Antioxidant Peptides from Food: What the Science Shows
Bioactive Peptides in Food Science
2-10 Amino Acids
Most food-derived antioxidant peptides are 2 to 10 amino acids long, under 1 kDa, and rich in hydrophobic residues like proline, valine, and tryptophan. They scavenge free radicals, chelate pro-oxidant metals, and upregulate the body's own antioxidant enzymes.
Zou et al., Molecules, 2016
Zou et al., Molecules, 2016
If you only read one thing
When you eat protein, your digestion chops it into tiny fragments. Some of those fragments act as antioxidants — they neutralize free radicals, pull iron and copper out of the blood before they cause damage, and even tell your body to make more of its own antioxidant enzymes. Research labs have found thousands of active sequences in fish skin, chia, lentils, mushrooms, and even insects. The big catch: most of these peptides get chewed up before they reach your bloodstream, so a lab result saying 'this peptide scavenges radicals at 0.3 mg/mL' doesn't automatically mean eating that food prevents disease. No human clinical trial has yet proven that food-derived antioxidant peptides prevent or treat any disease.
Every protein you eat is a potential source of antioxidant peptides. When the enzymes of your digestive tract, or the proteases used in food processing, cleave food proteins into shorter fragments, some of those fragments can scavenge free radicals, chelate pro-oxidant metal ions, and activate the body's own antioxidant defense systems. These food-derived antioxidant peptides have been isolated from milk, fish, soybeans, eggs, cereals, insects, mushrooms, and dozens of other protein sources. The research is extensive: a 2016 structure-activity review catalogued antioxidant peptides from virtually every major food protein family, identifying common structural features that predict activity.[1]
The interest is practical as well as scientific. The global bioactive peptide market reached an estimated $2.76 billion in 2025, with antioxidant and anti-hypertensive peptides driving demand for functional food ingredients. But the gap between laboratory antioxidant assays and proven health benefits in humans remains wide. This article examines what the research actually shows: which food sources produce the most active antioxidant peptides, how those peptides work at the molecular level, what reaches the bloodstream after digestion, and where the evidence falls short.
For how antioxidant peptides overlap with blood pressure-lowering effects, see ACE-Inhibitory Peptides in Food: Natural Blood Pressure Management. For the broader landscape of bioactive peptides as functional food ingredients, see Bioactive Peptides as Functional Food Ingredients: From Lab to Shelf.
Key Takeaways
- Every protein you eat is a source of potential antioxidant peptides. Digestion chops food proteins into tiny fragments, and some of those fight free radicals.
- Food-derived antioxidants work through three different strategies: neutralizing free radicals, grabbing dangerous metals, and telling your cells to make more of their own antioxidant enzymes.
- These peptides have been found in fish skin, chia seeds, lentils, mushrooms, soybeans, eggs, and even insects — basically every protein-rich food.
- The big catch: most peptides get chewed up by digestion before they reach your bloodstream. What works in a test tube may not work in you.
- No human clinical trial has yet proven that a food-derived antioxidant peptide prevents or treats any disease. The claims outrun the evidence.
- The market has exploded anyway — worth an estimated $2.76 billion in 2025 — driven by wellness marketing ahead of the actual science.
- Food waste is quietly becoming a peptide goldmine. Fish processing scraps, for instance, yield some of the strongest antioxidant sequences studied.
What Makes a Peptide an Antioxidant?
Oxidative stress occurs when reactive oxygen species (ROS), including superoxide anion, hydroxyl radical, and hydrogen peroxide, overwhelm the body's antioxidant defenses. ROS damage lipids, proteins, and DNA, contributing to aging, cardiovascular disease, neurodegeneration, and cancer. The body maintains endogenous antioxidant enzymes (superoxide dismutase, catalase, glutathione peroxidase) and small-molecule antioxidants (glutathione, vitamin C, vitamin E), but these systems can be overwhelmed by inflammation, UV exposure, pollution, or metabolic stress.
Three Modes of Protection
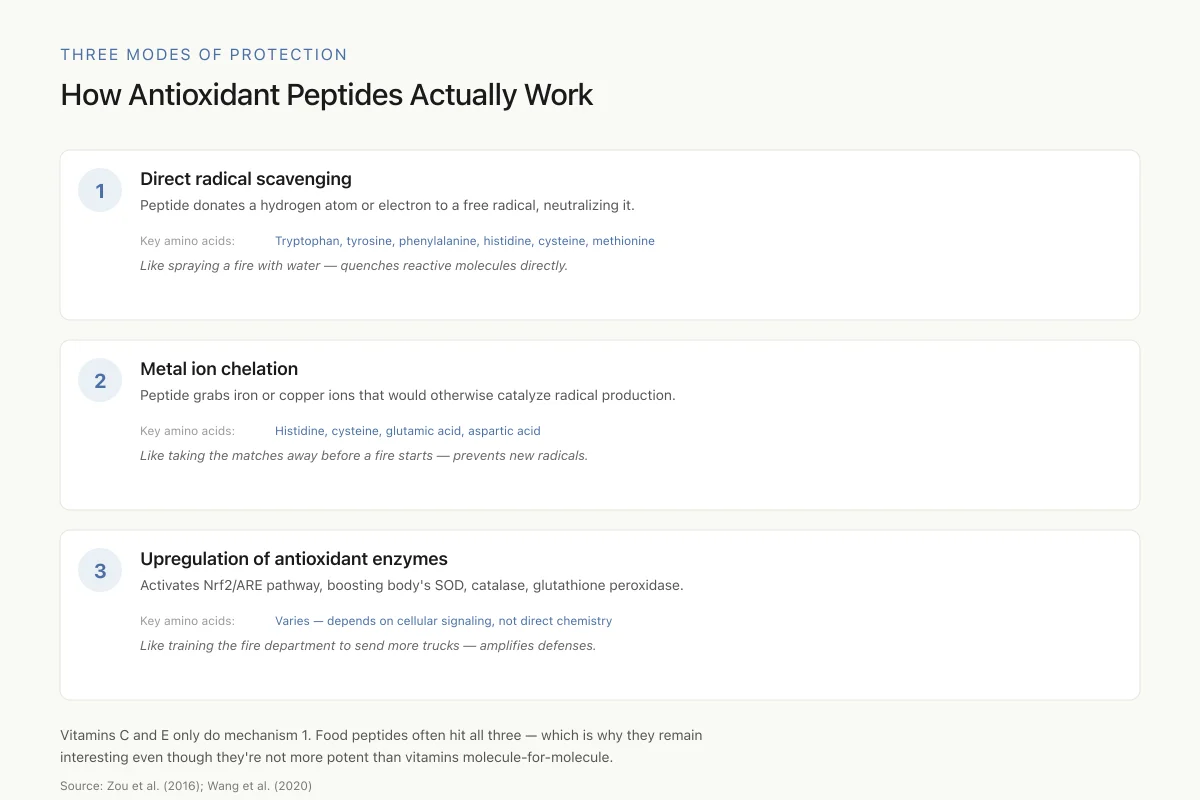
How Antioxidant Peptides Actually Work
Most dietary "antioxidants" (vitamins C, E) only do mechanism 1 — direct scavenging. Food-derived peptides often hit all three at once, which is why they remain interesting even though they're not more potent than vitamins molecule-for-molecule.
Source: Zou et al. (2016), Molecules; Wang et al. (2020), Marine Drugs
 View as image
View as imageFood-derived antioxidant peptides act through three distinct mechanisms, often simultaneously.[1]
Direct Radical Scavenging
Peptides donate hydrogen atoms or electrons to free radicals, converting them into stable, non-reactive molecules. The amino acids tyrosine, tryptophan, and phenylalanine are particularly effective donors because their aromatic ring structures can absorb the unpaired electron and stabilize it through resonance. Histidine contributes through its imidazole ring, which can both donate protons and chelate metals. Cysteine donates hydrogen from its thiol group. Methionine's sulfur atom can be oxidized to methionine sulfoxide, neutralizing radicals in the process.
Metal Ion Chelation
Transition metals like iron (Fe2+) and copper (Cu+) catalyze the Fenton reaction, which converts hydrogen peroxide into hydroxyl radicals, the most damaging ROS. Peptides containing histidine, cysteine, glutamic acid, or aspartic acid can chelate these metal ions, removing them from the reaction and preventing radical generation. This mechanism is distinct from direct scavenging: the peptide does not neutralize existing radicals but prevents new ones from forming.
Upregulation of Endogenous Antioxidant Enzymes
Some antioxidant peptides activate the Nrf2/ARE (nuclear factor erythroid 2-related factor 2/antioxidant response element) signaling pathway, increasing expression of superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px). This mechanism amplifies the body's own defenses rather than relying on the peptide itself to neutralize every radical. Collagen peptides from redlip croaker fish scales demonstrated this effect: treated liver cells showed increased SOD, CAT, and GSH-Px activity alongside reduced ROS and malondialdehyde (MDA, a lipid peroxidation marker).[2]
Structure-Activity Relationships
Not every protein fragment is an antioxidant peptide. The structural features that determine activity are well characterized.[1]
Size matters. Most active antioxidant peptides contain 2 to 10 amino acids with molecular weights below 1 kDa. Smaller peptides penetrate cell membranes more easily and are more likely to survive gastrointestinal digestion intact. Peptides under 3 kDa show enhanced intestinal epithelial membrane permeability via enterocytes, tight junctions, and passive diffusion.
Hydrophobicity is critical. Hydrophobic amino acids (leucine, valine, isoleucine, proline, alanine) at the N-terminus or within the sequence enhance radical scavenging by facilitating electron transfer. Proline's rigid pyrrolidine ring creates conformational constraints that can position other residues optimally for radical interaction.
Aromatic residues provide electron density. Tryptophan, tyrosine, and phenylalanine can stabilize free radicals through their aromatic ring systems. Tryptophan's indole ring is particularly effective because it offers multiple sites for electron donation and radical stabilization.
Sequence, not just composition. Two peptides with identical amino acid compositions but different sequences can have dramatically different antioxidant activities. The position of key residues relative to the N- and C-terminus, and the spatial arrangement of side chains, determines how effectively the peptide interacts with radicals or metal ions. This is why identifying active sequences requires empirical testing, not just compositional analysis.
Food Sources of Antioxidant Peptides
Validated Sources
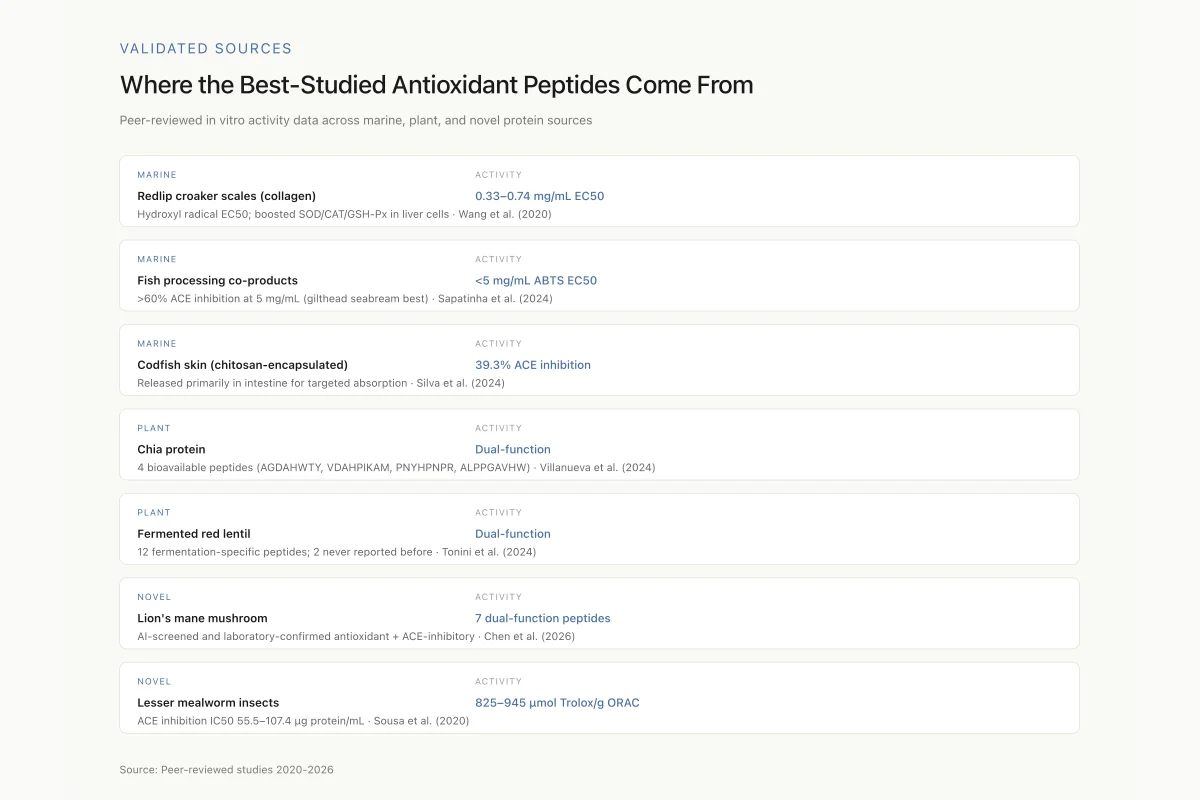
Where the Best-Studied Antioxidant Peptides Come From
Peer-reviewed in vitro activity data across marine, plant, and novel protein sources
Nothing on this list is a supplement you can walk into a pharmacy and buy. These are isolated, purified peptide fractions from research labs — the foods themselves may release some of these peptides during digestion, but much less consistently than isolated hydrolysates deliver.
Source: Peer-reviewed studies listed above (2020-2026)
 View as image
View as imageMarine Sources
Fish and shellfish are among the most productive sources of antioxidant peptides, partly because their proteins (especially collagen) are abundant and partly because enormous quantities of processing waste (heads, bones, skin, viscera) are available for hydrolysis.
Wang et al. isolated six antioxidant peptides (molecular weights 526-886 Da) from collagen hydrolysates of redlip croaker scales using neutrase, achieving 21.4% degree of hydrolysis.[2] The peptides showed EC50 values of 0.33-0.74 mg/mL for hydroxyl radical scavenging and protected HepG2 liver cells from hydrogen peroxide-induced oxidative damage, reducing intracellular ROS and MDA while increasing SOD, CAT, and GSH-Px enzyme activity.
Sapatinha et al. demonstrated that combining Alcalase and Protana enzymes in a single-stage hydrolysis of frozen fish processing co-products from three species (gilthead seabream, meagre, and sole) achieved 80% protein yield and 34-49% degree of hydrolysis.[3] The resulting hydrolysates showed ABTS antioxidant activity with EC50 below 5 mg/mL and over 60% ACE inhibition at 5 mg/mL. Gilthead seabream co-products produced the most promising bioactive peptides.
Silva et al. extracted collagen peptides from codfish skin using a novel deep eutectic solvent, producing hydrolysates with antioxidant activity and 39.3% ACE inhibition.[4] When encapsulated in chitosan capsules for gastrointestinal delivery, the peptides released primarily in the intestine, optimizing absorption. This study bridges two challenges simultaneously: extracting bioactive peptides using green chemistry and delivering them to the site of absorption intact. For more on how encapsulation technologies protect peptide bioactivity during digestion, see Peptide Encapsulation in Food Technology: Protecting Bioactivity Through Digestion.
Plant Sources
Plant proteins are increasingly attractive as antioxidant peptide sources because of sustainability, lower cost, and consumer preference for plant-based products.
Villanueva et al. hydrolyzed chia protein with Alcalase and identified the bioavailable peptidome using a Caco-2 cell transwell absorption model, confirming which peptides actually cross the intestinal barrier.[5] Twenty unique peptides under 1 kDa were characterized, and in silico analysis predicted that AGDAHWTY, VDAHPIKAM, PNYHPNPR, and ALPPGAVHW contribute to both antioxidant and ACE-inhibitory activity. The histidine-rich sequences (AGDAHWTY, VDAHPIKAM) are consistent with the known role of histidine's imidazole ring in metal chelation and radical scavenging.
Tonini et al. showed that fermentation of red lentil protein isolate with the yeast Hanseniaspora uvarum SY1 produced the highest abundance of bioactive peptides among all microorganisms tested.[6] From 2,039 peptide sequences identified in the low-molecular-weight fraction, 36 matched known bioactive peptides. Twelve peptides were fermentation-specific (not present in raw lentil protein), and two sequences (LVR and LVL) had never been reported in any fermented substrate. The tripeptide KVI, previously observed only in dried bonito, appeared in the fermented lentil product. This demonstrates that microbial fermentation does not merely break down existing proteins: it creates novel peptide sequences through specific enzymatic cleavage patterns.
Novel Sources: Mushrooms and Insects
Chen et al. used an AI-assisted workflow powered by DeepSeek to screen lion's mane mushroom (Hericium erinaceus) for functional peptides.[7] The approach identified 7 peptides with confirmed dual antioxidant and ACE-inhibitory activities, validated by LC-MS/MS and laboratory assays. This study represents one of the first applications of large language models to bioactive peptide discovery, demonstrating that AI tools can accelerate the screening process from months to days.
Sousa et al. hydrolyzed protein from the lesser mealworm (Alphitobius diaperinus) with Alcalase and Corolase PP, producing hydrolysates with antioxidant activity values of 95.0-95.7 micromol Trolox equivalent per gram by the ABTS method and 825.6-944.8 micromol Trolox equivalent per gram by the ORAC method.[8] ACE inhibition IC50 values were 55.5-107.4 micrograms of protein per mL. Edible insects represent a sustainable, high-protein source for bioactive peptide production, though consumer acceptance remains a barrier in Western markets.
How Antioxidant Peptides Are Produced
The production of food-derived antioxidant peptides follows three main routes.
Enzymatic Hydrolysis
The most common and controllable method. Food proteins are treated with proteolytic enzymes (Alcalase, pepsin, trypsin, papain, neutrase, or combinations) under controlled pH and temperature conditions. The enzyme, substrate concentration, pH, temperature, and hydrolysis duration all influence which peptides are released. Different enzymes cleave at different amino acid positions, producing different peptide profiles from the same starting protein. Alcalase, a broad-specificity endopeptidase, is the most widely used enzyme in antioxidant peptide research because it efficiently cleaves a range of peptide bonds and produces relatively small, bioactive fragments.
Microbial Fermentation
Lactic acid bacteria, yeasts, and other microorganisms secrete proteases during fermentation that break down food proteins into bioactive peptides. This is how traditional fermented foods (yogurt, cheese, miso, tempeh, kefir) naturally contain bioactive peptides. The advantage of fermentation is that it produces peptides as part of a food product, not as an isolated supplement. The disadvantage is less control over which peptides are produced: the microbial community, fermentation time, temperature, and substrate all affect the peptide profile.
Gastrointestinal Digestion
Human digestive enzymes (pepsin in the stomach, trypsin and chymotrypsin in the small intestine) naturally release bioactive peptides from dietary proteins during digestion. This means that eating protein-rich foods is itself a form of antioxidant peptide production. The clinical question is whether the peptides released during normal digestion reach the bloodstream in sufficient concentrations to exert systemic antioxidant effects, a question that in vitro hydrolysis studies cannot answer.
The Bioavailability Problem
The most important limitation in the antioxidant peptide field is the gap between in vitro activity and in vivo efficacy. A peptide that scavenges DPPH radicals in a test tube may never reach the bloodstream in active form.
The Bioavailability Gauntlet
How Much Peptide Actually Reaches Your Bloodstream?
Illustrative retention at each digestive checkpoint (exact values vary by peptide sequence)
A peptide that scores brilliantly on a DPPH radical scavenging assay in a test tube may end up at 3% of its starting concentration by the time it reaches the tissues where oxidative stress actually happens. This is the central gap between "in vitro antioxidant" headlines and real in-body effects.
Source: Villanueva et al. (2024); general oral peptide pharmacokinetics literature
 View as image
View as imageSurvival Through Digestion
Peptides must survive the acidic environment of the stomach (pH 1.5-3.5) and the proteolytic enzymes of the small intestine. Peptides containing proline or hydroxyproline are more resistant to gastrointestinal digestion because proline's cyclic structure makes the peptide bond preceding it resistant to cleavage by most proteases. Shorter peptides (di- and tripeptides) are generally more resistant than longer sequences.
Intestinal Absorption
Peptides must cross the intestinal epithelium to reach the bloodstream. Di- and tripeptides can be transported intact by the PepT1 transporter, a proton-coupled peptide transporter expressed on intestinal enterocytes. Larger peptides may cross via paracellular transport (between cells through tight junctions) or transcytosis, but absorption efficiency decreases sharply with increasing peptide size. The Caco-2 cell model used by Villanueva et al. simulates this barrier and identifies which peptides actually cross.[5]
Plasma Stability
Even peptides that reach the bloodstream face degradation by plasma peptidases. The half-life of a circulating peptide depends on its sequence, length, and susceptibility to specific peptidases. Few studies have measured the plasma concentrations of food-derived antioxidant peptides after oral intake in humans, making it difficult to determine whether physiologically relevant concentrations are achieved.
This chain of obstacles, digestion, absorption, plasma stability, explains why in vitro antioxidant activity does not reliably predict in vivo health effects. The field needs more studies that measure actual plasma peptide levels and downstream biomarkers of oxidative stress in humans after consuming peptide-rich foods, not just DPPH scavenging in a test tube.
Measuring Antioxidant Activity: A Methodological Caveat
The antioxidant peptide literature relies heavily on in vitro chemical assays that measure different aspects of antioxidant capacity. Results from different assays are not directly comparable, and high activity in one assay does not guarantee activity in another.
DPPH assay measures the ability to donate hydrogen atoms to the stable DPPH radical. It is simple and reproducible but uses an artificial radical not found in biological systems.
ABTS assay generates the ABTS cation radical and measures decolorization upon antioxidant addition. It works in both aqueous and lipophilic phases, making it more versatile than DPPH.
ORAC (Oxygen Radical Absorbance Capacity) measures the ability to protect a fluorescent probe from peroxyl radical damage over time. It is considered more physiologically relevant because it uses biologically relevant radicals and measures kinetics, not just endpoints.
Cellular assays (CAA, DCFH-DA) measure the ability to reduce ROS in living cells, which accounts for cellular uptake, metabolism, and compartmentalization. These are closer to biological reality than chemical assays but still far from whole-organism effects.
No single assay captures the full antioxidant potential of a peptide. Studies that report activity using multiple complementary assays (as Wang et al. did with their croaker collagen peptides[2]) provide more reliable characterization than single-assay results.
Dual-Function Peptides: Antioxidant and Beyond
A striking pattern in the research is that many food-derived antioxidant peptides also inhibit angiotensin-converting enzyme (ACE), the enzyme targeted by blood pressure medications like lisinopril and enalapril. This dual functionality is not coincidental: the structural features that enable radical scavenging (hydrophobic residues, aromatic amino acids, histidine) also facilitate binding to the ACE active site.
The chia peptides identified by Villanueva et al. showed predicted dual antioxidant and ACE-inhibitory activity.[5] The lion's mane peptides discovered by Chen et al. had confirmed dual activity.[7] The fish waste hydrolysates from Sapatinha et al. showed both antioxidant and ACE-inhibitory properties.[3] The fermented lentil peptides from Tonini et al. showed the same pattern.[6]
This dual functionality has implications for functional food development. A single peptide fraction from a food source could theoretically address both oxidative stress and hypertension, two interconnected cardiovascular risk factors. For a deeper look at the ACE-inhibitory side of this research, see ACE-Inhibitory Peptides in Food: Natural Blood Pressure Management.
Some food-derived peptides also show antimicrobial activity. The same structural features, hydrophobicity, cationic charge, amphipathic structure, that enable radical scavenging and ACE inhibition can also disrupt bacterial membranes. This multi-functionality has been observed in peptides from fermented milk, insect protein, and marine sources. The relationship between antimicrobial and antioxidant activity in peptides parallels what has been observed in dedicated antimicrobial peptides, though the mechanisms are not identical.
What the Evidence Does Not Show
The antioxidant peptide field has real achievements: thousands of active sequences identified, mechanisms characterized at the molecular level, and multiple food sources validated as peptide production substrates. But several claims commonly made in the literature outpace the evidence.
No food-derived antioxidant peptide has been proven to prevent or treat any disease in humans through randomized controlled trials. The disease-prevention claims (cardiovascular protection, cancer risk reduction, anti-aging) are extrapolated from in vitro antioxidant activity and animal studies, not from human clinical evidence.
In vitro antioxidant activity does not predict in vivo health effects. The history of antioxidant research is littered with compounds that showed strong in vitro activity but failed to produce health benefits (or even caused harm) in human trials. Beta-carotene supplements increased lung cancer risk in smokers despite strong in vitro radical scavenging. This cautionary precedent applies equally to antioxidant peptides.
Bioavailability data are sparse. Most studies stop at demonstrating antioxidant activity in a test tube or cell culture. Few measure whether the active peptides survive digestion, cross the intestinal barrier, and reach target tissues at concentrations sufficient to affect oxidative stress markers. The Caco-2 absorption model used by Villanueva et al. is a step in the right direction but does not replace human pharmacokinetic studies.[5]
Dose-response relationships in humans are largely unknown. How much peptide-rich food or supplement would a person need to consume to achieve a measurable antioxidant effect? This basic question remains unanswered for most food-derived antioxidant peptides.
Where the Field Is Heading
Three developments are reshaping antioxidant peptide research.
First, AI and computational tools are accelerating peptide discovery. The use of deep learning models to predict antioxidant activity from amino acid sequences, combined with quantum chemistry calculations of radical interaction energies, can screen millions of candidate peptides in silico before any laboratory work begins. Chen et al.'s use of a large language model for lion's mane mushroom peptide discovery is an early example of this trend.[7]
Second, encapsulation and delivery technologies are addressing the bioavailability problem. Nanoencapsulation in liposomes, chitosan nanoparticles, and protein-polysaccharide complexes can protect peptides from gastrointestinal degradation and improve intestinal absorption. Silva et al.'s work with chitosan-encapsulated codfish collagen peptides demonstrated targeted intestinal release.[4] For the full picture of how encapsulation technologies are being applied to bioactive peptides, see Peptide Encapsulation in Food Technology: Protecting Bioactivity Through Digestion.
Third, sustainability is driving interest in alternative protein sources. Fish processing waste, insect protein, algae, and agricultural by-products represent underutilized reservoirs of bioactive peptides. Converting these waste streams into high-value functional ingredients addresses both economic and environmental goals.
The transition from laboratory discovery to validated functional food ingredients requires answering the question that most studies still avoid: do these peptides actually reduce oxidative stress markers in humans at the doses achievable through food? Until human clinical trials provide that answer, food-derived antioxidant peptides remain scientifically interesting but clinically unproven.
The Bottom Line
Food-derived antioxidant peptides, typically 2-10 amino acids long and under 1 kDa, scavenge free radicals, chelate pro-oxidant metals, and upregulate endogenous antioxidant enzymes. Active peptides have been isolated from fish collagen, chia, lentils, mushrooms, insects, and dozens of other food proteins using enzymatic hydrolysis, fermentation, or simulated digestion. Structure-activity relationships are well characterized: hydrophobic and aromatic amino acids drive radical scavenging, while histidine and cysteine enable metal chelation. Many antioxidant peptides also inhibit ACE, creating dual-function candidates for cardiovascular health. The principal limitation is bioavailability: few studies confirm that active peptides survive digestion, cross the intestinal barrier, and reach target tissues at effective concentrations in humans. AI-driven screening and encapsulation technologies are advancing the field, but human clinical evidence of health benefits remains scarce.
Sources & References
- 2RPEP-05192·Wang, Wan-Yi et al. (2020). “Antioxidant Peptides From Fish Scale Collagen Protect Liver Cells From Damage.” Marine drugs.Study breakdown →PubMed →↩
- 3RPEP-09207·Sapatinha, Maria et al. (2024). “Fish Waste Yields Bioactive Peptides with Blood Pressure-Lowering and Antioxidant Properties.” Marine drugs.Study breakdown →PubMed →↩
- 4RPEP-09269·Silva, Isa et al. (2024). “Fish Skin Collagen Peptides Show Antioxidant and Blood Pressure-Lowering Properties When Delivered Through Gut-Targeted Capsules.” Food research international (Ottawa.Study breakdown →PubMed →↩
- 5RPEP-09444·Villanueva, Alvaro et al. (2024). “Chia Seeds Contain Peptides That May Lower Blood Pressure and Act as Antioxidants After Digestion.” Journal of agricultural and food chemistry.Study breakdown →PubMed →↩
- 6RPEP-09395·Tonini, Stefano et al. (2024). “Fermented Lentils Produce Bioactive Peptides With Antioxidant and Blood Pressure-Lowering Potential.” Microbial biotechnology.Study breakdown →PubMed →↩
- 7RPEP-14991·Chen, Rongheng et al. (2026). “AI Discovers Peptides in Lion's Mane Mushroom That Lower Blood Pressure and Fight Oxidation.” Food chemistry.Study breakdown →PubMed →↩
- 8RPEP-05146·Sousa, Pedro et al. (2020). “Insect Protein Broken Down Into Peptides Shows Antioxidant and Blood Pressure-Lowering Activity.” Food & function.Study breakdown →PubMed →↩