Collagen Peptides for Bone Density: The Research
Collagen Peptides
+4.2% spine BMD
In a 12-month RCT, postmenopausal women taking 5 g of specific collagen peptides daily had 4.2% higher spine BMD and 7.7% higher femoral neck BMD compared to placebo.
König et al., Nutrients, 2018
König et al., Nutrients, 2018
If you only read one thing
Collagen peptides can nudge bone density up in postmenopausal women. In the main year-long trial, 5 grams a day raised spine density about 4% and hip density about 8% compared to a placebo group that held flat. The effect is real but smaller than prescription osteoporosis drugs, and no study has shown it prevents actual fractures yet. Think of it as a gentle assist to the bone's collagen scaffold — helpful, not a replacement for medication when treatment is needed.
Bone is roughly one-third collagen by weight. Type I collagen forms the organic scaffold onto which calcium and phosphate minerals are deposited, creating the composite material that gives bone both flexibility and strength. When bone is lost in osteoporosis, both the mineral content and the collagen scaffold degrade. Standard osteoporosis treatments target mineral density directly (bisphosphonates, denosumab) or stimulate bone formation through hormonal pathways (abaloparatide, teriparatide). Collagen peptide supplementation targets a different question: can providing the building blocks of bone's organic matrix support density from the structural side?
The answer from clinical trials is cautiously positive. A 2018 randomized controlled trial demonstrated increased bone mineral density in postmenopausal women after 12 months of collagen peptide supplementation.[1] A 2025 meta-analysis confirmed moderate but consistent BMD improvements across multiple trials, particularly when collagen peptides were combined with calcium and vitamin D.[2] The effect sizes are smaller than pharmaceutical interventions, but the safety profile is favorable, and the mechanism addresses a different aspect of bone biology.
Key Takeaways
- In a 12-month RCT of 131 postmenopausal women, 5 g/day of specific collagen peptides increased spine BMD by 4.2% and femoral neck BMD by 7.7% compared to placebo[1]
- The bone formation marker P1NP increased in the collagen group while the bone resorption marker CTX-1 increased in the placebo group, indicating a favorable shift in bone turnover[1]
- A 2025 meta-analysis found standardized mean differences in BMD of 0.40-0.58 favoring collagen peptide supplementation, particularly when combined with calcium and vitamin D[2]
- Collagen peptides stimulate osteoblast differentiation and increase the OPG/RANKL ratio, potentially reducing bone resorption while promoting formation[3]
- Hydroxyproline-containing dipeptides (Pro-Hyp, Hyp-Gly) are the bioactive metabolites that reach bone tissue after oral collagen intake[4]
- Most trials use 5-15 g daily doses of hydrolyzed collagen; no serious adverse events have been reported across published bone density studies
How Collagen Relates to Bone Biology
Bone is a dynamic tissue composed of two major components. The inorganic component (approximately 65-70% of bone weight) consists primarily of hydroxyapatite crystals, the calcium phosphate mineral that provides rigidity and compressive strength. The organic component (approximately 30-35% of bone weight) is predominantly type I collagen, which forms a flexible fibrillar scaffold that provides tensile strength and fracture resistance.
This dual structure is essential. Bone without adequate mineral is too flexible (as in rickets). Bone without adequate collagen is too brittle (as in osteogenesis imperfecta). Age-related bone loss involves deterioration of both components, but most diagnostic tools (DEXA scans) and most treatments focus on the mineral side.
Bone turnover is maintained by two cell types working in balance. Osteoblasts build new bone by first secreting collagen fibrils (primarily type I collagen) that form the organic matrix (osteoid), then facilitating mineral deposition onto this scaffold. Osteoclasts break down existing bone through acidification and enzymatic degradation of both the mineral and collagen components. In osteoporosis, osteoclast activity exceeds osteoblast activity, resulting in net bone loss.
The hypothesis behind collagen peptide supplementation is that providing specific collagen-derived peptides can shift this balance by stimulating osteoblast activity, inhibiting osteoclast-mediated resorption, or both. For a primer on what happens when you consume collagen supplements and how they're absorbed, see Hydrolyzed Collagen: How Your Body Absorbs Collagen Peptides. For the broader question of whether collagen supplements produce measurable effects, see Do Collagen Supplements Work? What Clinical Trials Actually Show.
The König Trial: The Landmark Bone Density RCT
The most cited study on collagen peptides and bone density is the 2018 randomized, placebo-controlled, double-blinded trial by König and colleagues, published in Nutrients.[1]
Study Design
131 postmenopausal women with age-related reduction in BMD were enrolled. Mean age was 64.3 years (SD 7.2). Mean T-score at the spine was -2.4 (osteopenia/osteoporosis range). Participants were randomized to receive 5 g of specific collagen peptides (SCP) or placebo daily for 12 months. The primary endpoint was change in BMD at the femoral neck and lumbar spine.
Results
102 women completed the study, but all 131 were included in the intention-to-treat analysis:
- Spine BMD: T-score increased by +0.1 in the SCP group versus -0.03 in the control group (p=0.030). This translates to the SCP group having 4.2% higher BMD than controls at 12 months.
- Femoral neck BMD: T-score increased by +0.09 in the SCP group versus -0.01 in the control group (p=0.003). The SCP group had 7.7% higher femoral neck BMD than controls.
- Bone formation marker (P1NP): Increased in the SCP group (p=0.007), indicating enhanced bone formation.
- Bone resorption marker (CTX-1): Increased in the control group (p=0.011) but not in the SCP group, suggesting the placebo group continued normal age-related bone degradation while the collagen group did not.
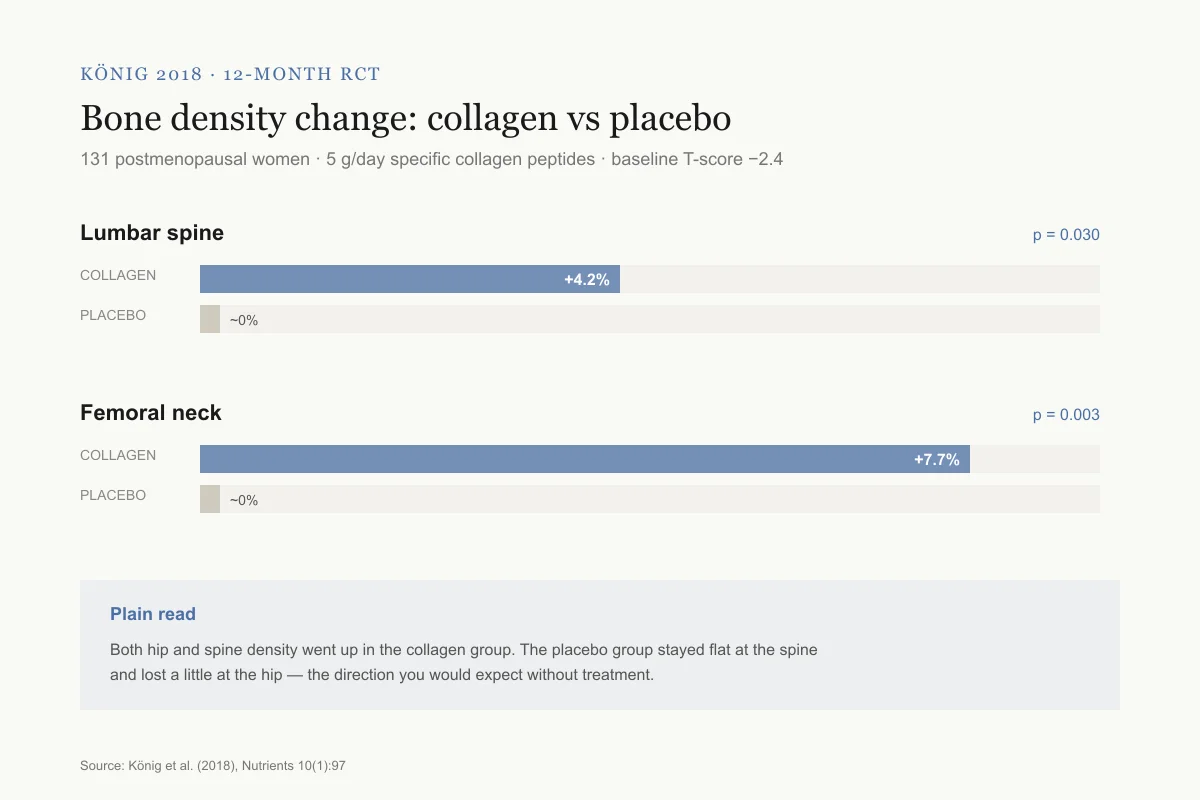
König 2018 · 12-month RCT
Bone density change: 5 g collagen peptides vs placebo
131 postmenopausal women, mean age 64, baseline T-score −2.4
Lumbar spine
p = 0.030+4.2% vs placebo — a meaningful gain in a group that usually loses 1–2% per year.
Femoral neck
p = 0.003+7.7% vs placebo — a meaningful gain in a group that usually loses 1–2% per year.
Plain read: Both hip and spine density went up in the collagen group. The placebo group stayed flat at the spine and lost a little at the hip, the direction you would expect without treatment.
Source: König et al. (2018), Nutrients 10(1):97
 View as image
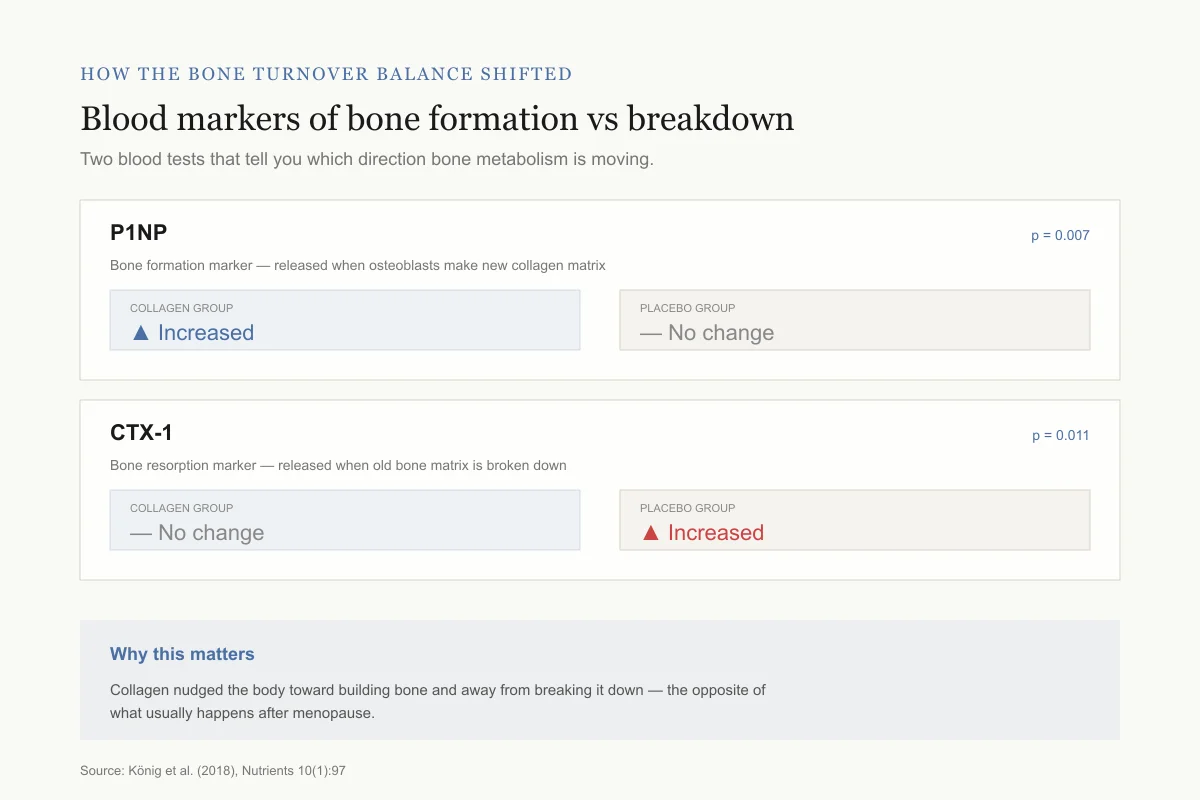
View as imageHow the bone turnover balance shifted
Blood markers of bone formation vs breakdown at 12 months
Two blood tests that tell you which direction bone metabolism is moving.
P1NP
p = 0.007Bone formation marker — released when osteoblasts make new collagen matrix
Collagen group
Placebo group
What it means: Collagen group was building new bone faster
CTX-1
p = 0.011Bone resorption marker — released when old bone matrix is broken down
Collagen group
Placebo group
What it means: Placebo group kept losing bone; collagen group held the line
Why this matters: The markers tell the same story as the density scans. Collagen nudged the body toward building bone and away from breaking it down — the opposite of what usually happens after menopause.
Source: König et al. (2018), Nutrients 10(1):97
 View as image
View as imageInterpretation
The results indicate that collagen peptide supplementation produced a favorable shift in bone metabolism: increasing formation markers while preventing the rise in resorption markers seen in the control group. The BMD improvements were statistically significant at both clinically relevant sites (spine and femoral neck).
The limitations are real. This was a single-center study with 131 participants and moderate dropout (22%). The participants were aware they were taking a supplement (though not whether active or placebo). No fracture outcomes were assessed; the study measured surrogate endpoints (BMD and markers), not clinical events.
The effect sizes deserve context. A 4.2% improvement in spine BMD over 12 months is meaningful in the context of age-related bone loss, where postmenopausal women typically lose 1-2% of spine BMD per year without intervention. The collagen peptide group not only halted this expected decline but reversed it. The 7.7% improvement at the femoral neck is particularly notable because femoral neck fractures (hip fractures) carry the highest morbidity and mortality of osteoporotic fractures. However, BMD improvement does not automatically translate to fracture prevention, and the relationship between BMD change and fracture risk reduction is not linear.
The bone marker data provided mechanistic support for the BMD findings. P1NP (procollagen type I N-terminal propeptide) is a specific marker of new collagen synthesis by osteoblasts, so its increase in the collagen group suggests that the supplementation stimulated new bone matrix formation rather than simply preventing mineral loss. The increase in CTX-1 (a collagen degradation product) in the control group but not the collagen group suggests that collagen supplementation may also have slowed the rate of bone matrix breakdown.
Additional Bone Density Trials
The König trial is not the only evidence. Multiple subsequent studies have examined collagen peptides and bone outcomes.
Calcium and Vitamin D Combination (2025)
Duangjai and colleagues examined whether adding collagen peptides to calcium and vitamin D supplementation improved outcomes beyond calcium and vitamin D alone in postmenopausal women with osteopenia. The combination group showed greater BMD improvements than calcium and vitamin D alone, and also demonstrated skin elasticity benefits, suggesting systemic effects from collagen supplementation.[5]
Type-Specific Collagen Supplementation (2025)
Genc and colleagues investigated different collagen types for bone and joint outcomes. Their study compared type 1/type 3 collagen peptides with type 2 hydrolyzed collagen, finding that both improved bone markers and joint function but through different mechanisms, consistent with the distinct tissue distributions of collagen types.[6] For more on how collagen types differ, see Type I, II, and III Collagen: What's the Difference and Does It Matter?.
Hydrolyzed Collagen Peptide Safety Trial (2025)
Demir-Dora and colleagues evaluated the efficacy and safety of a type I and type III hydrolyzed collagen peptide product in a clinical trial setting, reporting no serious adverse events and improvements in musculoskeletal parameters. The study added to the safety evidence for commercial collagen peptide products at standard doses.[7]
The 2025 Meta-Analysis
Sun and colleagues published a meta-analysis in Frontiers in Nutrition in 2025, pooling data from randomized controlled trials examining collagen peptide supplementation on bone and muscle health.[2]
The meta-analysis found:
- Standardized mean differences in BMD (both volumetric and areal) of 0.40-0.58 favoring collagen supplementation, reflecting moderate but clinically relevant effects
- Bone turnover markers shifted in a direction consistent with enhanced formation and/or reduced resorption
- Effects were strongest when collagen peptides were combined with calcium and vitamin D
- The combination of collagen with established bone-supportive nutrients appeared to produce additive benefits
The meta-analysis confirmed the direction and approximate magnitude of effects seen in the König trial, while acknowledging that the total evidence base remains relatively small compared to pharmaceutical osteoporosis interventions.
Several features of the pooled analysis deserve attention. First, trial durations ranged from 12 weeks to 12 months, and longer trials tended to show larger effects, suggesting that collagen peptide benefits on bone may be cumulative over time. Second, the muscle health outcomes were analyzed alongside bone outcomes, and collagen supplementation showed modest benefits for lean mass preservation in older adults, an important consideration because muscle and bone health are mechanically and metabolically linked. Third, the heterogeneity across trials (different products, doses, populations, and outcome measures) limits the precision of the pooled estimates. The 0.40-0.58 standardized mean difference should be interpreted as an approximate range rather than a precise effect size.
A key question the meta-analysis could not fully address is the minimum effective dose and duration. Most included trials used 5 g/day for at least 6 months, but whether lower doses or shorter durations produce clinically meaningful bone effects remains unclear. Similarly, whether the BMD improvements translate to reduced fracture risk is the central unanswered question in this literature.
How Collagen Peptides Affect Bone Cells
The mechanism by which ingested collagen peptides influence bone metabolism has been investigated in both cell culture and animal models.
Osteoblast Stimulation
In vitro studies have demonstrated that specific collagen-derived peptides, particularly those containing hydroxyproline (Hyp), stimulate osteoblast differentiation and activity. Wang and colleagues showed that antioxidant collagen peptides promote osteogenic activity in MC3T3-E1 pre-osteoblast cells through activation of ERK/MAPK signaling pathways, increasing expression of osteogenic markers including alkaline phosphatase, osteocalcin, and type I collagen.[3]
Osteoclast Inhibition
Collagen peptides also appear to influence osteoclast activity indirectly. Studies have shown that specific collagen peptides increase the ratio of osteoprotegerin (OPG) to RANKL (receptor activator of nuclear factor kappa-B ligand) in osteoblasts. This ratio is a key regulator of osteoclast differentiation: higher OPG/RANKL ratios inhibit osteoclast formation and bone resorption. This dual action (stimulating formation while inhibiting resorption) aligns with the bone marker data from clinical trials, where P1NP (formation) increased while CTX-1 (resorption) did not.
Bioavailability: What Reaches Bone Tissue
A critical question for oral supplementation is whether ingested collagen peptides survive digestion and reach target tissues in bioactive form. Virgilio and colleagues characterized the bioavailability of amino acids and bioactive peptides from collagen hydrolysate, identifying prolyl-hydroxyproline (Pro-Hyp) and hydroxyproline-glycine (Hyp-Gly) as the primary dipeptides that enter circulation after oral collagen intake.[4]
These hydroxyproline-containing dipeptides are resistant to further enzymatic degradation in blood and have been detected in bone tissue after oral administration in animal studies. They are the candidates for the direct osteoblast-stimulating effects observed in cell culture experiments. Importantly, hydroxyproline is almost unique to collagen in the human diet, so these peptides are specific markers of collagen intake and metabolism.
The pharmacokinetics of collagen peptide absorption follow a predictable pattern. After oral intake of hydrolyzed collagen, plasma levels of Pro-Hyp and Hyp-Gly peak within 1-2 hours and return to baseline within 4-6 hours. This rapid absorption and clearance is consistent with collagen peptides acting as signaling molecules that trigger cellular responses rather than accumulating as structural building blocks. The body does not take ingested collagen and directly insert it into bone matrix. Instead, the bioactive peptide fragments signal osteoblasts to increase their own collagen production and matrix mineralization activity.
For more on collagen absorption and bioavailability, see Hydrolyzed Collagen: How Your Body Absorbs Collagen Peptides. For the question of how much to take, see How Much Collagen Do You Need? What Dosing Studies Show.
Collagen Peptides Versus Pharmaceutical Osteoporosis Treatments
The BMD improvements from collagen peptide supplementation are modest compared to pharmaceutical interventions:
| Intervention | Spine BMD Change | Time Frame |
|---|---|---|
| Collagen peptides (5 g/day) | +3-7% vs placebo | 12 months |
| Alendronate (bisphosphonate) | +5-8% absolute | 3 years |
| Denosumab | +6-9% absolute | 3 years |
| Teriparatide/Abaloparatide | +9-13% absolute | 18-24 months |
| Romosozumab | +13-15% absolute | 12 months |
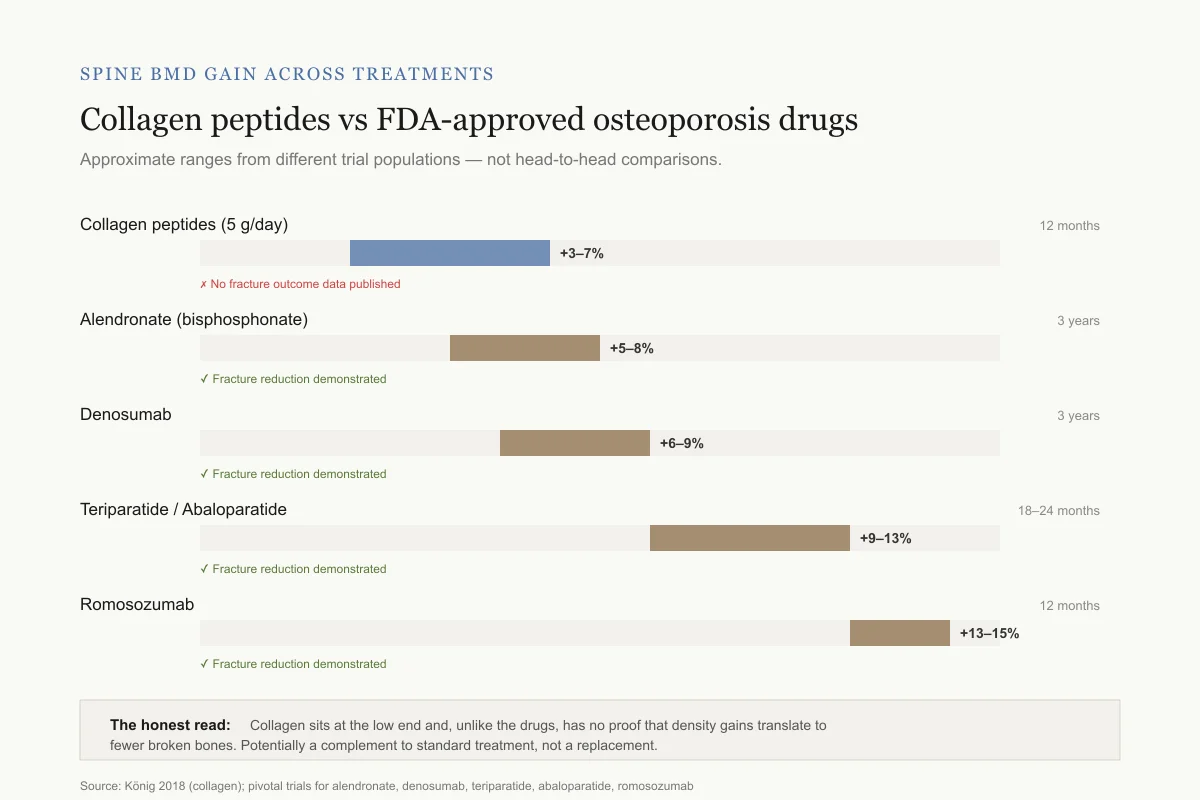
Spine BMD gain across treatments
Collagen peptides vs FDA-approved osteoporosis drugs
Approximate ranges from different trial populations — not head-to-head comparisons.
Collagen peptides (5 g/day)
12 months✗ No fracture outcome data published
Alendronate (bisphosphonate)
3 years✓ Fracture reduction demonstrated in large trials
Denosumab
3 years✓ Fracture reduction demonstrated in large trials
Teriparatide / Abaloparatide
18–24 months✓ Fracture reduction demonstrated in large trials
Romosozumab
12 months✓ Fracture reduction demonstrated in large trials
The honest read: Collagen peptides sit at the low end of this range and, unlike the drugs, have no proof that the density gains translate to fewer broken bones. They target a different part of bone (the collagen scaffold) — potentially a complement to standard treatment, not a replacement.
Source: König 2018 (collagen); pivotal trials for alendronate, denosumab, teriparatide, abaloparatide, romosozumab
 View as image
View as imageThese comparisons are approximate and drawn from different trial populations. The pharmaceutical agents are FDA-approved for osteoporosis with demonstrated fracture reduction. Collagen peptides are food supplements with no FDA approval for any medical indication and no published fracture outcome data.
However, the comparison is not strictly either/or. Collagen peptides target the organic matrix of bone, while most pharmaceuticals target the mineral component or hormonal regulation. Some clinicians and researchers have proposed that collagen supplementation could complement standard osteoporosis treatment, addressing different aspects of bone quality simultaneously. No combination trial has been published.
For more on peptide-based osteoporosis treatment, see Abaloparatide: The Newer PTH Analog for Osteoporosis.
What the Evidence Does Not Show
Several important limitations apply to the collagen peptide bone density literature.
No fracture outcome data. No published trial has demonstrated that collagen peptide supplementation reduces fracture risk. BMD is a surrogate endpoint that correlates with fracture risk, but the correlation is not perfect, and regulatory approval for osteoporosis drugs requires fracture outcome data.
Limited sample sizes. The König trial enrolled 131 women. Even the 2025 meta-analysis pooled a relatively small total number of participants. Pharmaceutical osteoporosis trials enroll thousands to tens of thousands.
Predominantly postmenopausal women. Most bone density trials studied postmenopausal women, the highest-risk group. Whether collagen peptide supplementation benefits other populations (men with osteoporosis, glucocorticoid-induced bone loss, younger adults) is largely untested.
Dosing varies across studies. Most trials used 5 g/day, but doses range from 2.5 to 15 g. The optimal dose for bone effects is not established.
Product heterogeneity. "Collagen peptides" is not a single product. Hydrolysate molecular weight, source species (bovine, porcine, marine), collagen type (I, II, III), and processing methods vary. The König trial used a specific branded product (FORTIBONE). Results may not generalize to all collagen supplements on the market. For more on source differences, see Marine Collagen vs Bovine Collagen: How Sources Compare.
Blinding challenges. Collagen peptide supplements have a distinctive taste and texture that may compromise blinding in some studies, potentially inflating effect sizes through expectation effects.
Industry funding. Several key trials, including the König study, were funded by collagen peptide manufacturers (GELITA AG). Industry funding does not invalidate results, but it is a relevant consideration when evaluating the literature. The absence of large independent academic trials funded by public health agencies is a gap that limits the strength of the evidence base.
No head-to-head comparisons with pharmaceutical agents. No trial has directly compared collagen peptides with bisphosphonates, denosumab, or other approved osteoporosis treatments. This makes it impossible to directly quantify relative efficacy from the same patient population.
Bone quality versus bone quantity. BMD measured by DEXA captures mineral density but does not fully characterize bone quality, which includes microarchitecture, collagen cross-linking, and material properties. It is theoretically possible that collagen peptides improve bone quality (particularly the organic matrix component) in ways not fully captured by standard BMD measurements. However, this remains speculative without dedicated bone quality imaging studies (e.g., high-resolution peripheral quantitative CT or bone turnover marker panels beyond P1NP and CTX-1).
Collagen Beyond Bone: The Broader Picture
The collagen peptide evidence extends well beyond bone density. The same class of supplements has been studied for joint health, skin elasticity, and muscle mass preservation, creating a broad evidence base for what is essentially a food-derived supplement. For the joint evidence, see Collagen for Joint Pain: The Osteoarthritis Clinical Data. For skin effects, see Collagen Peptides for Skin: Elasticity, Hydration, and Wrinkle Evidence.
The shared mechanism across tissues is that hydroxyproline-containing peptides from digested collagen reach target cells and stimulate tissue-specific matrix production. In bone, they promote osteoblast activity. In cartilage, they stimulate chondrocytes. In skin, they activate fibroblasts. The tissue specificity comes from the target cell's response, not from the peptide itself.
The Bottom Line
Clinical trial evidence demonstrates that 5 g/day of specific collagen peptides increases spine and femoral neck BMD in postmenopausal women, with a favorable shift in bone turnover markers toward formation and away from resorption. A 2025 meta-analysis confirmed moderate but consistent effects, particularly when combined with calcium and vitamin D. The mechanism involves stimulation of osteoblasts and inhibition of osteoclast differentiation through increased OPG/RANKL ratios. Effect sizes are smaller than pharmaceutical osteoporosis treatments, and no fracture outcome data exist. Collagen peptide supplementation targets the organic matrix component of bone, which is not directly addressed by standard mineral-focused therapies.
Sources & References
- 1RPEP-03765·König, Daniel et al. (2018). “Collagen Peptide Supplements Increased Bone Density in Postmenopausal Women Over 12 Months.” Nutrients.Study breakdown →PubMed →↩
- 2RPEP-13708·Sun, Chongxiao et al. (2025). “Collagen peptide supplements improve bone density, bone turnover, and muscle function, especially with vitamin D and calcium.” Frontiers in nutrition.Study breakdown →PubMed →↩
- 3RPEP-14066·Wang, Yali et al. (2025). “Unraveling the Osteogenic Activity and Molecular Mechanism of an Antioxidant Collagen Peptide in MC3T3-E1 Cells..” Nutrients.Study breakdown →PubMed →↩
- 4RPEP-13932·Virgilio, N et al. (2025). “Does Mixing Collagen With Coffee Reduce Its Absorption? A Clinical Trial Has Answers.” Food research international (Ottawa.Study breakdown →PubMed →↩
- 5RPEP-10782·Duangjai, Acharaporn et al. (2025). “Collagen Peptides With Calcium and Vitamin D Improved Skin and Reduced Hair Loss in Menopausal Women.” Clinics and practice.Study breakdown →PubMed →↩
- 6RPEP-11082·Genç, Ahmet Serhat et al. (2025). “Effect of supplementation with type 1 and type 3 collagen peptide and type 2 hydrolyzed collagen on osteoarthritis-related pain, quality of life, and physical function: A double-blind, randomized, placebo-controlled study..” Joint diseases and related surgery.Study breakdown →PubMed →↩
- 7RPEP-10686·Demir-Dora, Devrim et al. (2025). “Evaluation of the Efficacy and Safety of CollaSel PRO® Type I and Type III Hydrolyzed Collagen Peptides in the Treatment of Osteoarthritis: A Double-Blind, Placebo-Controlled, Randomized Clinical Trial..” Journal of clinical medicine.Study breakdown →PubMed →↩