Oxytocin and Female Sexual Response
Oxytocin and Female Sexuality
83% higher at orgasm
Plasma oxytocin levels were significantly higher during orgasm than baseline in women, with the temporal pattern suggesting a role in uterine contractions and subjective sexual experience.
Carmichael et al., Journal of Clinical Endocrinology & Metabolism, 1987
Carmichael et al., Journal of Clinical Endocrinology & Metabolism, 1987
If you only read one thing
Oxytocin — the 'bonding hormone' — does rise during female arousal and peaks at orgasm. It's real biology, not just a nickname. In couples studies, a nasal spray version made sex feel more emotionally connected and orgasms more intense. But when researchers ran a proper clinical trial in women with sexual dysfunction, oxytocin performed no better than placebo. The peptide clearly plays a role in how sex feels, but spraying it up your nose doesn't reliably fix sexual problems — at least not with the doses and timing tested so far.
Oxytocin is a nine-amino-acid peptide synthesized in the hypothalamus that has been measured in the bloodstream of women during every phase of the sexual response cycle. Carmichael et al. (1987) provided the first rigorous measurement of this phenomenon: plasma oxytocin levels rose during arousal and peaked during orgasm in women, with levels significantly higher than pre-stimulation baseline.[1] Since that landmark study, research has expanded in three directions. First, mechanistic work has identified the specific neural circuits through which oxytocin modulates female sexual behavior, including a 2014 Cell paper demonstrating that oxytocin acts through a specific class of prefrontal cortical interneurons to regulate sociosexual behavior in females.[2] Second, clinical trials have tested intranasal oxytocin as a treatment for female sexual dysfunction, with mixed results that highlight the complexity of translating a neuropeptide's physiological role into therapy. Third, the relationship between oxytocin and pair bonding has been mapped in detail, revealing how sexual activity and emotional attachment reinforce each other through shared peptide signaling. This article covers all three domains and their intersections. For related peptide mechanisms involved in female arousal, see kisspeptin and female sexual arousal. For a broader view of peptide-based approaches to sexual dysfunction, see peptide therapeutics for female sexual dysfunction.
Key Takeaways
- Oxytocin starts rising during arousal and peaks right at orgasm — that's measurable, not metaphor.
- In a proper trial for female sexual dysfunction, oxytocin spray worked no better than placebo.
- It's called the "bonding hormone," but its effects depend heavily on context — safe partners, not strangers.
- Couples using oxytocin spray reported more intense orgasms and stronger emotional connection afterward.
- Scientists traced oxytocin's sexual effects to specific cells in the brain's decision-making center.
- The peptide also boosts dopamine in the reward circuits, which is why bonding feels rewarding.
- Some women show big oxytocin surges during sex; others barely any — individual variation is huge.
The Physiology: What Happens During Female Sexual Response
The connection between oxytocin and female sexual response was first measured systematically by Carmichael et al. (1987). Using continuous blood sampling through indwelling venous catheters during private self-stimulation to orgasm, the researchers tracked plasma oxytocin levels in 13 women and 9 men. In women, oxytocin began rising during arousal, before any genital contact, and reached peak concentrations during orgasm. The temporal pattern suggested that oxytocin contributes to both the subjective experience of arousal and the uterine and vaginal smooth muscle contractions that characterize orgasm.[1]
Blaicher et al. (1999) replicated and extended this finding. In 12 healthy women, oxytocin serum levels measured 1 minute after orgasm were significantly higher than baseline. The study also detected oxytocin increases during arousal prior to orgasm, confirming that the peptide is not merely a byproduct of climax but participates in the arousal process itself.[3] The mechanism involves both central nervous system effects (oxytocin neurons in the paraventricular nucleus project to brain regions involved in sexual motivation and reward) and peripheral effects (oxytocin receptors in the uterus and vaginal smooth muscle mediate contractions during orgasm).
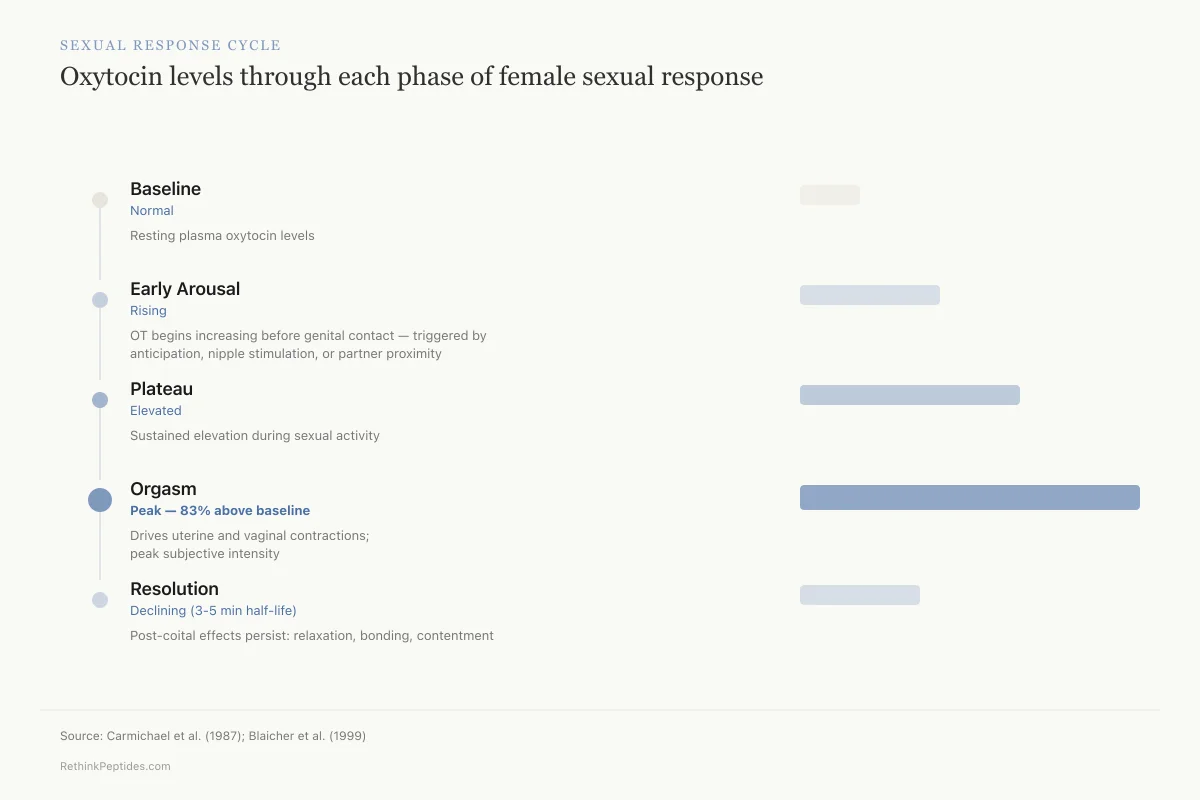
Sexual Response Cycle
Oxytocin levels through each phase of female sexual response
Resting plasma oxytocin levels before any stimulation
Oxytocin begins increasing before genital contact — triggered by anticipation, nipple stimulation, or partner proximity
Sustained elevation during sexual activity; varies with type and intensity of stimulation
Highest concentration — drives uterine and vaginal smooth muscle contractions and subjective intensity
Rapid decline (3-5 minute half-life) but post-coital effects persist — promotes relaxation, bonding, and contentment
Source: Carmichael et al. (1987); Blaicher et al. (1999)
 View as image
View as imageThe relationship between oxytocin and female arousal is bidirectional. Sexual stimulation triggers oxytocin release, and oxytocin in turn enhances the physiological components of arousal. Nipple stimulation, genital contact, and cervical stimulation each activate distinct afferent pathways that converge on hypothalamic oxytocin neurons. This convergence explains why different types of sexual stimulation can each independently raise plasma oxytocin levels and why combined stimulation produces the strongest oxytocin response.
What remains unclear is the precise dose-response relationship. Plasma oxytocin levels vary substantially between individuals and between sexual encounters in the same individual. Some women show large oxytocin spikes during orgasm; others show modest increases. Whether these differences correlate with subjective sexual satisfaction, orgasm intensity, or orgasmic consistency has not been adequately studied. The measurement challenge is real: oxytocin has a plasma half-life of approximately 3-5 minutes, meaning that sampling timing relative to orgasm critically affects results. Studies using less frequent sampling intervals likely underestimate peak concentrations.
The peripheral effects of oxytocin during female sexual response deserve separate attention. Oxytocin receptors are expressed throughout the female reproductive tract, including the uterine myometrium, fallopian tubes, and vaginal smooth muscle. Receptor density in these tissues fluctuates with the menstrual cycle, peaking during the late follicular phase when estrogen levels are highest. This hormonal modulation of receptor expression means that oxytocin's effects on genital arousal and orgasmic contractions vary across the cycle, potentially explaining why some women report cycle-dependent variation in orgasm quality. The same receptor upregulation occurs during pregnancy and lactation, creating the strong uterine contractions of labor and the milk ejection reflex during breastfeeding. These reproductive functions share the same molecular machinery as sexual response, a fact that connects oxytocin's roles across the full spectrum of female reproductive biology.
The distinction between central and peripheral oxytocin effects matters for therapeutic development. Central effects (arousal enhancement, emotional bonding, reduced anxiety) require oxytocin to reach specific brain regions. Peripheral effects (smooth muscle contraction, genital blood flow) can be achieved with lower doses that act locally. Intranasal delivery was initially pursued because it was thought to bypass the blood-brain barrier via olfactory nerve transport, but the extent of this transport in humans remains debated.
The Neural Circuit: How Oxytocin Acts in the Brain
Nakajima et al. (2014) identified a specific neural mechanism through which oxytocin regulates female sexual behavior. Published in Cell, the study discovered a population of oxytocin-responsive interneurons in the prefrontal cortex that modulate sociosexual behavior in females. These interneurons express oxytocin receptors and, when activated, alter the balance of excitation and inhibition in prefrontal circuits that control approach behavior, social decision-making, and sexual receptivity.[2]
This finding is significant because it moves the field beyond "oxytocin as a hormone" toward "oxytocin as a neuromodulator with circuit-level specificity." The prefrontal cortex is the brain region most involved in context-dependent decision-making, and the fact that oxytocin acts there through a specific interneuron class suggests that its effects on sexual behavior are not simply pro-sexual or anti-sexual but context-dependent. Oxytocin may enhance sexual receptivity in a safe, bonded context while having different or no effects in threatening or unfamiliar contexts.
Petersson et al. (2024) mapped the interaction between oxytocin and dopamine systems in regulating sexual behavior and reward. Oxytocin neurons project to the ventral tegmental area (VTA) and nucleus accumbens, key components of the brain's reward circuitry where dopamine operates. Oxytocin release during sexual activity potentiates dopamine transmission in these regions, amplifying the rewarding aspects of sexual experience. Disruptions in this oxytocin-dopamine interaction are associated with anhedonia, reduced sexual motivation, and depression.[4] This connection to the dopamine system is one reason why sexual dysfunction frequently co-occurs with mood disorders, and why interventions targeting only one system often produce incomplete responses. For more on how neuropeptides interact with mood circuits, see neuropeptides and depression: the biology beyond serotonin.
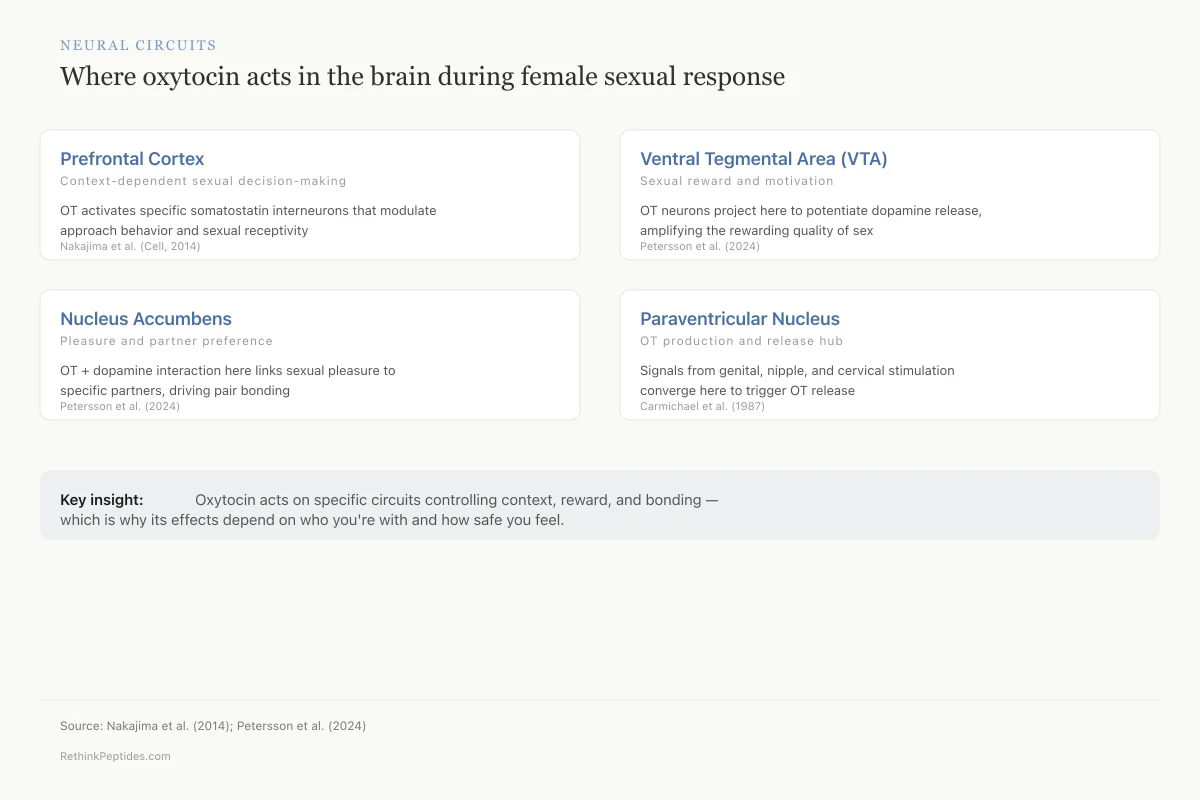
Neural Circuits
Where oxytocin acts in the brain during female sexual response
OT activates specific somatostatin interneurons that modulate approach behavior and sexual receptivity
Nakajima et al. (Cell, 2014)
OT neurons project here to potentiate dopamine release, amplifying the rewarding quality of sex
Petersson et al. (2024)
OT + dopamine interaction here links sexual pleasure to specific partners, driving pair bonding
Petersson et al. (2024)
Afferent signals from genital, nipple, and cervical stimulation converge here to trigger OT release
Carmichael et al. (1987)
Key insight: Oxytocin doesn't just flood the brain with a “feel good” signal. It acts on specific circuits that control context, reward, and bonding — which is why its effects depend on who you're with and how safe you feel.
Source: Nakajima et al. (2014); Petersson et al. (2024)
 View as image
View as imageYao et al. (2025) provided a comprehensive framework for understanding how oxytocin modulates behavior in Molecular Psychiatry. Their review synthesized evidence that oxytocin's behavioral effects depend on receptor density in specific brain regions, which varies with genetics, epigenetic modifications, and life experience. Women with higher oxytocin receptor density in reward and social cognition areas may experience stronger bonding effects from sexual activity, while those with lower receptor density may require stronger or more sustained oxytocin signals to achieve the same effects.[5]
Intranasal Oxytocin and Sexual Function: Clinical Evidence
The physiological link between oxytocin and female sexual response prompted clinical trials of exogenous oxytocin administration. The results have been instructive but not straightforward.
Acute Effects in Healthy Women
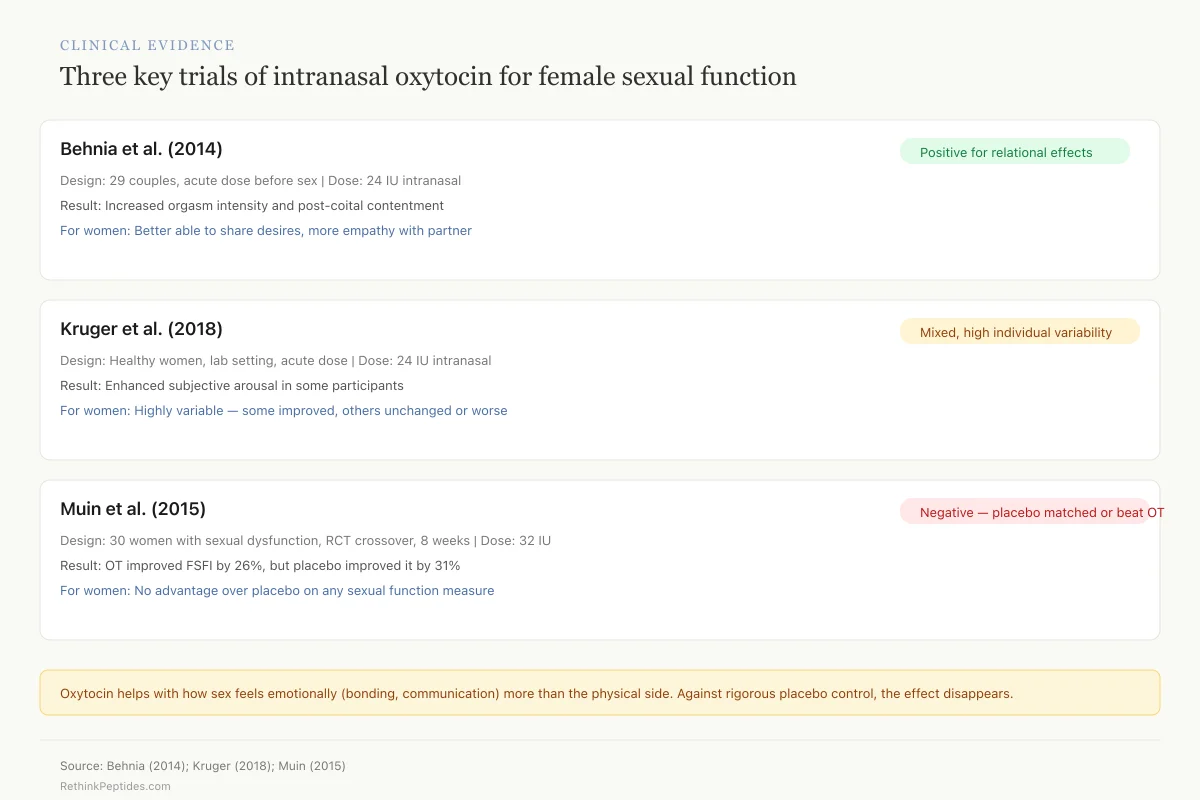
Kruger et al. (2018) conducted a laboratory study of intranasal oxytocin effects on sexual function in healthy women. Participants received either 24 IU of intranasal oxytocin or placebo before viewing erotic stimuli and engaging in self-stimulation. The study measured both subjective sexual response (arousal, desire, orgasm intensity) and physiological parameters. Results showed that intranasal oxytocin enhanced subjective arousal ratings and orgasm intensity in a subset of participants, though the overall effect was modest and not uniform across all women.[6]
The variability in individual response is a recurring theme. Some women showed clear enhancement of arousal and orgasm with oxytocin; others showed no change or slight decreases. This heterogeneity likely reflects individual differences in oxytocin receptor density, baseline oxytocin levels, and the complex interaction between oxytocin and other neurochemical systems. The laboratory setting itself may also modulate effects: sexual arousal in an unfamiliar clinical environment involves different neural circuitry than arousal in a private, comfortable setting with a familiar partner. Context dependence is not a confound but a feature of how oxytocin operates in the brain.
Couple-Based Studies
Behnia et al. (2014) tested intranasal oxytocin (24 IU) in 29 heterosexual couples (58 individuals). The study evaluated effects on sexual experience during partnered intercourse rather than laboratory stimulation, increasing ecological validity. Oxytocin increased the intensity of orgasm and contentment after sexual intercourse. The effects showed sex differences: men reported higher sexual satiety, while women reported feeling more relaxed and better able to share sexual desires and empathize with their partners.[7]
The sex difference in response is consistent with oxytocin's known role in communication and empathy rather than purely genital function. For women, the primary benefit was relational (improved communication, partner empathy) rather than physiological (increased lubrication, faster arousal). This pattern suggests that oxytocin's effects on female sexual satisfaction operate partly through enhanced emotional connection, a finding with implications for how we conceptualize female sexual dysfunction and its treatment.
Long-Term Treatment for Sexual Dysfunction
Muin et al. (2015) conducted the most rigorous clinical trial of intranasal oxytocin for female sexual dysfunction. In a randomized, double-blind, placebo-controlled crossover trial, 30 pre- and postmenopausal women with sexual dysfunction self-administered intranasal oxytocin (32 IU) or placebo within 50 minutes before intercourse over 8 weeks, followed by a 2-week washout and crossover. The Female Sexual Function Index (FSFI) increased by 26% with oxytocin and 31% with placebo. Sexual Quality of Life scores improved by 144% with oxytocin and 125% with placebo. Depression symptoms improved in both groups.[8]
The absence of a treatment effect beyond placebo is the central finding. Both groups improved substantially, but oxytocin did not outperform placebo on any sexual function measure. Several interpretations are possible. First, the placebo effect in sexual dysfunction trials is large because expectation, attention, and behavioral activation (scheduling sexual encounters around study visits) are themselves therapeutic. Second, the dose and timing may not have been optimal: 32 IU administered 50 minutes before intercourse may not achieve sufficient brain concentrations at the time of actual sexual activity. Third, intranasal delivery produces variable absorption, and plasma oxytocin levels were not measured in this study, so it is unknown whether the administered oxytocin actually reached relevant brain regions at pharmacologically active concentrations.
The trial also raised a methodological question about crossover designs in sexual dysfunction research. Because the crossover design exposed all participants to both conditions, any learning effects or relationship improvements from the first treatment period carried over into the second. If the act of participating in a structured sexual health study produced genuine therapeutic benefit (through increased communication with partners, reduced avoidance of sexual activity, or simply increased attention to sexual experience), this benefit would persist across both treatment arms, obscuring any drug-specific effect. Future trials may benefit from parallel-group designs that avoid this confound, even though they require larger sample sizes.
Clinical Evidence
Three key trials of intranasal oxytocin for female sexual function
The pattern: Oxytocin helps with how sex feels emotionally (bonding, communication) more than it helps with the physical side. And when tested rigorously against placebo, the effect disappears — possibly because simply participating in a structured sexual health study is itself therapeutic.
Source: Behnia (2014); Kruger (2018); Muin (2015)
 View as image
View as imageOxytocin and Pair Bonding: The Post-Coital Connection
The relationship between oxytocin and sexual behavior extends beyond the sexual encounter itself. Oxytocin released during sex contributes to pair bonding, the formation and maintenance of exclusive partner attachments.
Razo et al. (2022) studied the long-term effects of chronic intranasal oxytocin on adult pair bonding behavior and brain glucose uptake. Chronic oxytocin administration altered activity in brain regions associated with partner preference and social reward, suggesting that sustained elevation of oxytocin can modify bonding circuits over time.[9] The relevance to female sexual response is that pair bonding and sexual satisfaction are reciprocally linked: secure attachment promotes sexual openness, and satisfying sexual experiences strengthen attachment. Oxytocin sits at the nexus of both processes.
Kendrick et al. (2018) reviewed the broader landscape of human oxytocin research, documenting how oxytocin's effects on social cognition, trust, and emotional processing create the psychological context within which sexual behavior occurs. Their overview found that oxytocin enhances sensitivity to social cues, increases gaze toward partner's eyes, and promotes the interpretation of ambiguous social signals as positive rather than threatening.[10] In a sexual context, these effects translate to enhanced emotional intimacy, reduced performance anxiety, and greater attunement to a partner's responses, all factors that contribute to female sexual satisfaction independently of genital arousal.
The many faces of oxytocin extend well beyond sexuality. Zik and Roberts (2015) documented oxytocin's roles in maternal behavior, trust, stress buffering, and social memory, all of which interact with sexual function.[11] A woman's experience of oxytocin during sex is influenced by her baseline oxytocin tone, which reflects her broader social and emotional environment. Chronic stress, social isolation, and attachment insecurity all reduce baseline oxytocin levels, potentially diminishing the sexual response and pair bonding effects that depend on this peptide. Understanding this broader context is essential for interpreting clinical trial results and designing effective interventions. For how kisspeptin triggers puberty and drives desire through a different peptide pathway, see our dedicated article.
The Broader Neuropeptide Context
Oxytocin does not act alone in regulating female sexual response. It operates within a network of neuropeptides, neurotransmitters, and hormones that together produce the complex experience of sexual desire, arousal, and satisfaction.
Chaulagain et al. (2025) reviewed the neurobiological impact of oxytocin in the context of mental health, highlighting how oxytocin deficiency or receptor dysfunction contributes to conditions that secondarily impair sexual function: depression, anxiety, PTSD, and social withdrawal.[12] Treating oxytocin-related sexual dysfunction therefore requires addressing the broader neurobiological context rather than targeting oxytocin alone.
Paul et al. (2026) advanced this framework in a Neuropeptides review, mapping oxytocin's roles in neuromodulation, synaptic plasticity, and epigenetic regulation. Their work demonstrated that oxytocin does not merely modulate existing neural activity but can reshape neural circuits through sustained exposure, a finding with implications for long-term therapeutic strategies.[13]
The neuropeptide network approach also explains why bremelanotide, a melanocortin receptor agonist that acts on different peptide pathways, has shown efficacy for hypoactive sexual desire disorder in women where oxytocin alone has not. Bremelanotide activates desire circuits through melanocortin-4 receptors in the hypothalamus, complementing rather than replacing oxytocin's role in arousal and bonding. The distinction matters clinically: oxytocin appears to modulate how sex feels (arousal quality, orgasm intensity, emotional connection) while melanocortin agonists modulate whether sex is wanted (desire, motivation, initiation). These are distinct components of the sexual response cycle, and effective treatment may require addressing both. For more on melanocortin pathway effects, see melanocortin peptides and blood pressure.
Nowacka et al. (2025) added another dimension by identifying vagal oxytocin receptors as molecular targets in gut-brain signaling. The vagus nerve expresses oxytocin receptors that mediate bidirectional communication between the gut and the brain, influencing appetite, satiety, and emotional state. This vagal pathway may contribute to the "butterflies in the stomach" sensation associated with romantic and sexual arousal, providing a physiological substrate for the visceral component of sexual desire that extends beyond genital sensation alone.
Current Limitations and Open Questions
The field faces several unresolved challenges. Measurement remains problematic: peripheral plasma oxytocin levels may not reflect central nervous system concentrations, and the relationship between the two is inconsistent across studies. Some research uses salivary oxytocin, which introduces additional variability from oral enzyme degradation. Standardized, validated measurement methods for oxytocin in clinical settings do not yet exist.
Intranasal delivery pharmacokinetics are poorly characterized in women. Most pharmacokinetic studies of intranasal oxytocin have been conducted in men or mixed populations. The nasal mucosal absorption, blood-brain barrier penetration, and dose-response curves specific to women remain incompletely defined. This uncertainty makes it difficult to design clinical trials with confidence that the administered dose produces relevant brain concentrations.
Individual variability in oxytocin receptor expression adds another layer of complexity. Genetic polymorphisms in the oxytocin receptor gene (OXTR) influence receptor density and signaling efficiency. Epigenetic modifications to OXTR, driven by early life experiences and chronic stress, further modulate receptor function. A single dose of intranasal oxytocin may produce robust effects in a woman with high OXTR expression and minimal effects in one with epigenetically downregulated receptors. Personalized dosing based on receptor status is theoretically possible but practically beyond current clinical capabilities.
The placebo effect in female sexual dysfunction trials presents an ongoing interpretive challenge. The Muin et al. (2015) trial found improvement rates of 26-31% regardless of treatment assignment. Any future oxytocin trial must either control for this effect (difficult in crossover designs where participants experience both conditions) or demonstrate effects large enough to clearly exceed placebo (requiring larger samples and more sensitive outcome measures).
Emerging Directions
Several research directions could resolve the current limitations. First, biomarker-guided treatment selection: measuring baseline oxytocin receptor expression (via genetic testing of OXTR polymorphisms or plasma oxytocin levels) could identify women most likely to respond to exogenous oxytocin. This precision medicine approach would avoid the dilution effect seen in population-level trials where non-responders mask treatment effects in responders.
Second, combination therapies that pair oxytocin with agents acting on complementary pathways may prove more effective than oxytocin alone. An approach combining intranasal oxytocin (for arousal and bonding enhancement) with bremelanotide (for desire activation) has not been tested but has pharmacological rationale. The two peptides act on distinct receptor systems with non-overlapping mechanisms, and their combined effects on the sexual response cycle would be broader than either alone.
Third, non-intranasal delivery routes deserve investigation. Sublingual tablets, transdermal patches, and intravaginal formulations could provide more consistent absorption than intranasal sprays while potentially targeting peripheral reproductive tissue oxytocin receptors more directly. Intravaginal delivery is particularly interesting because it could achieve high local concentrations at oxytocin receptors in vaginal and cervical tissue without requiring systemic absorption or blood-brain barrier penetration, potentially enhancing the genital arousal component of sexual response.
Fourth, timing optimization remains an untapped variable. The 50-minute pre-coital administration window used in the Muin trial was chosen for practical reasons rather than pharmacokinetic optimization. Studies measuring real-time plasma and central oxytocin concentrations after intranasal administration, combined with continuous physiological monitoring during sexual activity, could identify the optimal administration-to-activity window for maximizing effects on arousal and orgasm.
The Bottom Line
Oxytocin rises during female sexual arousal and peaks at orgasm, acting through specific prefrontal cortical circuits and interacting with dopamine reward pathways to modulate both physiological response and emotional bonding. Intranasal oxytocin enhances orgasm intensity and relational connection in couple-based studies, but a rigorous clinical trial found no advantage over placebo for treating female sexual dysfunction. The gap between oxytocin's clear physiological role in sexual response and its uncertain therapeutic value reflects the complexity of translating neuropeptide biology into clinical intervention, a challenge shared across the peptide therapeutics field.
Sources & References
- 1RPEP-00041·Carmichael, M S et al. (1987). “Plasma oxytocin increases in the human sexual response..” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 2RPEP-02456·Nakajima, Miho et al. (2014). “Oxytocin modulates female sociosexual behavior through a specific class of prefrontal cortical interneurons..” Cell.Study breakdown →PubMed →↩
- 3RPEP-00512·Blaicher, W et al. (1999). “The role of oxytocin in relation to female sexual arousal..” Gynecologic and obstetric investigation.Study breakdown →PubMed →↩
- 4RPEP-09067·Petersson, Maria et al. (2024). “How Oxytocin and Dopamine Work Together to Control Social Behavior, Reward, and Mental Health.” Biomedicines.Study breakdown →PubMed →↩
- 5RPEP-14355·Yao, Shuxia et al. (2025). “How Oxytocin Shapes Human Behavior: A New Model from Attention to Social Bonds.” Molecular psychiatry.Study breakdown →PubMed →↩
- 6RPEP-03761·Kruger, Tillmann H C et al. (2018). “Effects of Intranasal Oxytocin Administration on Sexual Functions in Healthy Women: A Laboratory Paradigm..” Journal of clinical psychopharmacology.Study breakdown →PubMed →↩
- 7RPEP-02330·Behnia, Behnoush et al. (2014). “Differential effects of intranasal oxytocin on sexual experiences and partner interactions in couples..” Hormones and behavior.Study breakdown →PubMed →↩
- 8RPEP-02747·Muin, Dana A et al. (2015). “Effect of long-term intranasal oxytocin on sexual dysfunction in premenopausal and postmenopausal women: a randomized trial..” Fertility and sterility.Study breakdown →PubMed →↩
- 9RPEP-06450·Razo, Rocío Arias-Del et al. (2022). “Long term effects of chronic intranasal oxytocin on adult pair bonding behavior and brain glucose uptake in titi monkeys (Plecturocebus cupreus)..” Hormones and behavior.Study breakdown →PubMed →↩
- 10RPEP-03745·Kendrick, Keith M et al. (2018). “Overview of Human Oxytocin Research..” Current topics in behavioral neurosciences.Study breakdown →PubMed →↩
- 11RPEP-02855·Zik, Jodi B et al. (2015). “The many faces of oxytocin: implications for psychiatry..” Psychiatry research.Study breakdown →PubMed →↩
- 12RPEP-10367·Chaulagain, Ram Prasad et al. (2025). “The neurobiological impact of oxytocin in mental health disorders: a comprehensive review..” Annals of medicine and surgery (2012).Study breakdown →PubMed →↩
- 13RPEP-15879·Paul, Supratim et al. (2026). “Oxytocin beyond social bonding: Advancing neuromodulation, synaptic plasticity, and epigenetic precision in CNS disorders..” Neuropeptides.Study breakdown →PubMed →↩