Autoimmune Diabetes and GLP-1: The Research
Autoimmune Diabetes and GLP-1
15-20% insulin reduction
A 2026 meta-analysis of randomized controlled trials found GLP-1 receptor agonists reduced total daily insulin dose by 15-20% in type 1 diabetes patients.
Abdel-Rahman et al., Diabetes Obesity and Metabolism, 2026
Abdel-Rahman et al., Diabetes Obesity and Metabolism, 2026
If you only read one thing
Doctors used to assume GLP-1 drugs like Ozempic and Mounjaro couldn't help type 1 diabetes because there are no beta cells left for the drugs to stimulate. That turned out to be wrong. GLP-1 has five other effects that don't need beta cells — suppressing the liver's glucose output, slowing digestion, reducing appetite, calming inflammation, and protecting the heart. In the first Phase 2 tirzepatide trial in T1D, patients lost weight and cut their insulin dose. These drugs aren't FDA-approved for type 1 yet, but off-label use is growing so fast that consensus guidelines are due in mid-2026. The big safety catch: combining GLP-1 with SGLT2 inhibitors can trigger euglycemic DKA — DKA that shows up with normal-looking glucose readings. Ketone monitoring is essential.
Type 1 diabetes is an autoimmune disease that destroys insulin-producing beta cells in the pancreas. For a century, exogenous insulin has been the only treatment. But insulin replacement does not address the weight gain, cardiovascular risk, and residual beta-cell decline that complicate long-term management. GLP-1 receptor agonists, peptide-based drugs originally developed for type 2 diabetes, are now being studied as adjuncts to insulin in type 1 diabetes. A 2026 systematic review and meta-analysis of randomized controlled trials found that GLP-1 receptor agonists reduced total daily insulin dose, lowered body weight, and improved glycemic markers in type 1 diabetes patients, though safety concerns around diabetic ketoacidosis remain unresolved.[1] This article maps the full evidence landscape: what GLP-1 agonists do in autoimmune diabetes, what they do not do, and where the science stands.
Key Takeaways
- For years, doctors assumed Ozempic and similar drugs couldn't help type 1 diabetes because there are no beta cells left to stimulate. That turned out to be wrong.
- GLP-1 drugs do five things that don't need beta cells: cut appetite, slow digestion, block liver glucose release, reduce inflammation, and protect the heart.
- In type 1 patients, adding a GLP-1 to insulin cuts the daily insulin dose by 15-20% and produces real weight loss — especially in adults with obesity.
- The first tirzepatide trial in type 1 showed significant weight loss and reduced insulin needs. Real-world data confirm tirzepatide works better than earlier GLP-1s.
- There's one critical safety trap. Combining these drugs with SGLT2 inhibitors can trigger ketoacidosis at normal-looking glucose readings. Ketone monitoring is essential.
- None of these drugs are FDA-approved for type 1 yet. Off-label use is growing fast, and expert consensus guidelines are expected in mid-2026.
- This matters because type 1 patients have been stuck with insulin alone for a century. GLP-1 is the first serious add-on with measurable real-world benefit.
Why GLP-1 agonists are being studied in type 1 diabetes
GLP-1 (glucagon-like peptide-1) is a 30-amino acid incretin hormone released by intestinal L-cells after eating. It stimulates insulin secretion, suppresses glucagon release, slows gastric emptying, and reduces appetite. GLP-1 receptor agonists like semaglutide, liraglutide, and the dual GIP/GLP-1 agonist tirzepatide were developed and approved for type 2 diabetes and obesity.
Type 1 diabetes was historically considered off-limits for incretin therapy because the autoimmune destruction of beta cells eliminates the primary target of GLP-1's insulin-secreting action. If you have no beta cells, stimulating insulin release from beta cells is pointless. But this framing is too simple.
Most people with type 1 diabetes retain some residual beta-cell function, particularly in the years following diagnosis. Even long-standing T1D patients may have measurable C-peptide levels indicating ongoing, though diminished, beta-cell activity. GLP-1 receptor agonists could theoretically support these surviving beta cells while providing insulin-independent benefits: glucagon suppression during hyperglycemia, appetite reduction, weight loss, and cardiovascular protection.[11]
Vogt et al. (2026), writing in the journal Diabetes, argued that GLP-1 receptor agonists should be viewed as "integrative regulators of beta-cell function" rather than simple insulin secretagogues. They explored evidence that GLP-1 signaling promotes beta-cell survival, enhances glucose sensing, and modulates the secretory machinery through mechanisms that extend beyond acute insulin release.[11]
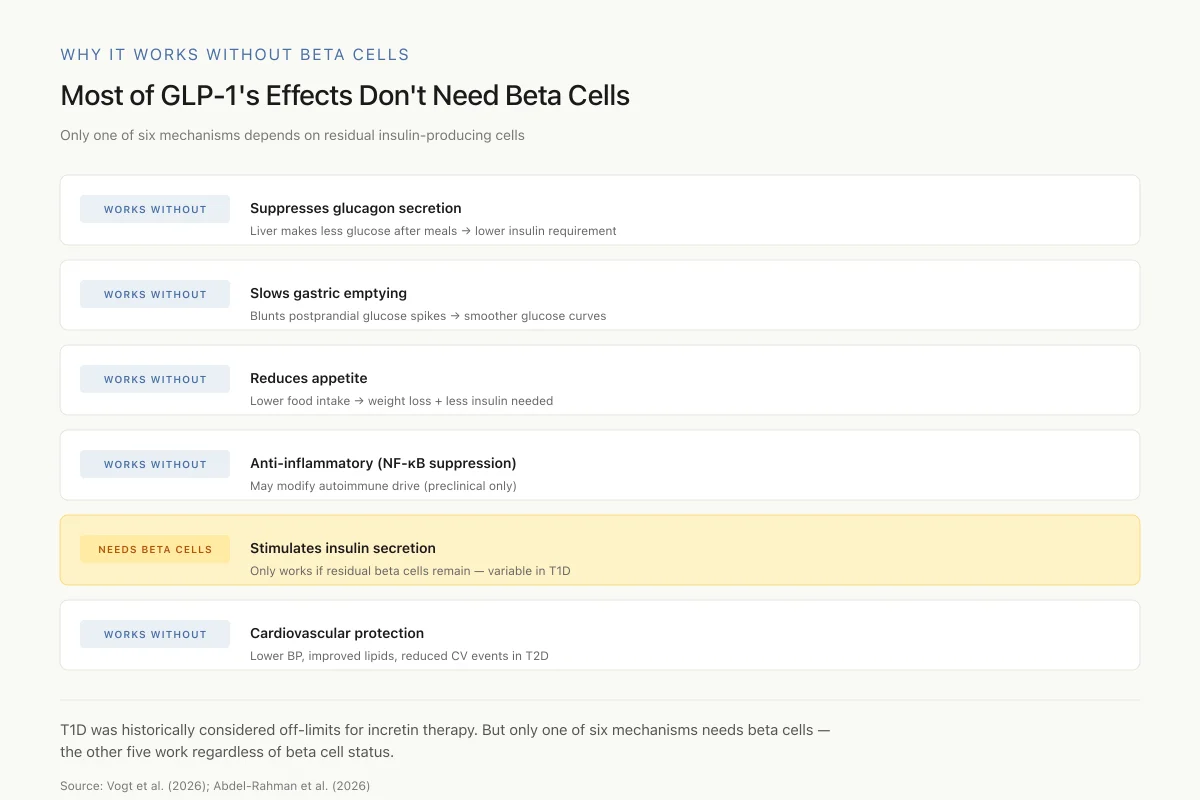
Why It Works Without Beta Cells
Most of GLP-1's Effects Don't Need Beta Cells
Only one of six mechanisms depends on residual insulin-producing cells
Historically, T1D was considered off-limits for incretin therapy because the insulin-stimulating action can't work without beta cells. But that's one mechanism out of six. The other five — glucagon suppression, slowed gastric emptying, appetite reduction, anti-inflammation, cardiovascular protection — all work regardless of beta cell status.
Source: Vogt et al. (2026); Abdel-Rahman et al. (2026)
 View as image
View as imageThe practical driver, though, is simpler. Nearly two-thirds of adults with type 1 diabetes are now overweight or obese. These patients face the same metabolic syndrome, cardiovascular risk, and weight-related complications as type 2 diabetes patients, on top of their autoimmune disease. GLP-1 agonists address these comorbidities directly.
The evidence: insulin dose reduction and glycemic control
Meta-analyses and systematic reviews
Abdel-Rahman et al. (2026) conducted a systematic review and meta-analysis of randomized controlled trials evaluating GLP-1 receptor agonists and SGLT2 inhibitors as adjuncts to insulin in type 1 diabetes. Across the included RCTs, GLP-1 receptor agonists significantly reduced total daily insulin dose and body weight. However, the effect on HbA1c, the standard measure of long-term glycemic control, was more modest and inconsistent across studies. The analysis also flagged an elevated risk of diabetic ketoacidosis, particularly with SGLT2 inhibitor use.[1]
Alhowiti et al. (2025) reviewed the effects of GLP-1 agonists specifically on HbA1c and insulin dose in type 1 diabetes patients. They found that while insulin dose reductions were consistent across studies, the HbA1c improvements were limited, and a high risk of bias across the available literature made definitive conclusions difficult. The authors noted that the inconsistency in glycemic outcomes may reflect the heterogeneity of residual beta-cell function across T1D populations.[6]
Chen et al. (2026) focused specifically on cardiometabolic risk factors. Their meta-analysis of RCTs found that GLP-1 receptor agonists improved body weight, waist circumference, and lipid profiles in type 1 diabetes patients, though blood pressure effects were not statistically significant. These cardiometabolic benefits mirror what has been established in type 2 diabetes, suggesting that the metabolic effects of GLP-1 agonists are largely independent of beta-cell stimulation.[4]
Clinical trials
The most rigorous evidence to date comes from the phase 2 tirzepatide trial published by Snaith et al. (2026) in Diabetes Care. This randomized, double-blind, placebo-controlled trial enrolled adults with type 1 diabetes and BMI above 30 kg/m2. Participants received once-weekly subcutaneous tirzepatide or placebo for 12 weeks, in addition to their existing insulin regimen. Tirzepatide produced significant reductions in body weight and total daily insulin dose compared to placebo.[2]
This trial is significant because tirzepatide is a dual GIP/GLP-1 receptor agonist, and this was its first evaluation in type 1 diabetes. The dual-incretin mechanism may offer advantages over GLP-1-only agonists: GIP receptor activation provides additional glucose-dependent insulin secretion and glucagon modulation through pathways that complement GLP-1 signaling. Whether these dual-receptor effects translate to clinically meaningful differences over GLP-1 receptor agonists alone in the T1D population is an active area of investigation. The 12-week duration limits conclusions about long-term efficacy and safety, and larger phase 3 trials will be needed before regulatory evaluation.
Ahmed et al. (2025) evaluated the effect of metformin and/or GLP-1 receptor agonists on total daily insulin dose in adult type 1 diabetes patients. They found that GLP-1 receptor agonists, including agents not routinely used in type 1 diabetes, reduced insulin requirements. The study also assessed dual GLP-1/GIP receptor agonists (tirzepatide), finding consistent insulin dose reduction across incretin classes.[7]
Can GLP-1 drugs reduce insulin requirements in type 1 diabetes?
The evidence consistently shows they can. Across meta-analyses and individual studies, GLP-1 receptor agonists reduce total daily insulin dose by approximately 15-20% when used as adjuncts to insulin therapy in type 1 diabetes. This reduction has practical significance: lower insulin doses mean less weight gain from insulin itself, fewer hypoglycemic episodes, and reduced injection burden.
Gonzalez et al. (2026) extended this finding to pediatric patients, showing that GLP-1 receptor agonists reduced both BMI and total daily insulin dose in youth with type 1 diabetes. The study, published in the Journal of Pediatric Endocrinology and Metabolism, reported that youth with T1D and obesity experienced meaningful weight reduction alongside insulin dose decreases, addressing a population with particularly limited treatment options.[3]
The mechanism behind insulin dose reduction in type 1 diabetes is not entirely through residual beta-cell stimulation. GLP-1 agonists suppress postprandial glucagon secretion, which reduces hepatic glucose output and lowers the insulin needed to maintain glycemic control. They also slow gastric emptying, blunting postprandial glucose spikes that require bolus insulin coverage. These glucagon-suppressing and gastric effects operate independently of beta-cell function, which is why they work even in patients with minimal or no residual C-peptide.
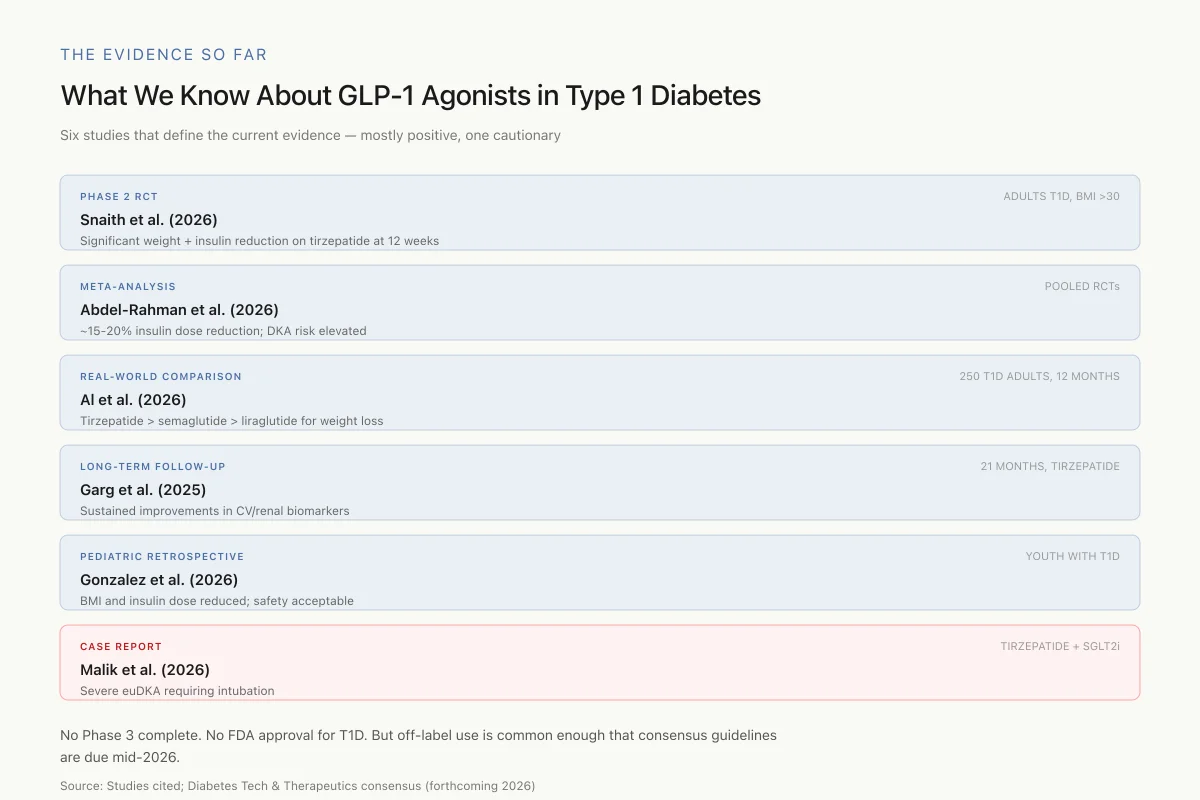
The Evidence So Far
What We Know About GLP-1 Agonists in Type 1 Diabetes
Six studies that define the current evidence — mostly positive, one cautionary
No Phase 3 trial is complete. No GLP-1 agonist is FDA-approved for T1D. But off-label use has become common enough that consensus guidelines are due in mid-2026.
Source: Studies cited; Diabetes Technology & Therapeutics consensus report (forthcoming 2026)
 View as image
View as imageWeight management: real-world comparisons
Al et al. (2026) published the first real-world head-to-head comparison of tirzepatide, semaglutide, and liraglutide for weight loss in type 1 diabetes. The study followed 250 people with T1D and BMI above 27 kg/m2 for 12 months, comparing those treated with tirzepatide (n=35), semaglutide (n=36), liraglutide (n=97), or usual care (n=82). Tirzepatide produced the greatest weight loss, followed by semaglutide, then liraglutide. All three active treatments outperformed usual care for body weight reduction and metabolic risk markers.[8]
Garg et al. (2025) assessed cardiovascular and renal biomarkers in overweight and obese adults with type 1 diabetes treated with tirzepatide for 21 months. The extended follow-up showed sustained improvements in metabolic parameters, with reductions in markers of cardiovascular and kidney disease risk. This study, published in Diabetes Technology and Therapeutics, provides the longest follow-up data for a dual-incretin agonist in type 1 diabetes to date.[9]
GLP-1 and autoimmune inflammation
Beyond glucose and weight, GLP-1 receptor agonists display anti-inflammatory properties that are relevant to the autoimmune pathophysiology of type 1 diabetes.
Birda et al. (2026) conducted a systematic review of GLP-1 analogues in immune-mediated inflammatory diseases, published in Autoimmunity Reviews. They found evidence of GLP-1 receptor agonist efficacy across multiple autoimmune and inflammatory conditions beyond diabetes, including inflammatory bowel disease, psoriasis, and other immune-mediated disorders. The anti-inflammatory mechanisms include suppression of NF-kB signaling, reduction of pro-inflammatory cytokine production, and modulation of immune cell activity.[5]
Whether these immunomodulatory effects can slow or modify the autoimmune destruction of beta cells in type 1 diabetes is an open question. Preclinical data are more encouraging than clinical data. Huang et al. (2026) demonstrated that oral delivery of GLP-1 analogues via recombinant Lactococcus lactis bacteria restored pancreatic islet structure in diabetic mice, with GLP-1 analogues crossing the intestinal mucosal barrier to exert systemic effects on islet architecture.[12]
Jagomae et al. (2025) found that combining GABA with a GLP-1 receptor agonist modified both diabetes progression and Langerhans islet cytoarchitecture in a rat model of Wolfram syndrome, a rare genetic form of diabetes. The combination therapy was more effective than either agent alone, suggesting that GLP-1 receptor stimulation may synergize with other pathways to preserve islet structure and function.[13]
These preclinical findings are intriguing but must be interpreted with caution. Animal models of autoimmune diabetes, particularly chemically-induced models, do not fully replicate the complex autoimmune environment of human type 1 diabetes. The gap between restoring islet structure in mice and preserving beta-cell function in humans with active autoimmunity is substantial.
Off-label use: what clinicians are already doing
While no GLP-1 receptor agonist is approved for type 1 diabetes, off-label prescribing has become increasingly common, particularly for T1D patients with obesity and insulin resistance. The diabetes community has not waited for regulatory approval.
The upcoming consensus report on "Adjunctive Treatment with GLP/GIPs for Patients with T1D," scheduled for the June 2026 issue of Diabetes Technology and Therapeutics, reflects the urgency of establishing clinical guidelines for a practice that is already widespread. This guidance aims to standardize safe use, address ketoacidosis risk, and define patient selection criteria.
The off-label pattern is consistent: clinicians prescribe GLP-1 agonists to T1D patients who are overweight or obese and struggling with insulin dose escalation. The primary goals are weight reduction and insulin dose reduction, with glycemic improvement as a secondary benefit. The approach is pragmatic, driven by the reality that type 1 diabetes patients with obesity face the same metabolic burden as type 2 diabetes patients but have fewer approved treatment options.
The growing prevalence of obesity in the T1D population has accelerated this trend. Data from diabetes registries indicate that rates of overweight and obesity among T1D adults now mirror the general population, fundamentally changing the clinical profile of a disease historically associated with leanness. For these patients, the cardiovascular and metabolic benefits demonstrated by GLP-1 agonists in the SUSTAIN, LEADER, and SURPASS trials for type 2 diabetes are directly relevant, even though the underlying pathophysiology differs. The challenge for clinicians is managing the unique risks of type 1 diabetes, particularly DKA and hypoglycemia from insulin adjustments, within a framework designed for type 2 diabetes pharmacotherapy.
Endocrinology societies have recognized the gap between clinical practice and regulatory approval. The American Diabetes Association's Standards of Care have begun acknowledging adjunctive therapy in type 1 diabetes, and several centers of excellence have developed institutional protocols for GLP-1 agonist use in T1D with specific ketone monitoring requirements and insulin reduction guidelines.
Safety: the euglycemic DKA concern
The most serious safety signal for GLP-1 agonists in type 1 diabetes is euglycemic diabetic ketoacidosis (euDKA), a form of DKA that occurs at normal or near-normal blood glucose levels.
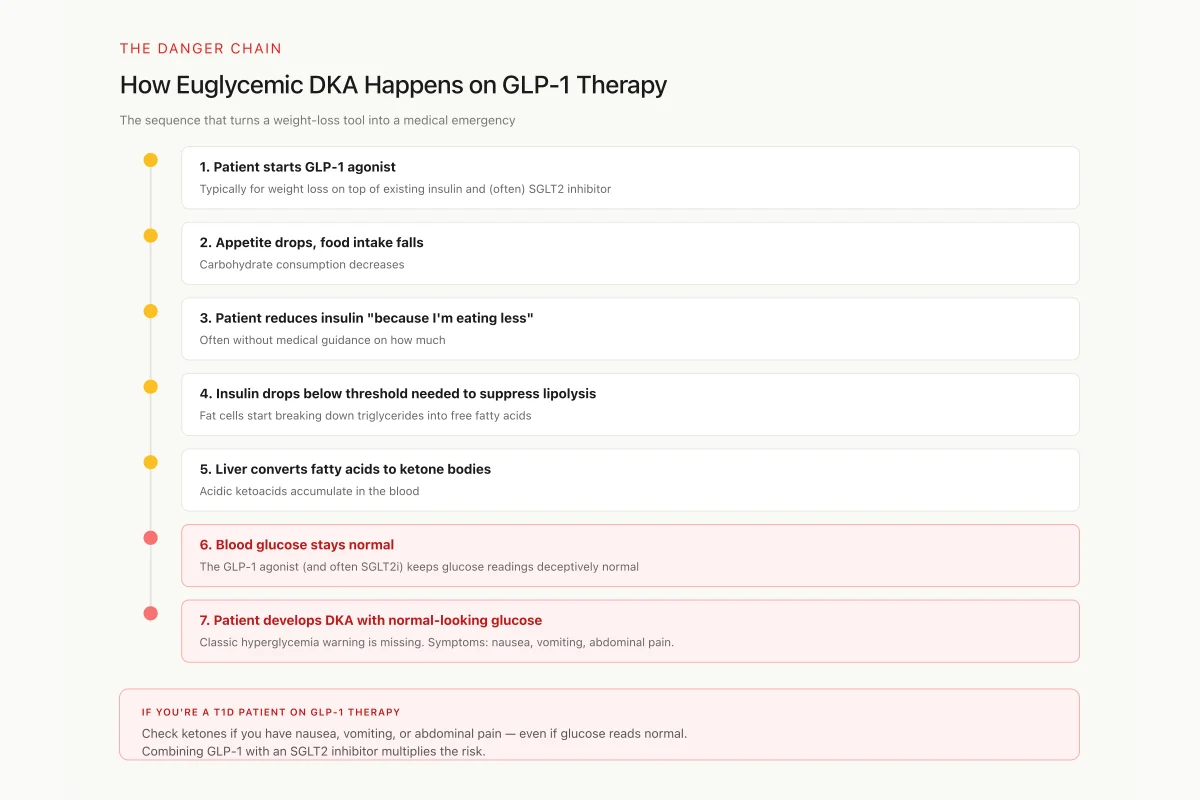
The Danger Chain
How Euglycemic DKA Happens on GLP-1 Therapy
The sequence that turns a weight-loss tool into a medical emergency
Check ketones (blood or urine) if you have nausea, vomiting, or abdominal pain — even if your glucose reads normal. Combining GLP-1 with an SGLT2 inhibitor multiplies the risk.
Source: Malik et al. (2026); Abdel-Rahman et al. (2026) meta-analysis
 View as image
View as imageSafety
CriticalEuglycemic DKA: the risk you won't see coming from the glucose meter
Concern
GLP-1 agonists suppress appetite and reduce insulin needs. T1D patients who reduce insulin too aggressively — especially if also on an SGLT2 inhibitor — can develop DKA at normal-looking glucose levels. The classic hyperglycemia warning is absent.
What the research says
Check ketones (blood or urine) when you have nausea, vomiting, or abdominal pain regardless of glucose reading. Reduce insulin conservatively, not aggressively. Be especially cautious when combining GLP-1 + SGLT2 inhibitor + significant carb restriction.
Particularly relevant for: All T1D patients on GLP-1 agonist therapy, especially those also taking SGLT2 inhibitors or low-carb diets
What to do
Keep ketone test strips on hand. Know the symptoms of euDKA: nausea, vomiting, abdominal pain, deep/rapid breathing. Seek emergency care immediately if ketones are elevated, even if glucose is normal.
Malik et al. (2026); Abdel-Rahman et al. (2026) meta-analysis
Malik et al. (2026) reported a case of severe euDKA requiring intubation in a 41-year-old woman with type 1 diabetes who had initiated tirzepatide for weight loss while also taking the SGLT2 inhibitor empagliflozin and basal-bolus insulin. The combination of insulin dose reduction (from the GLP-1 agonist effect), carbohydrate restriction (from reduced appetite), and urinary glucose excretion (from the SGLT2 inhibitor) created conditions for ketoacid accumulation despite normal blood glucose readings.[10]
This case illustrates the core risk: GLP-1 agonists suppress appetite and reduce insulin requirements, which can lead patients to reduce insulin doses too aggressively. In the absence of sufficient insulin, lipolysis accelerates and ketone bodies accumulate. The classic warning sign of DKA, hyperglycemia, is masked because the GLP-1 agonist and any concurrent SGLT2 inhibitor maintain glucose in the normal range.
The meta-analysis by Abdel-Rahman et al. (2026) confirmed that DKA risk is elevated when these agents are used in type 1 diabetes, though the absolute incidence remains low across RCTs. The risk is highest when GLP-1 agonists are combined with SGLT2 inhibitors, a common off-label combination.[1]
Managing this risk requires ketone monitoring, conservative insulin dose reduction (rather than aggressive titration), and patient education about euDKA symptoms, which differ from classical DKA. Nausea, vomiting, and abdominal pain in a T1D patient on GLP-1 therapy should prompt ketone measurement regardless of blood glucose level.
The connection to other pancreatic peptides
GLP-1 is not the only peptide hormone involved in glucose regulation. Amylin (IAPP), co-secreted with insulin from beta cells, is also deficient in type 1 diabetes. Pramlintide, a synthetic amylin analogue, is the only non-insulin peptide approved for adjunctive use in T1D, providing a precedent for peptide-based adjunctive therapy. The emerging data on GLP-1 agonists in T1D echoes the amylin story: a peptide hormone that was originally considered irrelevant to autoimmune diabetes turns out to address real clinical needs when used alongside insulin.
Current limitations and open questions
The evidence base for GLP-1 agonists in type 1 diabetes, while growing rapidly, has several gaps. No phase 3 trial has been completed. The longest published follow-up (Garg et al., 21 months with tirzepatide) provides encouraging sustained benefit data, but multi-year outcomes including hard cardiovascular endpoints are unavailable.[9]
Whether GLP-1 agonists preserve residual beta-cell function in newly diagnosed type 1 diabetes remains untested in adequately powered clinical trials. The preclinical evidence supports this possibility, but translating islet preservation in rodent models to autoimmune beta-cell protection in humans requires dedicated studies with C-peptide and immunological endpoints.
Patient selection criteria are poorly defined. Which T1D patients benefit most from GLP-1 adjunctive therapy: those with residual C-peptide? Those with obesity? Those with insulin resistance features? The current evidence does not distinguish well between these subgroups.
The gastrointestinal side effects of GLP-1 agonists (nausea, vomiting, diarrhea) are well characterized in the type 2 diabetes population but may interact differently with the glycemic variability and insulin management complexity of type 1 diabetes. A patient with type 1 diabetes who vomits after a meal faces acute hypoglycemia risk from pre-meal bolus insulin in a way that a type 2 diabetes patient on oral medications does not. This interaction between GLP-1-induced nausea, unpredictable food absorption from delayed gastric emptying, and tight insulin dosing creates a management challenge that has not been systematically studied.
Cost and access remain practical barriers. GLP-1 receptor agonists are expensive, and insurance coverage for off-label use in type 1 diabetes is inconsistent. Patients who cannot access these medications through their diabetes coverage sometimes obtain them through obesity indications, but this workaround is not universally available. If phase 3 trials eventually support a T1D indication, regulatory approval and payer coverage would substantially expand access for the patients most likely to benefit.
The Bottom Line
GLP-1 receptor agonists show consistent benefits as adjuncts to insulin in type 1 diabetes: reduced total daily insulin dose, weight loss, and improved cardiometabolic risk markers. The evidence includes a phase 2 RCT of tirzepatide, multiple meta-analyses of smaller RCTs, and growing real-world data. Anti-inflammatory properties raise the possibility of immunomodulatory effects relevant to autoimmune diabetes pathophysiology, though this remains preclinical. Euglycemic DKA is the primary safety concern, particularly in combination with SGLT2 inhibitors. No GLP-1 agonist is approved for type 1 diabetes, but off-label use is expanding rapidly enough that consensus guidelines are expected in mid-2026.
Sources & References
- 1RPEP-14689·Abdel-Rahman, Sama M et al. (2026). “Efficacy and safety of GLP-1 receptor agonists and SGLT2 inhibitors as adjuncts to insulin in type 1 diabetes: Systematic review and meta-analysis..” Diabetes.Study breakdown →PubMed →↩
- 2RPEP-16147·Snaith, Jennifer R et al. (2026). “Tirzepatide Produces Significant Weight Loss in Adults With Type 1 Diabetes and Obesity.” Diabetes care.Study breakdown →PubMed →↩
- 3RPEP-15222·Gonzalez, Frances et al. (2026). “GLP-1 Drugs Reduce BMI and Insulin Needs in Youth with Type 1 Diabetes and Obesity.” Journal of pediatric endocrinology & metabolism : JPEM.Study breakdown →PubMed →↩
- 4RPEP-15008·Chen, Yizhu et al. (2026). “Meta-Analysis: Do GLP-1 Drugs Improve Heart Risk Factors in Type 1 Diabetes Too?.” Diabetes/metabolism research and reviews.Study breakdown →PubMed →↩
- 5RPEP-14885·Birda, Chhagan L et al. (2026). “GLP-1 Drugs Show Anti-Inflammatory Promise for Autoimmune Diseases.” Autoimmunity reviews.Study breakdown →PubMed →↩
- 6RPEP-09861·Alhowiti, Amirah et al. (2025). “GLP-1 Drugs in Type 1 Diabetes: Effects on HbA1c and Insulin Dose.” Frontiers in endocrinology.Study breakdown →PubMed →↩
- 7RPEP-09802·Ahmed, Mayeesha et al. (2025). “Tirzepatide in Type 1 Diabetes: Does It Reduce Daily Insulin Needs?.” Innovations in pharmacy.Study breakdown →PubMed →↩
- 8RPEP-14724·Al Ozairi, Ebaa et al. (2026). “Weight loss in people with type 1 diabetes over 12 months: Real-world data comparing tirzepatide, semaglutide and liraglutide..” Diabetes.Study breakdown →PubMed →↩
- 9RPEP-11056·Garg, Satish K et al. (2025). “Tirzepatide in Type 1 Diabetes: 23% Weight Loss Plus Heart and Kidney Benefits Over 21 Months.” Diabetes technology & therapeutics.Study breakdown →PubMed →↩
- 10RPEP-15652·Malik, Maliha et al. (2026). “Severe Life-Threatening Ketoacidosis After Combining Tirzepatide With an SGLT2 Inhibitor in a Type 1 Diabetes Patient.” Clinical case reports.Study breakdown →PubMed →↩
- 11RPEP-16325·Vogt, Éverton L et al. (2026). “How GLP-1 Drugs Actually Affect Your Insulin-Producing Cells — And What We Still Don't Know.” Diabetes.Study breakdown →PubMed →↩
- 12RPEP-11553·Jagomäe, Toomas et al. (2025). “GABA Plus Liraglutide Reverses Diabetes and Restores Pancreatic Islets in Wolfram Syndrome Rats.” Diabetology & metabolic syndrome.Study breakdown →PubMed →↩
- 13RPEP-15331·Huang, Yuanjian et al. (2026). “Oral Probiotic Delivering GLP-1 Restores Pancreatic Function and Gut Barrier in Diabetic Mice.” EBioMedicine.Study breakdown →PubMed →↩