, plasma levels remain unchanged. This intensity dependence explains why light walking does not produce a "runner's high" but sustained running, cycling, or rowing can.
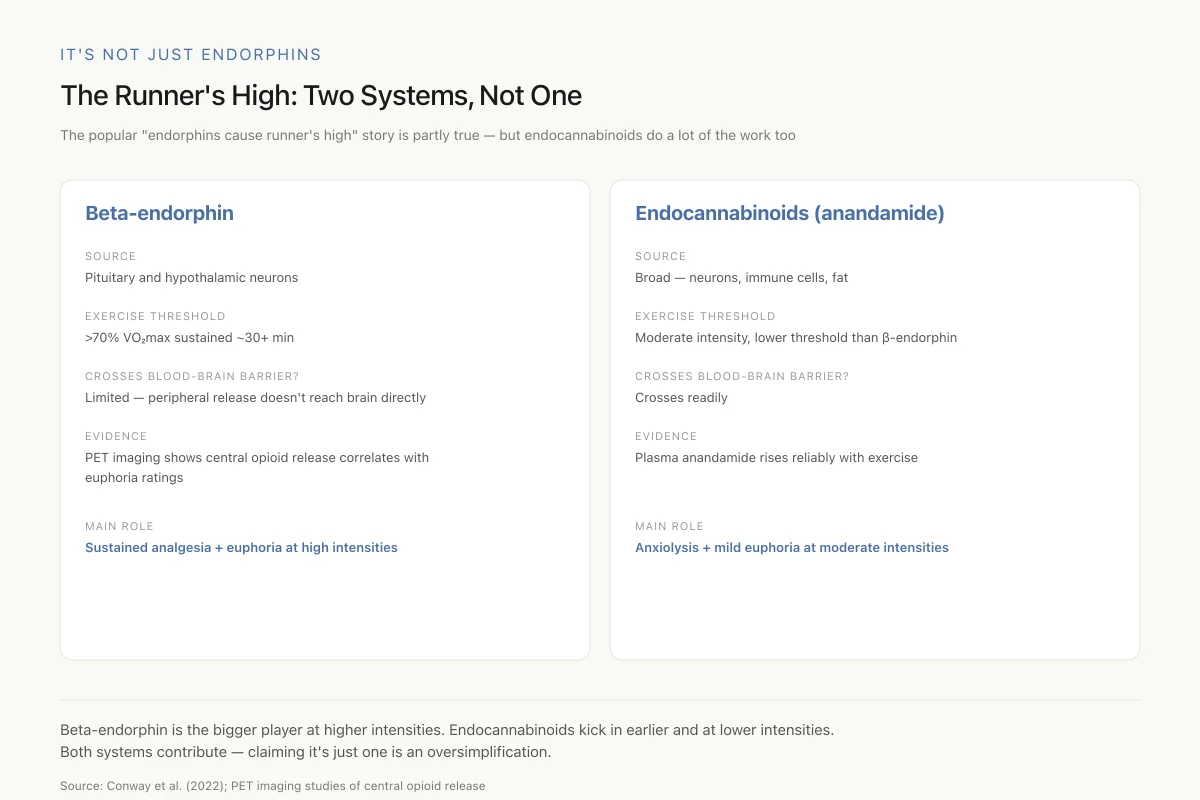
It's Not Just Endorphins
The Runner's High: Two Systems, Not One
The popular "endorphins cause runner's high" story is partly true — but endocannabinoids do a lot of the work too
Beta-endorphin is the bigger player at higher intensities and durations. Endocannabinoids kick in earlier and at lower intensities, which is why a 20-minute jog can produce mild euphoria even before you reach "runner's high" territory. Both systems contribute — claiming it's just one is an oversimplification.
Source: Conway et al. (2022); PET imaging studies of central opioid release
 View as image
View as imageA complication in interpreting exercise-endorphin research is that peripheral plasma beta-endorphin levels may not reflect central nervous system levels. Beta-endorphin does not cross the blood-brain barrier efficiently, so the beta-endorphin measured in blood samples after exercise originates primarily from the pituitary rather than from central POMC neurons. The euphoria-producing effects are driven by centrally released beta-endorphin acting on brain opioid receptors, which is a separate pool from peripheral release. This methodological issue has plagued exercise-endorphin research for decades: hundreds of studies report plasma beta-endorphin increases after exercise, but the correlation between peripheral levels and the subjective runner's high is inconsistent precisely because the two compartments are partially independent.
The peripheral beta-endorphin released during exercise is not without function. It contributes to exercise-induced analgesia through peripheral mu-opioid receptors on sensory nerve endings, which is why long-distance runners can sustain running despite accumulating tissue stress and inflammation. It also modulates cardiovascular responses during exercise, with evidence that beta-endorphin reduces exercise-induced increases in blood pressure and heart rate through central autonomic mechanisms.
Conway et al. (2022) reviewed the methodological challenges of detecting and quantifying endogenous opioid peptides, including beta-endorphin, in the context of reward signaling. They noted that traditional immunoassay methods often lack the specificity to distinguish beta-endorphin from its fragments and other POMC-derived peptides. Mass spectrometry-based approaches are improving measurement precision but remain technically demanding. These analytical limitations mean that quantitative claims about beta-endorphin levels in response to exercise or other stimuli should be interpreted with caution.[3]
Kraemer et al. (2024) demonstrated that exercise is not the only route to endogenous opioid peptide release. In resistance-trained men, floatation therapy (sensory deprivation in a float tank) produced measurable increases in endogenous opioid peptides. This finding suggests that relaxation-based interventions can engage the opioid system through mechanisms distinct from physical exertion, potentially through stress reduction and autonomic nervous system modulation.[4]
Beta-endorphin and pain modulation
Beta-endorphin is one of the body's most potent endogenous analgesics. Its pain-modulating effects operate through both peripheral and central mechanisms. In the brain and spinal cord, beta-endorphin activates mu-opioid receptors on pain-transmitting neurons, inhibiting the release of excitatory neurotransmitters and reducing the transmission of nociceptive signals. In the periphery, beta-endorphin released by immune cells at sites of inflammation contributes to local pain suppression.
Jorgensen et al. (1993) provided clinical evidence linking beta-endorphin deficiency to pain syndromes. They found that patients with functional abdominal pain had significantly decreased cerebrospinal fluid (CSF) beta-endorphin levels compared to healthy controls, and that these decreased levels correlated with increased pain sensitivity. This observation supports the concept that endogenous opioid tone, the baseline level of beta-endorphin in the central nervous system, serves as a buffer against pain perception. When this buffer is diminished, pain sensitivity increases.[7]
The relationship between beta-endorphin and other endogenous opioid peptides in pain modulation is complex. Beta-endorphin, met-enkephalin, leu-enkephalin, and dynorphin each have preferred receptors (mu, delta, and kappa respectively) and produce overlapping but distinct analgesic effects. In the dorsal horn of the spinal cord, these peptides are co-localized in overlapping neural populations, creating a multi-layered pain control system where the balance between different opioid peptides determines the net analgesic output.
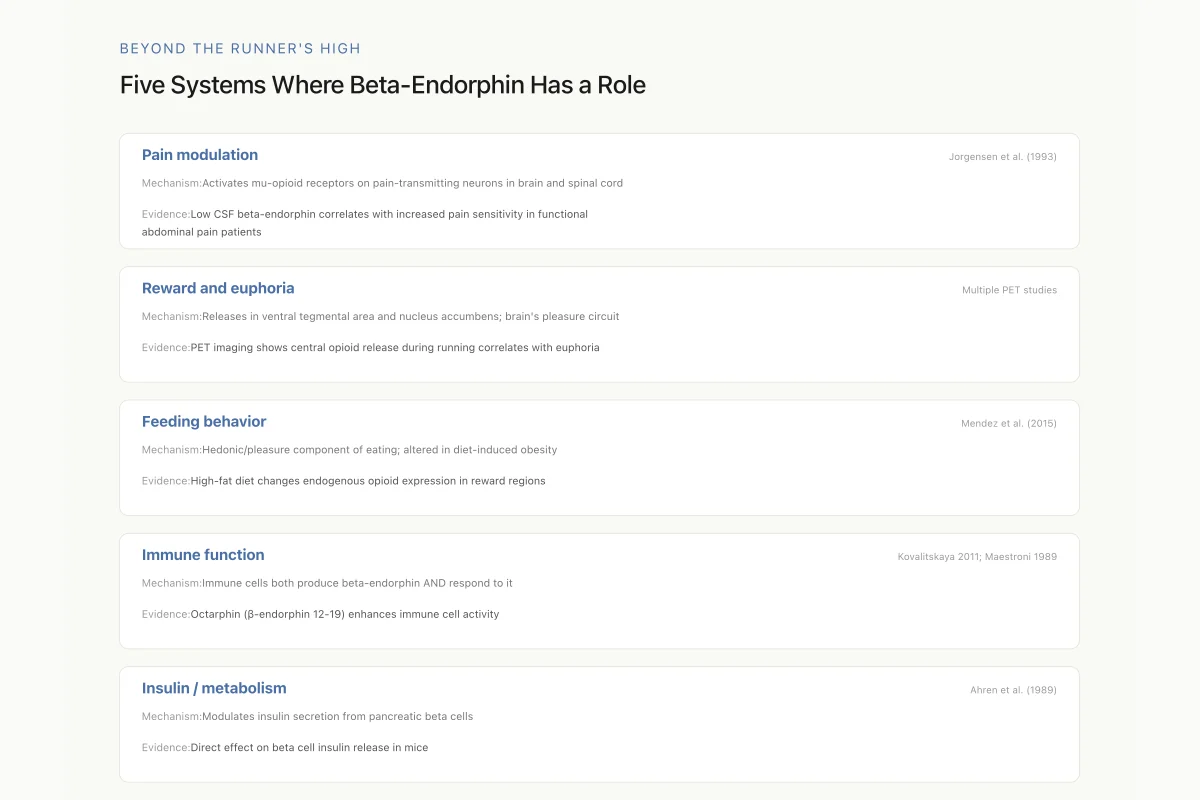
Beyond the Runner's High
Five Systems Where Beta-Endorphin Has a Role
Beta-endorphin is best known for the runner's high, but that's a narrow slice of its biology. It's a full-spectrum neuropeptide — operating in pain circuits, reward circuits, feeding circuits, the immune system, and metabolic tissue. Many effects once attributed to "opioids" in general are specifically beta-endorphin doing its many jobs.
Source: Studies cited in each row (1989-2024)
 View as image
View as imageBeyond pain: feeding, metabolism, and reward
Beta-endorphin's role extends well beyond analgesia. It is a central player in the brain's reward circuitry and has direct effects on feeding behavior and metabolic regulation.
Mendez et al. (2015) investigated the involvement of endogenous enkephalins and beta-endorphin in feeding and diet-induced obesity. They found that both peptide systems are activated during food consumption and that chronic exposure to high-fat diets alters the expression patterns of these endogenous opioids in brain regions associated with reward processing. The hedonic component of eating, the pleasure derived from palatable food, is at least partly mediated by beta-endorphin release in the nucleus accumbens and ventral tegmental area.[8]
Ahren et al. (1989) demonstrated direct metabolic effects of beta-endorphin, showing that it modulates basal and stimulated insulin secretion from pancreatic beta cells in mice. Met-enkephalin and dynorphin A also affected insulin release, but through different receptor mechanisms. This finding connects the endogenous opioid system to metabolic regulation, suggesting that beta-endorphin participates in the postprandial hormonal response alongside classical metabolic peptides like GLP-1 and insulin.[13]
Li et al. (2025) identified plasma beta-endorphin as a candidate biomarker for predicting obstructive sleep apnea syndrome, expanding the clinical relevance of beta-endorphin measurement beyond pain and exercise contexts. Their preliminary data suggest that circulating beta-endorphin levels, combined with neuropeptide Y levels, may help identify patients at risk for sleep-disordered breathing.[5]
Immune functions of beta-endorphin
One of the less-appreciated roles of beta-endorphin is its direct effect on the immune system. Beta-endorphin is produced not only by the pituitary and hypothalamus but also by immune cells themselves, including lymphocytes and monocytes. This immune-derived beta-endorphin acts in an autocrine and paracrine fashion, modulating immune cell activity at sites of inflammation and infection.
Kovalitskaya et al. (2011) demonstrated that a synthetic peptide corresponding to beta-endorphin fragment 12-19 (octarphin) had immunostimulating effects. This eight-amino acid fragment enhanced immune cell proliferation and activity through mechanisms that did not require full-length beta-endorphin or canonical mu-opioid receptor activation. The finding suggests that beta-endorphin's immune effects are mediated partly by shorter fragments generated by enzymatic processing, expanding the functional repertoire of the peptide beyond what the full-length molecule alone can achieve.[10]
Maestroni et al. (1989) showed that beta-endorphin and dynorphin could mimic the circadian immunoenhancing and anti-stress effects of melatonin. Both opioid peptides enhanced immune function when administered according to the circadian rhythm, with evening administration producing greater immunostimulation than morning administration. This temporal dependence links the endogenous opioid system to the circadian regulation of immunity and suggests that the immune-protective effects of beta-endorphin are time-of-day dependent.[9]
The immune connection has implications for the well-documented health benefits of regular exercise. If exercise-induced beta-endorphin release contributes to immune function enhancement, it provides a molecular mechanism linking physical activity to improved immune surveillance and reduced infection susceptibility. The bidirectional communication between the opioid and immune systems, with immune cells both producing and responding to beta-endorphin, represents one of the clearest examples of neuroimmune cross-talk mediated by a peptide hormone. This also explains why chronic stress, which initially elevates beta-endorphin but eventually depletes the system, is associated with immune suppression rather than enhancement.
The relationship to other endogenous opioid peptides
Beta-endorphin does not operate in isolation. The endogenous opioid system comprises three major peptide families: endorphins, enkephalins, and dynorphins, each derived from different precursor proteins and each with preferred receptor targets.
Enkephalins (met-enkephalin and leu-enkephalin), derived from proenkephalin, are the most abundant endogenous opioids in the brain. They preferentially activate delta-opioid receptors and are involved in pain modulation, mood regulation, and reward. Dynorphins, derived from prodynorphin, act primarily at kappa-opioid receptors and often produce effects opposite to beta-endorphin: dysphoria rather than euphoria, stress-like rather than stress-relieving.
Nociceptin/orphanin FQ represents a fourth endogenous opioid-like peptide that acts at its own receptor (NOP) and can either enhance or inhibit pain depending on the dose and site of action. Guan et al. (2023) identified nociceptin/orphanin FQ expression in endometriosis-associated nerve fibers, suggesting that this peptide contributes to the chronic pain of endometriosis through mechanisms distinct from classical opioid analgesia.[12]
The interactions between these peptide systems are reciprocal. Beta-endorphin can modulate enkephalin release, dynorphin can oppose beta-endorphin's effects, and nociceptin can inhibit beta-endorphin neurons in the hypothalamic arcuate nucleus. Understanding pain, mood, and reward in the brain requires considering the balance between all four systems rather than the activity of any single peptide. A shift toward dynorphin dominance, for instance, is associated with dysphoria and stress vulnerability, while a shift toward beta-endorphin and enkephalin activity is associated with positive affect and pain resilience. The opioid system functions as an integrated network, not a collection of independent pathways.
Current limitations and open questions
Despite decades of research, several fundamental questions about beta-endorphin remain unresolved.
The blood-brain barrier issue complicates all exercise-endorphin research. Peripheral plasma measurements are technically straightforward but may not reflect central nervous system opioid dynamics. PET imaging provides central data but is expensive, requires radioactive tracers, and has limited spatial and temporal resolution. Conway et al. (2022) emphasized that improved mass spectrometry methods are needed to accurately measure opioid peptide levels in different tissue compartments simultaneously.[3]
The relative contribution of beta-endorphin versus endocannabinoids to the runner's high remains debated. More recent research suggests that the endocannabinoid system, particularly anandamide, may play a larger role in exercise-induced euphoria than initially thought. Endocannabinoids cross the blood-brain barrier more readily than beta-endorphin, and their plasma levels rise with exercise at lower intensity thresholds. The current view is that both systems likely contribute, with endocannabinoids mediating anxiolysis and mild euphoria at moderate intensities, and beta-endorphin contributing additional analgesia and euphoria at higher intensities.
The therapeutic potential of beta-endorphin modulation is largely unexplored. If low CSF beta-endorphin correlates with increased pain sensitivity, could interventions that boost endogenous beta-endorphin production (exercise, acupuncture, stress management) serve as adjuncts to pain treatment? The concept is appealing but lacks the controlled clinical trial data needed to move from correlation to clinical recommendation.
The rapid enzymatic degradation of beta-endorphin (half-life measured in minutes) means that administering the peptide itself as a therapeutic is impractical without chemical modifications or encapsulation strategies. Enkephalinase inhibitors, which block the enzymes that degrade endogenous opioid peptides, represent an alternative approach: rather than adding exogenous beta-endorphin, they amplify the body's own production. This strategy avoids the tolerance, dependence, and respiratory depression risks of exogenous opioids, though clinical development of enkephalinase inhibitors has been slow.
The interaction between beta-endorphin and the endocannabinoid system also warrants investigation. Both systems are activated by exercise, both modulate pain and reward, and preclinical evidence suggests significant cross-talk between opioid and cannabinoid receptors. Understanding this interaction could inform combined therapeutic strategies for chronic pain that leverage both endogenous analgesic systems.
The Bottom Line
Beta-endorphin is a 31-amino acid opioid peptide derived from POMC that functions as the body's primary endogenous analgesic and reward signal. It binds mu-opioid receptors with high affinity, modulates pain through both central and peripheral mechanisms, participates in feeding behavior and metabolic regulation, and exerts direct immunostimulatory effects through both full-length and fragment forms. The exercise-endorphin connection, while real, is complicated by blood-brain barrier dynamics and the parallel contribution of endocannabinoids. The peptide operates within a larger network of endogenous opioids including enkephalins, dynorphins, and nociceptin, and understanding its biology requires considering the balance between all four systems.
Sources & References
- 1RPEP-05254·Abrimian, Anna et al. (2021). “How the Body's Own Opioid Peptides Work Through Different Versions of the Mu Opioid Receptor.” International journal of molecular sciences.Study breakdown →PubMed →↩
- 2RPEP-05429·Gupta, Achla et al. (2021). “Regulation of Opioid Receptors by Their Endogenous Opioid Peptides..” Cellular and molecular neurobiology.Study breakdown →PubMed →↩
- 3RPEP-06059·Conway, Sineadh M et al. (2022). “New Tools for Measuring the Brain's Own Opioid Peptides During Reward and Addiction.” Addiction neuroscience.Study breakdown →PubMed →↩
- 4RPEP-08598·Kraemer, William J et al. (2024). “Do Float Tanks Boost Your Endorphins After a Workout? A Study Says No.” Journal of strength and conditioning research.Study breakdown →PubMed →↩
- 5RPEP-12085·Li, Meng-Lin et al. (2025). “Plasma β-Endorphin and Neuropeptide Y as Candidate Biomarkers for Predicting Obstructive Sleep Apnea Syndrome: A Preliminary Study..” Canadian respiratory journal.Study breakdown →PubMed →↩
- 6RPEP-14302·Yamamoto, Hiroyuki et al. (2025). “Extracellular processing of proopiomelanocortin generates short beta endorphin that regulates rat keratinocytes via the delta opioid receptor..” Scientific reports.Study breakdown →PubMed →↩
- 7RPEP-00265·Jørgensen, L S et al. (1993). “People With Unexplained Abdominal Pain Have Lower Beta-Endorphin in Their Spinal Fluid.” Scandinavian journal of gastroenterology.Study breakdown →PubMed →↩
- 8RPEP-02740·Mendez, Ian A et al. (2015). “Enkephalins Drive Feeding Motivation and Weight Gain While β-Endorphin Controls Taste Pleasure.” Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology.Study breakdown →PubMed →↩
- 9RPEP-00123·Maestroni, G J et al. (1989). “Beta-Endorphin and Dynorphin Mimicked Melatonin Immune-Boosting Effects.” International journal of immunopharmacology.Study breakdown →PubMed →↩
- 10RPEP-01796·Kovalitskaya, Yu A et al. (2011). “A Beta-Endorphin Fragment (Octarphin) Stimulates the Immune System.” Biochemistry. Biokhimiia.Study breakdown →PubMed →↩
- 11RPEP-04925·LaVigne, Justin et al. (2020). “Body's Own Opioid Peptides Show Biased Signaling at the Mu Receptor — Implications for Safer Pain Drugs.” Pharmacological reports : PR.Study breakdown →PubMed →↩
- 12RPEP-06923·Guan, Qihui et al. (2023). “Pain-Modulating Nociceptin Receptors Found on Endometriosis Nerve Fibers — A New Treatment Target?.” Cells.Study breakdown →PubMed →↩
- 13RPEP-00102·Ahrén, B (1989). “Opioid Peptides Had Complex Effects on Insulin Release in Mice.” International journal of pancreatology : official journal of the International Association of Pancreatology.Study breakdown →PubMed →↩