Collagen for Exercise Joint Pain: Study Evidence
Collagen Peptides and Exercise
5 Randomized Controlled Trials
Five RCTs have directly tested collagen peptide supplementation for exercise-related joint pain in active adults, with consistent reductions in activity-related knee pain at doses of 5-20 g per day.
Zdzieblik et al., Appl Physiol Nutr Metab, 2017
Zdzieblik et al., Appl Physiol Nutr Metab, 2017
If you only read one thing
Collagen peptides at 5 to 20 grams a day modestly reduce exercise-related knee pain in active adults. You need to stay on it for at least 12 weeks before you notice anything, and the effect only shows up clearly in people who actually train often — over three hours a week. It is safe, the data is consistent, but the gap over placebo is small. Most studies were funded by collagen makers, so treat it as a supporting tool, not a fix.
Joint pain is the most common reason active adults reduce or stop exercise. Knee discomfort during or after physical activity affects young athletes, recreational runners, and aging fitness enthusiasts, often without a diagnosable joint disease like osteoarthritis. Collagen peptides, hydrolyzed fragments of collagen protein that can be absorbed orally, have been tested in multiple randomized controlled trials for this specific problem. A 2021 systematic review of 15 RCTs concluded that collagen peptides are "most beneficial in improving joint functionality and reducing joint pain," though exact mechanisms remain unclear.[1] This article covers every major trial, what the data shows, and where the evidence falls short. For how collagen peptides interact with tendons and ligaments specifically, see Collagen Peptides for Athletes: Tendon and Ligament Evidence.
Key Takeaways
- 5 g/day of specific collagen peptides reduced activity-related knee pain by 19.5 mm on VAS compared to 13.9 mm for placebo over 12 weeks in 139 athletes (Zdzieblik et al., Appl Physiol Nutr Metab, 2017)
- A confirmatory RCT in 180 young adults replicated the finding: 21.9 mm VAS reduction vs. 15.6 mm for placebo at the same dose (Zdzieblik et al., Nutrients, 2021)
- 10 g/day over 6 months improved pain scores only in high-frequency exercisers training more than 180 minutes per week (Kviatkovsky et al., JISSN, 2023)
- A systematic review of 15 RCTs found collagen peptides consistently reduced joint pain and improved functionality across different populations (Khatri et al., Amino Acids, 2021)
- No adverse effects were reported in any trial, even at doses up to 60 g/day, making collagen peptides one of the safest studied supplements for joint pain
- Effects require at least 12 weeks of daily supplementation; shorter protocols have not shown measurable joint pain improvements
What Are Collagen Peptides?
Collagen is the most abundant protein in the human body, comprising roughly 30% of total protein mass. It forms the structural scaffold of tendons, ligaments, cartilage, bone, and skin. When collagen protein is broken down through hydrolysis (enzymatic or chemical), the resulting fragments are called collagen peptides or collagen hydrolysate.
Commercial collagen peptides typically have a molecular weight between 2,000 and 6,000 daltons, small enough to survive digestion and appear in the bloodstream as di- and tripeptides. The most characteristic of these is hydroxyproline, an amino acid almost unique to collagen. Human studies have confirmed that hydroxyproline-containing peptides appear in blood plasma within 1-2 hours of oral collagen ingestion.
The collagen peptides used in joint pain research are typically derived from bovine or porcine type I collagen, with specific bioactive collagen peptides (marketed as FORTIGEL or similar) engineered to have a mean molecular weight around 3 kDa. These specific peptides are distinct from generic collagen supplements sold in bulk form, which may vary in peptide size, source, and bioactivity.
Type I collagen dominates tendons, ligaments, and bone. Type II collagen is the primary collagen of articular cartilage. Most joint pain studies use type I collagen peptides, which raises a question: how do type I fragments improve joint function when cartilage is primarily type II? The proposed answer involves signaling rather than direct structural replacement.
The Clinical Trial Evidence
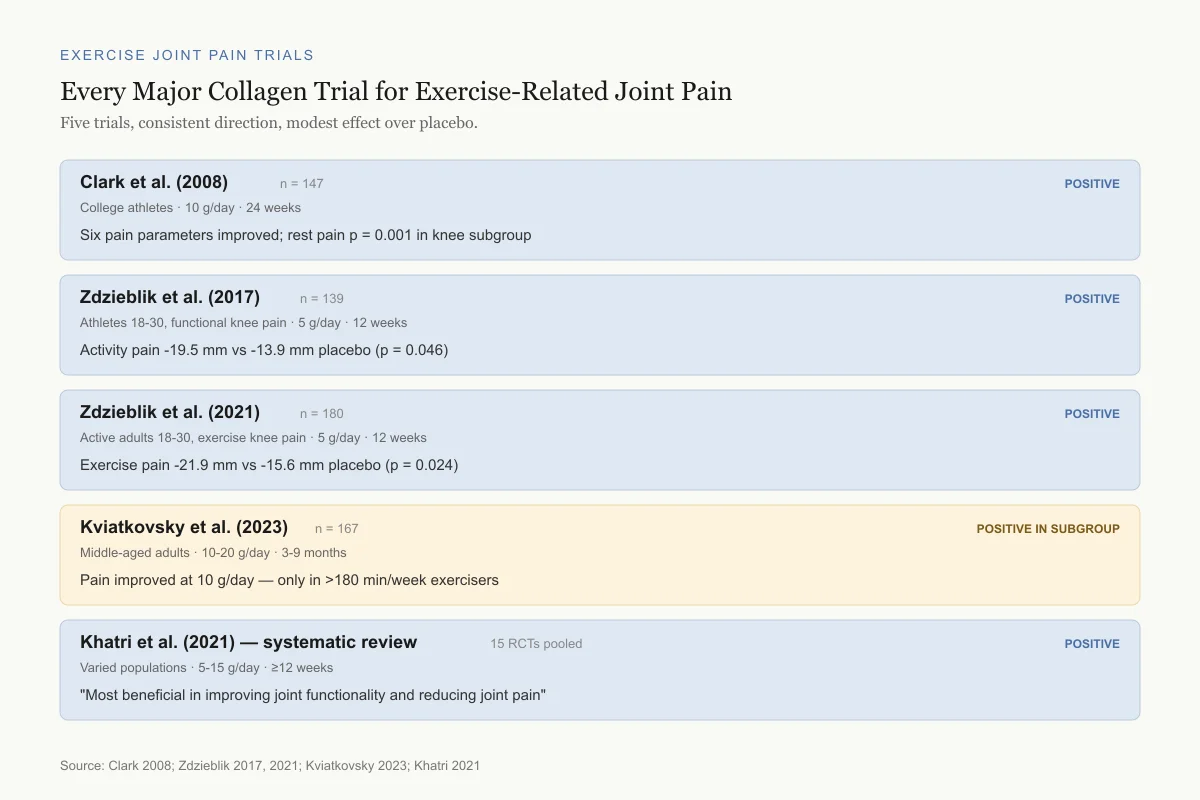
Exercise Joint Pain Trials
Every Major Collagen Trial for Exercise-Related Joint Pain
Five trials, consistent direction, modest effect over placebo.
Clark et al. (2008)
n = 147Who: College athletes with joint pain
Dose: 10 g/day
Length: 24 weeks
Six pain parameters improved; rest pain p = 0.001 in knee subgroup
Zdzieblik et al. (2017)
n = 139Who: Young athletes (18-30), functional knee pain
Dose: 5 g/day
Length: 12 weeks
Activity pain -19.5 mm vs -13.9 mm placebo (p = 0.046)
Zdzieblik et al. (2021)
n = 180Who: Active adults (18-30), exercise knee pain
Dose: 5 g/day
Length: 12 weeks
Exercise pain -21.9 mm vs -15.6 mm placebo (p = 0.024)
Kviatkovsky et al. (2023)
n = 167Who: Middle-aged active adults
Dose: 10 g or 20 g/day
Length: 3-9 months
Pain improved at 10 g/day — but only in >180 min/week exercisers
Khatri et al. (review) (2021)
n = 15Who: 15 RCTs pooled
Dose: 5-15 g/day
Length: ≥12 weeks
"Most beneficial in improving joint functionality and reducing joint pain"
Source: Clark 2008; Zdzieblik 2017, 2021; Kviatkovsky 2023; Khatri 2021
 View as image
View as imageZdzieblik 2017: The First Activity-Related Pain Trial
The first RCT specifically targeting exercise-induced joint pain (not osteoarthritis) was published by Zdzieblik and colleagues in Applied Physiology, Nutrition, and Metabolism in 2017.[2] They enrolled 139 athletic subjects (ages 18-30) with functional knee pain during sport but no diagnosed joint disease.
Participants received either 5 g of specific bioactive collagen peptides or placebo daily for 12 weeks. Pain was assessed using the Visual Analog Scale (VAS, 0-100 mm) during and after exercise.
Results:
- Activity-related pain decreased by 19.5 mm (VAS) in the collagen group vs. 13.9 mm in placebo (p = 0.046)
- Physician assessment confirmed: 16.7 mm improvement vs. 12.2 mm for placebo (p = 0.021)
- Resting pain improved but did not reach significance vs. placebo (10.2 mm vs. 7.4 mm, p = 0.209)
- Use of additional treatments (ointments, physiotherapy) was significantly reduced after collagen intake
The 5.6 mm difference between collagen and placebo on VAS may appear modest, but the study population had functional joint pain, not severe joint disease. The baseline pain levels were moderate, limiting the magnitude of possible improvement. The reduction in additional therapy use suggests practical quality-of-life benefits beyond the VAS score.
Pain Reduction Data
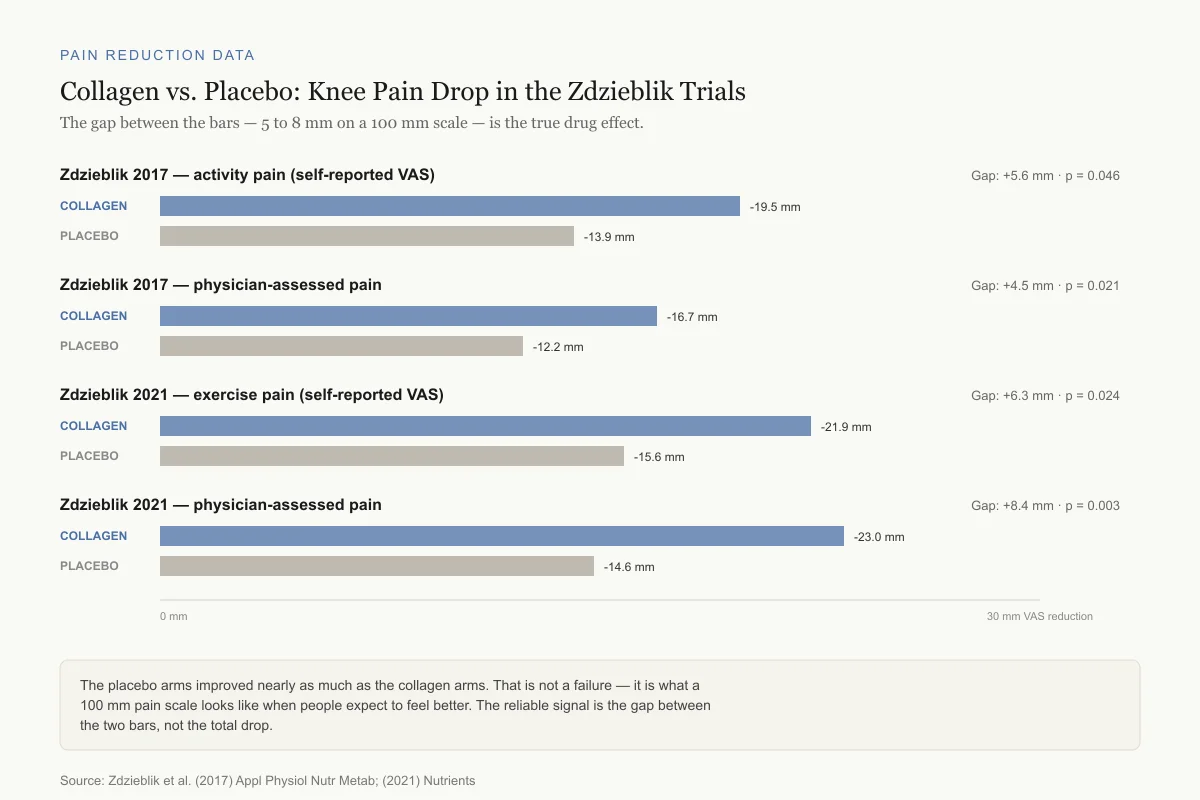
Collagen vs. Placebo: Knee Pain Drop in the Zdzieblik Trials
Both arms improve. The gap between them — 5 to 8 mm on a 100 mm scale — is the true drug effect.
Zdzieblik — activity pain · 2017
Gap: +5.6 mm · p = 0.046Self-reported VAS
Zdzieblik — physician pain · 2017
Gap: +4.5 mm · p = 0.021Physician assessment
Zdzieblik — exercise pain · 2021
Gap: +6.3 mm · p = 0.024Self-reported VAS
Zdzieblik — physician pain · 2021
Gap: +8.4 mm · p = 0.003Physician assessment
The placebo arms improved nearly as much as the collagen arms. That is not a failure — it is what a 100 mm pain scale looks like when people expect to feel better. The reliable signal is the gap between the two bars, not the total drop.
Source: Zdzieblik et al. (2017) Appl Physiol Nutr Metab; (2021) Nutrients
 View as image
View as imageZdzieblik 2021: The Confirmatory Trial
Zdzieblik et al. published a larger confirmatory trial in Nutrients in 2021 with 180 active men and women (ages 18-30) with exercise-related knee pain and no diagnosed joint disease.[3] The same protocol was used: 5 g of type I collagen peptides (mean molecular weight 3 kDa) vs. placebo for 12 weeks.
Results:
- Exercise-induced knee pain decreased by 21.9 mm (VAS) in the collagen group vs. 15.6 mm for placebo (p = 0.024)
- Physician-assessed pain: 23.0 mm vs. 14.6 mm reduction (p = 0.003)
- Resting pain and post-squat pain showed no significant differences (too few participants had pain under these conditions)
- Knee joint mobility was clinically unremarkable at baseline and did not change
The stronger physician-assessed significance (p = 0.003) compared to self-reported (p = 0.024) is noteworthy. Physician assessments used standardized examination criteria rather than subjective self-report, suggesting the treatment effect was detectable by clinical evaluation. This trial replicated and confirmed the 2017 findings in a larger cohort.
Kviatkovsky 2023: The Long-Term Dose-Ranging Study
Kviatkovsky et al. published in the Journal of the International Society of Sports Nutrition in 2023 the longest and most comprehensive collagen joint pain trial to date.[4] This was a double-blind RCT with three groups: placebo, 10 g/day collagen peptides, and 20 g/day collagen peptides, followed for 3, 6, and 9 months.
The population was middle-aged active adults (different from the younger Zdzieblik cohorts). Outcomes included the KOOS (Knee Injury and Osteoarthritis Outcomes Score) for pain and function, and VR-12 for physical and mental health.
Key findings:
- 10 g/day improved activities of daily living (p = 0.031, effect size 0.096) and pain (p = 0.037, effect size 0.164) over 6 months
- Pain improvements at 10 g/day appeared only in high-frequency exercisers (more than 180 minutes/week)
- Mental component scores improved with 10 g/day over 3-9 months (p = 0.017, effect size 0.309)
- Physical component scores improved with 20 g/day over 3-9 months, but only in females (p = 0.013, effect size 0.582)
- 20 g/day did not produce consistently better outcomes than 10 g/day for pain
The exercise frequency finding is important. Collagen peptides reduced pain only in people who exercised more than 3 hours per week. This suggests the supplement may specifically address exercise-induced joint stress rather than generalized joint discomfort. For how exercise interacts with collagen supplementation at the muscular level, see Does Collagen Help Muscle Recovery After Exercise?.
Exercise Threshold Finding
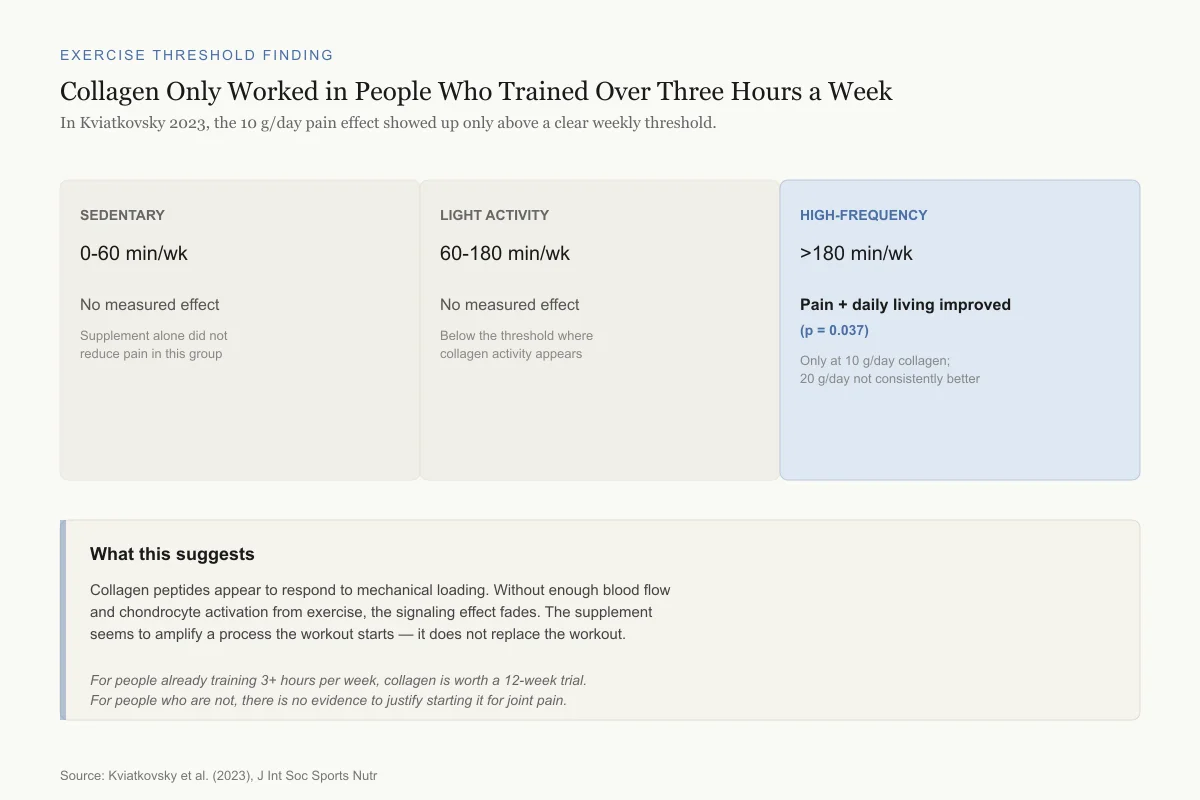
Collagen Only Worked in People Who Trained Over Three Hours a Week
In Kviatkovsky 2023, the 10 g/day effect on pain showed up only above a clear weekly threshold.
Sedentary
0-60 min/wk
No measured effect
Light activity
60-180 min/wk
No measured effect
High-frequency
>180 min/wk
Pain + daily living improved (p = 0.037)
This pattern suggests collagen peptides respond to mechanical loading. Without enough blood flow and chondrocyte activation from exercise, the signaling effect appears to fade. The supplement seems to amplify a process the workout starts, not replace it.
Source: Kviatkovsky et al. (2023), J Int Soc Sports Nutr
 View as image
View as imageThe mental health improvement (effect size 0.309 for 10 g/day) was unexpected and represents one of the largest effect sizes in the trial. Pain reduction that improves activity levels could plausibly improve mental health, but the mechanism is unclear. The female-specific physical health response at 20 g/day had the largest effect size in the study (0.582) but the narrowest subgroup, making it hypothesis-generating rather than conclusive.
The Clark 2008 Pioneer Study
The first major trial of collagen hydrolysate for athlete joint pain was a 24-week study at Penn State University by Clark et al. published in Current Medical Research and Opinion in 2008. Though conducted before the newer bioactive collagen peptides were available, it established the clinical precedent. The study enrolled 147 college athletes (72 male, 75 female) who received 10 g of collagen hydrolysate or placebo daily. When all subjects were analyzed, six pain parameters showed significant improvement with collagen. In the subgroup with knee pain (n = 63), the effect was more pronounced, with joint pain at rest reaching p = 0.001 significance. This was the first clinical trial to demonstrate collagen's potential for athlete joint pain and motivated the subsequent Zdzieblik program.
The Systematic Review Evidence
Khatri 2021: 15 RCTs Evaluated
Khatri et al. published a systematic review in Amino Acids in 2021, screening 856 articles and selecting 15 RCTs that tested collagen peptide supplementation combined with exercise.[1]
Their conclusions by outcome:
Joint pain and recovery: Collagen peptides are "most beneficial in improving joint functionality and reducing joint pain." The effective dose range was 5-15 g/day, with improvements appearing after at least 12 weeks. Five grams per day appeared as effective as 10 g/day for pain relief in the absence of degenerative joint disease.
Body composition: Some improvements in lean mass were observed, particularly when collagen was combined with resistance training (as in the Zdzieblik 2015 study of elderly sarcopenic men).[5] These effects were modest and not consistent across all trials.
Collagen synthesis: 15 g/day of collagen peptides elevated collagen synthesis rates (measured by procollagen I N-terminal peptide) but did not increase muscle protein synthesis compared to isonitrogenous higher-quality protein sources. This suggests collagen peptides specifically stimulate connective tissue synthesis rather than general protein synthesis.
Muscle recovery: Evidence was mixed. Some trials reported reduced muscle soreness, but the effects were inconsistent and smaller than those seen for joint pain.
The review noted important limitations: most studies used recreational athletes (not elite), few included female participants in adequate numbers, and outcome measures relied heavily on subjective pain scales rather than objective imaging or biomarkers.
Proposed Mechanisms: How Collagen Peptides Reduce Joint Pain
Collagen peptides do not rebuild cartilage directly. At 5-20 g per day, the amount reaching joint tissue is far too small for structural repair. The proposed mechanisms are signaling-based.
Stimulation of Extracellular Matrix Synthesis
When hydroxyproline-containing peptides are absorbed from the gut and reach joint tissue, they appear to stimulate resident chondrocytes and fibroblasts to increase their own collagen production. In vitro studies have shown that collagen-derived peptides upregulate type I, II, and IV collagen expression, as well as proteoglycan and elastin synthesis in cartilage explants. The peptides act as molecular signals, not building blocks.
Anti-Inflammatory Activity
Glycine, the most abundant amino acid in collagen (comprising roughly one-third of its sequence), has demonstrated anti-inflammatory properties. Glycine inhibits the release of pro-inflammatory cytokines including TNF-alpha and IL-6. Since exercise-induced joint pain involves local inflammation, glycine-mediated anti-inflammatory effects could contribute to pain reduction.
Timing with Exercise
The Kviatkovsky finding that pain reduction occurred only in high-frequency exercisers suggests a synergistic effect between collagen supplementation and exercise-induced mechanical loading. Exercise stimulates blood flow to joint tissue and activates mechanosensitive pathways in chondrocytes and tenocytes. Collagen peptides circulating during and after exercise may enhance this activation. This is consistent with the recommendation to take collagen peptides 30-60 minutes before exercise, allowing peak blood levels during the activity session. For the specific role of vitamin C in this process, see Vitamin C and Collagen: Why You Need Both for Synthesis.
Dose, Timing, and Duration: What the Trials Used
The clinical trials provide a narrow but consistent picture of effective protocols:
Dose: 5 g/day was effective for young athletes with functional knee pain (Zdzieblik 2017, 2021). 10 g/day was effective for middle-aged adults exercising more than 180 minutes per week (Kviatkovsky 2023). 20 g/day did not produce consistently superior outcomes to 10 g/day. The Khatri 2021 review concluded that 5 g/day may be as effective as 10 g/day in the absence of degenerative joint disease.
Type: All positive joint pain trials used specific bioactive collagen peptides (type I, mean molecular weight ~3 kDa). Whether generic collagen hydrolysate products produce the same effects is untested. The distinction between specific bioactive peptides and bulk collagen powder is significant and often overlooked.
Duration: No trial shorter than 12 weeks showed significant joint pain reduction. The Kviatkovsky trial found that 6-9 months produced the most robust effects. The 12-week minimum is consistent across all positive trials and matches the expected timeline for connective tissue turnover.
Timing: While the mechanism data suggests taking collagen 30-60 minutes before exercise, the clinical trials mostly used daily supplementation without specified timing relative to exercise sessions. The timing recommendation comes from collagen synthesis studies rather than pain outcome trials.
Safety: No adverse effects were reported in any trial. The Khatri review noted that doses up to 60 g/day showed no safety signals. Collagen peptides are generally recognized as safe food-grade ingredients.
How the Evidence Compares to Other Joint Supplements
The joint supplement market includes glucosamine, chondroitin, hyaluronic acid, omega-3 fatty acids, and curcumin. Collagen peptides occupy a distinct position in this landscape.
Glucosamine and chondroitin have the largest evidence base, with decades of osteoarthritis trials. However, recent large-scale trials (GAIT, LEGS) produced mixed results, and meta-analyses disagree on whether these supplements provide meaningful pain relief beyond placebo. The key difference: glucosamine/chondroitin data is primarily in OA patients, while collagen peptide data targets functional joint pain in active adults without degenerative disease.
Omega-3 fatty acids (EPA/DHA) reduce joint inflammation through resolvin and protectin synthesis. Their mechanism is purely anti-inflammatory, while collagen peptides combine anti-inflammatory activity (via glycine) with stimulation of connective tissue synthesis. The mechanisms are complementary rather than overlapping, suggesting they could theoretically be combined, though no trial has tested this combination for exercise-related joint pain.
Curcumin (from turmeric) has anti-inflammatory evidence in OA populations but limited data in exercise-induced joint pain specifically. Its poor oral bioavailability requires specialized formulations (liposomal, piperine-enhanced) that add complexity.
The practical advantage of collagen peptides is their safety profile and the specificity of evidence for exercise-related joint pain. The disadvantage is the industry funding concentration and the specificity of the tested products. A consumer purchasing generic collagen powder cannot assume the same results as trials using specific bioactive collagen peptides with a 3 kDa molecular weight.
Collagen peptides combined with resistance training also showed improvements in body composition in elderly sarcopenic men. Zdzieblik et al. (2015) reported that 15 g/day of collagen peptides plus resistance training 3 times per week produced greater increases in fat-free mass and greater decreases in fat mass compared to placebo plus the same training program over 12 weeks.[5] While this study focused on sarcopenia rather than joint pain, it demonstrates that collagen peptide supplementation interacts with exercise training to produce measurable tissue-level changes.
What Collagen Peptides Are Not
Collagen peptides for exercise joint pain occupy a specific evidence niche. Interpreting the data correctly requires understanding what the studies did not test:
Not a treatment for osteoarthritis: The Zdzieblik trials explicitly excluded participants with diagnosed joint disease. While some evidence exists for collagen in OA (including a separate RCT showing improvement in OA patients), the exercise joint pain data applies to functional joint discomfort in healthy joints.
Not a replacement for structural repair: Collagen peptides do not regenerate destroyed cartilage, repair torn ligaments, or heal fractures. They appear to reduce pain associated with exercise-related joint stress, not reverse structural damage. For peptides studied for structural tendon repair, see BPC-157 for Tendon Injuries: What the Animal Studies Show.
Not equivalent to generic collagen supplements: The bioactive collagen peptides used in clinical trials (specific molecular weight, specific source, specific enzymatic hydrolysis process) are different from generic collagen powder. Product-to-product variability is a concern that the systematic review highlighted.
Not a substitute for exercise modification: The trials tested collagen as an adjunct to continued physical activity, not as a replacement for load management, technique correction, or rehabilitation.
Limitations of the Current Evidence
The collagen joint pain literature has consistent positive results but also notable weaknesses:
Funding: Multiple trials were funded by GELITA AG, a collagen peptide manufacturer. The Zdzieblik studies, the most-cited positive results, were conducted with GELITA-supplied products. This does not invalidate the results, but it means independent replication by non-industry researchers is limited.
Subjective outcomes: All primary endpoints used self-reported or physician-assessed pain scales (VAS, KOOS). No trial used objective imaging (MRI of cartilage thickness), biomarkers of cartilage degradation (CTX-II, COMP), or functional performance tests as primary outcomes. Self-reported pain is valid but susceptible to expectation effects.
Population narrowness: Most trial participants were young European adults or college athletes. Data in elite athletes, non-European populations, and older active adults remains limited. The Kviatkovsky study partially addressed the age gap by including middle-aged adults.
Exercise type specificity: The trials enrolled participants with general "activity-related" joint pain. They did not test whether collagen peptides are equally effective for runners, weightlifters, cyclists, or team sport athletes. Different activities stress joint tissue through different loading patterns (compressive, tensile, shear), and whether collagen peptides address all types of exercise-induced joint stress is unknown.
No dose optimization: The trials tested fixed doses (5, 10, or 20 g/day) without a true dose-escalation design. No pharmacokinetic study has measured collagen peptide concentrations in joint tissue at different oral doses. The finding that 5 g/day matches 10 g/day for pain relief in young adults raises the possibility that even lower doses could be effective, but this has not been tested.
Mechanism gaps: While several plausible mechanisms have been proposed (ECM stimulation, glycine-mediated anti-inflammation, hydroxyproline signaling), no study has directly demonstrated which mechanism drives pain reduction in human joint tissue. The gap between the in vitro mechanistic evidence and the clinical pain outcomes remains largely bridged by inference rather than direct measurement.
Placebo response: The placebo groups in both Zdzieblik trials showed substantial pain improvement (13.9 mm and 15.6 mm on VAS). The treatment effect over placebo was 5-8 mm on a 100 mm scale. While statistically significant, this is a modest clinical difference. The strong placebo response makes the true drug effect difficult to isolate.
Generic product applicability: Since trials used specific bioactive peptides, consumers purchasing generic collagen hydrolysate cannot assume equivalent effects. No head-to-head trial has compared branded bioactive peptides to commodity collagen powder.
The Bottom Line
Collagen peptide supplementation at 5-20 g/day reduces exercise-related joint pain in active adults, based on five RCTs and a systematic review of 15 trials. The effect is consistent but modest (5-8 mm improvement over placebo on a 100 mm pain scale), requires at least 12 weeks of daily use, and appears strongest in people who exercise more than 3 hours per week. No adverse effects have been reported. The evidence is limited by industry funding, subjective outcome measures, and the use of specific branded collagen peptides that may not represent all commercial products.
Sources & References
- 1RPEP-05493·Khatri, Mishti et al. (2021). “The effects of collagen peptide supplementation on body composition, collagen synthesis, and recovery from joint injury and exercise: a systematic review..” Amino acids.Study breakdown →PubMed →↩
- 2RPEP-03542·Zdzieblik, Denise et al. (2017). “Collagen Peptide Supplements Reduced Knee Pain in Young Athletes.” Applied physiology.Study breakdown →PubMed →↩
- 3RPEP-05921·Zdzieblik, Denise et al. (2021). “Collagen Peptide Supplements Reduce Exercise-Related Knee Pain in Young Active Adults.” Nutrients.Study breakdown →PubMed →↩
- 4RPEP-07070·Kviatkovsky, Shiloah A et al. (2023). “9-Month Trial: Collagen Peptide Supplements Improve Pain, Function, and Mental Health in Active Middle-Aged Adults.” Journal of the International Society of Sports Nutrition.Study breakdown →PubMed →↩
- 5RPEP-02845·Zdzieblik, Denise et al. (2015). “Collagen peptide supplementation in combination with resistance training improves body composition and increases muscle strength in elderly sarcopenic men: a randomised controlled trial..” The British journal of nutrition.Study breakdown →PubMed →↩