Cost-Effectiveness of GLP-1s: Health Economics
GLP-1 Access & Economics
$53,400/QALY
ICER's 2025 report found tirzepatide the most cost-effective GLP-1 for obesity at $53,400 per quality-adjusted life year, below the $100,000 threshold.
ICER Evidence Report, 2025
ICER Evidence Report, 2025
If you only read one thing
Health economists agree GLP-1 drugs are a good deal per patient. The 2025 ICER report put tirzepatide at $53,400 per year of healthy life gained — well under the $100,000 level the US typically treats as 'worth it.' The problem is not whether they work or whether they are worth it. The problem is scale. At current US prices, fewer than 1 in 100 people who qualify could be covered before the national budget cracks. November 2025 price cuts helped. Europe and Japan have already solved the price side of this for a fraction of what Americans pay.
GLP-1 receptor agonists are among the most effective pharmacological treatments for obesity and type 2 diabetes ever developed. They are also among the most expensive. This creates a paradox that health economists have spent the past several years trying to quantify: drugs that deliver substantial clinical value at a price that health systems cannot scale. The 2025 ICER (Institute for Clinical and Economic Review) evidence report crystallized this tension, finding that all three major GLP-1s for obesity are cost-effective by conventional thresholds but that fewer than 1% of eligible patients could be treated at current net prices before crossing the $880 million annual budget impact threshold. This article examines the full body of cost-effectiveness evidence, from early diabetes analyses to the latest obesity and cardiovascular outcome data, and explains what cost-per-QALY figures actually mean for patients, payers, and the future of peptide therapeutics. For coverage of how much these drugs cost out of pocket, see how much do GLP-1 drugs actually cost.
Key Takeaways
- ICER's 2025 report found tirzepatide at $53,400/QALY, injectable semaglutide at $61,400/QALY, and oral semaglutide at $69,300/QALY for obesity, all below the $100,000 threshold
- Despite being cost-effective, fewer than 1% of eligible obesity patients can be treated at current prices before exceeding ICER's $880 million annual budget cap
- In diabetes, GLP-1RAs versus insulin cost $6,053 per QALY from the healthcare sector perspective in a real-world systematic review (Yang et al., 2021)
- Among four GLP-1RAs for obesity, semaglutide was the most cost-effective at $135,467/QALY versus no treatment at a $195,000 WTP threshold (Hu et al., 2022)
- Americans pay 2 to 4 times more for GLP-1s than patients in Europe, where monthly costs range from $83 to $144
- November 2025 deals between manufacturers and the US government brought Medicare GLP-1 prices to approximately $245/month with $50 copays
What Cost-Effectiveness Means (and Does Not Mean)
Health economists evaluate drugs using the incremental cost-effectiveness ratio (ICER), which divides the additional cost of a treatment by the additional health benefit it produces, measured in quality-adjusted life years (QALYs). One QALY represents one year of life lived in perfect health. A drug that extends life by two years at reduced quality might produce 1.5 QALYs. A drug that does not extend life but substantially improves daily functioning for five years might also produce a meaningful QALY gain.
The willingness-to-pay (WTP) threshold is the maximum cost per QALY that a health system considers acceptable. In the United States, this threshold is typically $100,000 to $150,000 per QALY, though ICER uses $100,000 to $200,000 as its range. The UK's NICE uses approximately $30,000 to $45,000 per QALY. These thresholds are not fixed rules but decision-making guides: a drug with an ICER of $60,000/QALY would generally be considered good value in the US, while one at $300,000/QALY would face significant pushback from payers.
Cost-effectiveness does not mean affordable. A drug can be highly cost-effective (delivering excellent value per dollar spent) while remaining unaffordable at scale (the total budget required to treat all eligible patients exceeds available resources). This distinction is central to the GLP-1 debate.
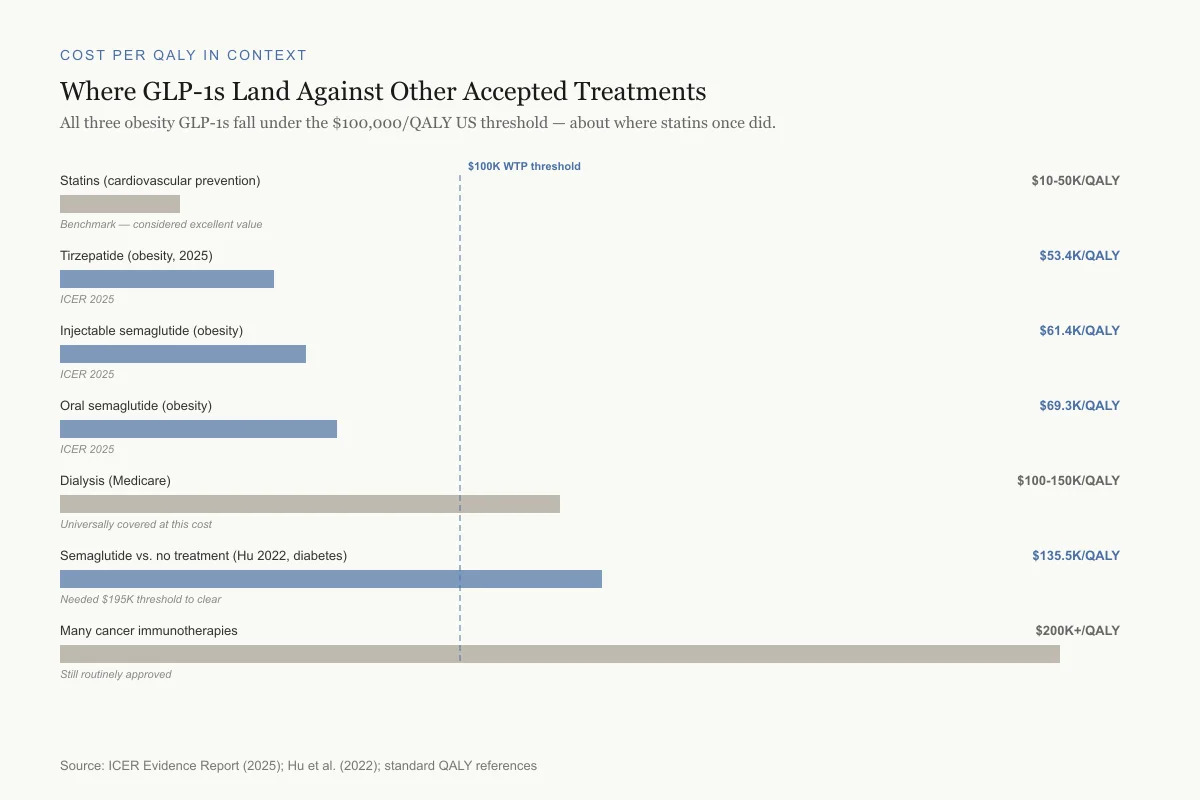
To put QALY values in context: statins for cardiovascular prevention typically cost $10,000 to $50,000 per QALY. Dialysis, which Medicare covers universally, costs approximately $100,000 to $150,000 per QALY. Cancer immunotherapies frequently exceed $200,000 per QALY and are still approved by most payers. Where GLP-1s fall on this spectrum depends on the indication, the specific drug, and critically, the price at which it is purchased.
Cost Per QALY in Context
Where GLP-1s Land Against Other Accepted Treatments
All three obesity GLP-1s fall under the $100,000/QALY US threshold — about where statins once did.
Statins (cardiovascular prevention)
$10-50K/QALYBenchmark — considered excellent value
Tirzepatide (obesity, 2025)
$53.4K/QALYICER 2025
Injectable semaglutide (obesity)
$61.4K/QALYICER 2025
Oral semaglutide (obesity)
$69.3K/QALYICER 2025
Dialysis (Medicare)
$100-150K/QALYUniversally covered at this cost
Semaglutide vs. no treatment (Hu 2022, diabetes)
$135.5K/QALYNeeded $195K threshold to clear
Many cancer immunotherapies
$200K+/QALYStill routinely approved
Source: ICER Evidence Report (2025); Hu et al. (2022); standard QALY references
 View as image
View as imageGLP-1s for Type 2 Diabetes: The Early Economics
The first cost-effectiveness analyses of GLP-1 receptor agonists focused on their original indication: type 2 diabetes. Van de Koppel et al. (2008) provided one of the earliest managed care perspectives, evaluating exenatide (the first GLP-1RA), sitagliptin (a DPP-4 inhibitor), and pramlintide (an amylin analog) for formulary positioning.[1] This analysis framed the economic conversation that would intensify as more potent GLP-1 drugs entered the market.
Yang et al. (2021) conducted the most comprehensive real-world cost-effectiveness analysis, combining individual-level data from 1,022 propensity-score-matched pairs of GLP-1RA and insulin users (from Taiwan's national health database, 2007-2016) with a systematic review of existing cost-effectiveness studies.[2]
From the payer perspective, using GLP-1RAs instead of insulin cost $54,851 per case of all-cause mortality prevented and $29,115 per case of hospitalized hypoglycemia prevented. From the healthcare sector perspective, GLP-1RAs were dominant (less costly and more effective), saving $19,391 per death prevented and $10,293 per hypoglycemia hospitalization prevented. The systematic review across all published studies found ICERs ranging from -$22,682 (cost-saving) to $90,646 per QALY from the payer perspective. The aggregate estimate: GLP-1RAs versus long-acting insulins cost approximately $6,053 per QALY from the healthcare sector perspective.
The number needed to treat (NNT) using a GLP-1RA versus insulin to prevent one death was 57 over 2.3 years, and to prevent one hospitalized hypoglycemia episode was 30. These NNTs are favorable compared to many widely accepted cardiovascular interventions. For comparison, the NNT for statins to prevent one cardiovascular event over five years is typically 20 to 100 depending on baseline risk.
The systematic review component of the Yang study is particularly valuable because it synthesized evidence across different healthcare systems and analytic perspectives. The consistent finding: when real-world costs are included (not just drug acquisition costs but also reduced hospitalizations, fewer hypoglycemia-related emergency visits, and avoided insulin titration appointments), GLP-1RAs are economically competitive with or superior to insulin therapy.
Oral vs. Injectable: The Cost-of-Control Framework
Hansen et al. (2020) introduced the cost-of-control framework, comparing oral semaglutide 14 mg against six injectable GLP-1 drugs.[3] For achieving HbA1c below 7.0%, injectable semaglutide was cheapest at $12,627 per patient achieving target, followed by oral semaglutide at $13,493. Oral semaglutide was cost-effective versus five of six injectable comparators, with only injectable semaglutide performing slightly better at the strictest glycemic target.
The cost-of-control framework adds nuance that traditional cost-effectiveness analyses miss. It accounts for the fact that drugs with different efficacy rates at different targets will have different "costs per success." A cheaper drug that brings fewer patients to target may have a higher cost per controlled patient than a more expensive drug with higher efficacy. This framework favors the semaglutide formulations because their higher efficacy rates effectively dilute the per-patient cost of achieving glycemic control.
This analysis also raised an important practical question: should cost-effectiveness drive the choice between oral and injectable GLP-1 formulations? Many patients strongly prefer oral medication, and the adherence advantage of a daily pill over a weekly injection may improve real-world outcomes beyond what clinical trial data suggest. If oral semaglutide achieves 95% adherence versus 80% for injectable versions, the real-world cost-effectiveness gap narrows or reverses.
GLP-1s for Obesity: Where the Economics Get Complicated
The Hu 2022 Analysis
When GLP-1RAs moved from diabetes to obesity treatment, the economic calculus changed. Hu et al. (2022) compared four GLP-1RAs for obesity in a US setting using a decision-tree model.[4] Among the four agents, semaglutide was the most cost-effective at an ICER of $135,467 per QALY versus no treatment. At a WTP threshold of $195,000/QALY (the upper range used in US analyses), semaglutide was cost-effective, but the other three GLP-1RAs were not.
This analysis highlights a critical point: the cost-effectiveness of GLP-1s for obesity depends heavily on the willingness-to-pay threshold used. At $100,000/QALY (a common US threshold), semaglutide for obesity at 2022 prices did not meet the bar. At $195,000/QALY, it did. The threshold chosen is ultimately a societal value judgment, not a scientific measurement.
The ICER 2025 Evidence Report
ICER's October 2025 evidence report on obesity treatments provided the most comprehensive independent assessment to date. The analysis evaluated three drugs: tirzepatide (Zepbound), injectable semaglutide (Wegovy), and oral semaglutide 50 mg.
The headline ICERs: $53,400/QALY for tirzepatide, $61,400/QALY for injectable semaglutide, and $69,300/QALY for oral semaglutide. All three fell well below the $100,000/QALY threshold, meaning they deliver good clinical value per dollar spent by conventional health economic standards.
The clinical efficacy data underlying these numbers: mean weight loss differences versus placebo were 17.8% with tirzepatide, 13.1% with injectable semaglutide, and 11.4% with oral semaglutide. A head-to-head trial showed tirzepatide producing greater weight loss than injectable semaglutide. The QALY gains come not just from weight loss itself but from downstream reductions in type 2 diabetes incidence, cardiovascular events, osteoarthritis progression, and sleep apnea severity.
The Affordability Paradox
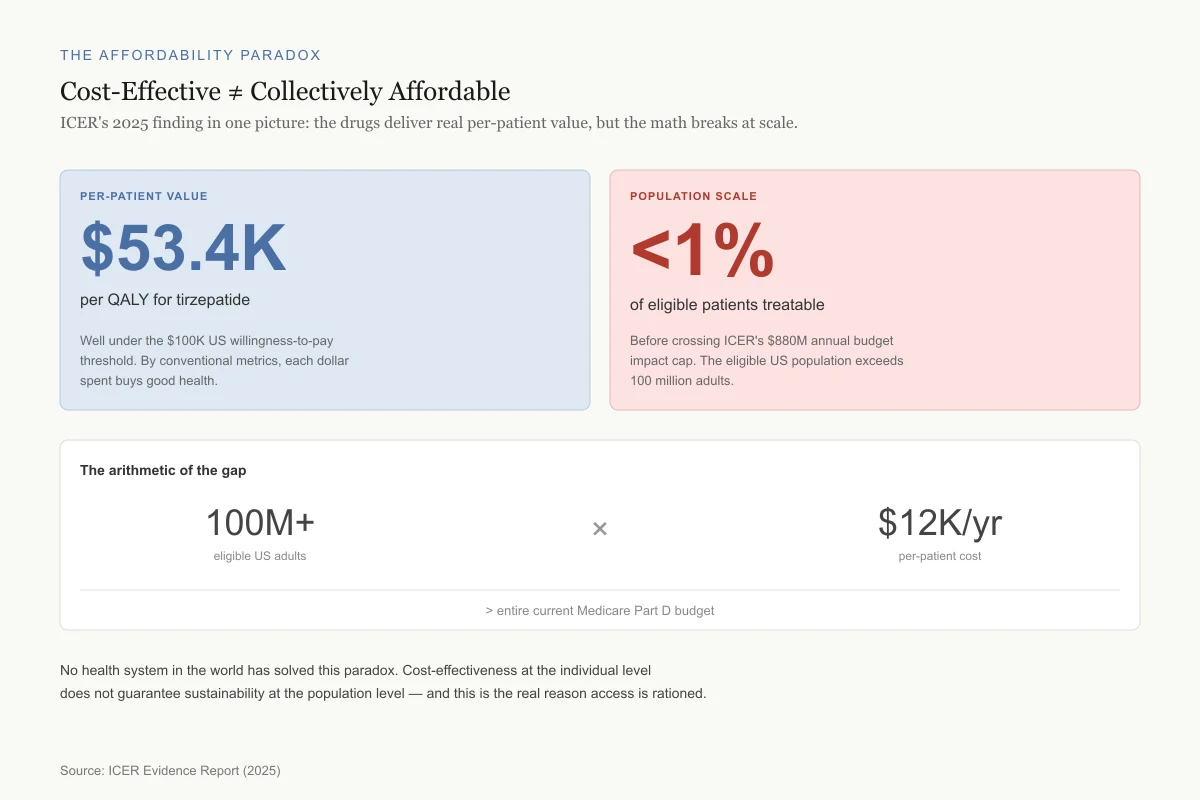
The ICER report's most important finding was not about cost-effectiveness but about affordability. Despite all three drugs being cost-effective by conventional metrics, ICER calculated that fewer than 1% of eligible patients could be treated at current net prices before crossing the organization's annual budget impact threshold of $880 million. The eligible population for GLP-1 obesity treatment in the US exceeds 100 million adults.
This creates what health economists call an "affordability gap." A drug can be worth every dollar spent on an individual level while being collectively unaffordable. If every eligible American received a GLP-1 for obesity, the annual drug cost would exceed the entire current Medicare Part D budget. No health system in the world has solved this problem.
The ICER report also compared GLP-1s to bariatric surgery and endoscopic sleeve gastroplasty. Bariatric surgery, which costs $20,000 to $35,000 as a one-time procedure and produces durable weight loss of 25-30%, has consistently favorable cost-effectiveness ratios over a lifetime horizon. GLP-1 drugs, which require ongoing treatment to maintain weight loss, must generate enough downstream health savings to justify their cumulative cost over years or decades of use. The longer a patient takes a GLP-1, the higher the bar for cost-effectiveness becomes.
The Affordability Paradox
Cost-Effective ≠ Collectively Affordable
ICER's 2025 finding in one picture: the drugs deliver real per-patient value, but the math breaks at scale.
Per-Patient Value
$53.4K
per QALY for tirzepatide
Well under the $100K US willingness-to-pay threshold. By conventional metrics, each dollar spent buys good health.
Population Scale
<1%
of eligible patients treatable
Before crossing ICER's $880M annual budget impact cap. The eligible US population exceeds 100 million adults.
The arithmetic of the gap
100M+
eligible US adults
$12K/yr
per-patient cost
> entire current Medicare Part D budget
No health system in the world has solved this paradox. Cost-effectiveness at the individual level does not guarantee sustainability at the population level — and this is the real reason access is rationed.
Source: ICER Evidence Report (2025)
 View as image
View as imageFor the related question of why insurance companies restrict coverage, see GLP-1 insurance coverage: why getting approved is so hard. For the supply side of this equation, see the GLP-1 shortage explained.
The Cardiovascular Value Proposition
The cost-effectiveness of GLP-1s improves substantially when cardiovascular outcomes are included. Meta-analyses of cardiovascular outcome trials show GLP-1RAs reduce major adverse cardiovascular events (MACE) by 14% (OR 0.86, 95% CI 0.80-0.94), all-cause mortality by 13%, cardiovascular mortality by 13%, and stroke by a similar magnitude.
These cardiovascular benefits transform the economic equation. A drug that only produces weight loss must justify its cost against the relatively modest QALY gains from being thinner. A drug that also prevents heart attacks, strokes, and cardiovascular death generates substantially more QALYs, improving its cost-effectiveness ratio. This is why analyses that include cardiovascular outcomes consistently produce more favorable ICERs than weight-loss-only analyses.
For patients with established cardiovascular disease, prior myocardial infarction, or stroke, the absolute risk reduction from GLP-1RAs is larger, and the cost per QALY gained often falls below $50,000. High-risk stratification consistently improves the economic case for GLP-1 treatment. This is the argument for risk-based prescribing: targeting GLP-1s to patients who will derive the greatest absolute benefit maximizes both clinical and economic value.
The cardiovascular data also introduces a temporal dimension to cost-effectiveness. Cardiovascular events are expensive: a myocardial infarction costs $30,000 to $100,000 in acute care, followed by years of medication and monitoring. A stroke can cost $50,000 to $200,000 in the first year alone, plus long-term disability costs. Each prevented event represents a substantial cost offset against the ongoing drug expense. Models that extend the time horizon to 10, 20, or 30 years consistently produce more favorable GLP-1 cost-effectiveness ratios than shorter analyses because the cardiovascular events being prevented are both costly and life-altering. For deeper coverage of GLP-1 effects in autoimmune diabetes contexts, see autoimmune diabetes and GLP-1.
International Price Comparisons
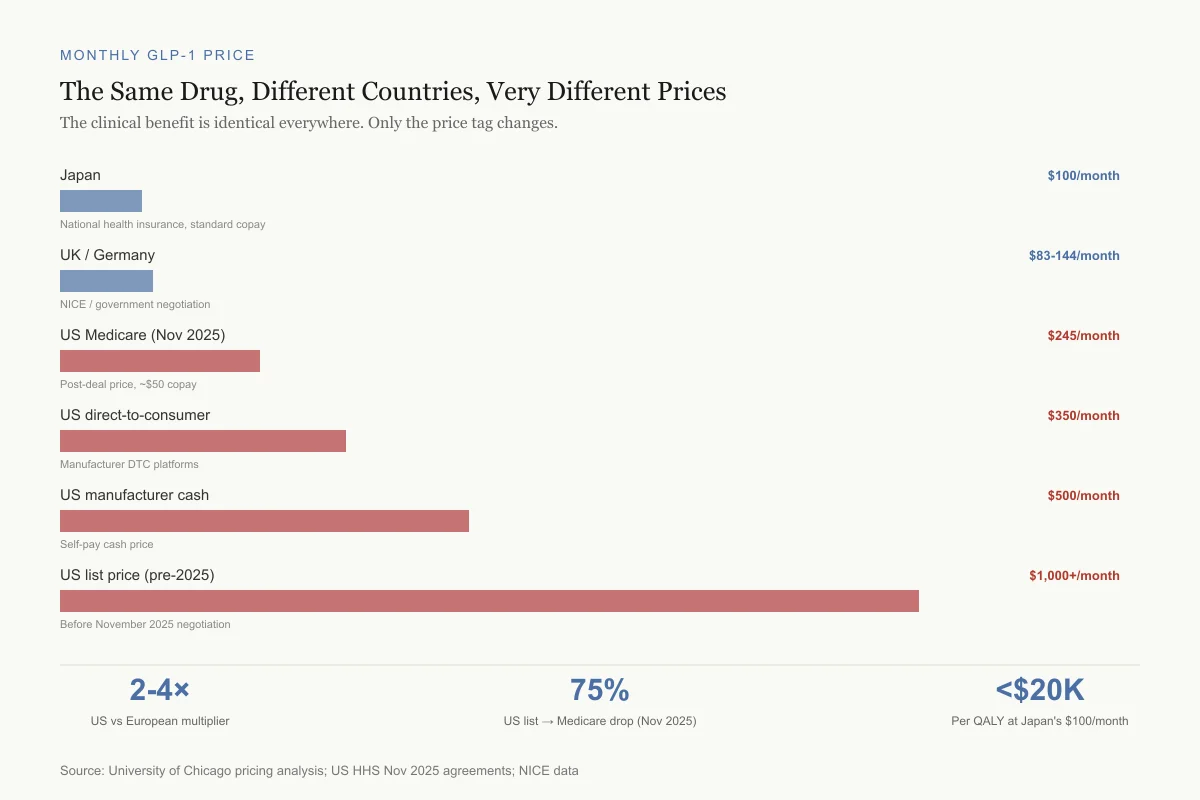
The cost-effectiveness of GLP-1s is heavily influenced by price, which varies dramatically across countries. Americans pay 2 to 4 times more than patients in peer European nations, where monthly GLP-1 costs range from $83 to $144. At European prices, the ICER for every GLP-1RA falls well below any commonly used WTP threshold.
This pricing disparity arises from structural differences in how countries negotiate drug prices. In the UK, Germany, and most European countries, government agencies negotiate directly with manufacturers based on cost-effectiveness evidence. NICE in the UK typically rejects drugs above $45,000 per QALY unless they meet specific criteria for innovation or end-of-life benefit. This forces manufacturers to offer lower prices to access European markets. In the US, Medicare has historically been prohibited from negotiating drug prices directly (though the Inflation Reduction Act of 2022 began changing this), and commercial insurers negotiate individually, resulting in higher list prices offset by complex rebate structures.
The implication for cost-effectiveness analysis is straightforward: the same drug delivering the same clinical benefit can be cost-effective in Europe and cost-ineffective in the US purely because of the price difference. This is a pricing problem, not a value problem. Health economists at the University of Chicago found that although GLP-1 drugs deliver impressive long-term health improvements, their US prices exceeded accepted cost-effectiveness thresholds by a wide margin before the 2025 price reductions.
Japan offers an instructive case. Monthly semaglutide costs are approximately $100 through the national health insurance system, and the drug is widely accessible with standard copayments. Cost-effectiveness analyses using Japanese prices produce ICERs well below $20,000/QALY for both diabetes and obesity indications. The clinical benefit is identical; only the price differs.
Monthly GLP-1 Price
The Same Drug, Different Countries, Very Different Prices
The clinical benefit is identical everywhere. Only the price tag changes.
Japan
$100/monthNational health insurance, standard copay
UK / Germany
$83-144/monthNICE / government negotiation
US Medicare (Nov 2025)
$245/monthPost-deal price, ~$50 copay
US direct-to-consumer
$350/monthManufacturer DTC platforms
US manufacturer cash
$500/monthSelf-pay cash price
US list price (pre-2025)
$1,000+/monthBefore November 2025 negotiation
2-4×
US vs European multiplier
75%
US price drop from list to Medicare (Nov 2025)
<$20K
Per QALY in Japan at $100/month
Source: University of Chicago pricing analysis; US HHS Nov 2025 agreements; NICE data
 View as image
View as imageThe Pricing Inflection Point: November 2025
In November 2025, the US government announced agreements with Eli Lilly and Novo Nordisk to reduce GLP-1 prices through multiple channels. Medicare prices for Ozempic, Wegovy, Mounjaro, and Zepbound fell to approximately $245 per month, with copays around $50. Direct-to-consumer pricing through the TrumpRx platform brought costs to roughly $350 per month. Manufacturers had already been selling directly at around $500 per month.
These price reductions substantially improve cost-effectiveness ratios. At $245/month versus $1,000+/month, the ICER for each GLP-1 drug drops by 50-75%, potentially bringing all agents below $30,000/QALY, a threshold at which nearly any health economist would consider them high-value interventions.
The practical impact remains uneven. Medicare beneficiaries with new coverage access see the largest immediate benefit, particularly those who previously faced the "doughnut hole" coverage gap that made GLP-1s unaffordable at list price. Commercially insured patients face varying coverage depending on employer plan design, with many plans still requiring prior authorization, step therapy through cheaper alternatives, or clinical criteria like BMI thresholds. Uninsured patients may access manufacturer programs or the direct-to-consumer platform but still face meaningful monthly costs that exceed what many households can sustain over years of treatment. For a comparison of brand versus compounded options, see compounded semaglutide vs brand Wegovy. For how telehealth is expanding access, see GLP-1 telehealth prescribing.
What the Economics Do Not Capture
Weight Regain After Discontinuation
Most cost-effectiveness models assume continuous treatment. Clinical trials show that weight regain begins within weeks of GLP-1 discontinuation, with patients recovering approximately two-thirds of lost weight within one year. If GLP-1s require lifelong use to maintain benefit, the lifetime cost per patient rises dramatically.
Consider a 45-year-old patient starting semaglutide for obesity. At $1,000/month, the drug cost over 30 years of continuous use would be $360,000. Even at the reduced Medicare price of $245/month, that is $88,200. Whether that cumulative investment generates enough health benefit to remain cost-effective depends on assumptions about sustained weight loss, cardiovascular risk reduction, and the probability that the patient would have developed obesity-related diseases without treatment. Models that assume finite treatment duration (for example, 5 years followed by discontinuation) may overestimate cost-effectiveness if weight regain erases the health gains.
Some researchers have proposed "intermittent" or "step-down" dosing strategies that might maintain partial weight loss at lower cost. These strategies have not been tested in formal cost-effectiveness models because the clinical evidence for dose reduction is limited. If validated, they could fundamentally change the economic picture.
Indirect Economic Benefits
Standard cost-effectiveness analyses focus on direct medical costs and QALYs. They typically exclude indirect benefits like increased workforce productivity, reduced disability claims, lower absenteeism, and improved mental health. For obesity, these indirect costs are substantial. The CDC estimates that obesity-related absenteeism costs US employers $3.38 billion annually. Total indirect costs of obesity in the US are estimated at $90 billion per year when including reduced productivity, increased disability, and premature mortality.
Including productivity gains would make GLP-1s substantially more cost-effective from a societal perspective. A 2024 microsimulation model that incorporated both direct and indirect costs found GLP-1 ICERs below $50,000/QALY across most scenarios. However, this does not help payers whose budgets cover only direct medical costs. Employers who self-insure may find GLP-1 coverage economically rational when workforce productivity is included, while traditional insurers operating on shorter time horizons may not.
The Equity Dimension
Cost-effectiveness analysis treats every QALY as equally valuable regardless of who receives it. In practice, GLP-1 access follows economic gradients: patients with better insurance, higher incomes, and greater health literacy are more likely to receive treatment. Obesity disproportionately affects lower-income populations and communities of color, yet these groups face the greatest barriers to GLP-1 access.
If GLP-1s primarily benefit the already-advantaged while being funded through insurance premiums shared by all, the equity implications deserve separate analysis beyond what ICER ratios capture. Some health economists have proposed "equity-weighted" cost-effectiveness analysis, which assigns higher value to health gains among disadvantaged populations. Under such frameworks, expanding GLP-1 access to underserved communities would generate even greater societal value. No major GLP-1 cost-effectiveness study has yet incorporated equity weighting. Until they do, conventional cost-effectiveness analyses will continue to answer the question "is this drug good value?" without addressing the equally important question "value for whom?"
For related coverage of the psychological dimensions of GLP-1 weight loss, see body image after GLP-1 weight loss.
The Bottom Line
GLP-1 receptor agonists are cost-effective by conventional health economic standards, with ICERs ranging from $53,400 to $69,300 per QALY for obesity and as low as $6,053 per QALY for diabetes versus insulin. The central tension is affordability: at pre-2025 US prices, fewer than 1% of eligible patients could be treated within budget constraints. November 2025 price reductions substantially improved the economics, but questions about lifelong treatment costs, weight regain after discontinuation, and equitable access remain unresolved. The drugs deliver clear value; the challenge is delivering that value at a price that health systems can sustain for the populations that need them.
Sources & References
- 1RPEP-01430·VanDeKoppel, Shawna et al. (2008). “GLP-1 Drugs for Diabetes in 2008: Exenatide, Sitagliptin, and Pramlintide From a Cost Perspective.” Journal of managed care pharmacy : JMCP.Study breakdown →PubMed →↩
- 2RPEP-05899·Yang, Chen-Yi et al. (2021). “Cost-effectiveness of GLP-1 receptor agonists versus insulin for the treatment of type 2 diabetes: a real-world study and systematic review..” Cardiovascular diabetology.Study breakdown →PubMed →↩
- 3RPEP-04840·Hansen, B B et al. (2020). “Oral Semaglutide Is Cost-Effective Compared to Most Injectable GLP-1 Drugs.” Journal of medical economics.Study breakdown →PubMed →↩
- 4RPEP-06202·Hu, Ying et al. (2022). “Cost-effectiveness analysis of 4 GLP-1RAs in the treatment of obesity in a US setting..” Annals of translational medicine.Study breakdown →PubMed →↩