Dopamine and Peptide Modulation
Reward & Motivation Peptides
12+ peptide families
At least twelve distinct peptide families converge on the brain's dopamine circuits to shape what you want, how badly you want it, and when you stop wanting it.
Jin et al., Neurochemical Research, 2023
Jin et al., Neurochemical Research, 2023
If you only read one thing
Dopamine is less about feeling good and more about wanting something. Peptides — ghrelin from your stomach, GLP-1 from your gut, endorphins from your brain, neuropeptide Y from your amygdala — are the signals that turn the wanting up or down. An empty stomach releases ghrelin, which doubles the firing of reward-circuit neurons. A meal releases GLP-1, which quiets the same circuit. Drugs like Ozempic (semaglutide) are already crossing into this territory in research: people on GLP-1 agonists report reduced interest not just in food but in alcohol and other rewards. The peptide-dopamine network is how your body ties hunger, stress, and social context to the urge to act.
Dopamine is not the "pleasure chemical." That framing, repeated across decades of pop science, misrepresents what dopamine actually does. The neurotransmitter's primary role in reward circuitry is closer to wanting than liking. It encodes the motivational salience of stimuli, the prediction errors that drive learning, and the effort calculations that determine whether you reach for the next bite or push back from the table. What controls dopamine itself? Peptides. A 2023 review in Neurochemical Research identified at least twelve peptide families that directly modulate dopamine neurons projecting from the ventral tegmental area (VTA) to the nucleus accumbens (NAc), the circuit most associated with motivated behavior.[1] For a broader look at how peptides shape brain reward processing, see our overview of how peptides control your brain's reward system.
This article maps the peptide systems that tune dopamine signaling: which ones amplify motivation, which ones suppress it, and what happens when the balance breaks down.
Key Takeaways
- Ghrelin increases dopamine neuron firing rates in the VTA by 2-fold, directly linking hunger to motivation (Abizaid et al., 2006)
- GLP-1 receptor activation reduces reward-related brain activity in the nucleus accumbens and insula in humans (van Bloemendaal et al., 2014)
- Opioid peptides (beta-endorphin, enkephalins) increase dopamine release in the nucleus accumbens by 50-100% above baseline (Spanagel et al., 1990)
- Nociceptin/orphanin FQ suppresses accumbal dopamine release, acting as a functional anti-opioid in reward circuits (Murphy et al., 1996)
- Neuropeptide Y neurons in the basolateral amygdala project directly to the nucleus accumbens to drive high-fat food intake (Yamada et al., 2025)
- Semaglutide (a GLP-1 analogue) reduced alcohol consumption in rodent models and modulated central GABA neurotransmission in reward areas (Chuong et al., 2023)
The VTA-to-NAc Circuit: Where Peptides Meet Dopamine
Who pushes which way
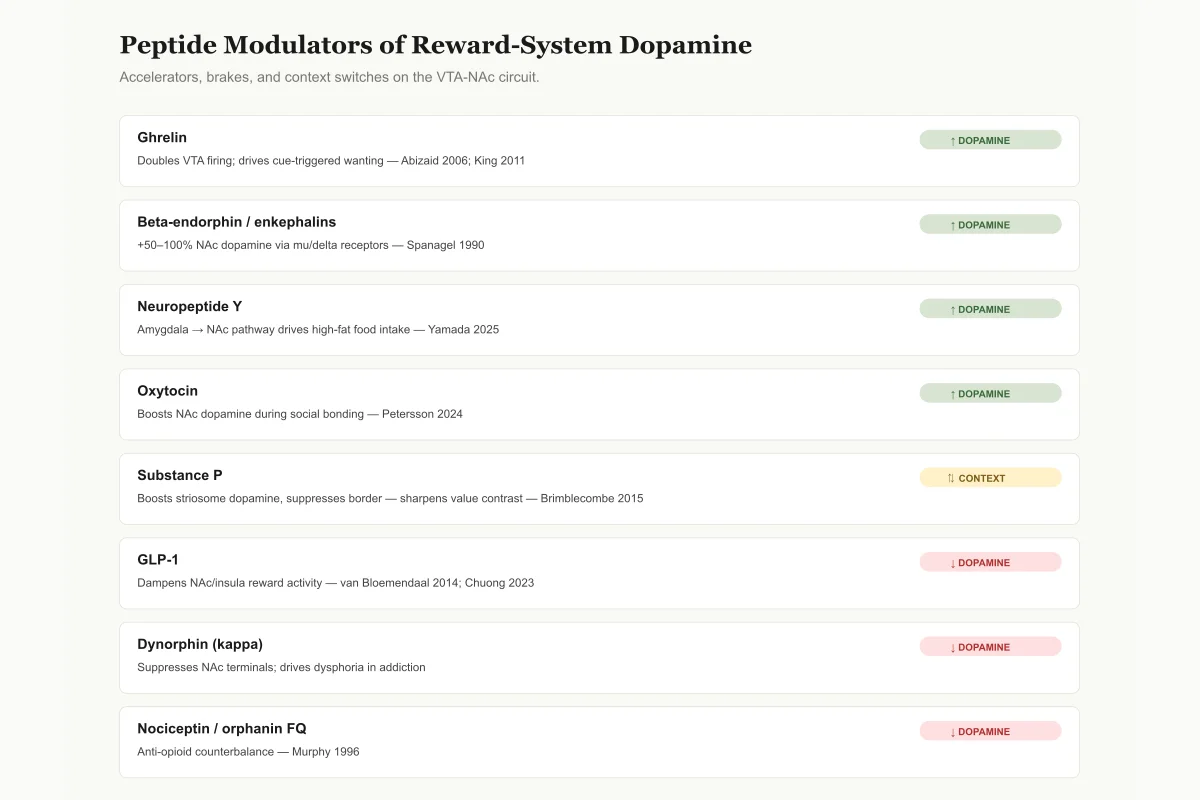
Peptide Accelerators, Brakes, and Context Switches on Dopamine
The dopamine system sums these inputs in real time. Net direction depends on which peptides are high right now.
Ghrelin
↑ DopamineDoubles VTA dopamine firing; drives cue-triggered wanting
Abizaid 2006; King 2011
Beta-endorphin / enkephalins
↑ Dopamine+50–100% NAc dopamine via mu/delta receptors (removes GABA brake)
Spanagel 1990
Neuropeptide Y (Y1/Y5)
↑ DopamineAmygdala → NAc pathway drives high-fat food intake
Tanaka 2021; Yamada 2025
Oxytocin
↑ DopamineBoosts NAc dopamine during social/bonding encounters
Petersson 2024
Substance P
⇅ Context-dependentBoosts striosome dopamine, suppresses border dopamine — sharpens value contrast
Brimblecombe 2015
GLP-1
↓ DopamineReduces NAc/insula reward activity; dampens cue-triggered wanting
van Bloemendaal 2014; Chuong 2023
Dynorphin (kappa)
↓ DopamineSuppresses NAc dopamine terminals; drives dysphoria in addiction
Classical opioid literature
Nociceptin / orphanin FQ
↓ DopamineAnti-opioid counterbalance; lowers NAc dopamine
Murphy 1996
Source: Jin (2023) and individual cited studies above
 View as image
View as imageThe mesolimbic dopamine pathway runs from the VTA in the midbrain to the NAc in the ventral striatum. This circuit does not operate in isolation. It receives convergent input from hypothalamic neurons, gut-derived hormones crossing the blood-brain barrier, and local neuropeptide-releasing interneurons. Each input adjusts dopamine release in the NAc, which in turn modulates approach behavior, effort expenditure, and reinforcement learning.
Jin et al. (2023) mapped how hypothalamic neuropeptides reach dopamine neurons through both direct synaptic connections and indirect humoral signaling.[1] Orexigenic peptides (those that promote feeding) generally increase dopamine neuron activity, while anorexigenic peptides (those that suppress feeding) generally decrease it. But "generally" carries weight here. The same peptide can have opposite effects depending on where in the circuit it acts, what receptor subtypes are expressed, and what metabolic state the organism is in.
This complexity is not a flaw. It is the mechanism by which the brain integrates metabolic need with environmental opportunity. A peptide signal arriving at a dopamine neuron is not processed in isolation. It interacts with the neuron's current firing state, the tonic dopamine level in the NAc, and the simultaneous input from other peptide and neurotransmitter systems. The output, a pattern of dopamine release in the NAc, encodes motivational value in a way that is context-dependent by design.
Ghrelin: The Hunger Signal That Hijacks Motivation
Ghrelin is a 28-amino-acid peptide released primarily by the stomach during fasting. Its receptor, GHS-R1a, is expressed on dopamine neurons in the VTA. When ghrelin reaches these neurons, it does something specific: it increases their firing rate.
Abizaid et al. (2006) demonstrated that ghrelin doubled the firing rate of VTA dopamine neurons and reorganized their synaptic inputs, increasing excitatory input while decreasing inhibitory tone.[2] This is not a subtle modulatory effect. It is a reconfiguration of the circuit's operating state, shifting the brain from "not seeking" to "actively seeking."
King et al. (2011) showed the behavioral consequence: when ghrelin was injected directly into the VTA, rats increased their motivation to obtain preferred foods, measured by how hard they would work (lever pressing) for a palatable reward.[3] Ghrelin did not change how much the rats seemed to enjoy the food once they got it. It changed how much they wanted it.
The ghrelin-dopamine connection extends beyond food. Jerlhag et al. (2011) found that blocking the ghrelin receptor (GHS-R1a) reduced nicotine-induced locomotor stimulation and dopamine release in the NAc.[4] This means ghrelin signaling amplifies dopamine responses to drugs of abuse, not just food.
Physical Receptor Coupling
The ghrelin-dopamine connection runs deeper than one receptor activating nearby neurons. Navarro et al. (2022) identified physical complexes between ghrelin receptors (GHS-R1a, GHS-R1b) and dopamine D1 receptors in the VTA.[5] These receptor heteromers create a molecular switch: when ghrelin binds, it changes how dopamine D1 receptors respond to dopamine. The implications for drug development are significant. Targeting the heteromer rather than individual receptors could modulate motivation without blunting dopamine signaling system-wide.
Abizaid (2009) reviewed the broader picture: ghrelin acts at multiple levels, from the stomach to the pituitary to the VTA, creating a coordinated hormonal signal that links energy deficit to motivated behavior through dopamine.[6] For a deeper look at how endocannabinoid and opioid systems interact with these circuits, see our article on endocannabinoid and opioid peptide interactions.
GLP-1: The Satiety Peptide That Brakes Reward
GLP-1 beyond diabetes
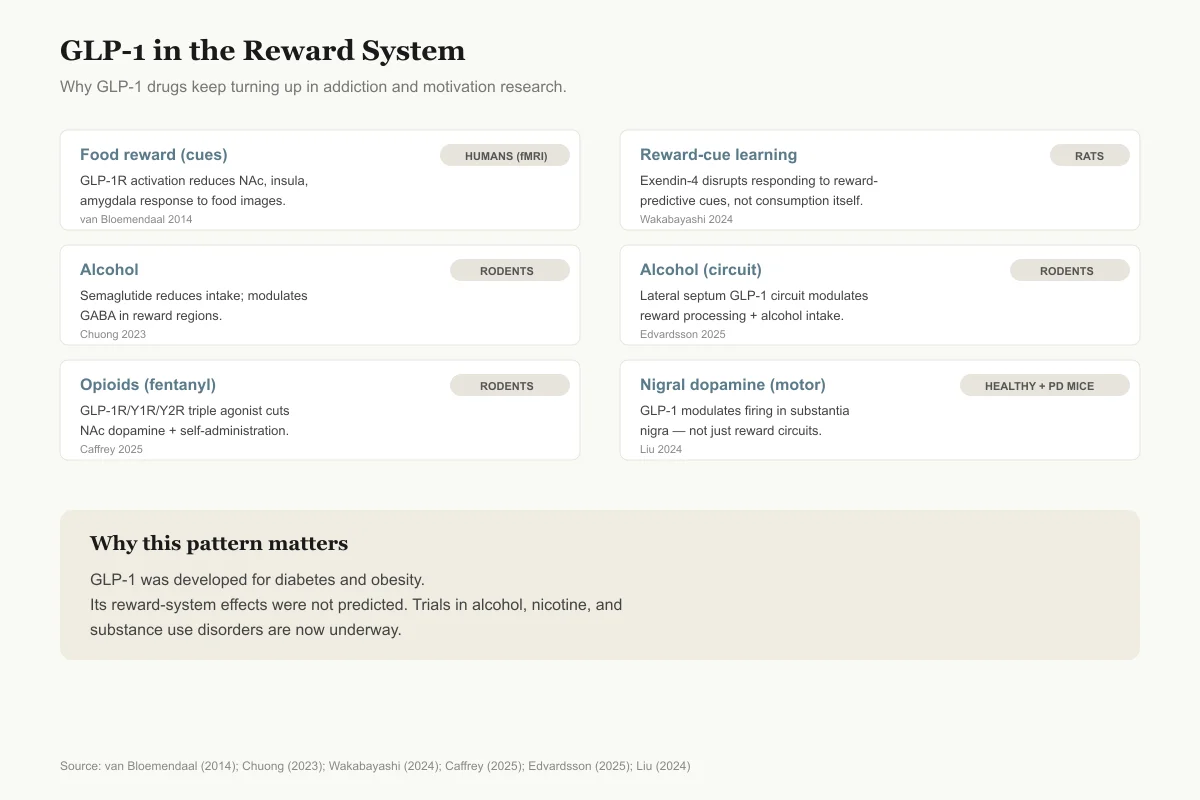
The Reward-System Evidence for GLP-1 Drugs
Why GLP-1 agonists keep showing up in addiction, motivation, and compulsive-behavior research.
Food reward (cues)
Humans (fMRI)GLP-1R activation reduces NAc, insula, amygdala response to high-calorie food images
van Bloemendaal 2014
Cue-triggered reward learning
RatsExendin-4 disrupts responding to reward-predictive cues without changing reward consumption
Wakabayashi 2024
Alcohol
RodentsSemaglutide reduces intake; modulates GABA in reward regions
Chuong 2023
Alcohol (circuit)
RodentsLateral septum GLP-1 circuit modulates reward + intake
Edvardsson 2025
Opioids (fentanyl)
RodentsGLP-1R/Y1R/Y2R triple agonist cuts NAc dopamine response + self-administration
Caffrey 2025
Motor dopamine (substantia nigra)
Healthy + parkinsonian miceGLP-1 modulates nigral dopamine firing — not limited to reward circuits
Liu 2024
GLP-1 was developed for diabetes and obesity. Its reward-system effects were not predicted — and clinical trials for alcohol, nicotine, and substance use are now underway.
Source: van Bloemendaal (2014); Chuong (2023); Wakabayashi (2024); Caffrey (2025); Edvardsson (2025); Liu (2024)
 View as image
View as imageGlucagon-like peptide-1 (GLP-1) is a 30/31-amino-acid incretin hormone released from intestinal L-cells after eating. While its metabolic effects (insulin secretion, gastric emptying) are well characterized, GLP-1's actions in the brain's reward system are a newer area of research with direct clinical relevance.
Van Bloemendaal et al. (2014) conducted a pivotal human neuroimaging study using fMRI. They showed that GLP-1 receptor activation (via the GLP-1 analogue exenatide) reduced activity in the NAc, insula, and amygdala in response to food images.[7] The effect was specific to high-calorie food stimuli and correlated with reduced food intake. GLP-1 was not simply reducing hunger signals. It was dampening the reward value of food cues.
Wakabayashi et al. (2024) extended this to reward-learning behavior. Exendin-4 (a GLP-1 analogue) disrupted how rats responded to cues that predicted rewards, without affecting the consumption of the reward itself once obtained.[8] This is a critical distinction: GLP-1 signaling appears to modulate wanting (cue-triggered motivation) rather than liking (hedonic response to the reward).
GLP-1 and Addiction
The clinical implications extend beyond appetite. Chuong et al. (2023) showed that the GLP-1 analogue semaglutide reduced alcohol consumption in rodent models, an effect associated with modulation of GABA neurotransmission in reward-processing brain regions.[9]
Two 2025 studies pushed this further. Caffrey et al. (2025) tested a novel GLP-1R/Y1R/Y2R triple agonist and found it decreased fentanyl-evoked dopamine release in the NAc and attenuated opioid self-administration.[10] Edvardsson et al. (2025) identified an inhibitory GLP-1 circuit in the lateral septum that modulates both reward processing and alcohol intake.[11]
Liu et al. (2024) showed that GLP-1 also modulated the firing activity of dopaminergic neurons in the substantia nigra, a structure primarily associated with motor control rather than reward, in both healthy and parkinsonian mice.[12] This suggests GLP-1's dopamine modulation is not limited to reward circuits.
These findings are reshaping how researchers think about GLP-1 drugs. Semaglutide and tirzepatide were developed for diabetes and obesity. Their effects on alcohol craving, drug reward, and compulsive behavior were not predicted by the metabolic framework in which they were developed.
Opioid Peptides: The Original Dopamine Modulators
The endogenous opioid system was among the first peptide systems shown to modulate dopamine release in reward circuits. Three major families of opioid peptides exist: endorphins (from pro-opiomelanocortin), enkephalins (from proenkephalin), and dynorphins (from prodynorphin). Each acts through different receptor subtypes, with different consequences for dopamine.
Spanagel et al. (1990) used in vivo microdialysis to measure dopamine release in the NAc while administering opioid peptides. Beta-endorphin and the enkephalin analogue DALA increased dopamine release by 50-100% above baseline, acting through mu and delta opioid receptors respectively.[13] The mechanism is indirect: opioid peptides inhibit GABAergic interneurons in the VTA that normally restrain dopamine neurons. Remove the brake, and the neurons fire faster.
Dynorphin, acting through kappa opioid receptors, does the opposite. It suppresses dopamine release in the NAc and produces aversive, dysphoric states. This is not a footnote. The dynorphin/kappa system is now understood to be central to the "dark side" of addiction, the negative emotional state that drives compulsive drug seeking to relieve withdrawal-induced dysphoria. Where mu-opioid activation in the VTA produces a permissive state for dopamine release (by removing inhibition), kappa-opioid activation directly inhibits dopamine terminals in the NAc. The balance between these two systems, one promoting approach and one promoting avoidance, is a core mechanism for calibrating motivated behavior.
Befort (2015) reviewed how the opioid and endocannabinoid systems interact in reward, finding that knockout of either system alters the other's contribution to dopamine-mediated reward.[14] The two systems do not operate independently. They form a cooperative network that fine-tunes the hedonic and motivational components of reward.
Nociceptin: The Anti-Opioid Peptide
Not all opioid-related peptides increase dopamine. Murphy et al. (1996) showed that nociceptin (orphanin FQ), a peptide structurally related to dynorphin but acting through its own receptor (NOP), suppressed dopamine release in the NAc.[15] This made nociceptin functionally opposite to classical opioids in the reward circuit. The nociceptin system appears to serve as an endogenous counterbalance to opioid-driven reward, a built-in check on excessive motivation.
Neuropeptide Y: Stress, Appetite, and Accumbal Dopamine
Neuropeptide Y (NPY), a 36-amino-acid peptide, is one of the most abundant neuropeptides in the brain. Its roles in anxiety, stress resilience, and appetite are well documented. Its direct modulation of dopamine in the NAc is less widely appreciated.
Tanaka et al. (2021) reviewed NPY's actions in the NAc, finding that NPY modulates dopamine-dependent behaviors including food motivation, ethanol preference, and anxiety-related responses.[16] NPY acts through Y1 and Y5 receptors in the NAc to influence the balance between approach and avoidance. Y1 receptor activation in the NAc shell increases feeding behavior, while Y5 receptor signaling modulates the dopamine response to palatable food. The combined effect is a peptide system that links the organism's stress state and energy reserves to the motivational value assigned to food and other rewards.
Yamada et al. (2025) identified a specific NPY circuit: NPY-expressing neurons in the basolateral amygdala (BLA) project directly to the NAc and stimulate high-fat food intake when activated.[17] This is not NPY acting diffusely. It is a defined anatomical pathway carrying a specific peptide signal from an emotional processing region to the brain's primary reward hub.
Thorsell (2017) reviewed NPY's role in alcohol addiction and affective disorders, finding that reduced NPY signaling is consistently associated with increased alcohol consumption and anxiety-like behavior, while restoring NPY reduces both.[18] For a detailed look at how neuropeptides drive compulsive behaviors, see our article on neuropeptides and compulsive behavior.
Oxytocin: Social Reward Through Dopamine
Oxytocin, a 9-amino-acid peptide, is often simplified as the "bonding hormone." Its connection to dopamine circuits provides a mechanism for why social interaction can be rewarding and why social isolation can be aversive.
Petersson et al. (2024) reviewed the interactions between oxytocin and dopamine systems, documenting how oxytocin modulates dopamine release in the NAc during social encounters, pair bonding, and parental behavior.[19] When oxytocin signaling is disrupted, social stimuli lose their rewarding properties. The dopamine response to a social partner diminishes, and the motivational drive to seek social contact fades.
This has implications for conditions marked by social reward deficits, including autism spectrum disorder and certain forms of depression. However, the therapeutic translation remains early-stage. Intranasal oxytocin trials have produced mixed results, partly because oxytocin's effects depend heavily on context: the same peptide can enhance or diminish social approach depending on the social environment and individual baseline.
Substance P: Compartment-Specific Dopamine Tuning
Substance P, an 11-amino-acid tachykinin peptide, modulates dopamine in a pattern that defies simple "up" or "down" descriptions. Brimblecombe and Cragg (2015) used carbon-fiber microelectrodes in mouse brain slices to map substance P's effects across the striatum's striosome-matrix compartments.[20]
The result was striking: substance P boosted dopamine release in striosome centers, diminished it at striosome-matrix borders, and had no effect in the matrix. This created a contrast enhancement pattern, sharpening the distinction between striosomal and matrix dopamine signals. The striosome compartment is enriched in circuits associated with motivation and value-based decision making. Substance P's selective enhancement of striosomal dopamine suggests it tunes the motivational component of striatal processing without altering the motor or habitual components. This compartment-specific action is unique among the peptide modulators discussed here. While ghrelin and GLP-1 shift the entire circuit's gain up or down, substance P reshapes the spatial pattern of dopamine signaling within the striatum. The functional consequence may be a sharpening of value-based decision making during states of high substance P release, such as stress or pain.
The Integration Problem: Competing Peptides, One Circuit
The dozen-plus peptide systems described above do not act sequentially. They act simultaneously. A hungry organism has elevated ghrelin, low GLP-1, high NPY, and specific opioid peptide tone, all converging on the same dopamine neurons. A satiated organism has the inverse profile. The dopamine neuron integrates these signals and produces a firing pattern that reflects the combined motivational state.
This creates a fundamental challenge for pharmacology. Blocking one peptide system to reduce pathological motivation (as in addiction) will shift the balance of all other inputs. GLP-1 agonists reduce alcohol seeking, but they also reduce food reward. Ghrelin antagonists could reduce drug craving, but they also blunt the motivational drive to eat. The clinical reports of reduced interest in food, alcohol, and compulsive behaviors during GLP-1 agonist therapy illustrate this point: the drug is not selectively targeting one form of wanting. It is reducing the dopamine system's responsiveness to reward cues broadly.
Understanding which peptide inputs are most dysregulated in a given condition could enable more targeted interventions. In alcohol use disorder, the NPY and opioid systems show distinct changes from those seen in binge eating. In compulsive gambling, the dopamine circuit responds to prediction errors without any metabolic peptide involvement. The challenge is matching the peptide pharmacology to the specific reward dysfunction.
The discovery of receptor heteromers, as with ghrelin and dopamine D1 receptors in the VTA,[5] points toward a possible solution: drugs that target specific receptor complexes rather than individual receptors. This approach could modulate one aspect of peptide-dopamine interaction (drug reward) while sparing another (food motivation). Such drugs do not yet exist outside laboratory settings, but the molecular targets have been identified.
What Remains Unknown
Several critical gaps persist. Most peptide-dopamine studies use rodent models, and the translation to human neurobiology is incomplete. The human VTA is structurally different from the rat VTA, with distinct subpopulations of dopamine neurons that may respond differently to peptide inputs. Human neuroimaging (like the GLP-1 fMRI studies) provides correlational data but cannot measure peptide concentrations at the synapse.
The temporal dynamics of peptide signaling in reward circuits are poorly characterized. Dopamine operates on a timescale of milliseconds to seconds. Peptides operate on timescales of minutes to hours. How these two timescales interact to produce coherent motivated behavior is not well understood.
The role of sex differences is understudied. Most studies cited here used male rodents. The few that included females found significant sex differences in peptide-dopamine interactions, but the data remain sparse. Estrogen modulates ghrelin receptor expression, progesterone alters opioid peptide sensitivity, and the oxytocin system shows marked sex differences in its interaction with dopamine. Any clinical application of peptide-based reward modulation will need to account for these differences.
Sagarkar et al. (2025) showed that epigenetic regulation of NPY expression in the amygdala promotes reward-seeking behavior, adding a layer of complexity: the peptide signals that modulate dopamine are themselves regulated by chromatin modifications that reflect the organism's history.[21] This means the peptide-dopamine circuit is not static. It is shaped by experience.
The Bottom Line
Dopamine's role in motivation is modulated by a network of peptides that link metabolic state, social context, and emotional processing to reward circuit function. Ghrelin amplifies wanting during hunger. GLP-1 suppresses reward drive during satiety and shows promise in reducing drug and alcohol seeking. Opioid peptides provide the hedonic overlay that distinguishes pleasure from mere desire. NPY connects stress and appetite to accumbal dopamine. Oxytocin links social experience to reward. These systems do not operate in isolation; they converge on the same neurons and produce behavior through their combined action.
Sources & References
- 1RPEP-07016·Jin, Ruijie et al. (2023). “Neuropeptides Modulate Feeding via the Dopamine Reward Pathway..” Neurochemical research.Study breakdown →PubMed →↩
- 2RPEP-01112·Abizaid, Alfonso et al. (2006). “Ghrelin Directly Activates Dopamine Reward Neurons: The Molecular Link Between Hunger and Pleasure.” The Journal of clinical investigation.Study breakdown →PubMed →↩
- 3RPEP-01792·King, S J et al. (2011). “Ghrelin in the Brain's Reward Center Increases Motivation to Get Preferred Foods.” Hormones and behavior.Study breakdown →PubMed →↩
- 4RPEP-01785·Jerlhag, Elisabet et al. (2011). “Blocking Ghrelin Receptors Reduces Nicotine's Rewarding Effects: Ghrelin in Nicotine Addiction.” Drug and alcohol dependence.Study breakdown →PubMed →↩
- 5RPEP-06393·Navarro, Gemma et al. (2022). “Complexes of Ghrelin GHS-R1a, GHS-R1b, and Dopamine D1 Receptors Localized in the Ventral Tegmental Area as Main Mediators of the Dopaminergic Effects of Ghrelin..” The Journal of neuroscience : the official journal of the Society for Neuroscience.Study breakdown →PubMed →↩
- 6RPEP-01449·Abizaid, Alfonso (2009). “Ghrelin and Dopamine: The Gut-Brain Appetite-Reward Connection.” Journal of neuroendocrinology.Study breakdown →PubMed →↩
- 7RPEP-02529·van Bloemendaal, Liselotte et al. (2014). “GLP-1 receptor activation modulates appetite- and reward-related brain areas in humans..” Diabetes.Study breakdown →PubMed →↩
- 8RPEP-09457·Wakabayashi, Ken T et al. (2024). “Exendin-4 Reduces Rats' Motivation to Respond to Reward-Predicting Cues — Implications for Addiction.” Frontiers in behavioral neuroscience.Study breakdown →PubMed →↩
- 9RPEP-06805·Chuong, Vicky et al. (2023). “Semaglutide Reduces Alcohol Drinking in Mice and Rats by Modulating Brain GABA Signaling.” JCI insight.Study breakdown →PubMed →↩
- 10RPEP-10268·Caffrey, Antonia et al. (2025). “A Triple-Acting Peptide Drug Reduced Fentanyl Use and Craving in Rats.” British journal of pharmacology.Study breakdown →PubMed →↩
- 11RPEP-10811·Edvardsson, Christian E et al. (2025). “An inhibitory GLP-1 circuit in the lateral septum modulates reward processing and alcohol intake in rodents..” EBioMedicine.Study breakdown →PubMed →↩
- 12RPEP-08736·Liu, Cui et al. (2024). “GLP-1 Peptide Boosts Firing of Dopamine Brain Cells in Both Healthy and Parkinson's Disease Mice, Suggesting Neuroprotective Mechanism.” Neuropharmacology.Study breakdown →PubMed →↩
- 13RPEP-00170·Spanagel, R et al. (1990). “Mu and Delta Opioids Boost Dopamine in the Brain's Reward Center While Kappa Opioids Reduce It.” Journal of neurochemistry.Study breakdown →PubMed →↩
- 14RPEP-02582·Befort, Katia (2015). “How the Brain's Opioid Peptides and Cannabinoid Systems Work Together in Reward and Addiction.” Frontiers in pharmacology.Study breakdown →PubMed →↩
- 15RPEP-00373·Murphy, N P et al. (1996). “Nociceptin Peptide Suppresses Dopamine in the Brain's Reward Center.” Neuroscience.Study breakdown →PubMed →↩
- 16RPEP-05805·Tanaka, Masaki et al. (2021). “The Role of Neuropeptide Y in the Nucleus Accumbens..” International journal of molecular sciences.Study breakdown →PubMed →↩
- 17RPEP-14298·Yamada, Shunji et al. (2025). “Neuropeptide Y neurons in the basolateral amygdala project to the nucleus accumbens and stimulate high-fat intake..” Frontiers in cellular neuroscience.Study breakdown →PubMed →↩
- 18RPEP-03497·Thorsell, Annika et al. (2017). “Neuropeptide Y in Alcohol Addiction and Affective Disorders..” Frontiers in endocrinology.Study breakdown →PubMed →↩
- 19RPEP-09067·Petersson, Maria et al. (2024). “How Oxytocin and Dopamine Work Together to Control Social Behavior, Reward, and Mental Health.” Biomedicines.Study breakdown →PubMed →↩
- 20RPEP-02591·Brimblecombe, Katherine R et al. (2015). “Substance P Sharpens Dopamine Signals in the Brain's Action Center.” The Journal of neuroscience : the official journal of the Society for Neuroscience.Study breakdown →PubMed →↩
- 21RPEP-13363·Sagarkar, Sneha et al. (2025). “Histone Arginine Methylation Regulates Neuropeptide Y Expression in the Basolateral Amygdala to Promote Reward-Seeking Behaviour..” Cellular and molecular neurobiology.Study breakdown →PubMed →↩