Do Collagen Supplements Work? What Trials Show
Collagen Peptides
23 RCTs analyzed
A 2025 meta-analysis of 23 randomized controlled trials found that high-quality, independently funded studies showed no significant effect of collagen supplements on skin aging.
Myung & Park, American Journal of Medicine, 2025
Myung & Park, American Journal of Medicine, 2025
If you only read one thing
Collagen supplements for skin are mostly hype. When researchers separated industry-funded studies from independent ones in 2025, the skin benefits vanished — only the studies paid for by supplement companies showed improvements. But collagen for joint pain is a different story. Multiple trials, including one with 147 college athletes, found 10 grams a day reduced joint pain. So the honest answer: strong signal for joints, weak signal for skin, and the $9 billion market sells the opposite.
Collagen supplements are a $9 billion global market built on a simple premise: swallow collagen peptides, and your body will make more collagen in skin, joints, and bones. The evidence for this claim is more complicated than either proponents or skeptics acknowledge. Some clinical trials report measurable improvements in skin hydration, joint pain, and bone density markers. Others, particularly those with rigorous methodology and independent funding, find no significant effects. The difference between these two camps is not random. It follows the money. For a broader overview of collagen peptides and bone density research, this article focuses specifically on whether supplementation produces the outcomes consumers expect.
Key Takeaways
- Collagen supplements are a $9 billion global market — and the evidence for what you're actually buying splits down a surprising line.
- When researchers pooled 23 trials in 2025, the skin benefits only showed up in studies paid for by supplement companies. Independent ones found nothing.
- For joint pain, the story is different. Trials in college athletes and osteoarthritis patients keep showing real pain reduction at 10 grams a day.
- Collagen fragments do reach your blood within 1 to 2 hours of swallowing. Whether they actually reach your face or your knee is unproven.
- The cheaper explanation: collagen may just be working as a protein source. Your skin might respond the same to a good protein-rich meal.
- Most commercial supplements sell 1 to 5 grams per serving — less than half the dose used in the trials that showed joint benefit.
- Quality varies wildly. These are not regulated like drugs, so heavy metals and mismatched doses have turned up in independent testing.
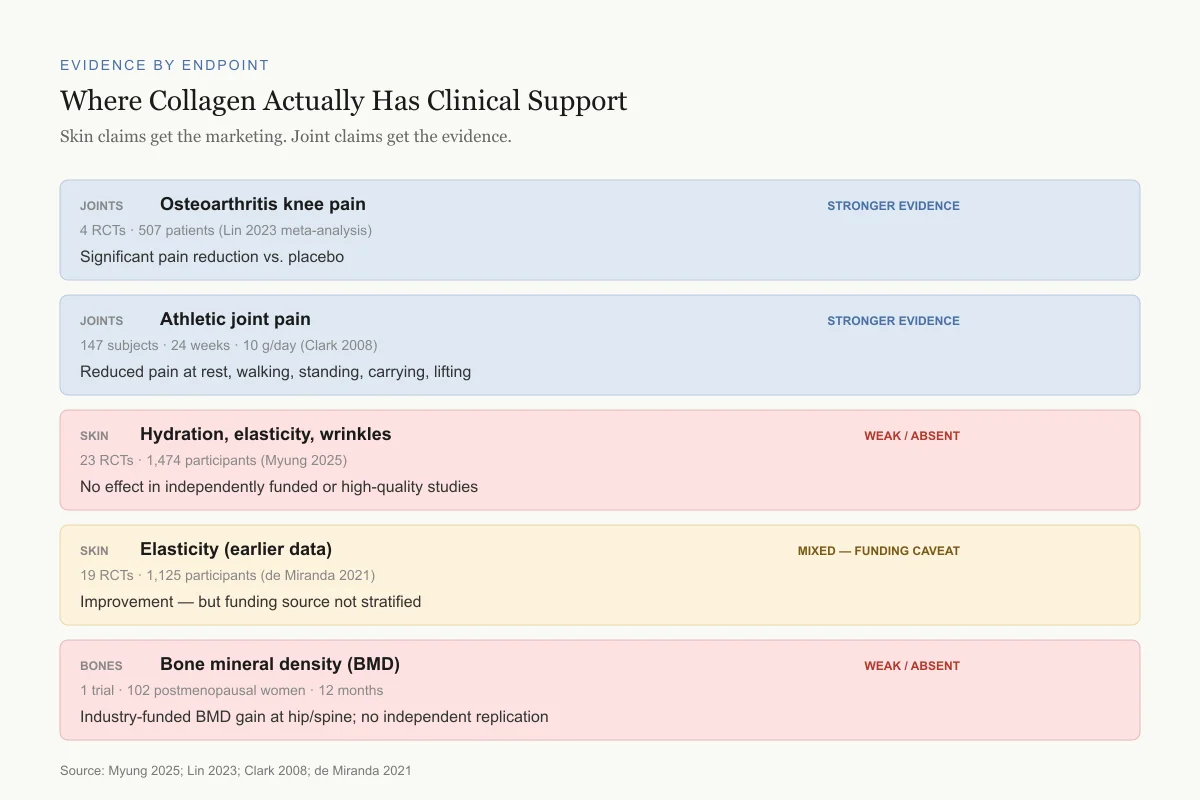
Evidence by Endpoint
Where Collagen Actually Has Clinical Support
Skin claims get the marketing. Joint claims get the evidence.
Osteoarthritis knee pain
4 RCTs · 507 patients (Lin 2023 meta-analysis)
Significant pain reduction vs. placebo
Athletic joint pain
147 subjects · 24 weeks · 10 g/day (Clark 2008)
Reduced pain at rest, walking, standing, carrying, lifting
Hydration, elasticity, wrinkles
23 RCTs · 1,474 participants (Myung 2025)
No effect in independently funded or high-quality studies
Elasticity (earlier data)
19 RCTs · 1,125 participants (de Miranda 2021)
Improvement — but funding source not stratified
Bone mineral density (BMD)
1 trial · 102 postmenopausal women · 12 months · 5 g/day
Industry-funded BMD gain at hip and spine; no replication
Source: Myung 2025; Lin 2023; Clark 2008; de Miranda 2021
 View as image
View as imageThe Funding Bias Problem
The most important collagen supplement study published in 2025 was not a trial of collagen itself. It was a meta-analysis that exposed a pattern in the existing evidence.
Myung and Park (2025), publishing in the American Journal of Medicine, systematically reviewed all 23 randomized controlled trials examining collagen supplements for skin aging, totaling 1,474 participants. When pooling all studies regardless of quality or funding, collagen supplements appeared to significantly improve skin hydration, elasticity, and wrinkles.[1]
Then they stratified by funding source. Studies funded by pharmaceutical or supplement companies showed significant benefits. Studies without industry funding showed no effect on hydration, elasticity, or wrinkles. The pattern held when they stratified by study quality: high-quality studies showed no significant effects in any category, while low-quality studies showed improvement in elasticity only.
This does not prove collagen supplements are ineffective. Industry-funded studies may use proprietary formulations that genuinely work better, or they may benefit from larger budgets that allow longer trial durations. But the correlation between who pays for the research and what the research finds is a red flag that the collagen supplement literature cannot ignore.
The Funding Split
What the 2025 Meta-Analysis Actually Found
23 RCTs · 1,474 participants · skin aging outcomes · Myung & Park, Am J Med 2025
By funding source
By study quality
The bottom line: remove industry-funded and low-quality trials from the evidence base, and the skin-aging benefit disappears entirely.
Source: Myung & Park (2025), American Journal of Medicine
 View as image
View as imageWhat Earlier Meta-Analyses Found
Before the 2025 analysis exposed the funding gap, the evidence appeared more uniformly positive.
De Miranda et al. (2021) conducted a systematic review of 19 randomized, double-blind, controlled trials with 1,125 participants aged 20-70 years. The meta-analysis found that hydrolyzed collagen supplementation for 90 days significantly improved skin hydration, elasticity, and wrinkles compared to placebo.[2]
The critical difference between this analysis and the 2025 work is that de Miranda et al. did not stratify results by funding source or study quality. The positive findings reflect the aggregate of all trials, most of which received industry funding. This does not invalidate the 2021 results, but it means they should be interpreted with the funding caveat in mind.
The Skin Evidence: Trial by Trial
The most cited individual trial is Proksch et al. (2014), a double-blind, placebo-controlled study of 69 women aged 35-55 who received 2.5 g or 5.0 g of collagen hydrolysate daily for 8 weeks. Skin elasticity improved significantly compared to placebo at both doses, and the improvement persisted four weeks after supplementation stopped. Skin moisture showed a positive trend but did not reach statistical significance.[3]
Several aspects of this trial warrant scrutiny. The sample size was small (23 per group). The collagen hydrolysate was a specific branded product (VERISOL), and the study was funded by the manufacturer (GELITA AG). The outcome measures (skin elasticity measured by cutometry) are objective, but the clinical significance of the magnitude of improvement is debatable. A statistically significant increase in a biophysical measurement does not necessarily translate to a visible or meaningful change in appearance.
Subsequent trials using different collagen products at various doses (2.5 g to 10 g daily) and durations (4 to 24 weeks) have reported improvements in skin hydration, wrinkle depth, and dermal collagen density measured by ultrasound. However, as the 2025 meta-analysis revealed, the positive findings cluster in industry-funded work.
For deeper analysis of the skin-specific evidence including elasticity, hydration, and wrinkle data, the individual trial results are examined in detail.
The Joint Pain Evidence: Stronger Ground
The joint pain literature is smaller but arguably more convincing, partly because the outcomes (pain scores, functional questionnaires) are harder to bias through measurement technique.
Clark et al. (2008) conducted the first major trial in athletes, a 24-week, double-blind, placebo-controlled study at Penn State University with 147 subjects who competed on varsity or club sports teams. Participants received 10 g of collagen hydrolysate or placebo daily. Among the 97 subjects with evaluable data, collagen hydrolysate produced statistically significant reductions in joint pain at rest (p = 0.025), when walking (p = 0.007), when standing (p = 0.011), when carrying objects (p = 0.014), and when lifting (p = 0.018) compared to placebo. In the subgroup with knee pain specifically (n = 63), effects were more pronounced across all parameters.[4]
This trial had a meaningful sample size, used validated pain assessments, and ran for 24 weeks, a duration sufficient to detect biological rather than placebo effects. The limitation: it was funded by GELITA AG, the same company that funded the Proksch skin trial.
Lin et al. (2023) pooled data from four RCTs involving 507 patients with diagnosed knee osteoarthritis and found collagen peptides produced significant pain reduction compared to placebo, with no difference in adverse event rates between groups.[5] The effect sizes were modest but consistent. The meta-analysis noted that collagen peptide supplementation at doses of 10 g/day for at least 12 weeks produced the most reliable pain outcomes.
For the full breakdown of the osteoarthritis-specific clinical data, including comparison to glucosamine and chondroitin, the joint evidence is more granular than what skin studies have produced.
How Absorption Works (and Where the Logic Gets Shaky)
The biological plausibility argument for collagen supplements goes like this: you ingest hydrolyzed collagen (broken into small peptides of 2-10 amino acids); these peptides are absorbed through the intestinal wall into the bloodstream; they reach target tissues (skin, joints, bones) where they stimulate fibroblasts and chondrocytes to produce new collagen.
The first two steps are supported by evidence. Hydrolyzed collagen peptides, primarily proline-hydroxyproline (Pro-Hyp) dipeptides, reach measurable blood concentrations within 1-2 hours of ingestion, peaking at 20-60 nmol/mL plasma. Approximately 90% of hydrolyzed collagen is absorbed, far higher than intact collagen protein. These fragments are detectable in the bloodstream as peptides, not just free amino acids, meaning they survive digestion relatively intact.
The third step, where blood-borne collagen peptides stimulate tissue-specific collagen production, is where the evidence thins. In vitro studies show that Pro-Hyp and other collagen-derived peptides can stimulate fibroblast proliferation and hyaluronic acid synthesis in cell culture. But cells in a dish respond to concentrations and conditions that may not reflect what happens in living tissue. No human study has directly demonstrated that oral collagen peptides reach a specific tissue (say, the dermis of facial skin or the cartilage of the knee) in concentrations sufficient to stimulate collagen synthesis. For more on the absorption and bioavailability science, the pharmacokinetic data is reviewed in detail.
The alternative explanation for any benefits: collagen hydrolysate simply provides amino acids (glycine, proline, hydroxyproline) that are rate-limiting for collagen synthesis in people with suboptimal protein intake. If this is the mechanism, collagen supplements work through the same pathway as any high-quality protein source, not through a collagen-specific signaling effect. This distinction matters because glycine and proline are available from many cheaper protein sources (bone broth, gelatin, meat, eggs).
Mechanism Audit
The Biological Plausibility Chain — Where It Holds and Where It Breaks
The argument has four links. The last two are weaker than the marketing suggests.
Step 1Ingestion of hydrolyzed collagen
Proven2–10 amino acid peptides enter the stomach — roughly 90% absorbed, far higher than intact collagen protein.
Step 2Di-peptides reach the bloodstream
ProvenPro-Hyp and similar fragments appear in plasma at 20–60 nmol/mL, peaking 1–2 hours after ingestion.
Step 3Peptides reach target tissues
Weak evidenceIn vitro: fragments can stimulate fibroblasts and chondrocytes in a dish. In vivo in humans: not directly measured.
Step 4Tissue makes new collagen via signaling
Unproven in humansThe collagen-specific signaling hypothesis — the reason people pay a premium for collagen over regular protein. Not confirmed in human tissue.
The alternative story: collagen supplements work because they're a high-quality protein source rich in glycine and proline — not because the peptides "signal" anything collagen-specific. If that's the real mechanism, bone broth, gelatin, and meat would work the same way, for less money.
Source: Collagen peptide pharmacokinetic literature; in vitro fibroblast studies
 View as image
View as imageWhat Collagen Types and Sources Matter
The supplement market distinguishes between Type I (skin, bones), Type II (cartilage), and Type III (blood vessels, organs) collagen, and between bovine, marine, porcine, and chicken sources. Some of these distinctions have biological basis; many are marketing. The clinical trials showing positive results have used various sources and types, and no head-to-head comparison has demonstrated superiority of one type over another for any specific outcome. For a full comparison of marine collagen vs bovine collagen, the source question is less settled than marketing suggests.
The molecular weight (degree of hydrolysis) may matter more than the source. More hydrolyzed products (lower molecular weight peptides) have better absorption. But "more hydrolyzed" also means the peptide fragments are less collagen-specific, further weakening the argument that these are signaling molecules rather than amino acid sources.
Bone Density: The Least Studied Claim
A small number of trials have examined collagen peptides for bone mineral density, primarily in postmenopausal women. The largest (102 participants, 12 months, 5 g/day specific collagen peptides) reported increased bone mineral density at the femoral neck and lumbar spine compared to placebo, along with favorable shifts in bone formation and resorption markers. These results are preliminary: the trial was industry-funded, the sample was small, and no independent replication exists. Animal studies consistently show collagen peptides increase bone formation markers and reduce bone loss, but animal-to-human translation in bone biology is notoriously unreliable. The bone density claim is the weakest of the three major collagen supplement categories (skin, joints, bones) because it rests on the fewest and smallest human trials.
The Dose Question
Effective doses in clinical trials have ranged from 2.5 g to 15 g daily, with most joint pain trials using 10 g/day and most skin trials using 2.5-5 g/day. Many commercial supplements contain 1-5 g per serving. Whether the lower commercial doses produce meaningful effects has not been rigorously tested. For a detailed analysis of what dosing studies actually show, the gap between trial doses and commercial product doses is substantial.
Who Might Actually Benefit
The evidence, with all its caveats, suggests collagen supplements are most likely to produce detectable effects in:
People with joint pain from osteoarthritis or athletic overuse. The joint evidence is more consistent across funding sources than the skin evidence, and the effect sizes (pain reduction on validated scales) are clinically meaningful even if modest.
Older adults with low protein intake. If collagen supplements are primarily working as a glycine/proline source, people with inadequate protein consumption would be the most likely responders.
Individuals with specific nutrient deficiencies. Collagen synthesis requires vitamin C, zinc, and copper as cofactors. Supplementing collagen in someone with adequate cofactors is different from supplementing in someone who is deficient. No trial has stratified results by baseline nutritional status.
The evidence does not support collagen supplements as anti-aging treatments for skin in the general population. The 2025 Myung meta-analysis found no effect in high-quality, independently funded studies. This does not mean individual consumers will never notice a difference, but it means the claimed effects cannot be distinguished from placebo in rigorous testing.
The gap between consumer expectation and clinical evidence is wide. Collagen supplement marketing implies visible anti-aging effects. The clinical data, when stripped of industry influence, does not support that claim. The joint pain data is more credible, but consumers purchasing collagen for skin rejuvenation are the largest market segment, and they are the ones least served by the evidence. For how elastin peptides complement collagen in aging skin biology, the structural protein story extends beyond collagen alone.
The Regulation Gap
Collagen supplements are classified as dietary supplements, not drugs, in most jurisdictions. This means they do not require proof of efficacy before sale, are not required to contain what the label claims, and are not routinely tested for contaminants. Independent testing has found collagen products with heavy metal contamination (particularly from marine-sourced collagen), amino acid profiles that differ from labeled claims, and hydroxyproline content inconsistent with the stated collagen quantity.
The gap between clinical trial conditions (pharmaceutical-grade collagen hydrolysate, verified dose, controlled administration) and consumer products (variable quality, unverified dose, no manufacturing oversight) means that even if clinical trials show real effects, the product a consumer purchases may not replicate those conditions. Cross-referencing with copper peptides in skincare, the supplement quality problem is not unique to collagen.
Safety
ModerateQuality varies sharply — and testing is not required
Concern
Collagen supplements are classified as dietary supplements, not drugs. They are not required to contain what the label claims, and they are not routinely tested for contamination. Independent testing has found heavy metals in marine-sourced collagen, mismatched amino acid profiles, and hydroxyproline content inconsistent with the labeled collagen quantity.
What the research says
Buy from brands that publish third-party test results for purity, heavy metals (especially for marine-sourced), and actual collagen content. Recognize that even verified products may not match the pharmaceutical-grade hydrolysate used in clinical trials.
Particularly relevant for: Anyone taking daily collagen — especially marine-sourced for people who monitor heavy-metal intake
What to do
Check for NSF Certified for Sport, Informed-Choice, or USP verification. If none is available, consider the dose you're actually receiving may be lower (or higher) than stated.
Independent supplement testing surveys; FDA dietary supplement framework
The Bottom Line
The collagen supplement evidence is split along a fault line: who paid for the research. Industry-funded trials consistently show benefits for skin hydration, elasticity, and wrinkles. Independently funded, high-quality studies show no significant effects on skin aging. The joint pain evidence is more consistent, with multiple RCTs and meta-analyses showing modest but significant pain reduction in osteoarthritis and athletic populations at 10 g/day doses. Collagen peptides are absorbed into the bloodstream as di- and tripeptides, but whether they stimulate collagen synthesis in target tissues or simply provide amino acids is unresolved. The $9 billion supplement market has outpaced the science.
Sources & References
- 1RPEP-12695·Myung, Seung-Kwon et al. (2025). “Collagen Supplements for Skin Aging: The Positive Results Disappear When You Remove Industry Funding Bias.” The American journal of medicine.Study breakdown →PubMed →↩
- 2RPEP-05335·de Miranda, Roseane B et al. (2021). “Effects of hydrolyzed collagen supplementation on skin aging: a systematic review and meta-analysis..” International journal of dermatology.Study breakdown →PubMed →↩
- 3RPEP-02478·Proksch, E et al. (2014). “Collagen Peptide Supplements Improve Skin Elasticity in a Placebo-Controlled Trial.” Skin pharmacology and physiology.Study breakdown →PubMed →↩
- 4RPEP-01324·Clark, Kristine L et al. (2008). “24-Week study on the use of collagen hydrolysate as a dietary supplement in athletes with activity-related joint pain..” Current medical research and opinion.Study breakdown →PubMed →↩
- 5RPEP-07117·Lin, Chun-Ru et al. (2023). “Analgesic efficacy of collagen peptide in knee osteoarthritis: a meta-analysis of randomized controlled trials..” Journal of orthopaedic surgery and research.Study breakdown →PubMed →↩