Edwin Lee: The Only Doctor Publishing Human BPC-157 Data
BPC-157 Human Evidence Gap
3 Human Studies
Every published human clinical study of BPC-157 comes from a single endocrinologist's clinic in Orlando, Florida, totaling 30 patients across three pilot reports.
Lee et al., Alternative Therapies in Health and Medicine, 2021-2025
Lee et al., Alternative Therapies in Health and Medicine, 2021-2025
If you only read one thing
BPC-157 is one of the most talked-about peptides on the internet. But in the entire medical literature, only three published human studies exist — and every one of them comes from the same endocrinologist's clinic in Orlando, Florida. Together they cover 30 patients, none of them given a placebo. The results look interesting (big majorities reported feeling better after knee or bladder injections), but without a control group we cannot tell how much of that is the peptide versus the shot itself or natural healing. This is the entire human evidence base for a compound being sold online to hundreds of thousands of people.
BPC-157 has been the subject of more than 544 published papers since 1992.[1] The overwhelming majority report results from rats, mice, and cell cultures. Only three papers report clinical outcomes in human patients. All three come from one physician: Edwin Lee, an endocrinologist in Orlando, Florida, who runs the Institute for Hormonal Balance.
That concentration matters. It means the entire published human evidence base for one of the most widely discussed peptides in the wellness space rests on 30 patients, a single clinic, and one researcher's judgment. This article examines who Lee is, what each of his studies found, where the designs fall short, and what the broader BPC-157 evidence landscape looks like. For deeper context on each subtopic, see the dedicated articles linked throughout: the human trial gap, the Sikiric replication problem, cancer and angiogenesis risk, supply chain and purity, and animal safety data.
Key Takeaways
- Only 3 published human clinical studies of BPC-157 exist as of March 2026, all from Dr. Edwin Lee's clinic in Orlando, Florida
- 16 patients with knee pain received intra-articular BPC-157: 87.5% (14/16) reported significant improvement at 6-12 month follow-up (Lee & Padgett, 2021)
- 12 women with interstitial cystitis received intravesical BPC-157 (10 mg): 10 of 12 reported complete symptom resolution, the remaining 2 reported 80% improvement (Lee et al., 2024)
- 2 healthy adults received intravenous BPC-157 up to 20 mg over two days with no adverse effects on cardiac, hepatic, renal, or thyroid biomarkers (Lee & Burgess, 2025)
- All 3 studies were published in the same journal (Alternative Therapies in Health and Medicine), none included a control group, and none used validated outcome measures for their primary endpoints
- The FDA classified BPC-157 as a Category 2 bulk drug substance in September 2023, restricting compounding pharmacies from supplying it
The entire published human evidence base
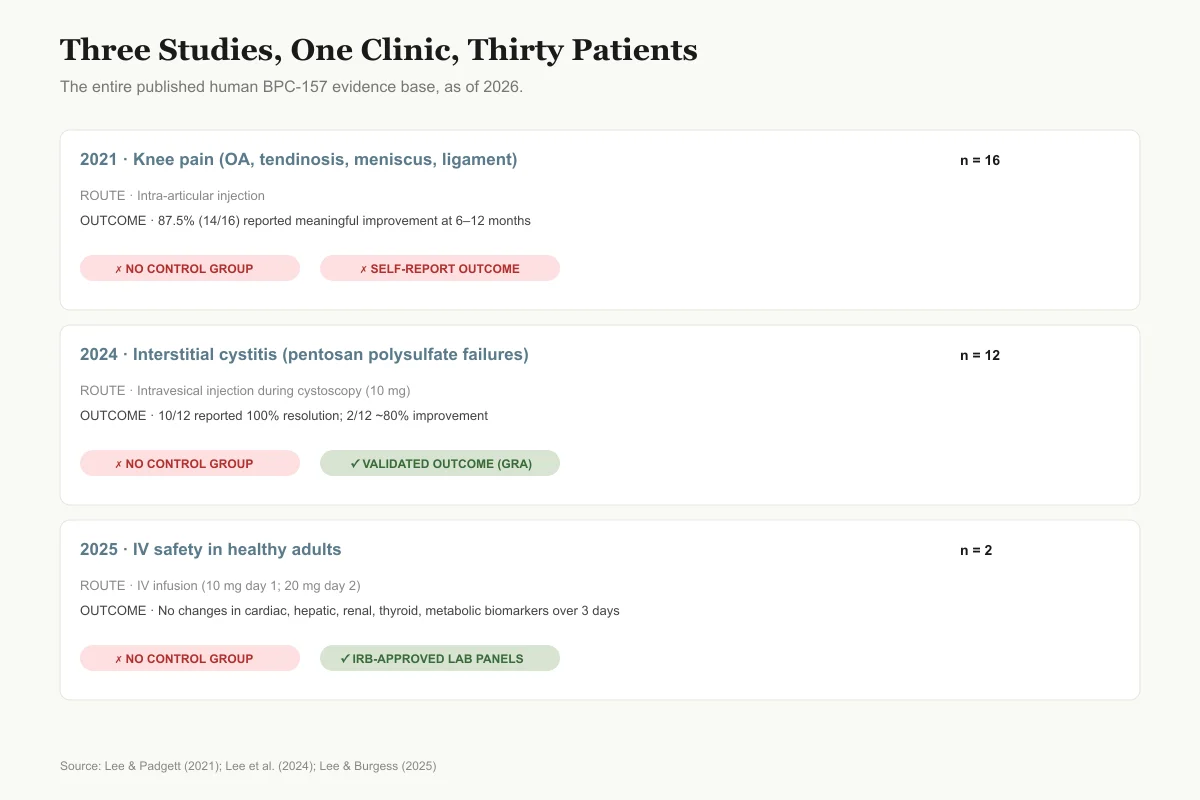
Three Studies, One Clinic, Thirty Patients
Every published human BPC-157 paper since 2021 comes from the same Orlando clinic.
2021 — Knee pain (osteoarthritis, tendinosis, meniscus tears, ligament injury)
n = 16Route
Intra-articular injection
Outcome
87.5% (14/16) reported meaningful improvement at 6–12 months
2024 — Interstitial cystitis (pentosan polysulfate failures)
n = 12Route
Intravesical injection during cystoscopy (10 mg)
Outcome
10/12 reported 100% symptom resolution; 2/12 reported ~80% improvement
2025 — IV safety in healthy adults
n = 2Route
IV infusion (10 mg day 1, 20 mg day 2)
Outcome
No changes in cardiac, hepatic, renal, thyroid, or metabolic biomarkers over 3 days
All three studies were published in the same journal (Alternative Therapies in Health and Medicine). No other site has replicated the findings.
Source: Lee & Padgett (2021); Lee et al. (2024); Lee & Burgess (2025)
 View as image
View as imageHuman Evidence Timeline
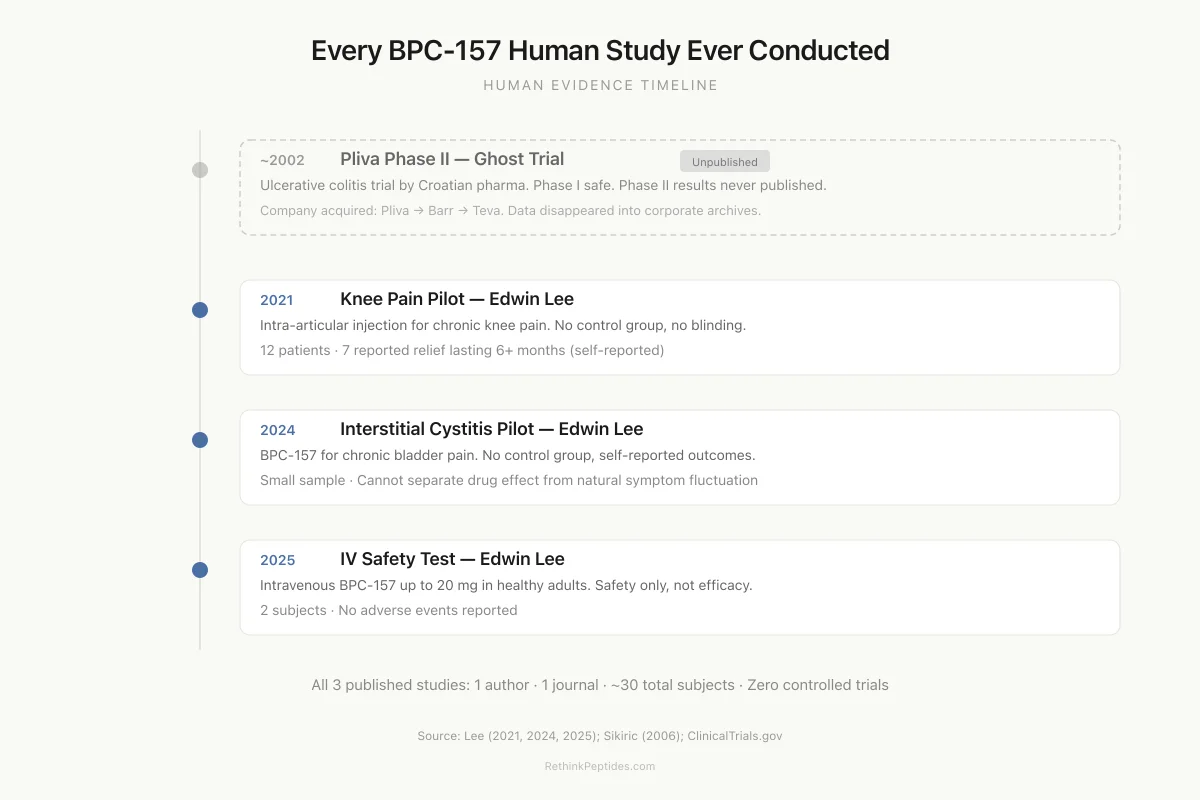
Every BPC-157 Human Study Ever Conducted
Ulcerative colitis trial by Croatian pharma company Pliva. Phase I was safe. Phase II results were never published.
Intra-articular BPC-157 injection for chronic knee pain. No control group, no blinding.
BPC-157 for chronic bladder pain. No control group, self-reported outcomes.
Intravenous BPC-157 up to 20 mg in healthy adults. Safety only, not efficacy.
All 3 published studies are by one author (Edwin Lee) in one journal. None are randomized controlled trials. The one trial that was controlled was never published.
Source: Lee (2021, 2024, 2025); Sikiric (2006); ClinicalTrials.gov
 View as image
View as imageWho Is Edwin Lee?
Edwin Lee is a board-certified endocrinologist who completed fellowships in both critical care and endocrinology at the University of Pittsburgh. He founded the Institute for Hormonal Balance in Orlando in 2008 and holds an assistant professorship at the University of Central Florida College of Medicine. His credentials include board certifications in internal medicine, endocrinology, diabetes, and metabolism, plus additional training in regenerative and functional medicine.
Lee co-founded the Clinical Peptide Society and started a nonprofit called SavePeptides.org, which has collected over 9,000 petition signatures opposing FDA restrictions on compounded peptides. He authored "The Fountain of Youth with Peptides" (2019) and lectures internationally on peptide therapeutics. He introduced BPC-157 into his clinical practice around 2016 and has stated publicly that he has used it on many more patients than the 30 who appear in his published studies.
These roles create overlapping interests. Lee is simultaneously a clinician prescribing BPC-157, a researcher publishing data supporting its use, an author selling books about peptides, a co-founder of an organization promoting peptide access, and a vocal opponent of the FDA's restrictions on compounding. That context does not invalidate his findings, but it means the data should be read with the same scrutiny applied to any researcher with strong financial and ideological stakes in their results.
Study 1: Knee Pain (2021)
Lee's first published human study was a retrospective chart review of 17 patients who received intra-articular BPC-157 injections for various types of knee pain at his Florida clinic between 2019 and 2020.[2] One patient could not be reached for follow-up, leaving 16 in the analysis.
Of those 16, twelve received BPC-157 alone. Eleven of those twelve (91.6%) reported significant pain improvement. Four patients received a combination of BPC-157 and thymosin beta-4 (TB4); three of those four (75%) reported improvement. Overall, 14 of 16 patients (87.5%) reported meaningful knee pain relief at 6 to 12 months post-injection.
The knee conditions varied across the group: osteoarthritis, meniscus tears, tendinosis, and ligament injuries. No standardized outcome tools were used. Patients were contacted by phone and asked to rate their pain improvement on a non-validated scale. There was no control group, no blinding, no imaging to document tissue changes, and no way to distinguish BPC-157's effect from placebo response, natural healing, or the injection procedure itself.
For context, saline injections into arthritic knees produce clinically meaningful pain relief in 30-50% of patients in placebo-controlled trials. An 87.5% response rate without a placebo comparison tells us less than the number suggests. The preclinical data showing BPC-157 accelerates tendon healing in transected rat Achilles tendons is mechanistically relevant,[3] but the leap from rat tendon transection models to human knee osteoarthritis is large, and this study was not designed to bridge it. Lee's paper acknowledged this, noting that future studies should include follow-up MRI scans to document tissue-level changes. None have been published. For more on the animal tendon evidence, see BPC-157 for Tendon Injuries.
Study 2: Interstitial Cystitis (2024)
Lee's second study tested intravesical BPC-157 injection in 12 women with moderate to severe interstitial cystitis (IC) who had failed pentosan polysulfate therapy.[4] The patients ranged from 39 to 76 years old (mean age 58.3). Ten were White, one Asian, and one Latina. Each received 10 mg of BPC-157 injected around the area of bladder inflammation during a single cystoscopy procedure.
Results: 10 of 12 patients reported complete symptom resolution (rating their success at 100%). The remaining 2 reported 80% improvement, with most symptoms resolved but approximately 20% lingering. All 12 scored 5 out of 5 on the Global Response Assessment. No adverse events were reported, and no patients dropped out.
This is a stronger design than the knee study in several respects. It used a validated outcome measure (Global Response Assessment), targeted a specific refractory population (pentosan polysulfate failures), involved a defined dose (10 mg) and route (intravesical injection during cystoscopy), and reported demographics. The condition itself is relevant: pentosan polysulfate, the only FDA-approved oral treatment for IC, has been linked to pigmentary maculopathy with long-term use, creating genuine unmet need.
The limitations remain substantial, however. Twelve patients without a control group cannot establish efficacy. IC is known for significant placebo response rates in clinical trials, and the invasive nature of cystoscopy with injection amplifies placebo effects. The study was conducted at a single clinic with no independent monitoring, and no long-term follow-up data has been published. The sample also lacked demographic diversity.
Study 3: Intravenous Safety (2025)
Lee's most recent publication assessed the safety of intravenous BPC-157 in two healthy adults: a 58-year-old Asian male and a 68-year-old Caucasian female.[5] Both had received IV BPC-157 previously. On day 1, each received 10 mg of BPC-157 in 250 cc of normal saline infused over one hour. On day 2, the dose increased to 20 mg. On day 3, fasting blood work and vital signs were repeated.
The infusions produced no measurable effects on cardiac, hepatic, renal, thyroid, or blood glucose biomarkers. Vital signs and electrocardiograms remained stable throughout. No side effects were reported at any point during the three-day monitoring period.
This was IRB-approved, which distinguishes it from most peptide case reports in the integrative medicine space. But a safety study in 2 patients who had already used the compound without incident provides minimal new information. The participants were essentially self-selected for tolerance. Two data points cannot detect adverse events that occur in even 5-10% of patients, and the study measured only a narrow panel of laboratory markers over 3 days. Longer-term safety signals, immunogenicity, cumulative dose effects, and dose-response relationships remain uncharacterized. For a full review of what animal safety data shows (and doesn't), see BPC-157 Safety Data.
What All Three Studies Share
Several patterns repeat across Lee's published work.
No control groups. None of the three studies included a placebo arm, sham procedure, or active comparator. Without controls, it is impossible to attribute outcomes to BPC-157 rather than placebo response, regression to the mean, natural disease course, or the act of injection itself.
Small sample sizes. The total across all three studies is 30 patients (16 + 12 + 2). Statistical power to detect anything other than a very large effect is effectively zero at these numbers.
One journal. All three appeared in Alternative Therapies in Health and Medicine, a publication focused on complementary and integrative medicine. The journal is PubMed-indexed but does not appear in the Journal Citation Reports and has a limited impact factor compared to journals that typically publish Phase I or Phase II clinical trial data.
One clinic. Every patient came from Lee's Institute for Hormonal Balance in Orlando. No other site, institution, or research group has attempted to replicate any of these findings in a published paper.
No validated primary endpoints (except the IC study's Global Response Assessment). The knee study used phone-based self-report without standardized scales. The safety study used laboratory panels but in only 2 subjects.
These are not fatal flaws for pilot studies. Pilot studies exist to generate hypotheses and signal whether a larger trial is worth funding. The problem is that Lee's data is being cited in clinical practice, podcasts, and advocacy campaigns as though it establishes BPC-157's efficacy and safety in humans. Pilot data cannot do that. For a detailed analysis of why human trial data is so scarce, see The BPC-157 Human Trial Gap.
Safety
ModerateA 2-patient, 3-day IV study is not 'safety data'
Concern
Lee's 2025 IV safety study enrolled two healthy adults who had already taken BPC-157 without incident and tracked them for three days. Even if the full pharmaceutical industry signed off on that design, the numbers are not sufficient to detect adverse events occurring in 1 in 10, 1 in 100, or 1 in 1000 users — let alone immunogenicity, long-term angiogenesis effects, or cumulative-dose toxicity. This is a preliminary signal, not evidence of safety.
What the research says
Genuine safety data would require hundreds to thousands of patients, multiple sites, dose escalation, long-term follow-up, and immunogenicity assays. None of that exists for BPC-157 in humans.
Particularly relevant for: Anyone using BPC-157 based on the premise that it has been 'shown safe in humans'
What to do
Treat claims of human BPC-157 safety as working hypotheses, not established facts. Discuss any self-administration with a physician aware of the evidence gaps.
Lee & Burgess (2025); standard Phase I safety trial design expectations
The Broader BPC-157 Evidence Landscape
The animal literature on BPC-157 is large. Sikiric's pharmacological review described the compound's organ-protective effects across gastrointestinal, musculoskeletal, cardiovascular, and neurological systems as early as 1999.[6] A 2025 literature and patent review cataloged BPC-157's pleiotropic beneficial effects across dozens of preclinical disease models, from inflammatory bowel disease to CNS disorders.[7] A separate 2025 systematic review of BPC-157 in orthopaedic sports medicine screened 544 articles from 1993 to 2024, ultimately including 36 studies in its analysis: 35 preclinical and 1 clinical.[1]
That ratio (35:1 preclinical to clinical) captures the state of BPC-157 research. The animal data shows accelerated healing in transected tendons,[3] modulated angiogenesis via VEGF pathways,[8] improved wound healing across multiple tissue types,[9] cytoprotective activity linked to nitric oxide system interactions,[10] and early indications of healing across bone, skin, and gastrointestinal tissue.[11]
But over 80% of these papers list Predrag Sikiric or Sven Seiwerth of the University of Zagreb as first or senior author. Sikiric has been named on BPC-157-related patent applications dating to at least 1989 and has ownership interests in companies connected to the peptide's commercialization. A compound whose evidence base comes overwhelmingly from one lab faces a replication problem regardless of how consistent the internal findings appear. For the full analysis, see The Sikiric Problem. For an overview of the full BPC-157 story, see The Real BPC-157 Story: 544 Papers, 30 Human Subjects, 50 Million Views.
The Missing Trial: BPC-157 in Ulcerative Colitis
Before Lee's work, there was another attempt at human BPC-157 data. Croatian pharmaceutical company Pliva conducted two trials testing BPC-157 enemas for ulcerative colitis in the early 2000s. These trials were referenced in subsequent Sikiric papers,[10] which described BPC-157 as being "in clinical trials" for IBD, but the trial results do not appear to be indexed in PubMed or published in peer-reviewed journals accessible through standard databases.
A separate trial registered on ClinicalTrials.gov in 2015 (NCT02637284) listed an oral BPC-157 formulation. According to reporting by Undark, the team submitted data and then withdrew it before outside review. No published paper or shared data resulted from that registration. The safety claims made in Sikiric's papers citing these trials remain unsubstantiated by publicly available data.
This history is relevant to Lee's work because it means the only BPC-157 clinical data that has survived peer review and reached PubMed is Lee's three pilot studies. The Pliva trials and the withdrawn 2015 registration represent attempts at human evidence that, for various reasons, never reached the published literature. Lee's 30 patients are not just the only published human data; they are the only human data available for independent evaluation by the scientific community.
Financial Interests and Advocacy
Lee's roles extend well beyond the clinic. As co-founder of the Clinical Peptide Society, he has a platform for promoting peptide use in medical practice. SavePeptides.org, his nonprofit, actively campaigns against FDA restrictions on compounded peptides, including BPC-157. His book "The Fountain of Youth with Peptides" positions peptides as cutting-edge anti-aging therapeutics.
His clinic offers peptide injection services. The broader peptide market includes gray-market BPC-157 nasal sprays priced at $375 and patches at $575 per month, with an estimated user base in the hundreds of thousands. Lee operates within this ecosystem as both a researcher generating evidence and a clinician generating revenue from the same compound.
Disclosure standards in clinical research exist for this exact situation. The three published studies do not provide detailed conflict-of-interest statements addressing the revenue generated from BPC-157 prescribing, the book, or the advocacy organizations. This does not mean the data is fabricated. It means readers lack the information needed to fully evaluate potential bias. For the related supply chain and purity concerns, see BPC-157 Supply Chain.
The Regulatory Backdrop
In September 2023, the FDA added BPC-157 to its Category 2 list of bulk drug substances that may present significant safety risks. This effectively prohibited compounding pharmacies from manufacturing BPC-157 for patient use. The FDA cited potential immunogenicity, manufacturing impurities, and the absence of adequate human safety data as its rationale. For a complete regulatory analysis, see BPC-157 and the FDA.
The Department of Justice subsequently prosecuted Tailor Made Compounding LLC for distributing unapproved peptides including BPC-157, resulting in a $1.79 million forfeiture. Lee's response has been public opposition: SavePeptides.org frames the FDA's action as overreach, and Lee has appeared on podcasts arguing that BPC-157's safety profile in animal studies supports continued clinical use.
The regulatory question intersects directly with the evidence question. The FDA's position is that BPC-157 lacks sufficient human safety data. Lee's published studies total 30 patients with no control groups. Whether those 30 patients constitute "sufficient" evidence is a judgment call, but by conventional pharmaceutical standards (typically thousands of patients across Phase I-III trials), the gap is vast.
What Stronger Evidence Would Look Like
The BPC-157 field needs specific types of studies that do not yet exist.
Randomized controlled trials. Any condition where Lee reports benefit (knee pain, interstitial cystitis) could be tested against sham injection. Knee osteoarthritis trials routinely enroll 50-100 patients per arm. The IC study's refractory population would be an especially valuable RCT target given the limited treatment options.
Multicenter replication. A second clinic, independent of Lee, reproducing any of his findings would dramatically increase confidence. The entire human evidence base rests on one investigator's patients at one site.
Validated outcome measures. Standardized tools like the WOMAC index for knee osteoarthritis, or the O'Leary-Sant Interstitial Cystitis Symptom Index, would allow comparison to existing treatments and produce data that regulatory agencies can evaluate.
Imaging endpoints. MRI documentation of cartilage changes, tendon repair, or bladder wall inflammation before and after BPC-157 treatment would provide objective evidence beyond patient self-report.
Dose-response studies. Lee's studies used fixed doses without systematic dose escalation across a range of patients. Determining the minimum effective dose, the maximum tolerated dose, and the shape of the dose-response curve is a basic pharmacological requirement for any therapeutic candidate.
Long-term safety monitoring. BPC-157 promotes angiogenesis (blood vessel growth) via VEGF pathways.[8] Compounds that promote angiogenesis raise theoretical questions about tumor vascularization. A three-day safety window in 2 patients does not address this. The animal literature reports no toxicity even at high doses,[6] but animal safety data does not always predict human outcomes. For more on the angiogenesis question, see BPC-157 and Cancer Risk.
The gap between what exists and what would be needed for regulatory approval is not a criticism of Lee specifically. Clinical trials are expensive, and BPC-157's gray-market availability makes patient recruitment for placebo-controlled studies genuinely difficult when prospective subjects can obtain the compound without enrolling. But the gap exists, and acknowledging it honestly is the starting point for closing it.
The Bottom Line
Three published human clinical studies of BPC-157 exist. All come from one doctor in Orlando, Florida, totaling 30 patients with no control groups. The knee pain and interstitial cystitis results are directionally promising but fall far short of the evidence threshold needed to establish efficacy. The safety study in 2 patients provides minimal new information. Previous attempts at human BPC-157 data, including Pliva's ulcerative colitis trials and a withdrawn 2015 registration, never reached the published literature. The BPC-157 field has an enormous preclinical literature, a severe human data deficit, and a regulatory environment shaped more by advocacy and market forces than by clinical trial evidence.
Sources & References
- 1RPEP-13892·Vasireddi, Nikhil et al. (2025). “BPC-157 for Sports Injuries: What a Systematic Review of the Evidence Actually Shows.” HSS journal : the musculoskeletal journal of Hospital for Special Surgery.Study breakdown →PubMed →↩
- 2RPEP-05534·Lee, Edwin et al. (2021). “BPC-157 Knee Injections Relieved Pain in 88% of Patients Across Multiple Knee Conditions.” Alternative therapies in health and medicine.Study breakdown →PubMed →↩
- 3RPEP-00860·Staresinic, M et al. (2003). “BPC-157 Accelerates Achilles Tendon Healing and Directly Stimulates Tendon Cell Growth.” Journal of orthopaedic research : official publication of the Orthopaedic Research Society.Study breakdown →PubMed →↩
- 4RPEP-08643·Lee, Edwin et al. (2024). “BPC-157 Injection Resolved Bladder Pain Symptoms in 10 of 12 Women: A Pilot Study.” Alternative therapies in health and medicine.Study breakdown →PubMed →↩
- 5RPEP-12011·Lee, Edwin et al. (2025). “First Study of Intravenous BPC-157 in Humans Shows No Side Effects in Two Patients.” Alternative therapies in health and medicine.Study breakdown →PubMed →↩
- 6RPEP-00561·Sikiric, P (1999). “BPC-157: A Comprehensive Review of Its Healing Effects Across Multiple Organ Systems.” Inflammopharmacology.Study breakdown →PubMed →↩
- 7RPEP-11666·Józwiak, Michalina et al. (2025). “BPC 157: Comprehensive Review of the Body Protection Compound's Benefits, Safety, and Regulatory Status.” Pharmaceuticals (Basel.Study breakdown →PubMed →↩
- 8RPEP-01458·Brcic, L et al. (2009). “BPC-157 Promotes Healing Through Angiogenesis in Both Muscle and Tendon.” Journal of physiology and pharmacology : an official journal of the Polish Physiological Society.Study breakdown →PubMed →↩
- 9RPEP-05748·Seiwerth, Sven et al. (2021). “BPC-157: A Gastric Peptide That Accelerates Wound Healing Across Nearly Every Tissue Type.” Frontiers in pharmacology.Study breakdown →PubMed →↩
- 10RPEP-01366·Klicek, Robert et al. (2008). “BPC-157 (PL14736 IBD Drug) Also Heals Healing-Impaired Wounds: Expanded Clinical Potential.” Journal of pharmacological sciences.Study breakdown →PubMed →↩
- 11RPEP-00426·Seiwerth, S et al. (1997). “BPC-157 Accelerates Wound Healing by Boosting Granulation, Blood Vessels, and Collagen.” Journal of physiology.Study breakdown →PubMed →↩