Abaloparatide for Osteoporosis: The PTHrP Analog
Bone-Building Peptides
86% fewer vertebral fractures
In the ACTIVE trial, abaloparatide reduced new vertebral fractures by 86% versus placebo over 18 months in 2,463 postmenopausal women.
Miller et al., JAMA, 2016
Miller et al., JAMA, 2016
If you only read one thing
Abaloparatide is a bone-building injection approved for osteoporosis. It's the newer alternative to teriparatide (Forteo) and works through the same receptor, but in a way that builds bone faster at the hip with fewer side effects. In the main clinical trial, it cut spinal fractures by 86% over 18 months. You take it as a daily shot for up to 2 years, then switch to a different drug to lock in the gains.
Osteoporosis affects roughly 200 million people worldwide, yet only about 20% of high-risk patients receive treatment. For decades, the only anabolic option was teriparatide, a fragment of parathyroid hormone. Then in 2017, the FDA approved abaloparatide (brand name Tymlos), a 34-amino-acid synthetic analog of parathyroid hormone-related protein (PTHrP). In the landmark ACTIVE trial, abaloparatide cut new vertebral fractures by 86% and nonvertebral fractures by 43% versus placebo over 18 months.[1] This article covers every major clinical trial, the receptor mechanism that explains its advantage, and where the evidence stands today. For deeper dives on individual topics, see our articles on teriparatide, how abaloparatide and teriparatide compare head-to-head, parathyroid hormone biology, and calcitonin.
Key Takeaways
- Most osteoporosis drugs slow bone breakdown. Abaloparatide is different — it's an injection that actually builds new bone.
- In the pivotal trial, 86% fewer spinal fractures over just 18 months versus placebo in nearly 2,500 postmenopausal women.
- It's the newer alternative to teriparatide (Forteo), and builds bone faster at the hip — a 5x advantage in one head-to-head study.
- You take it as a daily shot for up to two years, then switch to a different drug to lock the new bone in place. Without the switch, gains fade.
- The drug nudges a single bone-building receptor the way ringing a doorbell briefly differs from holding the button. Short pulses build; sustained signal breaks down.
- Men approved in 2023 saw bigger spine-bone gains than women — about 8.5% in a year versus 1.2% on placebo.
- The two-year lifetime limit comes from rat tumors on continuous infusion, not from any human cases. No human has developed bone cancer from this class of drug.
What Is Abaloparatide?
Abaloparatide is a 34-amino-acid peptide that shares 76% sequence homology with human PTHrP(1-34) and 41% homology with human PTH(1-34).[2] Both PTH and PTHrP act through the same receptor, PTH1R, but they bind to it differently. That difference in binding is what gives abaloparatide its distinct pharmacological profile.
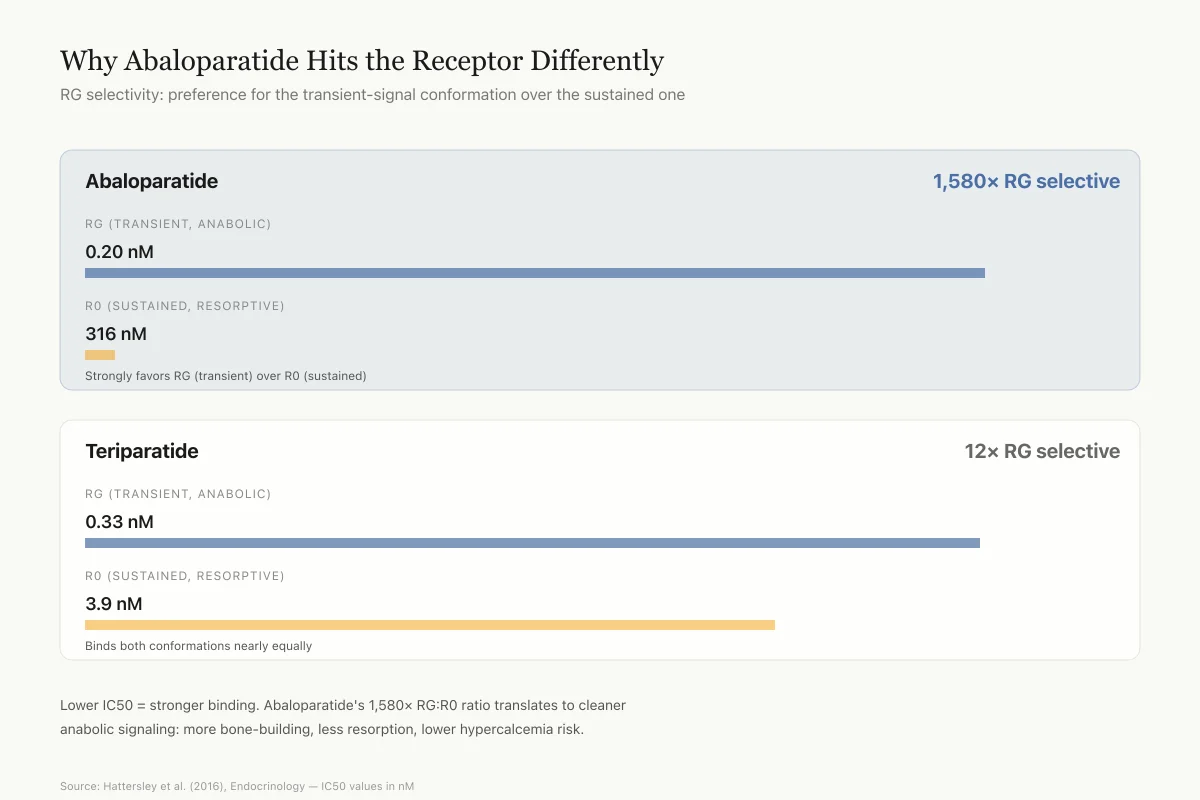
The PTH1R receptor exists in two high-affinity conformations. The R0 conformation binds ligands independently of G proteins and produces prolonged intracellular signaling. The RG conformation depends on G protein coupling and produces shorter, more transient signals.[3] Teriparatide (PTH 1-34) binds well to both conformations. Abaloparatide binds 1,580 times more strongly to RG than to R0 (IC50 values of 0.20 nM versus 316 nM), while teriparatide shows only a 12-fold difference (0.33 nM versus 3.9 nM).[3]
Receptor Binding Selectivity
Why Abaloparatide Hits the Receptor Differently
RG selectivity: how much a drug prefers the transient-signal conformation over the sustained-signal one
RG (transient, anabolic)
0.2 nM
R0 (sustained, resorptive)
316 nM
Strongly favors RG (transient) over R0 (sustained)
RG (transient, anabolic)
0.33 nM
R0 (sustained, resorptive)
3.9 nM
Binds both conformations nearly equally
Lower IC50 = stronger binding. Abaloparatide's RG:R0 ratio (1,580×) translates to a cleaner anabolic signal: more bone-building, less bone-breakdown, lower hypercalcemia risk.
Source: Hattersley et al. (2016), Endocrinology — IC50 values in nM
 View as image
View as imageThis RG selectivity matters because intermittent, transient PTH1R activation favors osteoblast-driven bone formation, while sustained activation tips the balance toward osteoclast-driven bone resorption and elevated blood calcium.[4] Abaloparatide's transient signaling pattern produces a net anabolic effect with less of the resorption and hypercalcemia that can accompany PTH(1-34). A 2021 study by Sato and colleagues confirmed that abaloparatide, teriparatide, and long-acting PTH all initially engage the same intracellular signaling pathways through PTH1R, but differ in how long those signals persist.[5] For a full explanation of how a hormone known for dissolving bone can also build it, see our article on parathyroid hormone and bone biology.
The ACTIVE Trial: Pivotal Evidence
The Abaloparatide Comparator Trial In Vertebral Endpoints (ACTIVE) was the phase 3 study that led to FDA approval. Miller and colleagues randomized 2,463 postmenopausal women (mean age 69, range 49-86) with osteoporosis to receive daily subcutaneous injections of abaloparatide 80 mcg, placebo, or open-label teriparatide 20 mcg for 18 months.[1]
Fracture outcomes
New morphometric vertebral fractures occurred in 0.58% of the abaloparatide group versus 4.22% of the placebo group, an 86% relative risk reduction (risk difference, -3.64 percentage points; 95% CI, -5.42 to -2.10). Nonvertebral fractures occurred at a rate of 2.7% with abaloparatide versus 4.7% with placebo, a 43% relative risk reduction (hazard ratio, 0.57; 95% CI, 0.32 to 0.99).[1]
Teriparatide also reduced vertebral fractures versus placebo (0.84% versus 4.22%), but its 28% reduction in nonvertebral fractures (2.9% versus 4.7%) did not reach statistical significance in this trial.[1] The trial was not powered to directly compare abaloparatide against teriparatide for fracture endpoints. For a detailed side-by-side comparison, see our article on teriparatide vs abaloparatide.
BMD changes
A post hoc analysis of the ACTIVE data by Miller et al. (2019) found that a higher percentage of abaloparatide-treated patients achieved clinically meaningful BMD gains compared to both placebo and teriparatide at the total hip and femoral neck.[6] At 18 months, more patients in the abaloparatide group crossed the threshold of >3% BMD gain at the total hip than in the teriparatide group. The difference was most pronounced at the hip and femoral neck, which are the fracture sites that carry the highest mortality risk in elderly patients.
Safety in the ACTIVE trial
Hypercalcemia occurred in 3.4% of abaloparatide patients versus 6.4% of teriparatide patients, a 47% lower rate. The most common adverse events were injection site reactions, dizziness, and nausea. Serious adverse events were similar across groups.[1]
A limitation worth noting: teriparatide was given open-label, not blinded. This could introduce bias in reporting of subjective adverse events, though it would not affect the objective fracture and BMD endpoints.
ACTIVExtend: What Happens After Abaloparatide?
Anabolic bone therapy does not work as a standalone lifetime treatment. Stopping without follow-up antiresorptive therapy leads to rapid BMD loss. The ACTIVExtend trial addressed this by enrolling ACTIVE completers into 24 months of alendronate treatment.
Early results (6 months of alendronate)
Cosman and colleagues (2017) reported the first ACTIVExtend results after 18 months of abaloparatide followed by 6 months of alendronate. BMD gains achieved during ACTIVE were preserved and continued to increase slightly during the alendronate phase. Fracture incidence remained low.[7]
Full 43-month data
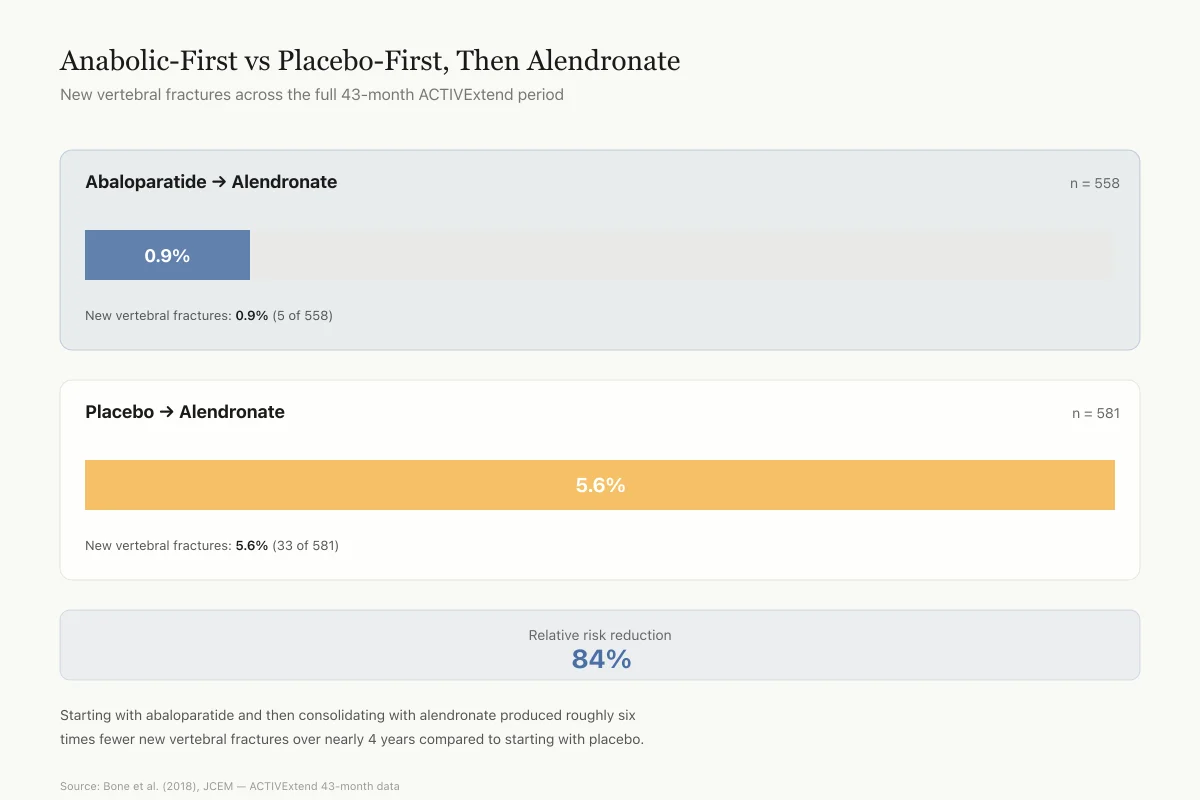
Bone et al. (2018) reported the complete ACTIVExtend results at 43 months (18 months anabolic + 24 months alendronate). Among 558 women in the abaloparatide-then-alendronate group and 581 in the placebo-then-alendronate group, new vertebral fractures occurred in 0.9% versus 5.6%, an 84% relative risk reduction.[8] Nonvertebral, clinical, and major osteoporotic fracture rates were all lower in the abaloparatide-first group.
43-Month Outcomes
Anabolic-First vs Placebo-First, Then Alendronate
New vertebral fractures across the full ACTIVExtend period
New vertebral fractures: 0.9%
New vertebral fractures: 5.6%
This is the core evidence for the "build first, lock in second" strategy. Starting with abaloparatide and then consolidating with alendronate produced roughly 6 times fewer new vertebral fractures over nearly 4 years compared to starting with placebo.
Source: Bone et al. (2018), Journal of Clinical Endocrinology & Metabolism — ACTIVExtend 43-month data
 View as image
View as imageThis finding is clinically important. It shows that the bone built during the abaloparatide phase is durable when consolidated with a bisphosphonate. The sequence of anabolic-first, antiresorptive-second produced better outcomes than antiresorptive alone.
Fracture reduction by baseline risk
Leder et al. (2019) analyzed ACTIVExtend outcomes by baseline fracture risk. The abaloparatide-then-alendronate sequence reduced fractures across all nine baseline risk subgroups tested, including women with the highest baseline fracture probability.[9] This addresses a common clinical concern: whether anabolic therapy works in the sickest patients. The data suggest it does.
Women aged 80 and older
Greenspan et al. (2020) performed a post hoc analysis of ACTIVExtend focusing on women aged 80 and older. This age group is often underrepresented in osteoporosis trials despite having the highest fracture rates. The abaloparatide-then-alendronate sequence produced BMD gains and fracture reductions consistent with the overall trial population in this elderly subgroup.[10] The numbers were small (sub-group analysis), so the confidence intervals were wide, but the direction of effect was consistent.
Earlier Clinical Development
Phase 2 dose-ranging study
Before the ACTIVE trial, Leder et al. (2015) conducted a phase 2, double-blind, placebo-controlled trial in 222 postmenopausal women. Participants received daily subcutaneous injections of abaloparatide at 20, 40, or 80 mcg, teriparatide 20 mcg, or placebo for 24 weeks.[2]
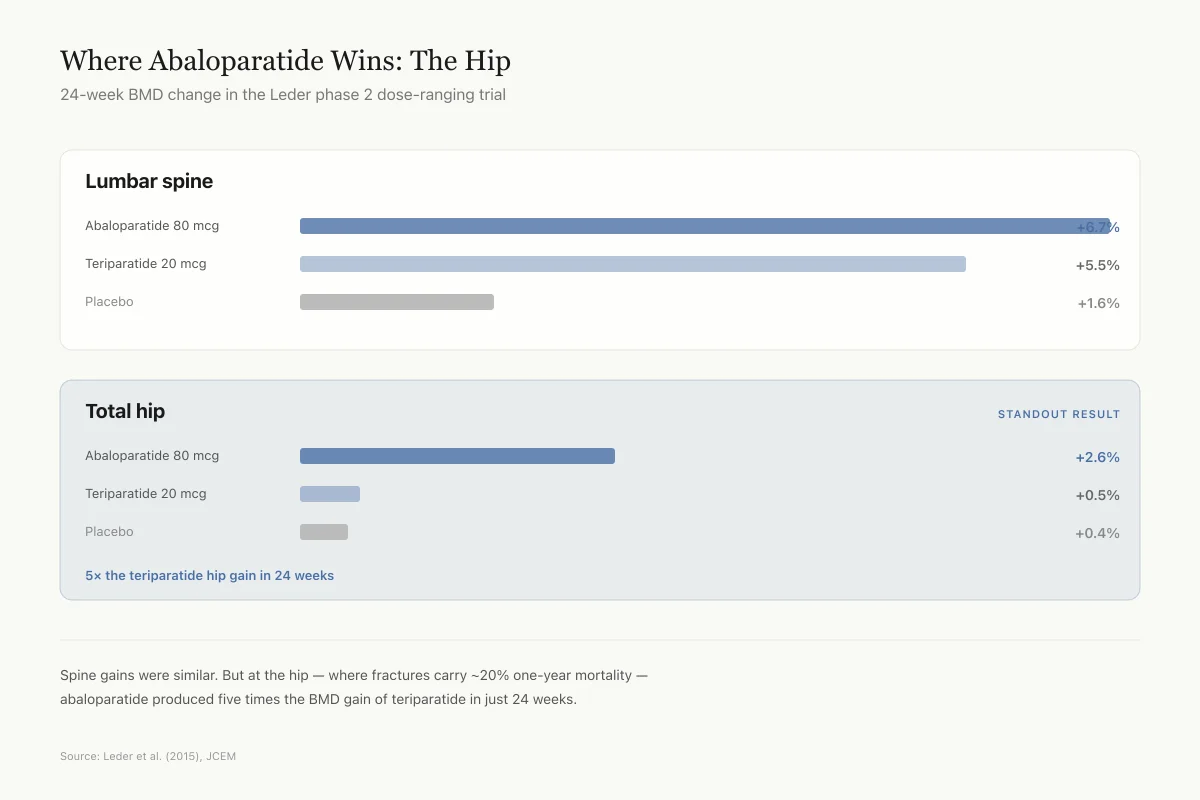
At 24 weeks, lumbar spine BMD increased by 6.7% with abaloparatide 80 mcg versus 5.5% with teriparatide and 1.6% with placebo. Total hip BMD increased by 2.6% with abaloparatide 80 mcg versus 0.5% with teriparatide and 0.4% with placebo.[2]
The total hip difference was the standout finding. Abaloparatide produced five times the hip BMD gain of teriparatide in just 24 weeks. Hip fractures are the most devastating osteoporotic fractures, with roughly 20% mortality within a year in elderly patients, so faster hip BMD improvement is clinically meaningful.
Head-to-Head BMD
Where Abaloparatide Wins: The Hip
24-week BMD change in the Leder phase 2 dose-ranging trial
Spine gains were similar. But at the hip — where fractures carry roughly 20% one-year mortality — abaloparatide produced five times the BMD gain of teriparatide in just 24 weeks.
Source: Leder et al. (2015), Journal of Clinical Endocrinology & Metabolism
 View as image
View as imageThis study is limited by its short duration (24 weeks) and small sample size (222 women). The phase 3 ACTIVE trial confirmed the BMD advantage at the hip with a much larger population.
Japanese phase 2 study
Matsumoto et al. (2023) conducted a dose-ranging study in Japanese postmenopausal women, testing abaloparatide at 40, 80, and 160 mcg daily versus placebo for 28 weeks. BMD increases were dose-dependent at the lumbar spine, total hip, and femoral neck, confirming the dose-response relationship in a non-Western population.[11] At the 80 mcg dose matching the approved U.S. formulation, lumbar spine BMD increased by 7.0% versus 0.6% for placebo. This cross-population replication strengthens confidence in the drug's efficacy beyond the predominantly White populations studied in earlier trials.
Abaloparatide in Men: The ATOM Trial
Osteoporosis in men is underdiagnosed and undertreated. About 2 million American men have the condition, and one in four men over 50 will have an osteoporotic fracture. Until 2023, abaloparatide was only approved for postmenopausal women.
The ATOM trial (Czerwinski et al., 2022) randomized 228 men aged 40-85 with osteoporosis to abaloparatide 80 mcg or placebo daily for 12 months. At 12 months, lumbar spine BMD increased by 8.48% with abaloparatide versus 1.17% with placebo. Total hip BMD increased by 2.14% versus 0.01%, and femoral neck BMD by 2.98% versus 0.15%. All differences reached p<0.0001.[12]
The most common adverse events in the ATOM trial were injection site reactions, dizziness, nasopharyngitis, arthralgia, bronchitis, and headache, consistent with the safety profile seen in women.[12]
The trial was not powered for fracture endpoints. It measured BMD as a surrogate. BMD is a validated predictor of fracture risk, but the absence of direct fracture data in men is a gap. Based on the ATOM results, the FDA expanded abaloparatide's approval to include men with osteoporosis at high risk for fracture in June 2023.
PTH and PTHrP Biology: Why the Receptor Matters
Understanding why abaloparatide works requires understanding the PTH1R receptor system. Martin (2021) published a comprehensive review of PTH and PTHrP biology in Endocrine Reviews, covering the shared receptor and divergent physiological roles of these two ligands.[4]
PTH is an endocrine hormone released by the parathyroid glands when blood calcium drops. Its primary job is to raise calcium by pulling it from bone, increasing kidney reabsorption, and boosting intestinal absorption via vitamin D activation. PTHrP, by contrast, acts mostly as a local paracrine factor. In bone, PTHrP produced by osteoblast-lineage cells helps regulate the rate of bone turnover in a localized, tightly controlled manner.[4]
Both hormones signal through PTH1R, but their different binding characteristics produce different signaling durations. Abaloparatide was engineered to mimic the transient, PTHrP-like signaling pattern while avoiding the prolonged, PTH-like pattern that favors resorption. This is the pharmacological basis for its favorable bone-formation-to-resorption ratio.
The distinction between endocrine and paracrine PTH1R signaling also explains the osteosarcoma concern. Continuous endocrine-level PTH1R stimulation in rats (achieved by implanted slow-release pumps) drove bone tumors. The brief, once-daily pulse of abaloparatide mimics the physiological paracrine pattern of PTHrP, which is how bone cells normally communicate with each other. This difference between sustained and pulsatile receptor activation is central to both the drug's efficacy and its safety profile.
For a deeper exploration of this topic, see parathyroid hormone and bone.
Beyond Injection: The Transdermal Patch
Daily self-injection is a barrier to adherence. About 30-40% of patients prescribed teriparatide discontinue within the first year. Abaloparatide developers pursued a transdermal delivery system using solid microstructured technology (sMTS), essentially a small patch with microscopic projections that deliver the peptide through the skin.
Miller et al. (2021) published phase 1b results of abaloparatide-sMTS in 22 postmenopausal women with low BMD. Participants self-administered a 300 mcg patch to the thigh daily for 29 days. The patch delivered abaloparatide with pharmacokinetic profiles suitable for once-daily dosing, and bone turnover markers responded positively. P1NP (a marker of bone formation) increased while CTX (a marker of bone resorption) showed a smaller rise, mirroring the favorable formation-to-resorption ratio seen with the injection.[13]
The phase 3 wearABLe trial compared the patch against subcutaneous abaloparatide in approximately 470 women. However, the patch did not meet its primary endpoint of non-inferiority, with spine BMD increasing 7.1% with the patch versus 10.9% with the injection. The transdermal program has not advanced further.
This is an honest account of a clinical failure. The patch concept was sound, but the technology did not deliver enough abaloparatide to match injection efficacy. Future transdermal peptide delivery systems may solve this problem, but for now, abaloparatide remains injection-only.
How Abaloparatide Fits in the Treatment Landscape
A 2020 review by Miller summarized abaloparatide's place among osteoporosis therapies.[14] The current anabolic options are:
- Teriparatide (Forteo): PTH(1-34) fragment, approved 2002, daily injection, 24-month limit. The first bone anabolic. Builds bone at the spine and hip, but with more resorption and higher hypercalcemia rates than abaloparatide.
- Abaloparatide (Tymlos): PTHrP(1-34) analog, approved 2017, daily injection, 24-month limit. Faster hip BMD gains, lower hypercalcemia rates. The RG-selective mechanism produces a cleaner anabolic signal.
- Romosozumab (Evenity): Anti-sclerostin antibody, approved 2019, monthly injection, 12-month limit. Different mechanism entirely (Wnt pathway), strongest spine BMD gains of the three, but carries a cardiovascular warning based on the ARCH trial.
All three share a critical constraint: limited treatment duration. Cumulative use of abaloparatide and teriparatide together cannot exceed 24 months in a patient's lifetime. This limit exists because long-term continuous PTH receptor stimulation in rats caused osteosarcoma (bone cancer). No osteosarcoma cases have been linked to these drugs in humans across decades of clinical use and post-marketing surveillance, but the regulatory restriction remains.
Safety
Moderate24-month lifetime limit on PTH-type bone drugs
Concern
Rats given continuous PTH receptor stimulation developed bone tumors (osteosarcoma). The FDA limits total cumulative use of abaloparatide and teriparatide combined to 24 months per lifetime.
What the research says
No osteosarcoma cases have ever been linked to these drugs in humans across decades of use and post-marketing surveillance. The rat studies used continuous infusion, not the brief daily pulse humans receive. The limit is precautionary.
Particularly relevant for: All patients taking abaloparatide or teriparatide
What to do
Track cumulative months on any PTH-type drug. If you've used teriparatide before, that time counts toward your 24-month total. Plan the transition to a maintenance drug (like alendronate) with your doctor.
FDA prescribing information; Miller (2020) review
The standard treatment sequence is: anabolic therapy first, followed by a bisphosphonate (like alendronate) or denosumab to consolidate gains. The ACTIVExtend data support this approach specifically for abaloparatide followed by alendronate. Starting with antiresorptive therapy and switching to anabolic therapy later (the reverse sequence) is less effective, as shown by studies with romosozumab, because antiresorptives slow the bone remodeling that anabolics depend on.
Dosing and Administration
Abaloparatide is supplied as a prefilled pen injector containing a 30-day supply. The approved dose is 80 mcg injected subcutaneously once daily into the periumbilical region (around the navel) of the abdomen. The pen requires refrigeration. Patients self-administer the injection after training.
The first dose should be given where the patient can sit or lie down, because orthostatic hypotension (a temporary drop in blood pressure upon standing) can occur, usually within four hours of injection. This effect tends to diminish with continued use.
Calcium and vitamin D supplementation is typically used alongside abaloparatide, as adequate calcium and vitamin D are prerequisites for any bone-building therapy to work effectively. The ACTIVE trial required participants to take 500-1,000 mg of calcium and 400-800 IU of vitamin D daily throughout the study.[1]
Limitations of the Evidence
The abaloparatide evidence base has real gaps that are worth naming directly.
Head-to-head fracture comparison: The ACTIVE trial was not powered to compare fracture rates between abaloparatide and teriparatide directly. The BMD data favor abaloparatide at the hip, and the fracture trends favor it for nonvertebral fractures, but a definitive superiority claim for fractures requires a dedicated trial that has not been conducted.
Open-label teriparatide arm: In ACTIVE, teriparatide was administered open-label while abaloparatide and placebo were blinded. This design choice was made for regulatory reasons but introduces potential bias.
Limited diversity: The ACTIVE trial population was predominantly White and postmenopausal. The ATOM trial in men was smaller (228 versus 2,463) and measured BMD, not fractures. Data in non-White populations remain limited outside the Japanese phase 2 study.
No fracture data in men: The ATOM trial used BMD as a surrogate endpoint. While BMD predicts fracture risk, direct fracture reduction has not been demonstrated in men.
Manufacturer involvement: Radius Health (now Ipsen) funded most of the clinical trials. This is typical for pharmaceutical development but should be noted when evaluating the evidence.
Long-term safety ceiling: The 24-month treatment cap means long-term continuous safety data do not exist and likely never will for this drug class.
The Bottom Line
Abaloparatide is a 34-amino-acid PTHrP analog that builds bone through transient, RG-selective activation of the PTH1 receptor. The ACTIVE trial demonstrated 86% vertebral and 43% nonvertebral fracture reduction versus placebo over 18 months in 2,463 women. Sequential treatment with alendronate preserves these gains through at least 43 months. The drug offers faster hip BMD improvements and lower hypercalcemia rates compared to teriparatide, though a definitive head-to-head fracture superiority trial has not been conducted. Approval expanded to men in 2023 based on strong BMD data from the ATOM trial.
Sources & References
- 1RPEP-03050·Miller, Paul D et al. (2016). “Effect of Abaloparatide vs Placebo on New Vertebral Fractures in Postmenopausal Women With Osteoporosis: A Randomized Clinical Trial..” JAMA.Study breakdown →PubMed →↩
- 2RPEP-02702·Leder, Benjamin Z et al. (2015). “Effects of abaloparatide, a human parathyroid hormone-related peptide analog, on bone mineral density in postmenopausal women with osteoporosis..” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 3RPEP-02960·Hattersley, Gary et al. (2016). “Why Abaloparatide May Build Bone With Fewer Side Effects Than Teriparatide: A Receptor Binding Study.” Endocrinology.Study breakdown →PubMed →↩
- 4RPEP-05582·Martin, T John et al. (2021). “PTH and PTHrP Peptide Hormones: Dual Roles in Bone Formation and Breakdown.” Endocrine reviews.Study breakdown →PubMed →↩
- 5RPEP-05742·Sato, Tadatoshi et al. (2021). “Comparable Initial Engagement of Intracellular Signaling Pathways by Parathyroid Hormone Receptor Ligands Teriparatide, Abaloparatide, and Long-Acting PTH..” JBMR plus.Study breakdown →PubMed →↩
- 6RPEP-04372·Miller, P D et al. (2019). “More Osteoporosis Patients Gained Bone on Abaloparatide Than on Teriparatide or Placebo.” Bone.Study breakdown →PubMed →↩
- 7RPEP-03253·Cosman, Felicia et al. (2017). “Eighteen Months of Treatment With Subcutaneous Abaloparatide Followed by 6 Months of Treatment With Alendronate in Postmenopausal Women With Osteoporosis: Results of the ACTIVExtend Trial..” Mayo Clinic proceedings.Study breakdown →PubMed →↩
- 8RPEP-03591·Bone, Henry G et al. (2018). “ACTIVExtend: 24 Months of Alendronate After 18 Months of Abaloparatide or Placebo for Postmenopausal Osteoporosis..” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 9RPEP-04308·Leder, Benjamin Z et al. (2019). “Fracture and Bone Mineral Density Response by Baseline Risk in Patients Treated With Abaloparatide Followed by Alendronate: Results From the Phase 3 ACTIVExtend Trial..” Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research.Study breakdown →PubMed →↩
- 10RPEP-04826·Greenspan, Susan L et al. (2020). “Abaloparatide followed by alendronate in women ≥80 years with osteoporosis: post hoc analysis of ACTIVExtend..” Menopause (New York.Study breakdown →PubMed →↩
- 11RPEP-07165·Matsumoto, Toshio et al. (2023). “Bone-Building Peptide Abaloparatide Increases Bone Density Up to 11.5% in Osteoporosis Trial.” Journal of bone and mineral metabolism.Study breakdown →PubMed →↩
- 12RPEP-06070·Czerwinski, Edward et al. (2022). “The Efficacy and Safety of Abaloparatide-SC in Men With Osteoporosis: A Randomized Clinical Trial..” Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research.Study breakdown →PubMed →↩
- 13RPEP-05610·Miller, Paul D et al. (2021). “Osteoporosis Peptide Drug Delivered by Skin Patch Shows Promise in Early Trial.” Clinical drug investigation.Study breakdown →PubMed →↩
- 14RPEP-04999·Miller, Paul D et al. (2020). “Abaloparatide: an anabolic treatment to reduce fracture risk in postmenopausal women with osteoporosis..” Current medical research and opinion.Study breakdown →PubMed →↩