Epithalon: The Telomerase-Activating Peptide
Epithalon
4 amino acids (Ala-Glu-Asp-Gly)
Epithalon is a synthetic tetrapeptide that activated telomerase and extended telomeres in human cell cultures. Animal studies showed lifespan increases of 11-31% in mice and rats. No randomized controlled trials in humans exist.
Khavinson et al., Bulletin of Experimental Biology and Medicine, 2003
Khavinson et al., Bulletin of Experimental Biology and Medicine, 2003
If you only read one thing
Epithalon is a four-amino-acid peptide that did two interesting things: it turned on telomerase in human cells in a dish, and it extended the lifespan of mice by about 11%. Those are real findings. But nearly every study comes from one Russian laboratory, and no placebo-controlled human trial has ever been run. The FDA hasn't approved it and banned it from compounding pharmacies in 2023. It's sold only through gray-market 'research use only' suppliers with no quality control. The science is worth watching. The case for taking it today is not strong enough to call it evidence-based.
Epithalon (also spelled Epitalon) is a synthetic tetrapeptide with the sequence Ala-Glu-Asp-Gly, developed at the St. Petersburg Institute of Bioregulation and Gerontology in Russia. Its primary claim to interest is that it activated telomerase, the enzyme that maintains chromosome-protecting telomere caps, in human cell cultures.[1] It has also extended lifespan in several animal models. These findings have made epithalon one of the most discussed peptides in the longevity space, but the evidence base has significant gaps that are often glossed over. For how epithalon connects to pineal gland function and melatonin, see our guide to Epithalon and melatonin: the pineal gland connection.
Key Takeaways
- Epithalon is a tiny four-amino-acid peptide built to switch on telomerase — the enzyme that rebuilds the protective caps on your chromosomes.
- In a petri dish, human cells treated with it kept dividing past the point where normal cells stop. That's a real, measurable finding.
- In mice, it extended average lifespan by about 11% — meaningful for a longevity drug, but not a miracle.
- Nearly every human and animal study comes from a single Russian laboratory. Independent labs have barely touched it.
- No placebo-controlled human trial has ever been done. The widely cited "mortality reduction" study was neither randomized nor blinded.
- Cancer cells also use telomerase to become immortal. Giving pre-cancerous cells that same ability is a real theoretical risk nobody has studied long-term.
- The FDA banned it from compounding pharmacies in 2023. Anything sold as "epithalon" in the US comes from unregulated sources with no quality control.
Epithalon is a synthetic version of the naturally occurring tetrapeptide epithalamin, which was originally isolated from bovine pineal gland extracts. The pineal gland produces melatonin and several small peptides, and the hypothesis driving epithalon research is that pineal peptides regulate aging through gene expression modulation, particularly of telomerase.[2]
The peptide is only four amino acids long: alanine-glutamic acid-aspartic acid-glycine (AEDG). Its molecular weight is approximately 390 Da, making it one of the smallest bioactive peptides studied for anti-aging effects. The small size raises the question of how such a minimal structure can produce specific biological effects, though other short peptides (such as the tripeptide GHK-Cu) have well-documented receptor interactions.
Epithalon is distinct from epithalamin, its predecessor. Epithalamin is a crude extract of the pineal gland that contains epithalon along with other peptides and biological molecules. The early lifespan studies used epithalamin; the later, more mechanistic studies used the purified synthetic tetrapeptide epithalon. This distinction matters when evaluating the evidence because the effects of epithalamin may reflect contributions from multiple active components, not epithalon alone.
The telomerase evidence
Cell culture studies
The foundational finding was published in 2003 by Khavinson and colleagues. When epithalon was added to cultures of telomerase-negative human fetal fibroblasts, it induced expression of hTERT (the catalytic subunit of telomerase), activated telomerase enzymatic activity, and produced measurable telomere elongation.[1]
A follow-up study in 2004 demonstrated the functional consequence of this telomerase activation: epithalon-treated human pulmonary fibroblasts continued dividing past passage 44, while untreated control cells stopped dividing at passage 34, reaching the Hayflick limit. The treated cells showed no signs of malignant transformation.[3]
A 2026 study from a different research group provided partial independent confirmation. Sanchez and colleagues tested epitalon in human breast cancer cell lines (21NT and BT474) and found that it increased telomere length through two mechanisms: telomerase upregulation in some lines (with hTERT expression upregulated 12-fold at 1 ug/mL in 21NT cells) and alternative lengthening of telomeres (ALT) in others.[4] This study is the first non-Russian confirmation of epithalon's telomere effects, though it used cancer cell lines rather than normal somatic cells.
What the cell data does not prove
Activating telomerase in a petri dish and extending human lifespan are separated by an enormous translational gap. Cancer cells routinely activate telomerase as part of their immortalization program. The question is whether epithalon-induced telomerase activation in normal cells would produce beneficial effects without increasing cancer risk. The 2004 study reported no malignant transformation in treated fibroblasts, but cell culture experiments lasting dozens of passages cannot rule out long-term oncogenic potential in a living organism.
Animal lifespan studies
Epithalamin (pineal extract) studies
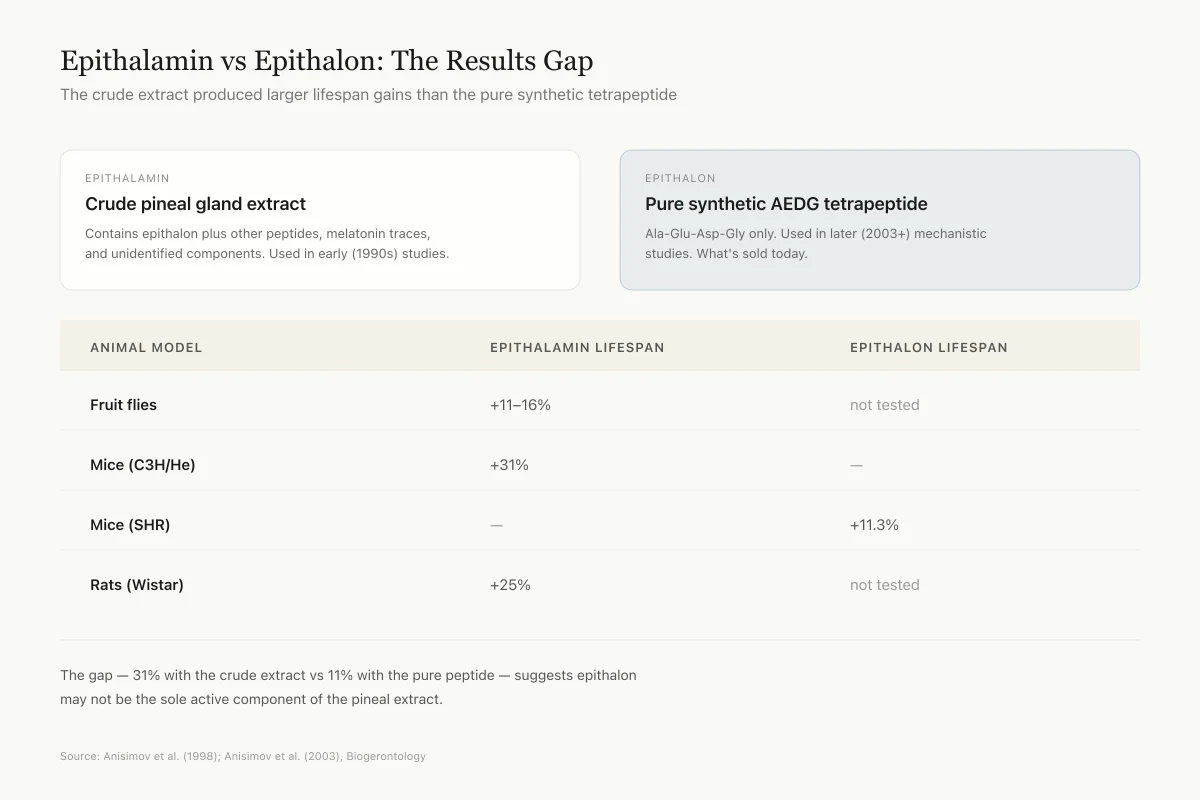
Before the synthetic tetrapeptide was available, Anisimov and colleagues tested epithalamin (the crude pineal extract) in multiple species. A 1998 study reported lifespan increases of 11-16% in Drosophila fruit flies, 31% in C3H/He mice, and 25% in Wistar rats.[5] These are substantial effects by the standards of longevity research, where interventions that reliably extend lifespan by more than 10% are rare.
The limitation is that epithalamin is not a pure compound. The lifespan extension could result from epithalon, from other pineal peptides, from melatonin contamination, or from interactions between multiple components. Attributing the effect specifically to the AEDG tetrapeptide requires testing with the synthetic version.
Synthetic epithalon studies
A 2003 study tested synthetic epithalon (not the crude extract) in female Swiss-derived SHR mice. The treated group showed an 11.3% increase in mean lifespan compared to controls. Epithalon also delayed age-related changes in estrous cycling, suggesting effects on neuroendocrine aging.[6]
The 11.3% increase with the pure tetrapeptide is notably smaller than the 31% seen with epithalamin in C3H/He mice, supporting the interpretation that the crude extract contains additional bioactive components beyond epithalon itself.
Pure Peptide vs Crude Extract
Epithalamin vs Epithalon: The Results Gap
The crude pineal extract produced larger lifespan gains than the pure synthetic tetrapeptide
Epithalamin
Crude pineal gland extract
Contains epithalon plus other peptides, melatonin traces, and unidentified components. Used in early (1990s) lifespan studies.
Epithalon
Pure synthetic AEDG tetrapeptide
Ala-Glu-Asp-Gly only. Used in later (2003+) mechanistic and lifespan studies. What's sold today.
The size of the gap — 31% with the crude extract, 11% with the pure peptide in the only direct test — suggests epithalon may not be the main active component of the pineal extract. Other pineal peptides or melatonin may carry the rest of the effect.
Source: Anisimov et al. (1998); Anisimov et al. (2003), Biogerontology
 View as image
View as imageHuman evidence
The Khavinson prospective study
The most cited human evidence comes from a prospective cohort study of 266 people over age 60, published in 2003. Treatment with epithalamin (the pineal extract, not synthetic epithalon) produced a reported 1.6-1.8-fold reduction in mortality over the following 6 years. The same study also reported that both epithalon and epithalamin increased telomere lengths in blood cells of patients aged 60-65 and 75-80.[7]
This study has several limitations that are not always acknowledged. It was not randomized or placebo-controlled. The sample size of 266 is small for mortality endpoints. The study was conducted and reported by the same group that developed the peptide. No independent group has replicated these mortality findings in a controlled trial design.
Retinitis pigmentosa studies
Epithalon has been tested in 162 patients with retinitis pigmentosa, with reported improvements in retinal function. These studies were conducted in Russia and published in Russian-language journals, limiting their accessibility for independent evaluation.
The evidence gap
There are no Phase I, II, or III clinical trials of epithalon registered on ClinicalTrials.gov or equivalent Western trial registries. The absence of controlled human trials means that all human evidence comes from observational studies and case reports, which cannot establish causation.
Evidence Summary
What We Know About Epitalon's Telomere Effects
Epitalon activated telomerase in human fibroblasts, extended lifespan by 10 extra doublings beyond Hayflick limit
Confirmed telomere extension via both telomerase upregulation AND alternative lengthening (ALT) — dual mechanism
Epithalamin increased mean lifespan: +31% in fruit flies, +12.3% in mice, +24% in female rats
Epitalon reduced spontaneous tumor incidence ~30% in female mice and extended maximum lifespan
Increased telomere length in blood cells of elderly patients (ages 60-80), statistically significant
Does not exist. No placebo-controlled human trial for anti-aging endpoints.
Source: Khavinson (2003); Sanchez (2026); Anisimov (1998, 2003)
 View as image
View as imageThe single-laboratory problem
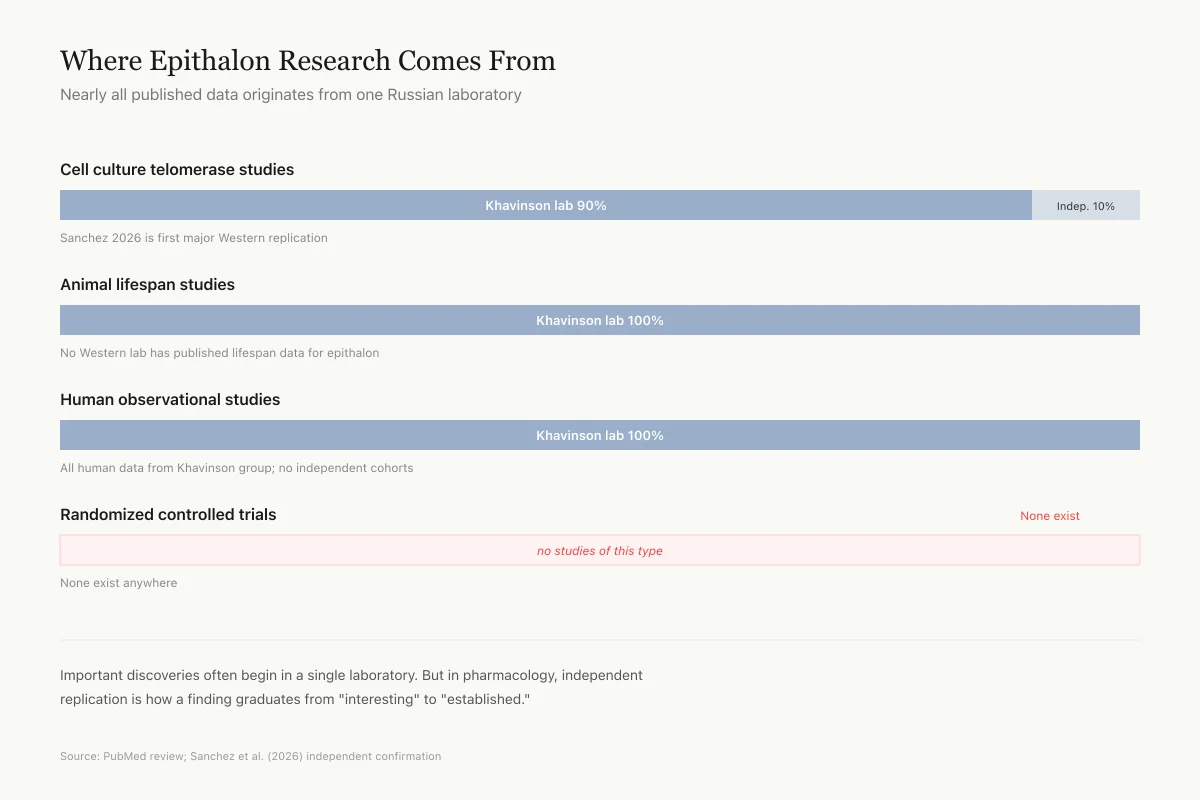
Nearly all published epithalon research, spanning cell culture, animal, and human studies, originates from the laboratory of Vladimir Khavinson at the St. Petersburg Institute of Bioregulation and Gerontology. This is not inherently disqualifying; important discoveries often begin in a single laboratory. But in pharmacology, independent replication is the standard for establishing an effect as real and robust.
The 2026 Sanchez et al. study represents the first significant independent work on epithalon's cellular mechanisms.[4] It confirmed telomere elongation in cancer cell lines but did not test lifespan extension, pineal gland function, or in vivo effects. More independent research is needed before the broader claims about epithalon can be evaluated on their merits.
A 2025 review in Molecules acknowledged that epitalon research is "promising" but explicitly noted the need for large-scale controlled studies to validate the findings from Khavinson's group.[8]
Source of Evidence
Where Epithalon Research Comes From
Nearly all published data originates from one Russian laboratory
Khavinson group dominates; Sanchez 2026 is first major Western replication
No Western lab has published lifespan data for epithalon
All human data from Khavinson group; no independent cohorts
None exist anywhere
Important discoveries often begin in a single laboratory. But in pharmacology, independent replication is how a finding graduates from "interesting" to "established." Epithalon has not cleared that bar for most of its claims.
Source: PubMed review of epithalon/epitalon publications; Sanchez et al. (2026) independent confirmation
 View as image
View as imageMechanism: how a tetrapeptide might activate telomerase
A 2021 systematic review of peptide regulation of gene expression proposed that short peptides like epithalon interact directly with DNA through sequence-specific binding to the minor groove, influencing gene transcription without traditional receptor-mediated signaling.[9]
This is an unconventional mechanism. Most peptide drugs work by binding cell-surface receptors (like GLP-1 agonists) or intracellular targets (like some antimicrobial peptides). Direct DNA interaction by a four-amino-acid peptide, if confirmed, would represent a distinct pharmacological mechanism. The hypothesis has been supported by computational modeling studies, but direct experimental proof that epithalon binds to specific DNA sequences in living cells and thereby activates hTERT transcription has not been published.
The proposed mechanism for how epithalon may influence telomere length is covered in more detail in our dedicated article.
Epithalon within the peptide bioregulation framework
Khavinson's broader research program proposes that short peptides (2-4 amino acids) serve as natural bioregulators that modulate gene expression across tissues. Epithalon is one of several peptides in this framework. Thymalin, a thymus-derived dipeptide, is claimed to restore immune function in aging. Thymulin, another thymic peptide, has documented zinc-dependent immunomodulatory effects. A 2022 review examined how peptide bioregulators may influence the senescence-associated secretory phenotype (SASP) of cardiovascular cells, proposing that short peptides could modulate inflammatory aging processes.[10]
The scientific question is whether a four-amino-acid peptide can have specific biological effects or whether the observed results reflect non-specific cellular responses to amino acid supplementation. Several lines of evidence support specificity: the effects are sequence-dependent (other tetrapeptides do not activate telomerase), they occur at low concentrations, and they involve upregulation of specific genes rather than global transcriptional changes. The counterargument is that the specificity data comes primarily from one laboratory.
The broader field of peptide regulation of telomere maintenance provides context for evaluating epithalon's claims against the wider evidence for peptide-based anti-aging interventions.
Regulatory status and availability
Epithalon is not approved by the FDA, EMA, or any other major regulatory agency for any medical indication. In September 2023, the FDA classified epithalon among peptides that cannot be used in compounding pharmacies, citing insufficient safety data and potential immunogenicity risks.
The peptide remains available through "research use only" suppliers, which is a regulatory gray area that allows purchase without a prescription but also means no quality control standards, no batch testing requirements, and no guarantee that the product contains what the label claims. The connection between sirtuins and peptide regulation provides additional context on the broader longevity peptide landscape.
Safety
ModerateTelomerase activation carries a theoretical cancer signal
Concern
Cancer cells use telomerase activation as one of the steps to become immortal. Any drug that systemically activates telomerase raises the theoretical question of whether pre-cancerous cells — which nearly everyone harbors — could be given a survival advantage. Long-term in vivo cancer-risk data for chronic epithalon dosing does not exist.
What the research says
The 2003 Anisimov mouse study did not find increased tumor incidence, and the 2004 cell study saw no malignant transformation. These are reassuring data points but not a full safety profile. Cell culture can't model years of systemic exposure, and the mouse cohort was modest.
Particularly relevant for: Anyone considering epithalon, especially those with personal or family cancer history, known precancerous lesions, or active surveillance
What to do
Do not use epithalon if you have an active cancer, a history of cancer that could recur, or a strong family history. Discuss with an oncologist before any use. Gray-market products additionally carry contamination and identity risks that a pharmacist would normally catch.
Shay & Wright reviews on telomerase and cancer; Anisimov 2003; Khavinson 2004 cell study
What is and is not established
Established: Epithalon activates telomerase and extends telomeres in human cell cultures. This has been demonstrated by the original Russian group and partially confirmed by an independent 2026 study. Animal studies using both epithalamin and synthetic epithalon show lifespan extension in mice, rats, and fruit flies, though the magnitude varies substantially between the crude extract and the pure peptide.
Not established: Whether epithalon extends human lifespan, prevents age-related disease, or is safe for chronic use. No randomized controlled trial has been conducted. The observational human data comes exclusively from the developing group's own studies. The mechanism of action, specifically whether epithalon directly binds DNA to activate hTERT transcription, remains a hypothesis supported by indirect evidence.
Unknown: Long-term cancer risk from telomerase activation in vivo. Optimal dosing for any proposed application. Whether the effects seen in cell culture and animal models translate to meaningful human outcomes. Whether epithalon from gray-market suppliers actually contains the AEDG sequence at the labeled concentration.
The Bottom Line
Epithalon (AEDG) is a synthetic tetrapeptide that activated telomerase and extended telomeres in human cell cultures, and increased lifespan by 11-31% in animal models depending on the preparation used. Nearly all evidence comes from a single Russian laboratory. The 2026 independent confirmation of telomere effects in cancer cell lines is encouraging but limited. No randomized human trials exist, the FDA has not approved the peptide, and gray-market availability raises quality concerns. The science is interesting; the evidence base for human use is insufficient.
Sources & References
- 1RPEP-00833·Khavinson, V Kh et al. (2003). “Epithalon Peptide Activates Telomerase and Lengthens Telomeres in Human Cells.” Bulletin of experimental biology and medicine.Study breakdown →PubMed →↩
- 2RPEP-00738·Khavinson, V Kh et al. (2002). “Peptides and Ageing..” Neuro endocrinology letters.Study breakdown →PubMed →↩
- 3RPEP-00933·Khavinson, V Kh et al. (2004). “Epithalon Peptide Helps Human Cells Overcome the Hayflick Division Limit.” Bulletin of experimental biology and medicine.Study breakdown →PubMed →↩
- 4RPEP-16052·Sanchez-Martin, Veronica et al. (2026). “Epitalon increases telomere length in human cell lines through telomerase upregulation or ALT activity..” Biochemical and biophysical research communications.Study breakdown →PubMed →↩
- 5RPEP-00447·Anisimov, V N et al. (1998). “Pineal peptide preparation epithalamin increases the lifespan of fruit flies, mice and rats..” Mechanisms of ageing and development.Study breakdown →PubMed →↩
- 6RPEP-00788·Anisimov, V N et al. (2003). “Epitalon Peptide Extended Lifespan by 12% and Reduced Cancer in Aging Mice.” Biogerontology.Study breakdown →PubMed →↩
- 7RPEP-00834·Khavinson, Vladimir Kh et al. (2003). “Russian Study Claims Thymus and Pineal Peptides Cut Death Rates Up to 4-Fold in Elderly Patients.” Neuro endocrinology letters.Study breakdown →PubMed →↩
- 8RPEP-11540·Ivko, Olga M et al. (2025). “Overview of Epitalon-Highly Bioactive Pineal Tetrapeptide with Promising Properties..” Molecules (Basel.Study breakdown →PubMed →↩
- 9RPEP-05495·Khavinson, Vladimir Khatskelevich et al. (2021). “How Tiny Peptides Can Switch Genes On and Off: A Systematic Review.” Molecules (Basel.Study breakdown →PubMed →↩
- 10RPEP-06247·Khavinson, Vladimir et al. (2022). “Senescence-Associated Secretory Phenotype of Cardiovascular System Cells and Inflammaging: Perspectives of Peptide Regulation..” Cells.Study breakdown →PubMed →↩