BPC-157 for Tendon Injuries: What Animal Studies Show
BPC-157 Tendon & Musculoskeletal Healing
35 preclinical studies
A 2025 systematic review found 35 preclinical and just 1 human study on BPC-157 for orthopaedic injuries, revealing a peptide with consistent animal results and almost no clinical data.
Vasireddi et al., HSS Journal, 2025
Vasireddi et al., HSS Journal, 2025
If you only read one thing
BPC-157 is a small peptide that has helped rats heal cut tendons in study after study since the 1990s. Researchers have worked out how it seems to do this — it helps repair cells move, grow, and build new blood vessels. The catch is huge: almost no humans have ever been studied. One survey of 12 people is the entire clinical record. What works beautifully in rats may or may not work in people, and nobody has run the trial to find out.
Tendons heal slowly. An Achilles rupture can sideline an athlete for six months or longer, and the repaired tissue often never matches the original in tensile strength. BPC-157, a 15-amino-acid peptide isolated from human gastric juice, has shown consistent effects on tendon healing across more than two decades of animal research. A 2025 systematic review identified 35 preclinical studies and only a single human survey on BPC-157 in orthopaedic contexts.[1] The results in rats are striking. The evidence in humans is nearly nonexistent. This article examines what the animal data actually demonstrates, how BPC-157 appears to work at the cellular level, and why the gap between rodent models and clinical medicine remains so wide. For a broader overview of this peptide's research across all organ systems, see BPC-157: The Body Protection Compound and What the Research Shows.
Key Takeaways
- BPC-157 restored full Achilles tendon integrity in rats after complete transection, improving load to failure, Young's modulus, and functional index scores across 14 days (Staresinic et al., 2003)
- The peptide promoted tendon fibroblast outgrowth from explants, protected cells from oxidative stress, and stimulated migration via the FAK-paxillin pathway (Chang et al., 2011)
- BPC-157 upregulated growth hormone receptor expression in tendon fibroblasts at both mRNA and protein levels in a dose-dependent manner (Chang et al., 2014)
- In Achilles detachment models, BPC-157 promoted tendon-to-bone healing that did not occur spontaneously in controls, and opposed corticosteroid-induced healing impairment (Krivic et al., 2006)
- A 2025 systematic review of 36 studies found improved structural, biomechanical, and functional outcomes across all preclinical tendon models, but only 1 human study exists (Vasireddi et al., 2025)
- BPC-157 enhanced angiogenesis in healing tendon tissue, with effects correlated to increased VEGF expression (Brcic et al., 2009)
What Is BPC-157?
BPC-157 (Body Protection Compound-157) is a pentadecapeptide with the amino acid sequence GEPPPGKPADDAGLV and a molecular weight of 1,419 Da. It was first identified as a partial sequence of a protein found in human gastric juice. Unlike many growth factors used in tissue engineering, BPC-157 is stable in gastric acid without requiring a carrier molecule, which has made it comparatively straightforward to study in animal models.[2]
The peptide entered clinical trials for inflammatory bowel disease under the designation PL 14736 (Pliva, Croatia), though those trials have not produced published Phase III results. Its musculoskeletal research, which spans tendon, ligament, muscle, and bone models, remains almost entirely preclinical. The peptide's sequence (GEPPPGKPADDAGLV) does not correspond to any known receptor ligand, and its precise binding target or receptor has not been identified, which is one of the lingering questions in BPC-157 pharmacology. For the full regulatory and research landscape, see The Real BPC-157 Story.
Achilles Tendon Transection: The Core Animal Model
The Achilles tendon transection model in rats has been the workhorse of BPC-157 tendon research. In the foundational 2003 study by Staresinic and colleagues, the right Achilles tendon was sharply cut 5 mm proximal to its calcaneal insertion in 72 Wistar rats, creating a large defect between the severed ends. Control animals received saline; treated animals received BPC-157 at doses of 10 microg, 10 ng, or 10 pg per kilogram body weight, injected intraperitoneally once daily starting 30 minutes after surgery.[3]
Assessments at days 1, 4, 7, 10, and 14 showed BPC-157 treated rats had increased load to failure, increased load to failure per area, and higher Young's modulus of elasticity compared to controls. Functionally, treated animals scored higher on the Achilles functional index (AFI), a standardized measure of gait recovery that tracks toe spread and print length. Histologically, BPC-157 treated tendons showed more mononuclear cells (associated with constructive remodeling) and fewer granulocytes (associated with destructive inflammation), along with superior formation of fibroblasts, reticulin, and collagen. By day 14, treated tendons showed reestablishment of full tendon integrity, while controls still had compromised healing.[3]
The same study included an in vitro component: BPC-157 opposed the growth-inhibiting effects of 4-hydroxynonenal (HNE), a lipid peroxidation product that suppresses tendocyte proliferation. HNE is produced during the oxidative stress that accompanies tissue injury, and its growth-inhibiting activity was reversed into growth stimulation when BPC-157 was present. This dual evidence of in vivo repair and direct cellular protection established the template for subsequent BPC-157 tendon research.
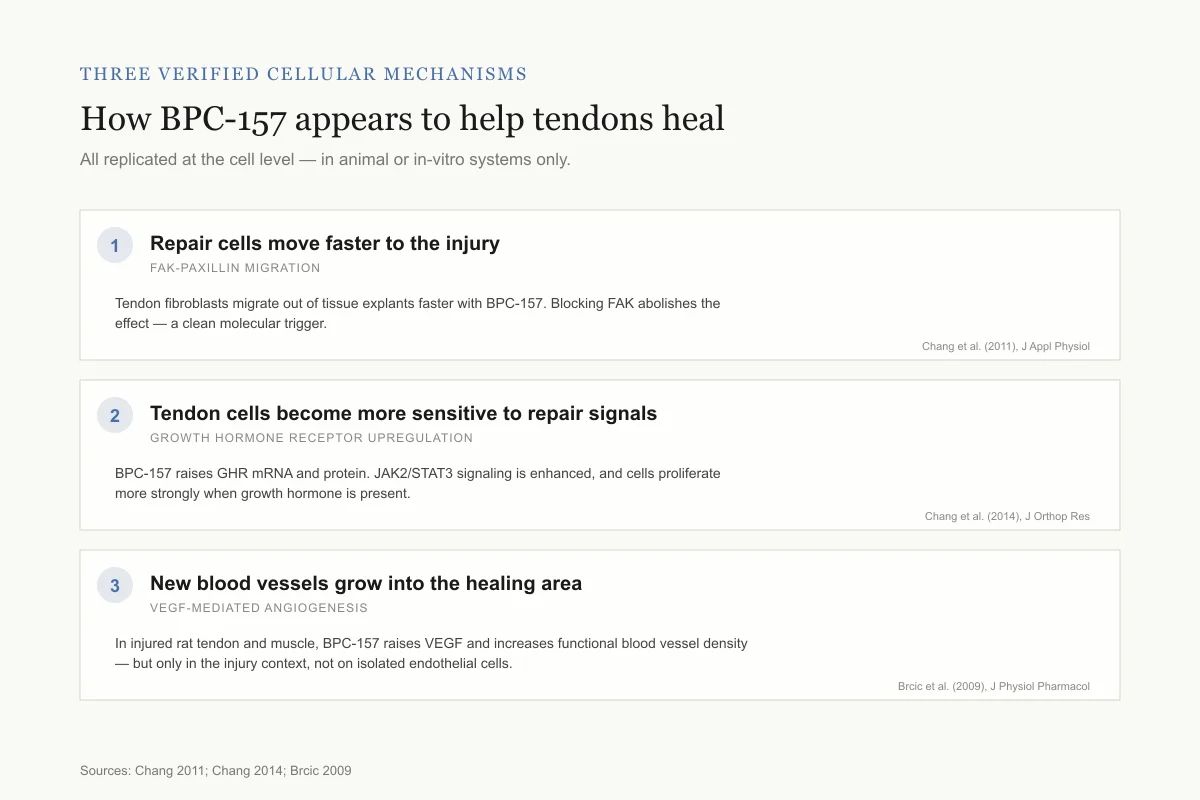
Three verified cellular mechanisms
How BPC-157 appears to help tendons heal
Each mechanism has been replicated in cell-level experiments — all in animal or in-vitro systems.
Repair cells move faster to the injury
FAK-paxillin migration
Tendon fibroblasts migrate out of tissue explants and through transwell membranes at a higher rate with BPC-157. Blocking FAK abolished the effect — a clean molecular trigger.
Tendon cells become more sensitive to repair signals
Growth hormone receptor upregulation
BPC-157 raises growth hormone receptor (GHR) mRNA and protein in tendon fibroblasts. Downstream JAK2/STAT3 signaling is enhanced, and cells then proliferate more strongly when growth hormone is present.
New blood vessels grow into the healing area
VEGF-mediated angiogenesis
In injured rat tendon and muscle, BPC-157 raises VEGF and increases functional blood vessel density. Notably, it does not act directly on endothelial cells in culture — the effect shows up in the injury context.
Plain read: The three mechanisms line up. Cells move to the wound, become more responsive to natural repair signals, and get better blood supply — all observed in rat and cell studies, none confirmed in a human trial.
Sources: Chang 2011; Chang 2014; Brcic 2009
 View as image
View as imageHow BPC-157 Promotes Tendon Healing at the Cellular Level
The 2003 Staresinic study demonstrated that BPC-157 accelerates tendon healing. Two follow-up studies from Chang and colleagues at Taipei Medical University investigated the specific cellular mechanisms responsible.
Tendon Outgrowth, Cell Survival, and Migration
In a 2011 study published in the Journal of Applied Physiology, Chang et al. investigated three distinct cellular processes. First, they cultured rat Achilles tendon explants with BPC-157 and found the peptide accelerated fibroblast outgrowth from the explant edges, meaning repair cells migrated out of the tissue fragment faster than untreated controls. Second, when tendon fibroblasts were exposed to hydrogen peroxide (an oxidative stress that mimics the inflammatory environment of an acute injury), BPC-157 improved cell survival in a dose-dependent manner. Third, in a transwell migration assay, BPC-157 increased tendon fibroblast migration, also dose-dependently.[4]
The migration effect was mediated through the FAK-paxillin signaling pathway. Focal adhesion kinase (FAK) and paxillin are proteins that regulate how cells attach to surfaces, spread, and move. BPC-157 increased the phosphorylation of both proteins without changing their total expression levels and induced F-actin formation, the cytoskeletal rearrangement cells need to physically relocate. When the researchers blocked FAK with a specific inhibitor, BPC-157's pro-migratory effect was abolished. This provided one of the first clear molecular targets for BPC-157's tendon healing action, transforming the evidence from "it works" to "here is one mechanism by which it works." For a deeper exploration of the fibroblast data, see BPC-157's Effect on Fibroblasts and Collagen Synthesis.
Growth Hormone Receptor Upregulation
In 2014, the same research group used cDNA microarray analysis to identify genes affected by BPC-157 in tendon fibroblasts. Growth hormone receptor (GHR) emerged as one of the most upregulated targets. BPC-157 increased GHR expression in a dose- and time-dependent manner at both the mRNA level (measured by RT-PCR) and protein level (measured by Western blot). At the functional level, cells treated with BPC-157 showed increased phosphorylation of JAK2 and STAT3, the downstream signaling molecules of the GH-GHR axis.[5]
When growth hormone was added to BPC-157-pretreated fibroblasts, cell proliferation increased in a dose- and time-dependent manner, confirmed by both MTT assay and PCNA expression. This suggests BPC-157 does not directly drive cell division in tendon fibroblasts but instead sensitizes them to growth hormone signaling by upregulating its receptor. Growth hormone signaling promotes collagen synthesis, stimulates cell proliferation, and drives new extracellular matrix formation. That BPC-157 enhances this pathway in tendon fibroblasts specifically (not just systemically) points to a local, tissue-level mechanism.
Angiogenesis: Building New Blood Supply to Injured Tendons
Tendon healing depends on blood vessel formation (angiogenesis) to deliver oxygen, nutrients, and repair cells to the injury site. Tendons are naturally hypovascular, meaning they have low blood vessel density under normal conditions, which contributes to their notoriously slow repair rates. Brcic and colleagues investigated BPC-157's angiogenic potential in both muscle and tendon healing models in a 2009 study.[6]
The study used immunohistochemistry with VEGF, CD34, and FVIII antibodies to assess blood vessel formation in healing tissue. In rats with transected Achilles tendons and crushed gastrocnemius muscles, BPC-157 treatment was associated with increased expression of vascular endothelial growth factor (VEGF) compared to controls. The increased VEGF translated to more functional blood vessels in the healing tissue, confirmed by vessel density measurements.
An important nuance: BPC-157 had no direct angiogenic effect on endothelial cells in culture. Its pro-angiogenic action appears to operate within the context of injured tissue rather than as a standalone growth factor. This distinction matters because it suggests BPC-157 modulates the healing environment rather than acting as a simple VEGF substitute. A 2018 review by Seiwerth et al. argued that BPC-157 implements its own angiogenic effect across multiple tissue types, distinct from (and in some contexts more consistent than) standard angiogenic growth factors like EGF, FGF, and VEGF administered exogenously.[7] For the full analysis of this mechanism, see How BPC-157 Promotes Angiogenesis: New Blood Vessel Formation.
Tendon-to-Bone Healing: The Achilles Detachment Model
One of the most clinically relevant tendon injuries is avulsion, where the tendon detaches from the bone. This creates a healing challenge distinct from mid-substance tendon tears because the repair must regenerate the enthesis, the specialized transitional zone between tendon and bone that includes fibrocartilage layers.
In a 2006 study, Krivic and colleagues sharply transected the rat Achilles tendon from the calcaneal bone and tested BPC-157 against saline controls and methylprednisolone-treated groups. The central finding: tendon-to-bone healing did not occur spontaneously in control animals, but was recovered in BPC-157 treated rats. BPC-157 improved AFI values, increased load to failure and stiffness, produced better collagen fiber organization, and increased collagen type I (the mature structural collagen). The improvements were observed at doses ranging from 10 micrograms to 10 picograms per kilogram.[8]
Critically, methylprednisolone (a corticosteroid commonly used to manage inflammation post-injury) aggravated the healing failure, and BPC-157 opposed this corticosteroid-induced impairment. This finding has practical implications because corticosteroid injections are widely used in tendon injury management despite accumulating evidence that they can compromise tissue repair. For more on BPC-157's effects on bone healing specifically, see BPC-157 and Bone Healing: Fracture Repair Research.
Early Functional Recovery Before Collagen Healing
A follow-up 2008 study from the same group isolated the first four days after Achilles tendon-to-bone transection, the early inflammatory phase before collagen healing begins. BPC-157 improved AFI values at all time points, decreased myeloperoxidase activity (a neutrophil enzyme that serves as an inflammatory marker), reduced histological inflammatory cell infiltration, and increased new blood vessel formation as measured by vascular index.[9]
Methylprednisolone reduced inflammation in the same model but also suppressed new blood vessel formation and did not improve early functional recovery. This distinction is important: BPC-157 appeared to combine anti-inflammatory action with pro-angiogenic activity, while the corticosteroid traded vascular healing capacity for inflammation control. Whether this dual action occurs in humans is untested, but the contrast highlights a potential advantage over standard anti-inflammatory approaches.
Myotendinous Junction Repair
The myotendinous junction (MTJ), where muscle fibers transition into tendon tissue, is a common injury site in sports medicine. Hamstring strains, quadriceps tears, and calf muscle injuries frequently occur at or near the MTJ. Japjec et al. (2021) tested BPC-157 on quadriceps tendon dissection from the quadriceps muscle in rats, a model where the MTJ defect does not heal spontaneously.[10]
BPC-157 was administered both intraperitoneally (10 microg/kg and 10 ng/kg) and orally in drinking water (0.16 microg/mL). Results were assessed at 7, 14, 28, and 42 days post-surgery. Both routes produced improvements in macroscopic and microscopic appearance, biomechanical properties, and functional recovery. Two findings stand out. First, BPC-157 counteracted the progressive muscle atrophy that develops after MTJ disruption, bringing muscle presentation close to normal. Second, by 28 and 42 days, treated rats showed complete defect disappearance with well-oriented recovered tissue and no inflammatory infiltrate.
The study also measured eNOS and COX-2 mRNA levels, oxidative stress markers, and nitric oxide levels, connecting BPC-157's MTJ healing to its broader effects on the nitric oxide system and inflammatory mediators. The oral administration finding is relevant because it suggests the peptide survives gastrointestinal transit in sufficient quantity to affect distant musculoskeletal tissue, consistent with BPC-157's documented stability in gastric acid. For the muscle-specific data, see BPC-157 Muscle Injury Recovery: Evidence from Preclinical Models.
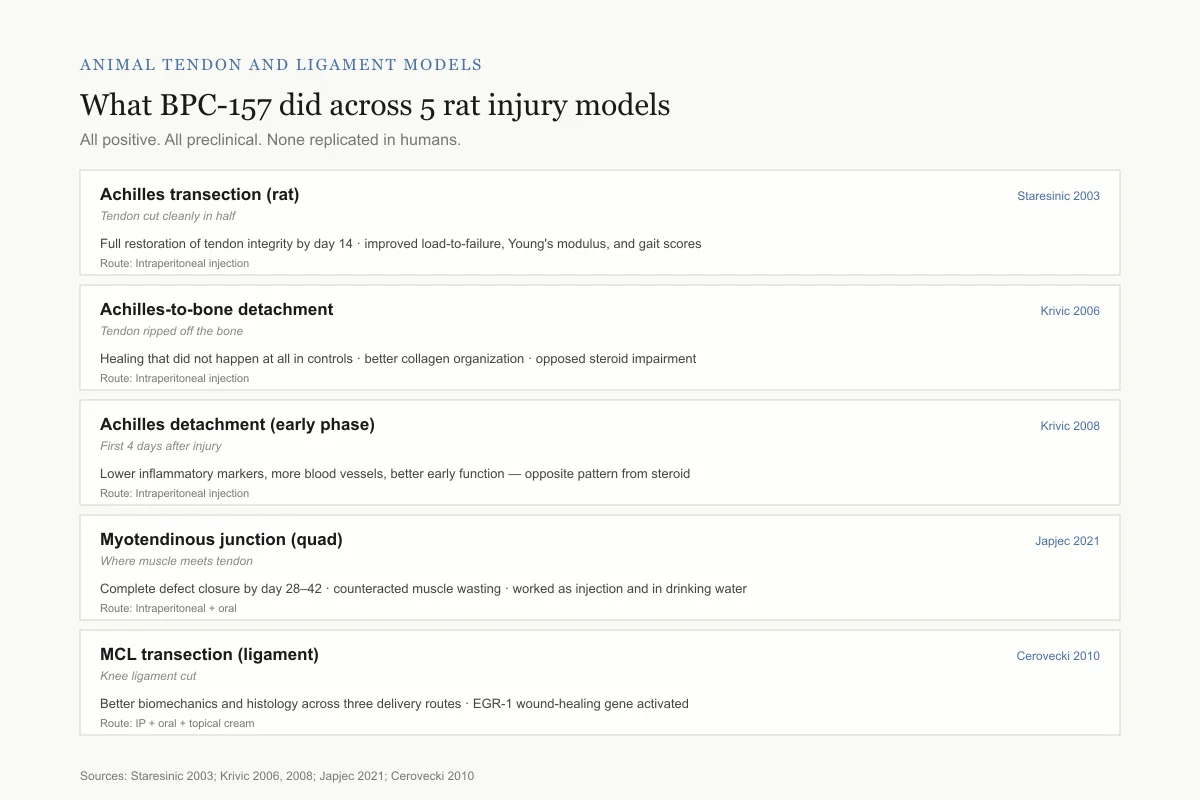
Animal tendon and ligament models
What BPC-157 did across 5 different rat injuries
All positive. All preclinical. None replicated in humans.
Achilles transection (rat)
Staresinic 2003Tendon cut cleanly in half
Full restoration of tendon integrity by day 14 · improved load-to-failure, Young's modulus, and gait scores
Route: Intraperitoneal injection
Achilles-to-bone detachment
Krivic 2006Tendon ripped off the bone
Healing that did not happen at all in controls · better collagen organization, more type I collagen · opposed steroid-induced impairment
Route: Intraperitoneal injection
Achilles detachment (early phase)
Krivic 2008First 4 days after injury
Lower inflammatory markers, more blood vessels, better early function — opposite pattern from steroid
Route: Intraperitoneal injection
Myotendinous junction (quad)
Japjec 2021Where muscle meets tendon
Complete defect closure by day 28–42 · counteracted muscle wasting · worked both as injection and in drinking water
Route: Intraperitoneal + oral
MCL transection (ligament)
Cerovecki 2010Knee ligament cut
Better biomechanics and histology across three delivery routes · EGR-1 wound-healing gene activated
Route: IP + oral + topical cream
The caveat: These are surgical transection models — clean cuts in rat tendons and ligaments. Most human tendon problems are slow overuse injuries and partial tears, which have never been tested.
Sources: Staresinic 2003; Krivic 2006, 2008; Japjec 2021; Cerovecki 2010
 View as image
View as imageFrom Tendons to Ligaments: Broadening the Connective Tissue Evidence
The Achilles tendon findings prompted investigation of BPC-157 in other connective tissue injuries. Cerovecki et al. (2010) tested the peptide on medial collateral ligament (MCL) transection in rats over 90 days, using three administration routes: intraperitoneal injection, per-oral (in drinking water), and topical (thin cream layer). All three routes were given without a carrier molecule.[11]
All three routes improved biomechanical and histological outcomes of ligament healing. The per-oral route is noteworthy because it adds to the Japjec 2021 MTJ data showing oral bioavailability. The topical route raises questions about local delivery for superficial connective tissue injuries. The study also noted increased EGR-1 (early growth response 1) gene expression, linking BPC-157's ligament healing to wound-healing gene activation pathways. For the full ligament data, see BPC-157 and Ligament Healing: Research on Connective Tissue Repair.
A 2019 review by Gwyer et al. in Cell and Tissue Research synthesized the musculoskeletal soft tissue evidence across tendon, ligament, and muscle models. The review highlighted BPC-157's practical advantage over exogenous growth factor approaches: stability without carriers, effectiveness across multiple administration routes, and consistent positive results across tissue types.[12] However, the same review noted that "only a handful of research groups have performed in-depth studies regarding this peptide," identifying the concentration of research as a limitation.
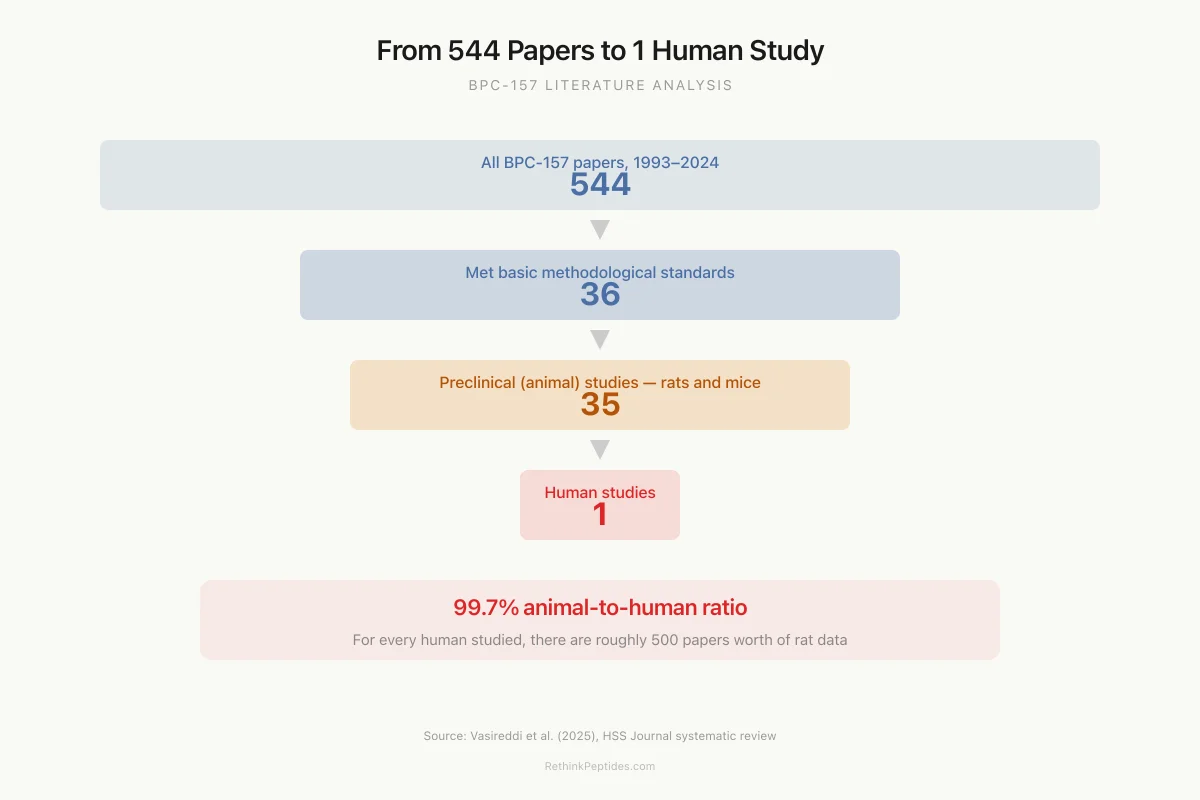
Literature Analysis
From 544 Papers to 1 Human Study
That is a 99.7% animal-to-human ratio. For every human who has been studied, there are roughly 500 papers worth of rat data.
Source: Vasireddi et al. (2025), HSS Journal
 View as image
View as imageThe Evidence Hierarchy: Where Tendon Research Actually Stands
The 2025 systematic review by Vasireddi and colleagues at the Hospital for Special Surgery provides the most current and rigorous assessment. Their analysis included 36 studies (35 preclinical, 1 human survey). Across eight tendon and ligament transection models, BPC-157 was associated with reduced instability and post-injury contracture, restored biomechanics, and restored motor function indices.[1]
The single human data point: 12 patients who received intra-articular BPC-157 injections for unspecified knee pain were surveyed at 6 to 12 months. Seven of the twelve reported subjective improvement in pain lasting more than 6 months. This is not a clinical trial. There was no control group, no blinding, no standardized outcome measure, and no imaging confirmation of structural change.
What the animal evidence demonstrates
- Consistent positive effects on tendon healing across multiple research groups (Zagreb, Taipei)
- Multiple verified cellular mechanisms (FAK-paxillin migration, GHR upregulation, VEGF-mediated angiogenesis)
- Efficacy across different tendon injury models (mid-substance transection, bone detachment, myotendinous junction)
- Maintained efficacy during concurrent corticosteroid treatment
- Dose-dependent effects at extremely low concentrations (10 pg/kg to 10 microg/kg)
- Oral bioavailability in two independent studies (MCL and MTJ models)
What the animal evidence does not demonstrate
- Safety or efficacy in humans at any dose for any tendon condition
- Optimal dosing, route, timing, or duration for human tendon injuries
- Long-term effects on tendon mechanical properties beyond 90 days
- Superiority to existing treatments (platelet-rich plasma, surgical repair, physical therapy)
- Whether complete transection models translate to the partial tears and tendinopathies that comprise most human tendon injuries
- Independent replication by research groups outside Zagreb and Taipei
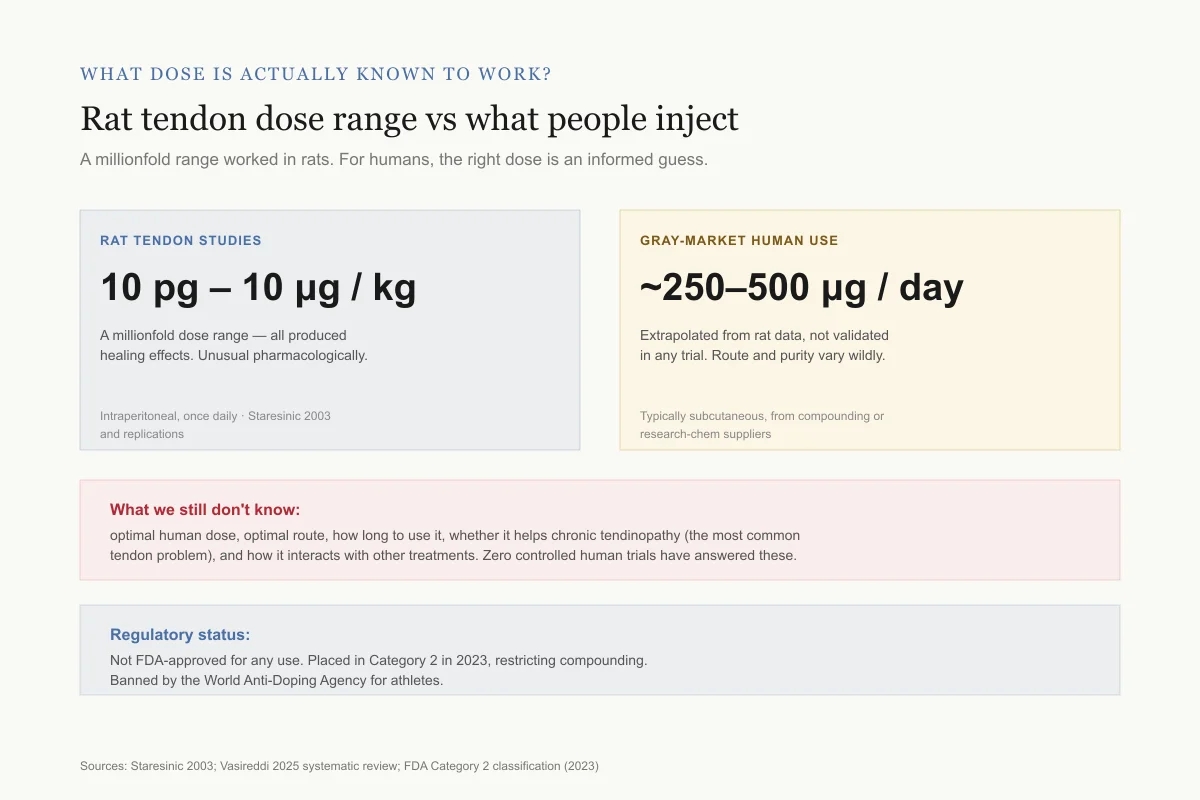
What dose is actually known to work?
Rat tendon dose range vs what people are actually injecting
A millionfold range worked in rats. For humans, the right dose is an informed guess.
Rat tendon studies
10 pg – 10 μg / kg
A millionfold dose range — all produced healing effects. Unusual pharmacologically.
Intraperitoneal, once daily · Staresinic 2003 and replications
Gray-market human use
~250–500 μg / day
Extrapolated from rat data, not validated in any trial. Route, purity, and frequency vary wildly.
Typically subcutaneous injection from compounding pharmacies or research-chem suppliers
What we still don't know: optimal human dose, optimal route, how long to use it, whether it helps chronic tendinopathy (the most common tendon problem), and how it interacts with other treatments. Zero controlled human trials have answered any of these.
Regulatory status: BPC-157 is not FDA-approved for any use. The FDA placed it in Category 2 in 2023, restricting compounding. It is banned by the World Anti-Doping Agency for athletes.
Sources: Staresinic 2003; Vasireddi 2025 systematic review; FDA Category 2 classification (2023)
 View as image
View as imageSafety
ModerateNo controlled human safety data — and a gray-market supply problem
Concern
Every therapeutic use of BPC-157 in humans today is off-label, unapproved, and drawn from animal-data extrapolation. Beyond the unknown biology, the actual vials sold online are not regulated for purity, potency, or sterility.
What the research says
Rat studies show a wide apparent safety margin and no organ toxicity. But absence of evidence is not evidence of absence — and even if BPC-157 itself is benign, a contaminated or under-dosed product is not.
Particularly relevant for: Anyone considering self-injecting peptides purchased online or from compounding sources
What to do
Recognize this is experimental self-treatment, not a medical therapy. The peptide is FDA Category 2 (restricted compounding) and WADA-banned for athletes. A clinician can help weigh risks and watch for adverse effects.
Vasireddi 2025; FDA 2023 Category 2 classification; WADA Prohibited List
The peptide lacks US Food and Drug Administration approval for any indication. The FDA classified it as a Category 2 compound in 2023, restricting its compounding. Its use is also prohibited in professional sports under World Anti-Doping Agency rules.[1]
How BPC-157 Compares to Standard Growth Factor Approaches
The Seiwerth 2018 review positioned BPC-157 against standard angiogenic growth factors (EGF, FGF, VEGF) used in tissue engineering and regenerative medicine.[7] The comparison raised a practical point: exogenous growth factors typically require biocompatible carriers, specialized delivery systems, and local application to achieve musculoskeletal healing effects. BPC-157, by contrast, showed consistent results across intraperitoneal, oral, and local routes without any carrier molecule.
However, this comparison has context that matters. The growth factor literature spans thousands of studies from hundreds of independent groups worldwide, which means it includes both positive and negative results across heterogeneous conditions. The BPC-157 literature, by contrast, is much smaller and more uniform in its positive findings. A smaller, more concentrated evidence base will almost always look more consistent than a larger, more diverse one. This does not mean BPC-157 is superior; it may mean the evidence has not yet been tested broadly enough to reveal where it fails.
Gwyer et al. (2019) provided an independent assessment from outside the Sikiric group. Their review acknowledged BPC-157's consistent positive results while noting that "only a handful of research groups have performed in-depth studies regarding this peptide."[12] They identified BPC-157's stability and multi-route efficacy as genuine advantages over fragile growth factors but stopped short of recommending it as a therapeutic without human trial data.
Why the Gap Between Animal and Human Data Persists
Nearly all BPC-157 musculoskeletal research originates from a small number of research groups, most prominently the laboratory of Predrag Sikiric at the University of Zagreb, which has produced the majority of published BPC-157 studies since the 1990s. The Chang laboratory in Taipei provided independent replication for the tendon-specific cellular mechanisms, which strengthens the mechanistic evidence. Still, the overall evidence base relies heavily on a concentrated group of investigators.
Several factors contribute to the human data gap. BPC-157 is a naturally occurring peptide fragment, making patent protection difficult and reducing pharmaceutical company incentive to fund expensive clinical trials. The peptide's 2023 Category 2 classification by the FDA further complicated the regulatory path. Meanwhile, a growing gray-market supply has created a situation where many people use BPC-157 for tendon injuries without controlled clinical evidence guiding dose, route, or duration.
The rat models themselves introduce limitations. Complete surgical transection of an Achilles tendon is a clean, reproducible injury, but it differs from the tendinopathy, partial tears, and chronic overuse injuries that constitute the vast majority of human tendon problems. Whether the cellular mechanisms BPC-157 activates in acute surgical wounds translate to chronic degenerative tendon disease has not been studied. The dose range tested in rats (10 pg/kg to 10 microg/kg) spans a millionfold range with effects at all levels, which is unusual pharmacologically and raises questions about the dose-response relationship that would need to be resolved before human trials could determine an appropriate dose. Additionally, most rat studies assessed healing over 14 to 42 days. Human tendon healing operates on a longer timeline, often 3 to 6 months for functional recovery and 12 to 18 months for full tissue remodeling, and whether BPC-157's early-phase effects translate to improved long-term outcomes is unknown.
The Bottom Line
BPC-157 has demonstrated consistent, positive effects on tendon healing across more than 20 years of animal research, with verified mechanisms including fibroblast migration via FAK-paxillin signaling, growth hormone receptor upregulation, and VEGF-mediated angiogenesis. The entire evidence base consists of 35 preclinical studies and a single 12-patient retrospective survey. The gap between rodent tendon transection models and human tendon injuries has not been bridged by any controlled clinical trial.
Sources & References
- 1RPEP-13892·Vasireddi, Nikhil et al. (2025). “BPC-157 for Sports Injuries: What a Systematic Review of the Evidence Actually Shows.” HSS journal : the musculoskeletal journal of Hospital for Special Surgery.Study breakdown →PubMed →↩
- 2RPEP-01187·Sikiric, P et al. (2006). “BPC-157 in Clinical Trials for IBD: Comprehensive Animal Evidence Supporting Human Testing.” Inflammopharmacology.Study breakdown →PubMed →↩
- 3RPEP-00860·Staresinic, M et al. (2003). “BPC-157 Accelerates Achilles Tendon Healing and Directly Stimulates Tendon Cell Growth.” Journal of orthopaedic research : official publication of the Orthopaedic Research Society.Study breakdown →PubMed →↩
- 4RPEP-01746·Chang, Chung-Hsun et al. (2011). “BPC-157 Promotes Tendon Healing Through Cell Outgrowth, Survival, AND Migration.” Journal of applied physiology (Bethesda.Study breakdown →PubMed →↩
- 5RPEP-02350·Chang, Chung-Hsun et al. (2014). “Pentadecapeptide BPC 157 enhances the growth hormone receptor expression in tendon fibroblasts..” Molecules (Basel.Study breakdown →PubMed →↩
- 6RPEP-01458·Brcic, L et al. (2009). “BPC-157 Promotes Healing Through Angiogenesis in Both Muscle and Tendon.” Journal of physiology and pharmacology : an official journal of the Polish Physiological Society.Study breakdown →PubMed →↩
- 7RPEP-03561·Unknown (2018). “Seiwerth 2018 Bpc 157 And Standard.” .Study breakdown →↩
- 8RPEP-01157·Krivic, Andrija et al. (2006). “BPC-157 Promotes Achilles Tendon-to-Bone Healing AND Opposes Steroid-Induced Healing Failure.” Journal of orthopaedic research : official publication of the Orthopaedic Research Society.Study breakdown →PubMed →↩
- 9RPEP-01371·Krivic, A et al. (2008). “BPC-157 Improves Achilles Tendon-to-Bone Healing Even During Steroid Treatment.” Inflammation research : official journal of the European Histamine Research Society ... [et al.].Study breakdown →PubMed →↩
- 10RPEP-05464·Japjec, Mladen et al. (2021). “BPC 157 Completely Healed Muscle-Tendon Junction Injuries That Don't Heal on Their Own in Rats.” Biomedicines.Study breakdown →PubMed →↩
- 11RPEP-01595·Cerovecki, Tomislav et al. (2010). “BPC-157 Improves Ligament Healing in Rats.” Journal of orthopaedic research : official publication of the Orthopaedic Research Society.Study breakdown →PubMed →↩
- 12RPEP-04213·Gwyer, Daniel et al. (2019). “BPC-157 for Tendon, Ligament, and Muscle Healing: What the Animal Research Shows.” Cell and tissue research.Study breakdown →PubMed →↩