Collagen Peptides for Joints: Clinical Evidence
Collagen Peptides and Joint Health
35 RCTs Analyzed
A 2024 meta-analysis pooled 35 randomized controlled trials involving 3,165 patients and found that collagen supplementation produced significant improvements in pain and functional scores in osteoarthritis compared to placebo.
Meta-analysis, BMC Musculoskeletal Disorders, 2024
Meta-analysis, BMC Musculoskeletal Disorders, 2024
If you only read one thing
Collagen peptides look like they help joint pain a little. A pooled analysis of 35 trials with more than 3,000 people found that taking collagen every day reduced osteoarthritis pain and stiffness better than a placebo pill. The effect is real but modest, and most trials were paid for by the companies selling collagen. It works better than doing nothing, but exercise therapy still works at least as well.
Collagen peptides are the most commercially successful peptide supplement in the world. The global collagen market exceeds $5 billion annually, with joint health as one of the primary claims driving sales. The question is whether that commercial success is backed by clinical evidence. The answer is more nuanced than either the supplement industry or its critics typically present. A 2024 meta-analysis pooled 35 randomized controlled trials involving 3,165 patients with osteoarthritis and found statistically significant improvements in both pain and functional scores for collagen supplementation versus placebo. But the effect sizes are modest, the trial quality is mixed, and the mechanism by which an orally ingested protein fragment reaches and benefits cartilage tissue is still being worked out.
Key Takeaways
- A 2024 meta-analysis of 35 RCTs (3,165 OA patients) found significant improvements in pain and function with collagen supplementation versus placebo
- Specific collagen peptides improved joint comfort by 39% versus 14% for placebo in a 2018 RCT of 160 participants with knee osteoarthritis (Konig et al., Applied Physiology, Nutrition, and Metabolism, 2018)
- Orally ingested collagen peptides are digested to small fragments, primarily the dipeptide prolyl-hydroxyproline (Pro-Hyp), which accumulates in cartilage tissue (Sato et al., Journal of Agricultural and Food Chemistry, 2020)
- Collagen hydrolysate stimulated type II collagen synthesis by chondrocytes in culture, suggesting a direct anabolic effect on cartilage cells (Saito et al., 2009)
- Trial designs vary widely: collagen types I, II, and III tested at different doses (2-13g/day), different durations (8-48 weeks), and in different populations (OA patients vs. athletes vs. healthy adults)
- The most common effective dose across positive trials is 10g/day of hydrolyzed collagen or 40mg/day of undenatured type II collagen
What Collagen Peptides Are
Collagen is the most abundant protein in the human body, comprising approximately 30% of total protein content. It forms the structural framework of skin, tendons, ligaments, cartilage, and bone. There are at least 28 identified collagen types, but types I, II, and III account for the vast majority of collagen in the body.
Type I collagen is the predominant collagen in skin, tendons, ligaments, and bone. It provides tensile strength. Most collagen peptide supplements marketed for skin and general health contain hydrolyzed type I collagen, often sourced from bovine hide or marine fish skin.
Type II collagen is the primary collagen in hyaline cartilage, the type that lines joint surfaces. It provides the compressive resilience of cartilage. Type II collagen supplements come in two forms: hydrolyzed (broken into small peptides, typically 2-5 kDa) and undenatured (native, intact triple-helix structure at low doses, typically 40mg/day).
Type III collagen is found alongside type I in skin, blood vessels, and internal organs. It contributes to tissue elasticity.
"Collagen peptides" refers to collagen that has been enzymatically hydrolyzed into smaller peptide fragments, typically 2,000 to 5,000 daltons in molecular weight.[1] This hydrolysis improves solubility and bioavailability compared to intact collagen or gelatin. The key insight from the past decade of research is that these fragments are not simply amino acid sources. Specific collagen-derived peptides, particularly the dipeptide prolyl-hydroxyproline (Pro-Hyp) and the tripeptide glycyl-prolyl-hydroxyproline (Gly-Pro-Hyp), appear to have bioactive signaling properties beyond their nutritional value.[2]
How Collagen Peptides Reach Joints
The central skepticism about oral collagen supplements has always been: how can an ingested protein improve a joint? Proteins are digested to amino acids. Why would collagen amino acids preferentially reach cartilage?
The answer, established over the past 15 years of research, is that collagen hydrolysates are not fully digested to individual amino acids. A significant fraction is absorbed as intact di- and tripeptides. The dipeptide Pro-Hyp is particularly important: hydroxyproline is almost unique to collagen in the human body, so collagen-derived Pro-Hyp can be tracked as a biomarker.
A 2020 study demonstrated that the collagen-derived dipeptide Pro-Hyp accumulates in blood and reaches cartilage tissue after oral ingestion, where it stimulates chondrocyte proliferation and extracellular matrix production.[2] A 2009 study showed that collagen hydrolysates stimulated type II collagen synthesis by chondrocytes in cell culture, providing direct evidence that collagen-derived peptides can influence cartilage metabolism.[3]
A 2016 study tracked orally administered collagen peptide distribution in mice and confirmed accumulation in cartilage, skin, and bone tissue, with higher concentrations in cartilage compared to plasma.[4] A separate study examining ingestion of bioactive collagen hydrolysates showed that the peptides increased expression of cartilage-protective genes, including aggrecan and type II collagen, in knee joint cartilage.[5]
The mechanism is not simple digestion and delivery. These collagen-derived peptides appear to act as signaling molecules that stimulate chondrocytes (cartilage cells) and fibroblasts (connective tissue cells) to increase their own collagen production. In other words, the exogenous peptides may function as triggers for endogenous repair, not as raw building materials.
This signaling hypothesis resolves a long-standing objection. Critics have argued that eating collagen to help joints makes no more sense than eating brain to help your brain. If collagen were simply digested to amino acids and redistributed, this objection would hold. But the evidence that specific collagen-derived dipeptides survive digestion, circulate in blood, accumulate in target tissues, and modulate gene expression in resident cells changes the calculus. The analogy is closer to eating a specific bioactive compound than to eating a structural material.
That said, the signaling model has its own limitations. The concentrations of Pro-Hyp measured in blood after oral collagen ingestion are in the micromolar range. Whether these concentrations are sufficient to produce the effects observed in cell culture studies (which often use higher concentrations) is an area of active investigation. The gap between in vitro dose-response and in vivo plasma concentrations is a common challenge in nutraceutical research.
Clinical Trial Evidence for Osteoarthritis
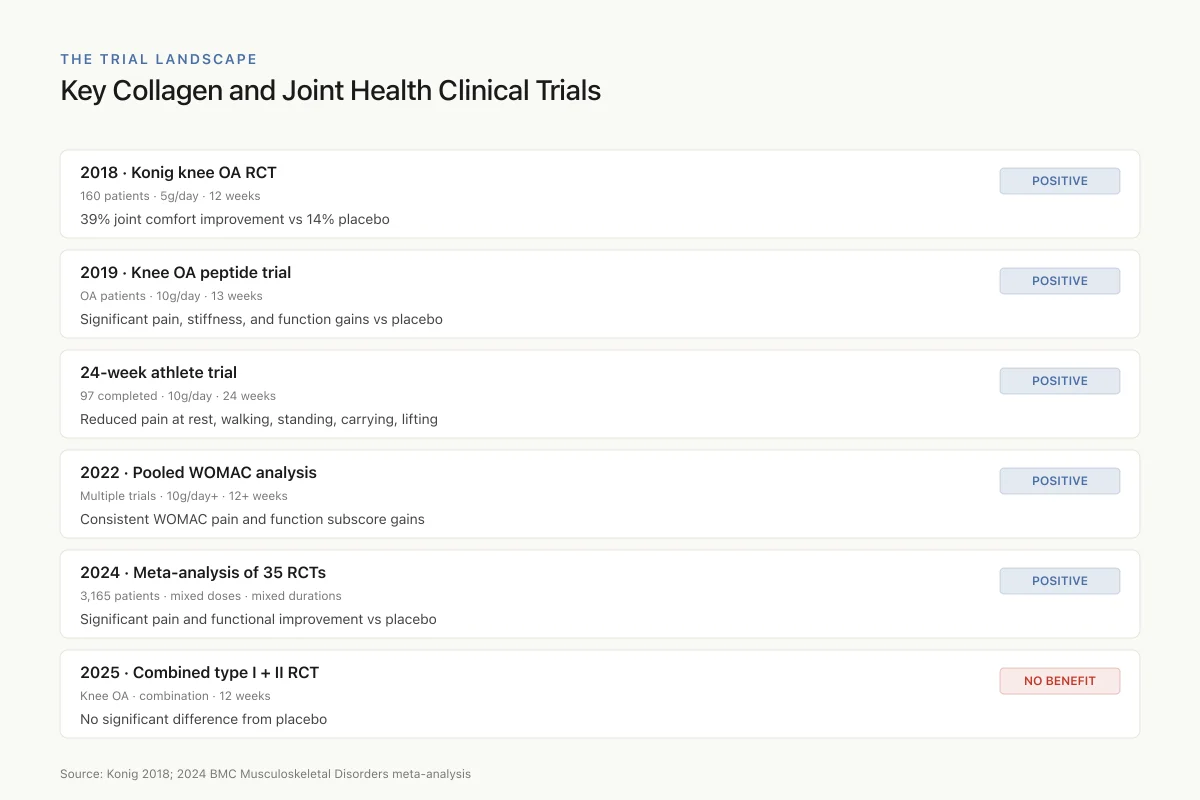
The Trial Landscape
Key Collagen and Joint Health Clinical Trials
Konig knee OA RCT
2018 · 160 patients
39% joint comfort improvement vs 14% placebo
Knee OA peptide trial
2019 · OA patients
Significant pain, stiffness, and function gains
24-week athlete trial
Athlete · 97 completed
Reduced pain at rest, walking, lifting
Pooled WOMAC analysis
2022 · Multiple trials
Consistent WOMAC pain and function gains
Meta-analysis of 35 RCTs
2024 · 3,165 patients
Significant pain and functional improvement
Combined type I + II RCT
2025 · Knee OA
No significant difference vs placebo
Pattern: most trials report modest pain and function gains at 10g/day for 12 weeks or longer. Effects are real but smaller than established therapies like exercise.
Source: Konig 2018; 2024 BMC Musculoskeletal Disorders meta-analysis
 View as image
View as imageThe clinical evidence for collagen peptides in osteoarthritis comes from multiple randomized controlled trials across different collagen types and preparations.
Hydrolyzed Collagen
A 2018 double-blind, placebo-controlled trial randomized 160 patients with knee osteoarthritis to receive 5g/day of specific collagen peptides or placebo for 12 weeks.[6] The collagen group showed a 39% improvement in joint comfort scores compared to 14% for placebo. The difference was statistically significant (p<0.01). The study used a specific collagen peptide preparation optimized for joint health, with a defined molecular weight distribution.
A 2019 study examining orally administered collagen peptide for knee osteoarthritis found significant improvements in pain, stiffness, and physical function over 13 weeks of supplementation at 10g/day compared to placebo.[7]
A 2022 analysis examining the effects of collagen hydrolysate on osteoarthritis symptoms found consistent improvements across pooled trials in WOMAC pain and function subscores, with the greatest benefit in trials using doses of 10g/day or higher and treatment durations of 12 weeks or longer.[8] The WOMAC (Western Ontario and McMaster Universities Osteoarthritis Index) is the most widely used outcome measure in OA trials, scoring pain, stiffness, and physical function on validated scales. Collagen peptides consistently improved all three WOMAC subscores, though the magnitude varied by study.
The most comprehensive pooled analysis came in 2024, when a meta-analysis combined 35 RCTs involving 3,165 patients. The primary analysis (25 RCTs, 2,856 patients) found statistically significant improvements in both visual analog scale (VAS) pain scores and WOMAC functional scores. Subgroup analyses suggested that hydrolyzed collagen performed better than undenatured collagen for pain reduction, that higher doses (10g/day or more) produced larger effects than lower doses, and that longer trial durations were associated with greater benefits. These findings support a dose-response and time-response relationship, which strengthens the case for a genuine biological effect rather than a placebo response.
Undenatured Type II Collagen
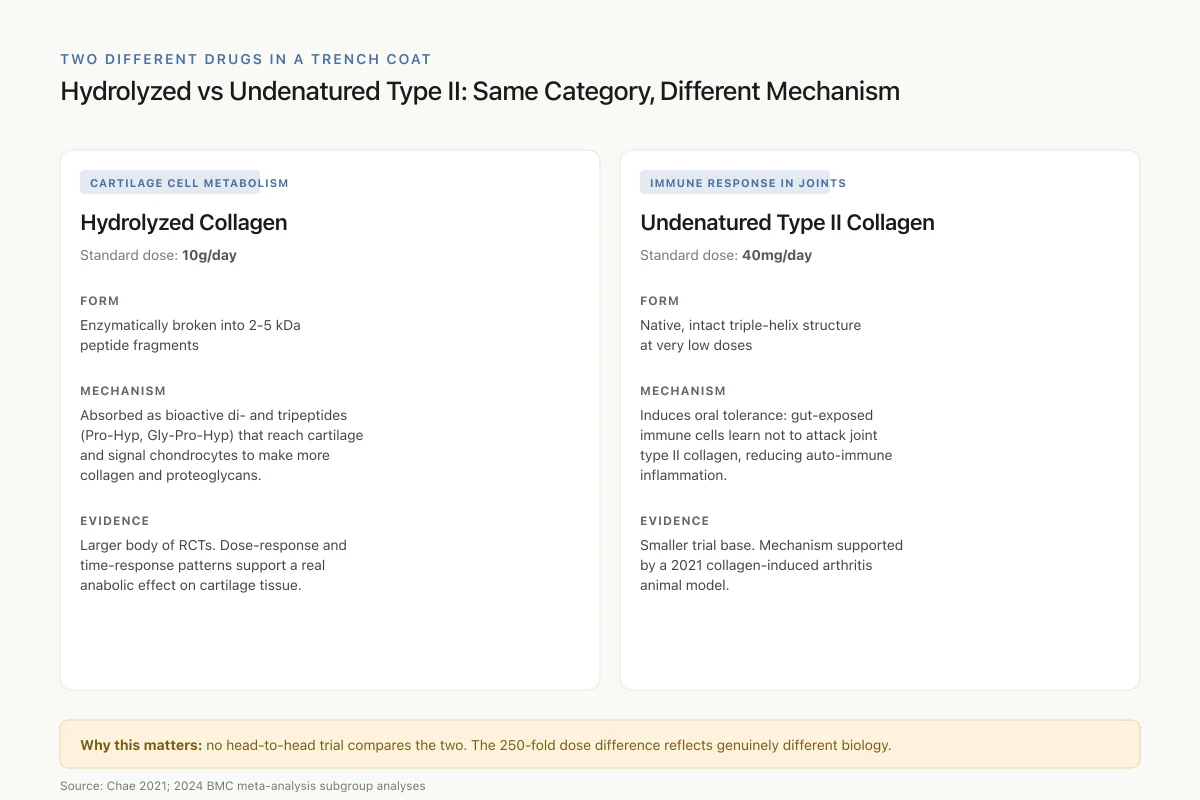
Two Different Drugs in a Trench Coat
Hydrolyzed vs Undenatured Type II: Same Category, Different Mechanism
Hydrolyzed Collagen
Standard dose: 10g/day
Form
Enzymatically broken into 2-5 kDa peptide fragments
Mechanism
Absorbed as bioactive di- and tripeptides (Pro-Hyp, Gly-Pro-Hyp) that reach cartilage and signal chondrocytes to make more collagen and proteoglycans.
Evidence
Larger body of RCTs. Dose-response and time-response patterns support a real anabolic effect.
Undenatured Type II Collagen
Standard dose: 40mg/day
Form
Native, intact triple-helix structure at very low doses
Mechanism
Induces oral tolerance: gut-exposed immune cells learn not to attack joint type II collagen, reducing auto-immune inflammation.
Evidence
Smaller trial base. Mechanism supported by a collagen-induced arthritis animal model (2021).
Why this matters: no head-to-head trial has compared the two. A positive study of one does not transfer to the other, and the 250-fold dose difference reflects genuinely different biology.
Source: Chae 2021; 2024 BMC meta-analysis subgroup analyses
 View as image
View as imageUndenatured type II collagen (UC-II) uses a fundamentally different mechanism. At low doses (typically 40mg/day), native type II collagen is thought to induce oral tolerance, a process by which the immune system is exposed to a small amount of an antigen (type II collagen) via the gut and subsequently reduces its inflammatory response to that same antigen in joints. This is relevant because autoimmune reactivity against type II collagen contributes to cartilage destruction in osteoarthritis and rheumatoid arthritis.
A 2021 study provided evidence that AP collagen peptides prevented joint inflammation in a collagen-induced arthritis model, supporting the immune-modulation mechanism for type II collagen.[9]
Negative and Mixed Results
Not all trials are positive. A 2025 RCT testing combined undenatured type II collagen plus hydrolyzed collagen over 12 weeks found no significant difference from placebo in managing knee osteoarthritis symptoms. This negative result is worth noting because it used a combination approach (two collagen types simultaneously) that may not be equivalent to either type alone.
The overall evidence picture: the majority of RCTs show positive results, but the effect sizes are modest (typically 20-40% improvement over placebo in pain scores), and the trials are heterogeneous in design, dose, collagen type, and population studied.
Evidence in Athletes and Active Populations
A separate body of evidence examines collagen peptides in athletes and active adults with activity-related joint pain rather than clinical osteoarthritis. This is an important distinction because athletes often have joint pain from overuse and microtrauma without the structural cartilage loss that defines OA.
A landmark 24-week randomized study enrolled 147 athletes (97 completed) with activity-related joint pain. Participants received 10g/day of collagen hydrolysate or placebo. The collagen group showed significant reductions in joint pain at rest and during walking, standing, carrying objects, and lifting compared to placebo. The effect was particularly pronounced in athletes with the highest baseline pain levels and in those with the worst pre-existing knee joint conditions.
This athlete study is frequently cited by the supplement industry, but its limitations deserve attention. The sample size was modest. The study was industry-funded (GELITA, a collagen manufacturer). And the 24-week duration, while longer than most collagen trials, is still short relative to the timescale of joint tissue adaptation.
Separate from pain outcomes, the molecular effects on connective tissue have been documented. A 2018 study showed that collagen peptides modulate the expression of matrix metalloproteinases (MMPs) and their tissue inhibitors (TIMPs) in connective tissue, shifting the balance toward tissue preservation rather than degradation.[10] MMPs are the enzymes responsible for breaking down extracellular matrix components, including collagen itself. By upregulating TIMPs and downregulating certain MMPs, collagen peptides may create a net anabolic environment in joint tissues. This shift has been demonstrated in cell culture and animal models but has not been confirmed in human joint tissue biopsies.
A 2021 study examining collagen-binding peptides demonstrated that specific peptide sequences can reverse bone loss in osteoporotic models, raising the possibility that collagen-derived peptides have effects on mineralized tissues as well as cartilage.[11] This is relevant because osteoarthritis involves subchondral bone remodeling alongside cartilage loss, and an intervention that addresses both compartments could have synergistic effects.
Dose, Type, and Duration: What the Trials Tell Us
The heterogeneity of collagen supplement trials makes direct comparison difficult, but patterns emerge:
Dose. The most commonly studied effective dose of hydrolyzed collagen for joint outcomes is 10g/day. Some trials show effects at 5g/day with specific peptide preparations. For undenatured type II collagen, 40mg/day is the standard dose.
Type. Hydrolyzed type I/III collagen and undenatured type II collagen appear to work through different mechanisms (anabolic stimulation vs. immune modulation). There is no head-to-head trial comparing the two approaches directly.
Duration. Positive outcomes typically emerge at 8 to 12 weeks, with some trials showing continued improvement at 24 weeks. The biology supports longer durations: cartilage turnover is slow, and any anabolic effect on chondrocytes would take weeks to manifest as measurable clinical change.
Source. Bovine, porcine, marine (fish), and chicken collagen have all been tested. No study has established superiority of one source over another for joint outcomes when matched for collagen type and molecular weight.
Limitations of the Evidence
Several limitations constrain the conclusions that can be drawn from the collagen-joint literature.
Funding bias. The majority of positive collagen-joint trials are funded by supplement manufacturers. This does not automatically invalidate results, but it is a pattern that warrants caution. Industry-funded nutrition trials have a well-documented tendency to produce more favorable results than independently funded trials.
Heterogeneous preparations. "Collagen peptides" is not a single standardized substance. Different hydrolysis processes produce different peptide profiles, different molecular weight distributions, and potentially different bioactivities. A positive result with one preparation does not necessarily generalize to all collagen peptide products.
Modest effect sizes. Even in positive trials, the improvement over placebo is typically 20-40% in pain scores. For comparison, exercise therapy for knee osteoarthritis produces similar or larger effect sizes. Collagen peptides may be additive to exercise, but no study has tested this combination rigorously.
No structural endpoint. Most trials measure pain and function (patient-reported outcomes). Few have examined cartilage structure via MRI or biomarkers of cartilage metabolism. The question of whether collagen peptides actually protect or regenerate cartilage, or simply reduce pain through anti-inflammatory mechanisms, remains open. For a deeper analysis of whether peptides can truly regenerate cartilage structure, see Can Peptides Regenerate Cartilage? The Current State of Research.
No long-term data. The longest published trial is 48 weeks. Osteoarthritis is a chronic, progressive disease that progresses over decades. Whether collagen supplementation slows structural progression over years is unknown. The absence of long-term data is particularly significant because the question is not just whether collagen peptides reduce pain (which they appear to, modestly), but whether they alter the natural history of the disease. Pain relief without structural benefit would position collagen peptides as symptomatic treatment, not disease modification.
Publication bias. As with many supplement categories, there is likely publication bias in the collagen-joint literature. Positive trials are more likely to be published than negative ones, particularly when funded by manufacturers. The 2025 negative trial (combined collagen types showing no benefit) received far less attention than the positive trials despite being methodologically sound.
Comparisons with established treatments. Few collagen trials include active comparators like exercise, NSAIDs, or physical therapy. Without these comparisons, it is difficult to position collagen peptides relative to treatments with stronger evidence bases. The 2018 trial showing 39% improvement is meaningful versus placebo, but how does it compare to a structured quadriceps strengthening program, which meta-analyses show produces 40-60% pain improvement in knee OA?
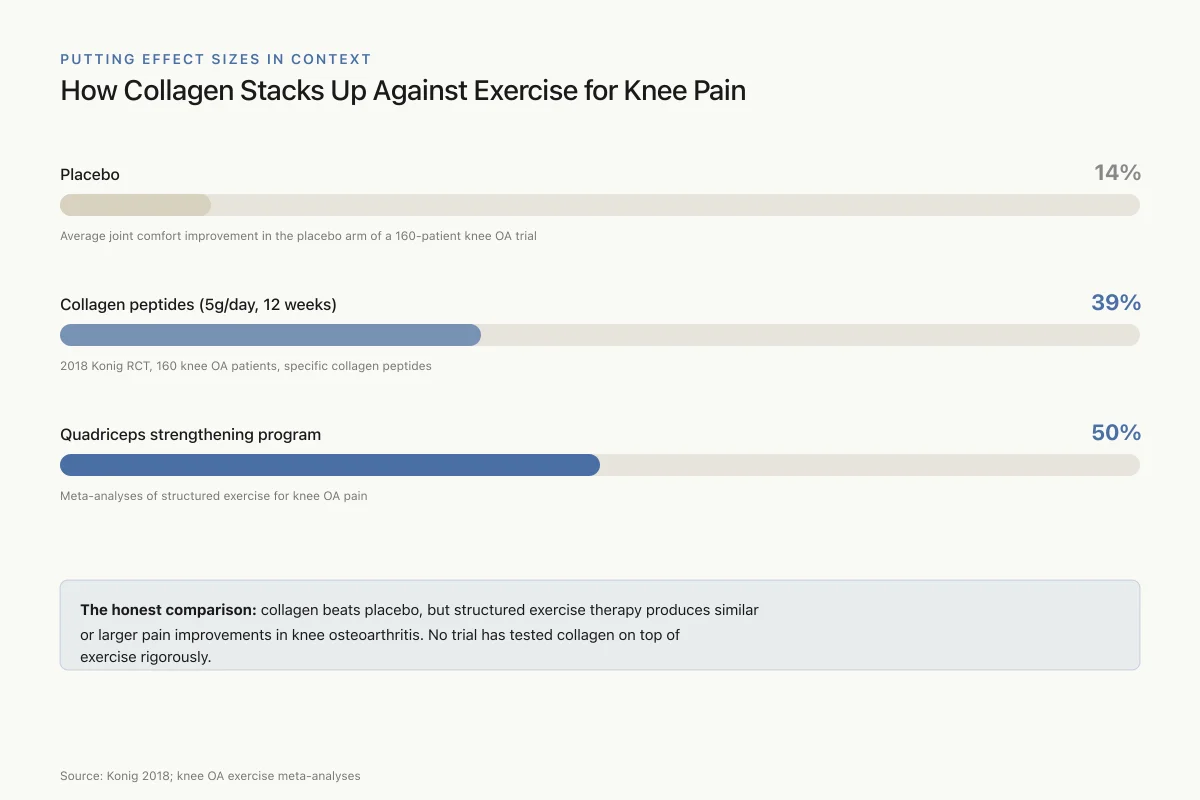
Putting Effect Sizes in Context
How Collagen Stacks Up Against Exercise for Knee Pain
Placebo
14%
Average joint comfort improvement in the placebo arm of a 160-patient knee OA trial
Collagen peptides (5g/day, 12 weeks)
39%
2018 Konig RCT, 160 knee OA patients, specific collagen peptides
Quadriceps strengthening program
50%
Meta-analyses of structured exercise for knee OA pain
The honest comparison: collagen beats placebo, but structured exercise therapy produces similar or larger pain improvements in knee osteoarthritis. No trial has tested collagen on top of exercise rigorously.
Source: Konig 2018; knee OA exercise meta-analyses
 View as image
View as imageWhere Collagen Peptide Research Stands
The evidence for collagen peptides in joint health has moved from theoretical to supported by a substantial, if imperfect, clinical trial base. The 2024 meta-analysis of 35 RCTs provides the strongest pooled evidence to date that collagen supplementation produces measurable improvements in osteoarthritis symptoms compared to placebo.
The mechanism has been partially elucidated: orally ingested collagen peptides are absorbed as bioactive di- and tripeptides, accumulate in cartilage, and stimulate chondrocyte metabolism. Whether this translates to structural cartilage preservation is the critical next question.
Collagen peptides are among the most well-tolerated supplements studied. Adverse event rates across trials are consistently comparable to placebo. They are generally recognized as safe (GRAS) by the FDA as food ingredients. The risk profile is fundamentally different from pharmacological peptides like BPC-157 or CJC-1295. The worst-case scenario with collagen supplementation is spending money on something that does not work; the worst-case scenario with unregulated injectable peptides involves contamination, infection, and unknown systemic effects.
The commercial landscape introduces its own complications. Because collagen peptides are sold as food supplements rather than drugs, they are not subject to the same manufacturing, labeling, and efficacy standards as pharmaceuticals. Product quality varies. The peptide profile of a collagen supplement depends on the source material, the hydrolysis process, and quality control during manufacturing. A positive clinical trial using a specific manufacturer's preparation does not validate all collagen peptide products on the market. Consumers have no practical way to verify that the product they purchase matches the preparation tested in clinical trials.
For those interested in the deeper cellular biology of how peptides interact with cartilage repair mechanisms, including growth factors like TGF-beta and IGF-1 that drive chondrocyte proliferation, see Growth Factor Peptides and Chondrocyte Proliferation: Rebuilding Joints. For the structural bone connection and peptides that address the mineralized tissue side of joint health, see the cross-cluster article on Abaloparatide: The Newer PTH Analog for Osteoporosis.
For the related question of how growth factor peptides interact with cartilage repair at a cellular level, see Growth Factor Peptides and Chondrocyte Proliferation: Rebuilding Joints.
The Bottom Line
Collagen peptide supplementation produces statistically significant improvements in joint pain and function in osteoarthritis, supported by a 2024 meta-analysis of 35 RCTs. The mechanism involves absorption of bioactive dipeptides (primarily Pro-Hyp) that accumulate in cartilage and stimulate chondrocyte metabolism. Effect sizes are modest (20-40% over placebo), most trials are industry-funded, and no study has demonstrated structural cartilage regeneration. The evidence supports collagen peptides as a well-tolerated supplement with modest joint benefits, not a replacement for established OA treatments.
Sources & References
- 1RPEP-02719·Liu, Dasong et al. (2015). “Collagen-Derived Peptides: From Food Ingredient to Bioactive Supplement with Cell-Level Effects.” Annual review of food science and technology.Study breakdown →PubMed →↩
- 2RPEP-05111·Sato, Kenji et al. (2020). “Collagen Dipeptide Pro-Hyp Identified as Growth Signal for Wound-Healing Fibroblasts.” Frontiers in cell and developmental biology.Study breakdown →PubMed →↩
- 3RPEP-01543·Saito, Masataka et al. (2009). “Fish Collagen Peptides Improve Cholesterol Profiles in Rats.” Journal of agricultural and food chemistry.Study breakdown →PubMed →↩
- 4RPEP-02973·Iba, Yoshinori et al. (2016). “Collagen Peptides Improve Blood Sugar Control in Mice Through GLP-1 and Other Pathways.” Journal of medicinal food.Study breakdown →PubMed →↩
- 5RPEP-02975·Inoue, Naoki et al. (2016). “Collagen Peptide Supplements Improve Skin Moisture, Elasticity, and Wrinkles in Randomized Controlled Trial.” Journal of the science of food and agriculture.Study breakdown →PubMed →↩
- 6RPEP-03765·König, Daniel et al. (2018). “Collagen Peptide Supplements Increased Bone Density in Postmenopausal Women Over 12 Months.” Nutrients.Study breakdown →PubMed →↩
- 7RPEP-04311·Lee, Hyun-Jun et al. (2019). “Oral Fish Collagen Peptides Protect Skin From UV Aging Through Absorbed Dipeptides Gly-Pro and Pro-Hyp.” Bioscience.Study breakdown →PubMed →↩
- 8RPEP-06038·Chen, Bei et al. (2022). “Effects of Collagen Hydrolysate From Large Hybrid Sturgeon on Mitigating Ultraviolet B-Induced Photodamage..” Frontiers in bioengineering and biotechnology.Study breakdown →PubMed →↩
- 9RPEP-05306·Chae, Minjung et al. (2021). “Fish Collagen Peptides Protect Skin Collagen From Stress Hormone Damage.” International journal of molecular sciences.Study breakdown →PubMed →↩
- 10RPEP-04006·Zague, Vivian et al. (2018). “Collagen Peptides Made Skin Cells Produce More Collagen and Break Down Less — Especially Sun-Damaged Cells.” Cell biology international.Study breakdown →PubMed →↩
- 11RPEP-05758·Shin, Yoon-Kyum et al. (2021). “Collagen-binding peptide reverses bone loss in a mouse model of cerebral palsy based on clinical databases..” Annals of physical and rehabilitation medicine.Study breakdown →PubMed →↩