Casomorphins: The Opioid Peptides in Cheese
Casomorphins and Dairy Peptides
1/10th morphine's potency
Human beta-casomorphin-7 binds mu-opioid receptors at roughly one-tenth to one-twentieth the potency of morphine, enough for measurable biological activity but far below pharmacological opioid effects.
Koch et al., Naunyn-Schmiedeberg's Archives of Pharmacology, 1985
Koch et al., Naunyn-Schmiedeberg's Archives of Pharmacology, 1985
If you only read one thing
Casomorphins are small peptide fragments that break off from milk protein during digestion. They latch onto opioid receptors in your gut — the same family of switches morphine uses — but at about one-tenth to one-twentieth morphine's strength. They probably slow gut transit and stimulate mucus locally in the intestine. Whether enough gets past the gut wall to affect your brain is still unclear. The 'cheese is addictive' story goes well beyond what the evidence supports.
When you eat cheese, yogurt, or drink milk, digestive enzymes break down beta-casein into smaller peptide fragments. Some of these fragments, called casomorphins, bind opioid receptors. The most studied is beta-casomorphin-7 (BCM-7), a seven-amino-acid peptide that preferentially binds mu-opioid receptors in the gut wall and, if absorbed systemically, potentially in the brain.[1] The word "opioid" makes casomorphins sound alarming, but their potency is low: roughly 1/10th to 1/20th that of morphine in receptor binding assays.[1] Whether this low-level opioid activity produces meaningful health effects in humans remains one of the most debated questions in food peptide science. This article reviews what is actually known about casomorphins: their chemistry, how they form, what they do at opioid receptors, and why the evidence for health effects remains difficult to establish despite four decades of research. For broader context on bioactive peptides in milk, see our companion article.
Key Takeaways
- Human and bovine beta-casomorphins bind opioid receptors with highest affinity for mu-receptors, lower for delta-receptors, and lowest for kappa-receptors (Koch et al., 1985).[1]
- BCM-7 is released primarily from the A1 variant of beta-casein; the A2 variant produces approximately 4-fold less BCM-7 during in vitro digestion (Kaminski et al., 2007).[6]
- In rat jejunum, BCM-7 at micromolar concentrations induced mucus release through mu-opioid receptor activation, an effect blocked by naloxone (Trompette et al., 2003).[5]
- Beta-casomorphin-5 stimulated neurite outgrowth in mouse neuroblastoma cells at picomolar concentrations in a naloxone-reversible manner (Sakaguchi et al., 1998).[3]
- The European Food Safety Authority found no established cause-and-effect relationship between oral BCM-7 intake and non-communicable diseases in its 2009 assessment.[10]
- Whether casomorphins cross the intestinal barrier in sufficient quantities to produce systemic or neurological effects in healthy adults remains unconfirmed.[9]
Milk-Derived Opioid Peptides
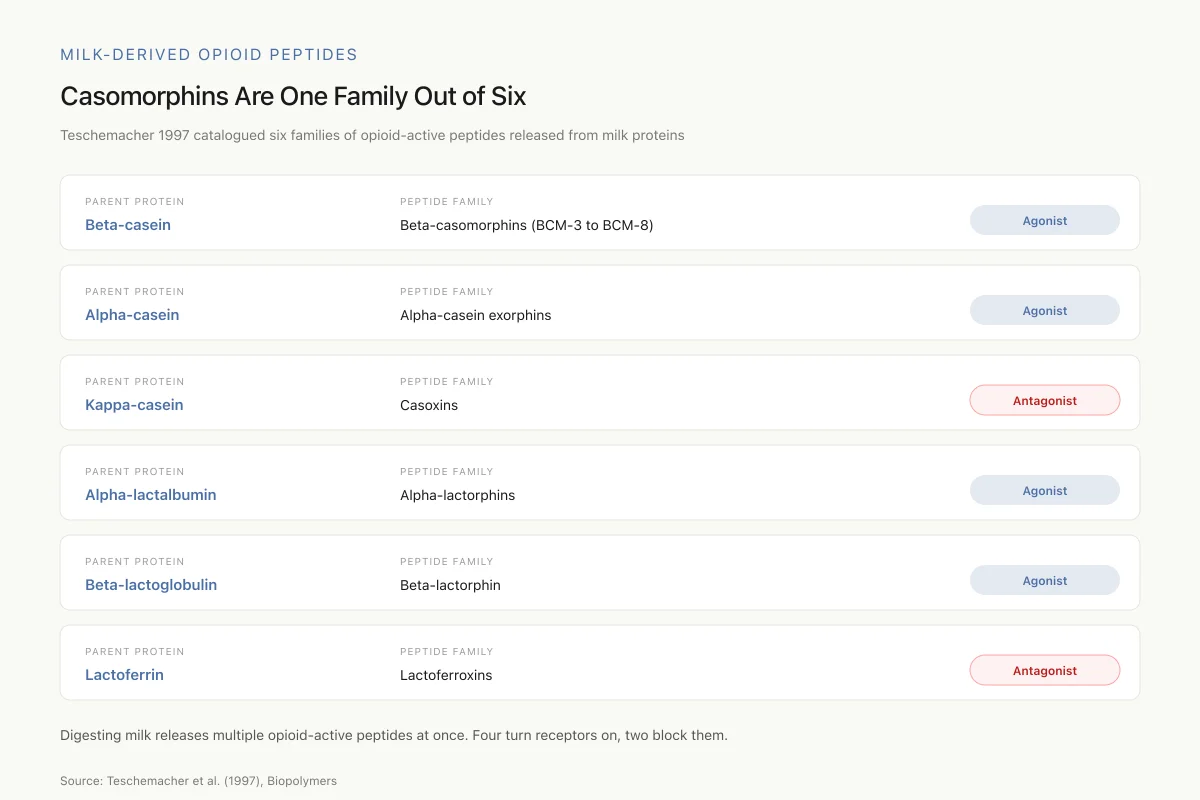
Casomorphins Are One Family Out of Six
Teschemacher (1997) catalogued six families of opioid-active peptides released from milk proteins. Four are agonists; two are antagonists.
Parent protein
Beta-casein
Peptide family
Beta-casomorphins (BCM-3 to BCM-8)
Parent protein
Alpha-casein
Peptide family
Alpha-casein exorphins
Parent protein
Kappa-casein
Peptide family
Casoxins
Parent protein
Alpha-lactalbumin
Peptide family
Alpha-lactorphins
Parent protein
Beta-lactoglobulin
Peptide family
Beta-lactorphin
Parent protein
Lactoferrin
Peptide family
Lactoferroxins
When casein digests, multiple opioid-active peptides come out at once. Some turn opioid receptors on; some block them. The net effect of drinking milk isn’t BCM-7 alone — it’s this whole mix.
Source: Teschemacher et al. (1997), Biopolymers
 View as image
View as imageWhat are casomorphins?
Casomorphins are peptide fragments released during the enzymatic digestion of casein, the primary protein in mammalian milk. The name combines "casein" and "morphine," reflecting their ability to bind opioid receptors. Teschemacher (1997) identified the full family of milk-derived opioid peptides: alpha-casein exorphins, beta-casomorphins, casoxins (from kappa-casein), alpha-lactorphins (from alpha-lactalbumin), beta-lactorphin (from beta-lactoglobulin), and lactoferroxins (from lactoferrin). Of these, only casoxins and lactoferroxins act as opioid antagonists. All others are agonists.[2]
The most studied casomorphin is beta-casomorphin-7 (BCM-7), with the amino acid sequence Tyr-Pro-Phe-Pro-Gly-Pro-Ile. The tyrosine residue at position 1 and the proline-rich backbone are characteristic of opioid peptides. BCM-7 exists in both human and bovine forms, with bovine BCM-7 estimated to have up to 30 times greater potency than human BCM-7 in some assays.
Shorter fragments also exist: BCM-5 (Tyr-Pro-Phe-Pro-Gly), BCM-4, and BCM-3. These retain opioid activity but at varying potencies. Teschemacher (1997) proposed that casomorphins function as "food hormones," carrying specific biological information from the milk-producing organism to the milk-receiving organism.[2]
How casomorphins form during digestion
Casomorphins are not present in intact milk. They form when digestive enzymes cleave beta-casein during gastrointestinal transit. The sequence that produces BCM-7 is embedded within beta-casein at positions 60-66.
Boutrou et al. (2013) performed the most direct study of this process in humans. Using double-lumen nasogastric tubes in healthy volunteers, they sampled jejunal contents after ingestion of casein or whey protein. Mass spectrometry identified multiple bioactive peptides released during digestion, confirming that casein hydrolysis in the human jejunum produces peptide fragments with known biological activities.[8]
The A1/A2 distinction
The amount of BCM-7 produced depends on which variant of beta-casein is present. At position 67, A1 beta-casein has histidine while A2 beta-casein has proline. This single amino acid difference profoundly affects BCM-7 release: histidine at position 67 allows enzymatic cleavage that liberates BCM-7, while proline at position 67 resists this cleavage.
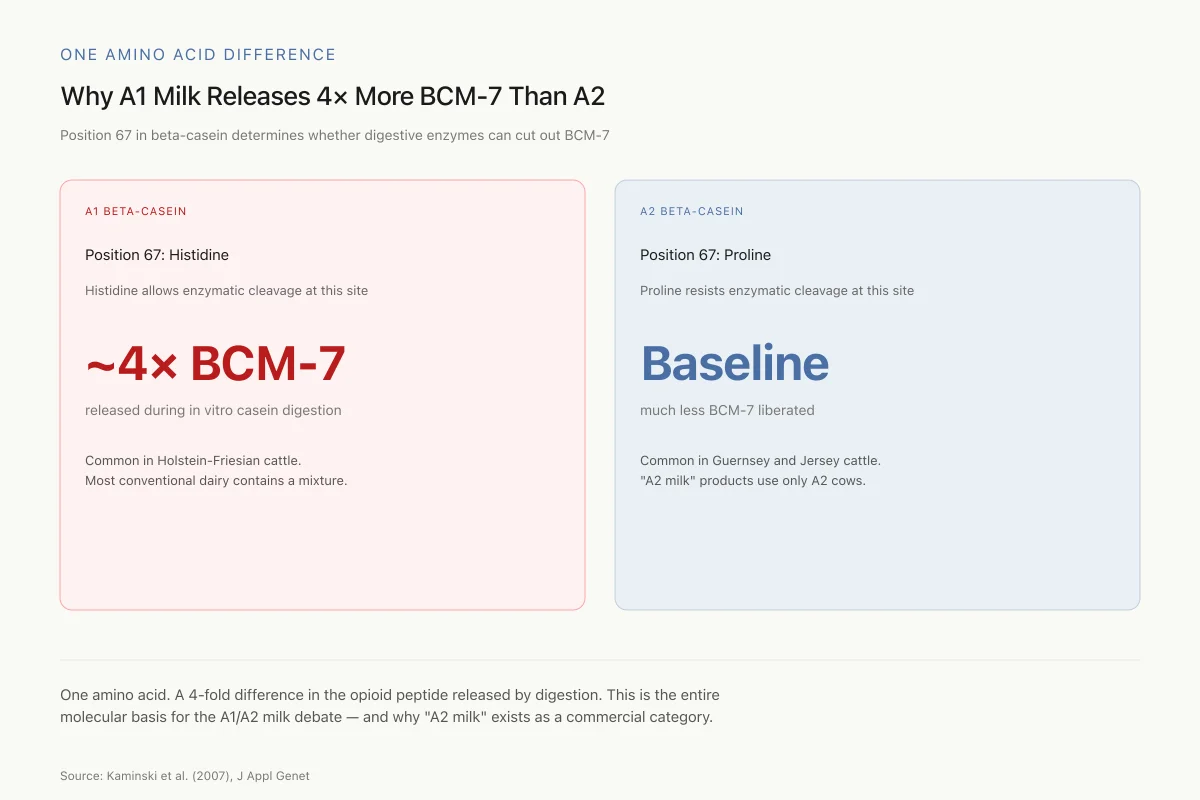
Kaminski et al. (2007) reviewed the evidence and reported that in vitro hydrolysis of A1 beta-casein produces approximately 4-fold more BCM-7 than A2 beta-casein. Most conventional cow's milk contains a mixture of A1 and A2 variants. Breeds like Holstein-Friesian tend to produce predominantly A1 milk, while Guernsey and Jersey cattle produce predominantly A2 milk. "A2 milk" products are specifically sourced from cows that carry only the A2 variant.[6]
One Amino Acid Difference
Why A1 Milk Releases 4× More BCM-7 Than A2
Position 67 in beta-casein determines whether digestive enzymes can cut out BCM-7.
A1 Beta-Casein
Position 67: Histidine
Histidine allows enzymatic cleavage at this site.
~4× BCM-7
released during in vitro casein digestion
Common in Holstein-Friesian cattle
A2 Beta-Casein
Position 67: Proline
Proline resists enzymatic cleavage at this site.
Baseline
much less BCM-7 liberated
Common in Guernsey and Jersey cattle
One amino acid. A 4-fold difference in the opioid peptide released by digestion. This is the entire molecular basis for the A1/A2 milk debate — and why “A2 milk” products exist as a commercial category.
Source: Kaminski et al. (2007), J Appl Genet
 View as image
View as imageCheese production concentrates casein, meaning cheese contains substantially more beta-casein per gram than liquid milk. During cheese aging, bacterial enzymes further hydrolyze casein, potentially releasing additional casomorphin fragments. The combination of higher casein concentration and prolonged enzymatic activity makes aged cheese the most concentrated dietary source of casomorphin precursors.
Human breast milk also contains beta-casein and produces casomorphins during infant digestion. Teschemacher (1997) noted that human beta-casomorphins differ in sequence from their bovine counterparts and generally display lower opioid potency. The evolutionary persistence of casomorphin-releasing sequences in mammalian milk proteins across species suggests a conserved biological function rather than a coincidental byproduct of digestion. What that function might be, whether related to infant gut maturation, feeding behavior regulation, or mother-infant bonding through opioid-mediated reward, remains speculative but biologically plausible.[2]
Opioid receptor binding
Receptor Binding Hierarchy
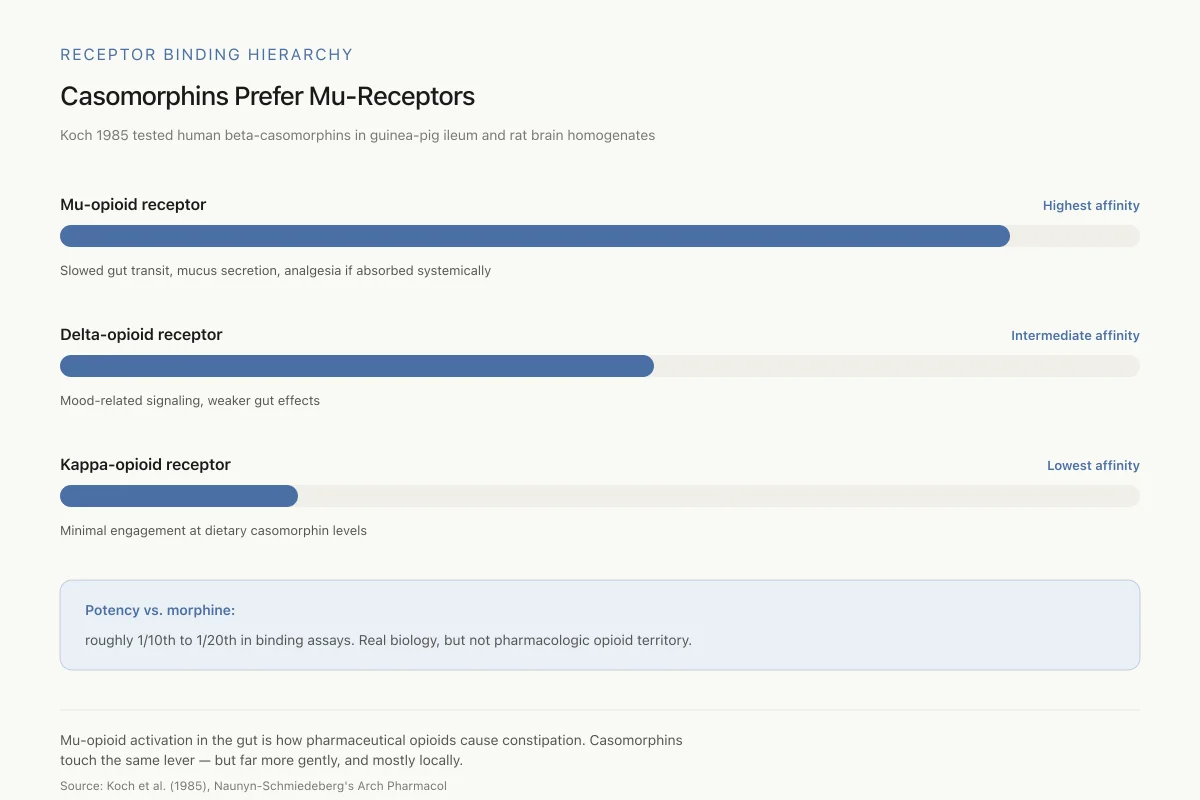
Casomorphins Prefer Mu-Receptors
Koch 1985 tested human beta-casomorphins in guinea-pig ileum and rat brain homogenates. Clear receptor preference hierarchy.
Slowed gut transit, mucus secretion, analgesia if absorbed
Mood-related signaling, weaker gut effects
Minimal engagement at dietary casomorphin levels
Potency vs. morphine: roughly 1/10th to 1/20th in binding assays. Real biology, but not pharmacologic opioid territory.
Source: Koch et al. (1985), Naunyn-Schmiedeberg’s Arch Pharmacol
 View as image
View as imageKoch et al. (1985) published the foundational opioid binding study for human beta-casomorphins. Using guinea-pig ileum preparations and rat brain homogenates, they measured the binding parameters of human BCM-4, BCM-5, BCM-7, and BCM-8 alongside their bovine counterparts. All beta-casomorphins examined displayed opioid activity, with a clear receptor preference hierarchy: highest affinity for mu-receptors, intermediate for delta-receptors, lowest for kappa-receptors.[1]
The mu-receptor preference is significant because mu-opioid receptors mediate the classic opioid effects: analgesia, euphoria, respiratory depression, and gut motility reduction. In the gastrointestinal tract, mu-receptor activation slows peristalsis, increases water absorption, and stimulates mucus secretion. These are the same mechanisms by which pharmaceutical opioids cause constipation.
Holzer (2009) reviewed the distribution and function of opioid receptors throughout the gastrointestinal tract. Mu-, kappa-, and delta-opioid receptors are expressed on enteric neurons, smooth muscle cells, and immune cells within the gut wall. Endogenous opioid peptides (met-enkephalin, leu-enkephalin, beta-endorphin, dynorphin) are produced locally by both neurons and endocrine cells in the gut.[7] Casomorphins entering the gut lumen during digestion therefore encounter a receptor system already actively engaged in regulating gut function. The question is whether dietary casomorphins contribute meaningfully to this signaling at the concentrations produced during normal digestion.
Effects in the gut
The best-documented biological effect of casomorphins is on intestinal function, where the peptides act locally before any absorption into the bloodstream is required.
Trompette et al. (2003) demonstrated that BCM-7 and shorter beta-casomorphins induced mucus discharge in isolated rat jejunum segments. The effect was dose-dependent at micromolar concentrations and was blocked by naloxone, confirming mu-opioid receptor mediation. An enzymatic casein hydrolysate (simulating digestion products) produced the same mucus-stimulating effect, suggesting that casomorphin concentrations achievable through normal casein digestion are sufficient to trigger this response. BCM-4-amide (a tetrapeptide variant) was the most potent mucus stimulant among the fragments tested.[5]
Shah (2000) reviewed the broader landscape of milk-derived bioactive peptides and noted that casomorphins' gastrointestinal effects include slowed intestinal transit, increased water and electrolyte absorption, and modulation of amino acid transport. These effects are consistent with mu-opioid receptor activation in the enteric nervous system and could explain the constipating effect some individuals report with dairy consumption.[4]
Effects beyond the gut: the absorption question
The central debate about casomorphins concerns whether they cross the intestinal barrier in quantities sufficient to produce systemic effects. Opioid receptor activation in the gut can produce local effects without absorption. Systemic and neurological effects would require intact casomorphin peptides to survive intestinal peptidases, cross the epithelial barrier, survive hepatic first-pass metabolism, and reach target tissues.
Woodford (2021) reviewed the evidence for systemic casomorphin effects and identified several mediating factors. Intestinal permeability varies between individuals and is influenced by inflammation, infection, age (neonates have higher permeability), and genetic differences. Dipeptidyl peptidase-4 (DPP4), an enzyme that degrades proline-containing peptides like casomorphins, shows variable activity across individuals. Low DPP4 activity could theoretically allow greater intact casomorphin absorption.[9]
De et al. (2023) reviewed the difficulties in establishing adverse effects of BCM-7 and concluded that the topic "remains controversial" primarily because most studies were performed in vitro or in animal models. A threshold of BCM-7 exposure sufficient to trigger physiological responses at gastrointestinal and systemic levels has not been established. The proportion of the population that may be more susceptible to BCM-7 effects, potentially those with increased intestinal permeability or reduced DPP4 activity, remains undefined.[10]
Neurological activity in cell models
The most striking in vitro finding comes from Sakaguchi et al. (1998), who demonstrated that beta-casomorphin-5 stimulated neurite outgrowth in mouse neuroblastoma cells (Neuro-2a) at picomolar concentrations. The effect was naloxone-reversible, confirming opioid receptor mediation. For comparison, the selective mu-agonist DAMGO produced similar effects only at micromolar concentrations, making BCM-5 approximately 1000-fold more potent than DAMGO for this specific cellular response.[3]
This finding is notable because it demonstrates biological activity at concentrations far below what would be needed for classical opioid pharmacology. It also suggests that casomorphins may have neurotrophic properties distinct from their opioid receptor-mediated effects. Whether this in vitro neurite outgrowth effect occurs in intact nervous tissue exposed to physiological casomorphin concentrations is unknown.
The A1 milk hypothesis and health claims
The observation that A1 beta-casein releases more BCM-7 than A2 beta-casein spawned what became known as the "A1/A2 milk hypothesis": that consumption of A1 milk (and its BCM-7 byproduct) contributes to various diseases, while A2 milk is safer.
Kaminski et al. (2007) reviewed the epidemiological and preclinical evidence. Population-level correlations between A1 beta-casein consumption and the incidence of type 1 diabetes and cardiovascular disease were reported in ecological studies. A 1999 retrospective analysis found a "strong correlation" between national casein A1 consumption levels and insulin-dependent diabetes incidence.[6]
These ecological correlations have significant limitations. They are population-level associations that cannot demonstrate causation, and they fail to control for the many confounding variables that differ between countries with different dairy consumption patterns. The European Food Safety Authority evaluated the clinical evidence in 2009 and found no established causal relationship between oral BCM-7 intake and non-communicable diseases including type 1 diabetes, cardiovascular disease, or autism.
De et al. (2023) updated this assessment and found the situation largely unchanged: "plausible indications" of a relationship between A1 beta-casein/BCM-7 and adverse health effects exist, but in vivo studies remain insufficient to confirm or refute the hypothesis.[10]
Gastrointestinal symptoms
The strongest clinical evidence for A1/A2 differences involves digestive discomfort rather than chronic disease. A 2016 crossover study (Jianqin et al.) found that milk containing only A2 beta-casein produced less gastrointestinal inflammation, softer stools, and reduced abdominal pain compared to A1-containing milk in a cohort of Chinese adults. Some researchers have proposed that individuals who self-diagnose as "lactose intolerant" may actually be reacting to BCM-7 from A1 beta-casein rather than to lactose itself. This hypothesis has clinical plausibility but has not been validated in diagnostic studies comparing the two mechanisms.
Casomorphins and immune function
Woodford (2021) reviewed evidence suggesting that casomorphins interact with immune cells independently of classical opioid receptor signaling. Mu-opioid receptors are expressed on macrophages, lymphocytes, and other immune cells throughout the gastrointestinal tract. In vitro studies have demonstrated that BCM-7 modulates cytokine production, with several reports showing proinflammatory effects through Th2 pathway activation in mouse gut models.[9]
The immune effects are relevant to the gut because the intestinal mucosa contains the body's largest concentration of immune tissue (gut-associated lymphoid tissue, or GALT). Opioid receptor activation on immune cells in this environment could theoretically influence local inflammatory responses, mucosal immunity, and tolerance to dietary antigens. Some researchers have proposed that BCM-7-mediated immune modulation could contribute to gastrointestinal inflammation in susceptible individuals, connecting the casomorphin hypothesis to conditions like inflammatory bowel disease and food sensitivity.
These immune effects remain largely documented in cell culture and animal models. The concentrations of BCM-7 used in immune cell studies often exceed what would be expected from normal dairy digestion, making physiological relevance uncertain. Whether immune modulation by casomorphins contributes to the gastrointestinal symptoms reported by some dairy consumers, or whether these symptoms are better explained by lactose intolerance or other mechanisms, has not been resolved through controlled clinical investigation.
The "cheese addiction" narrative
Popular media frequently claims that cheese is "addictive" because of casomorphins. This narrative typically invokes a 2015 University of Michigan study (Schulte et al.) that identified cheese as among the most "addictive" foods in a survey. The study used self-reported "food addiction" measures and found that processed foods with high fat and refined carbohydrate content scored highest, with cheese among them.
The study did not measure casomorphins, opioid receptor binding, or any neurobiological parameter. The association between cheese and self-reported addictive eating behavior more likely reflects cheese's combination of fat, salt, and umami flavors, the same palatability factors that drive overconsumption of many processed foods.
While casomorphins do bind opioid receptors, their potency is roughly 1/10th to 1/20th that of morphine.[1] Clinical opioid addiction involves mu-receptor activation at levels that produce euphoria, tolerance, and withdrawal. No evidence suggests that dietary casomorphin exposure reaches levels capable of producing these pharmacological effects. The endogenous opioid system, which includes peptides like beta-endorphin that are orders of magnitude more potent, provides a more relevant comparator for understanding opioid-mediated reward from food.
Teschemacher (1997) proposed the more measured term "food hormones" for casomorphins, suggesting a regulatory role in gut-brain signaling that is distinct from the addictive pharmacology of clinical opioids.[2]
Casomorphins in the broader dairy peptide landscape
Casomorphins are just one family within a large and varied group of bioactive peptides generated during milk protein digestion. Shah (2000) catalogued the major categories: opioid peptides (casomorphins, lactorphins), antihypertensive peptides (ACE-inhibitory fragments from casein), casein phosphopeptides (mineral-binding peptides), and antimicrobial peptides (lactoferricin from lactoferrin). Each category involves distinct enzymatic release patterns, receptor targets, and physiological effects.[4]
The lactotripeptides Ile-Pro-Pro and Val-Pro-Pro, for example, inhibit angiotensin-converting enzyme and have been studied in human blood pressure trials, making them some of the best-characterized bioactive milk peptides. Their clinical evidence base far exceeds that of casomorphins, illustrating the difference between peptides that have been tested in controlled human studies and those that remain primarily characterized through in vitro and animal work.
Boutrou et al. (2013) confirmed that multiple bioactive peptide families are generated simultaneously during casein digestion in the human jejunum, suggesting that the biological effects of dairy consumption reflect the combined activity of dozens of peptide fragments rather than any single molecule.[8] Isolating the specific contribution of casomorphins to dairy's overall health effects is therefore inherently difficult. The same protein hydrolysis that releases BCM-7 also releases antioxidant peptides, ACE-inhibitory peptides, and immunomodulatory fragments.
Understanding casomorphins in isolation, divorced from the other peptides co-released during digestion, may produce a distorted picture of their net biological impact. This is a systemic limitation in food-derived peptide research: the reductionist approach of studying individual peptides in purified form cannot fully capture the complex signaling environment of actual food digestion.
Safety
LowInfant dairy exposure
Concern
Newborns have higher intestinal permeability and lower DPP4 enzyme activity than adults. This could allow more intact casomorphins to cross the gut wall — from breast milk, cow's milk formula, or dairy introduction.
What the research says
No controlled evidence shows harm. Teschemacher proposed infant casomorphin exposure may be a normal part of gut maturation and mother-infant signaling. The concern is theoretical, not demonstrated.
Particularly relevant for: Infants, especially premature or very young
What to do
Follow standard pediatric dairy-introduction guidance. Don't avoid breast milk based on this concern — the signal isn't strong enough.
Woodford 2021; Teschemacher 1997
Outstanding questions and limitations
Four decades after their discovery, casomorphins remain poorly understood relative to their public profile. Several critical questions remain unresolved.
Absorption and bioavailability. Whether intact BCM-7 crosses the human intestinal barrier at physiologically relevant concentrations has not been definitively demonstrated. The proline-rich structure of casomorphins confers some resistance to peptidases, but quantitative absorption data in healthy adults is lacking.
Dose-response in humans. No clinical study has established a dose-response relationship between casomorphin intake and any measurable health outcome. Without this data, it is impossible to determine whether the amounts released during normal dairy consumption produce any effect beyond local gut signaling.
Individual variability. DPP4 activity, intestinal permeability, gut microbiome composition, and genetic polymorphisms likely create substantial between-individual variation in casomorphin exposure and response. Population-level studies may obscure effects that are significant in susceptible subgroups.
Infant vulnerability. Neonates have higher intestinal permeability and lower DPP4 activity than adults, potentially increasing their exposure to intact casomorphins from breast milk or cow's milk formula. Whether this has developmental implications, positive or negative, remains an open research question.
Cheese concentration. While cheese concentrates casein and its digestion extends over hours of bacterial enzymatic activity, the actual casomorphin content of different cheese varieties has been measured in only a small number of studies. Whether aged cheeses produce meaningfully different casomorphin exposures compared to fresh dairy products has not been systematically characterized.
The field suffers from a gap between the molecular biology (well-established receptor binding data) and clinical relevance (almost no controlled human outcome data). This gap allows both overclaiming (cheese is addictive, A1 milk causes disease) and underclaiming (casomorphins have no biological relevance) to persist without adequate evidence for either position.
The Bottom Line
Casomorphins are opioid peptides released during the digestion of milk casein. Beta-casomorphin-7 binds mu-opioid receptors at roughly 1/10th to 1/20th the potency of morphine, with documented effects on gut mucus secretion and motility in animal models. The A1 variant of beta-casein releases approximately 4-fold more BCM-7 than A2 during digestion. Whether dietary casomorphin exposure produces clinically meaningful effects in humans beyond local gut signaling remains unestablished after four decades of research.
Sources & References
- 1RPEP-00028·Koch, G et al. (1985). “Opioid activities of human beta-casomorphins..” Naunyn-Schmiedeberg's archives of pharmacology.Study breakdown →PubMed →↩
- 2RPEP-00433·Teschemacher, H et al. (1997). “Milk protein-derived opioid receptor ligands..” Biopolymers.Study breakdown →PubMed →↩
- 3RPEP-00489·Sakaguchi, M et al. (1998). “A Milk-Derived Opioid Peptide Promotes Nerve Cell Growth.” Neuroscience letters.Study breakdown →PubMed →↩
- 4RPEP-00618·Shah, N P (2000). “The Hidden Pharmacy in Milk: Opioid, Blood Pressure, and Mineral-Boosting Peptides.” The British journal of nutrition.Study breakdown →PubMed →↩
- 5RPEP-00865·Trompette, Aurélien et al. (2003). “Milk-Derived Beta-Casomorphin Triggers Protective Mucus Release in the Gut.” The Journal of nutrition.Study breakdown →PubMed →↩
- 6RPEP-01249·Kamiński, Stanisław et al. (2007). “A1 vs A2 Milk: How Beta-Casein Type Affects Opioid Peptide Release and Human Health.” Journal of applied genetics.Study breakdown →PubMed →↩
- 7RPEP-01492·Holzer, Peter (2009). “Opioid Receptors in the GI Tract: The Complete Guide to Gut Opioid Pharmacology.” Regulatory peptides.Study breakdown →PubMed →↩
- 8RPEP-02135·Boutrou, Rachel et al. (2013). “Your Gut Releases Opioid and Blood Pressure-Lowering Peptides When You Digest Milk Protein.” The American journal of clinical nutrition.Study breakdown →PubMed →↩
- 9RPEP-05876·Woodford, Keith Bernard (2021). “Casomorphins and Gliadorphins Have Diverse Systemic Effects Spanning Gut, Brain and Internal Organs..” International journal of environmental research and public health.Study breakdown →PubMed →↩
- 10RPEP-06831·de Vasconcelos, Marta Liliane et al. (2023). “Difficulties in Establishing the Adverse Effects of β-Casomorphin-7 Released from β-Casein Variants-A Review..” Foods (Basel.Study breakdown →PubMed →↩