Cerebrolysin for Vascular Dementia: The Research
Cerebrolysin
10.6 pts ADAS-cog+ improvement
In the largest vascular dementia RCT, cerebrolysin improved ADAS-cog+ scores by 10.6 points versus 4.4 for placebo at week 24, a statistically significant difference (P < 0.0001).
Guekht et al., Journal of Stroke and Cerebrovascular Diseases, 2011
Guekht et al., Journal of Stroke and Cerebrovascular Diseases, 2011
If you only read one thing
Cerebrolysin is a peptide mixture made from pig brain proteins, given as an IV drip. In six clinical trials, it helped people with vascular dementia think more clearly than placebo, and the largest trial showed a big difference. But the Cochrane reviewers rated all that evidence as very low quality because the studies weren't designed to modern standards. It's approved in 40+ countries but not in the US or UK, and no one has compared it head-to-head with the dementia drugs we already use.
Cerebrolysin is a porcine brain-derived peptide preparation that has been tested in randomized controlled trials for vascular dementia, a condition without definitive pharmacological treatment. The largest RCT, conducted by Guekht et al. (2011) with 242 patients, found that cerebrolysin improved ADAS-cog+ scores by 10.6 points versus 4.4 for placebo at 24 weeks (P < 0.0001).[1] Two Cochrane systematic reviews (2013 and 2019) identified six RCTs with a total of 597 participants and concluded that cerebrolysin showed "a beneficial effect" on cognitive function, but rated the evidence quality as "very low" due to methodological concerns across the included studies.[2][3] This article reviews the full clinical evidence for cerebrolysin in vascular dementia: the composition of the peptide preparation, how it works at the molecular level, what the trials actually showed, and why the evidence remains both promising and insufficient.
Key Takeaways
- In the Guekht 2011 RCT (n=242), 82.1% of cerebrolysin patients improved by 4+ points on ADAS-cog+ versus 52.2% on placebo, with combined cognitive-clinical response at 67.5% versus 27.0%.[1]
- The 2019 Cochrane review (6 RCTs, 597 participants) found beneficial effects on cognition and global function, but rated all evidence as "very low quality."[3]
- Cerebrolysin is a mixture of approximately 80% free amino acids and 20% active neuropeptides (all below 10 kDa), designed to mimic the activity of BDNF, NGF, GDNF, and CNTF.[4]
- Preclinical studies show cerebrolysin upregulates BDNF, VEGF, IGF-1, and NGF while downregulating TNF-alpha, suggesting both neuroprotective and neurorestorative mechanisms.[5]
- Cerebrolysin requires intravenous administration, which limits its practical application compared to oral treatments.
- No head-to-head trial has compared cerebrolysin to cholinesterase inhibitors or memantine in vascular dementia.
What cerebrolysin is
What's in the bottle
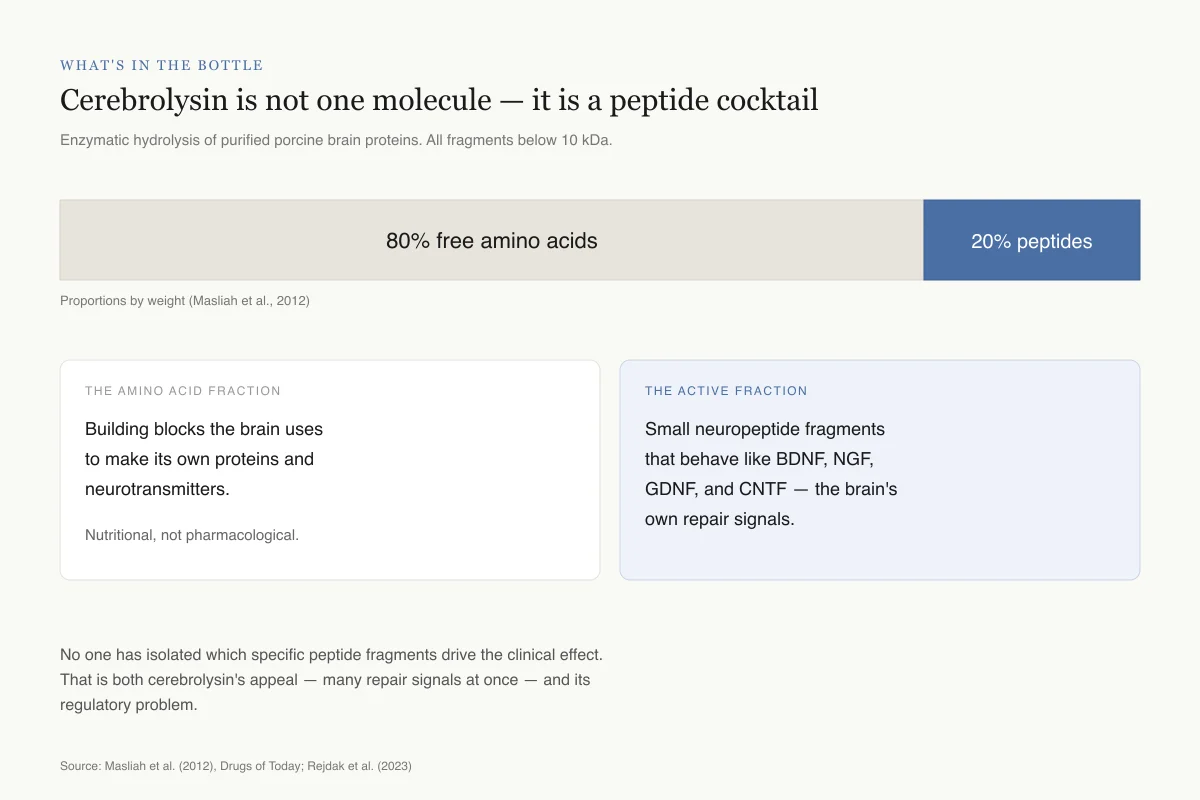
Cerebrolysin is not one molecule — it is a peptide cocktail
Produced by enzymatic hydrolysis of purified porcine brain proteins. All fragments are below 10 kilodaltons, small enough to cross the blood-brain barrier.
Proportions by weight (Masliah et al., 2012)
The amino acid fraction
The building blocks the brain uses to make its own proteins and neurotransmitters. Nutritional, not pharmacological.
The active fraction
Small neuropeptide fragments that behave like BDNF, NGF, GDNF, and CNTF — the brain's own growth and repair signals.
No one has isolated which specific peptide fragments drive the clinical effect. That is both cerebrolysin's appeal — many repair signals at once — and its regulatory problem.
Source: Masliah et al. (2012), Drugs of Today; Rejdak et al. (2023)
 View as image
View as imageCerebrolysin is produced through standardized enzymatic hydrolysis of purified porcine (pig) brain proteins. The process breaks brain proteins into small peptide fragments, all below 10 kilodaltons, mixed with free amino acids. The final product is approximately 80% free amino acids and 20% biologically active neuropeptides. Because the peptide fragments are small enough, they can cross the blood-brain barrier after intravenous administration.[4]
Unlike most pharmaceutical peptides that contain a single defined molecule, cerebrolysin is a complex mixture. Its composition has been characterized but individual active components have not been isolated and tested separately. This complexity is both a potential advantage (multiple neurotrophic activities in one preparation) and a scientific limitation (difficult to identify which components drive clinical effects).
Seidl et al. (2024) compared the biological activity of cerebrolysin to other commercially available peptide preparations and found significant differences in neurotrophic potency, highlighting that not all brain-derived peptide preparations are equivalent despite similar marketing claims.[7]
The preparation has been described as a "pharmacological cocktail" of neurotrophic peptides, distinguishing it from single-molecule drug development approaches that dominate modern pharmaceutical research.
Cerebrolysin has been manufactured by the Austrian pharmaceutical company EVER Neuro Pharma (formerly Ebewe Pharma) since the 1970s. It is approved in over 40 countries for various neurological indications, though not in the United States or United Kingdom. Its regulatory status varies: prescription medicine in some countries, investigational in others.
Neurotrophic mechanism of action
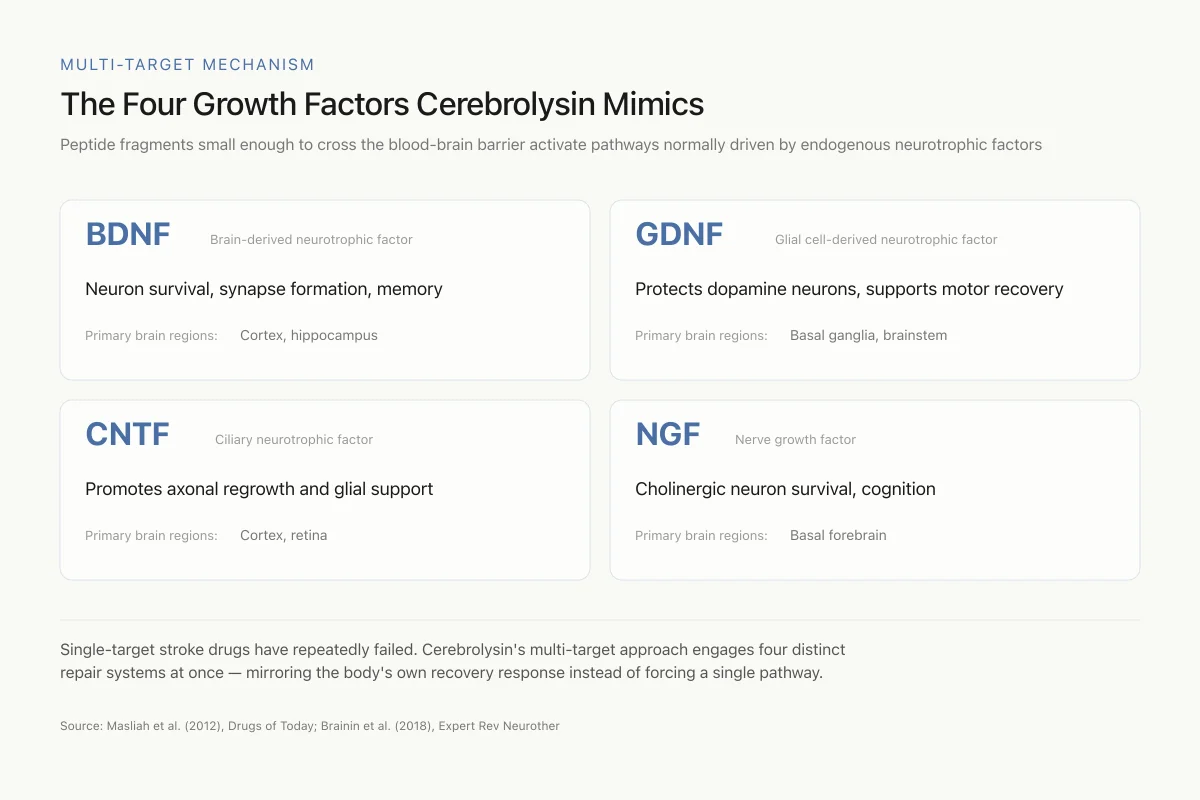
Multi-Target Mechanism
The Four Growth Factors Cerebrolysin Mimics
Peptide fragments small enough to cross the blood-brain barrier activate pathways normally driven by endogenous neurotrophic factors
Single-target stroke drugs have repeatedly failed. Cerebrolysin's multi-target approach engages four distinct repair systems at once — mirroring the body's own recovery response instead of trying to force a single pathway.
Source: Masliah et al. (2012), Drugs of Today; Brainin et al. (2018), Expert Rev Neurother
 View as image
View as imageCerebrolysin's peptide fragments are designed to mimic the biological functions of endogenous neurotrophic factors. Rejdak et al. (2023) reviewed the neurotrophic factor modulation evidence and identified five key pathways: nerve growth factor (NGF), insulin-like growth factor 1 (IGF-1), brain-derived neurotrophic factor (BDNF), vascular endothelial growth factor (VEGF), and tumor necrosis factor alpha (TNF-alpha). Cerebrolysin both mimics these factors directly and stimulates endogenous production.[5]
Masliah et al. (2012) reviewed the preclinical pharmacology and described several mechanisms relevant to vascular dementia. In dementia models, cerebrolysin decreased beta-amyloid deposition and tau phosphorylation by regulating glycogen synthase kinase-3beta and cyclin-dependent kinase 5 activity. It increased synaptic density and restored neuronal cytoarchitecture, protecting the integrity of neuronal circuits.[4]
Stepanichev et al. (2017) demonstrated that cerebrolysin affected brain neurotrophic factor systems in aged rats. Cerebrolysin treatment modified BDNF and NGF levels in specific brain regions and improved cognitive performance in aged animals, providing preclinical evidence for its use in age-related cognitive decline and dementia.[6]
For deeper coverage of cerebrolysin's neurotrophic mechanisms, see our companion article.
The Guekht 2011 trial: the primary evidence
The most substantial clinical evidence for cerebrolysin in vascular dementia comes from Guekht et al. (2011), a randomized, double-blind, placebo-controlled, multicenter trial published in the Journal of Stroke and Cerebrovascular Diseases.[1]
Study design. 242 patients meeting criteria for probable or possible vascular dementia (NINDS-AIREN criteria) with mild to moderate severity (MMSE 10-24) were randomized to cerebrolysin 20 mL IV daily or matching placebo, administered over two treatment cycles as add-on therapy to basic treatment with acetylsalicylic acid. The primary endpoint was combined improvement on cognition (ADAS-cog+) and overall clinical function (CIBIC+) at week 24.
Cognitive results. At week 24, ADAS-cog+ scores improved by 10.6 points in the cerebrolysin group compared with 4.4 points in the placebo group (least squares mean difference: -6.17; P < 0.0001). The improvement of 10.6 points on a cognitive scale is substantial. For context, a 4-point improvement on ADAS-cog is typically considered clinically meaningful in Alzheimer's trials.
Responder analysis. 82.1% of cerebrolysin patients showed ADAS-cog+ improvement of 4 or more points from baseline, compared with 52.2% of placebo patients. The combined responder rate (improvement on both ADAS-cog+ and CIBIC+) was 67.5% for cerebrolysin versus 27.0% for placebo.
Global clinical function. CIBIC+ showed a mean score of 2.84 in the cerebrolysin group versus 3.68 in the placebo group (lower scores indicate greater improvement on this 7-point scale).
Safety. The treatment was reported as well-tolerated, with adverse event rates comparable between cerebrolysin and placebo groups.
Largest RCT · Guekht 2011
Every primary outcome favored cerebrolysin
242 patients with probable or possible vascular dementia · 20 mL IV daily · two treatment cycles · 24-week endpoint
ADAS-cog+ improvement at 24 weeks
Cerebrolysin
10.6 pts
Placebo
4.4 pts
6.17-point advantage over placebo (P < 0.0001)
4+ point cognitive responders
Cerebrolysin
82.1%
Placebo
52.2%
Share of patients with clinically meaningful cognitive gain
Combined cognitive + clinical responders
Cerebrolysin
67.5%
Placebo
27.0%
Improved on both ADAS-cog+ and overall clinical function
CIBIC+ global score (lower is better)
Cerebrolysin
2.84
Placebo
3.68
Clinician-rated overall change in the patient
Why the size matters: In Alzheimer's trials, a 4-point ADAS-cog change is usually called clinically meaningful. Cerebrolysin produced more than 6 points of separation from placebo.
Source: Guekht et al. (2011), Journal of Stroke and Cerebrovascular Diseases
 View as image
View as imageThe Guekht trial is the strongest single piece of evidence for cerebrolysin in vascular dementia. Its strengths include the double-blind, placebo-controlled design, the multicenter implementation, and the clinically meaningful effect size. Its limitations include the add-on design (both groups received aspirin), the relatively short duration (24 weeks), and the absence of comparison to standard dementia treatments.
Cochrane systematic reviews
The Cochrane Collaboration has evaluated cerebrolysin for vascular dementia twice, providing the most rigorous independent assessment of the evidence.
Chen et al. (2013): first Cochrane review
The initial Cochrane review by Chen et al. (2013) searched through 2012 and assessed the available randomized evidence. The review found beneficial effects of cerebrolysin on cognitive function, but highlighted methodological limitations across the included trials. The review concluded that the evidence was insufficient to support definitive recommendations.[2]
Cui et al. (2019): updated Cochrane review
The updated review by Cui et al. (2019) searched through May 2019 and identified six randomized controlled trials with 597 participants. No new eligible studies had been published since the first review, though existing data was re-analyzed.[3]
Cognitive function. Combining MMSE and ADAS-cog+ data from three studies with 420 participants showed a beneficial effect of cerebrolysin, but the evidence was rated as "very low quality."
Global function. Two studies with 379 participants showed a beneficial effect on global function measures, also rated as "very low quality."
Evidence quality concerns. The Cochrane reviewers identified several issues across the included trials: potential selection bias, unclear allocation concealment, incomplete outcome reporting, and possible reporting bias. These methodological concerns, not contradictory results, drove the "very low quality" rating. The studies consistently showed benefit; the question is whether the study designs were rigorous enough to trust those results.
Cochrane conclusion. "Although the studies suggested a benefit of cerebrolysin treatment, the results are not definitive, as the included studies had several issues that may have led to misleading results, and even if the benefit is real, the effect was modest and may not be important to people living with dementia."
The gap between "consistent positive results" and "very low quality evidence" reflects the Cochrane methodology's emphasis on trial quality over effect direction. All six trials reported cerebrolysin benefit, but none met the highest standards of trial design and reporting. This is a common pattern for interventions studied primarily outside of the US/UK clinical trial ecosystem.
Why vascular dementia lacks treatments
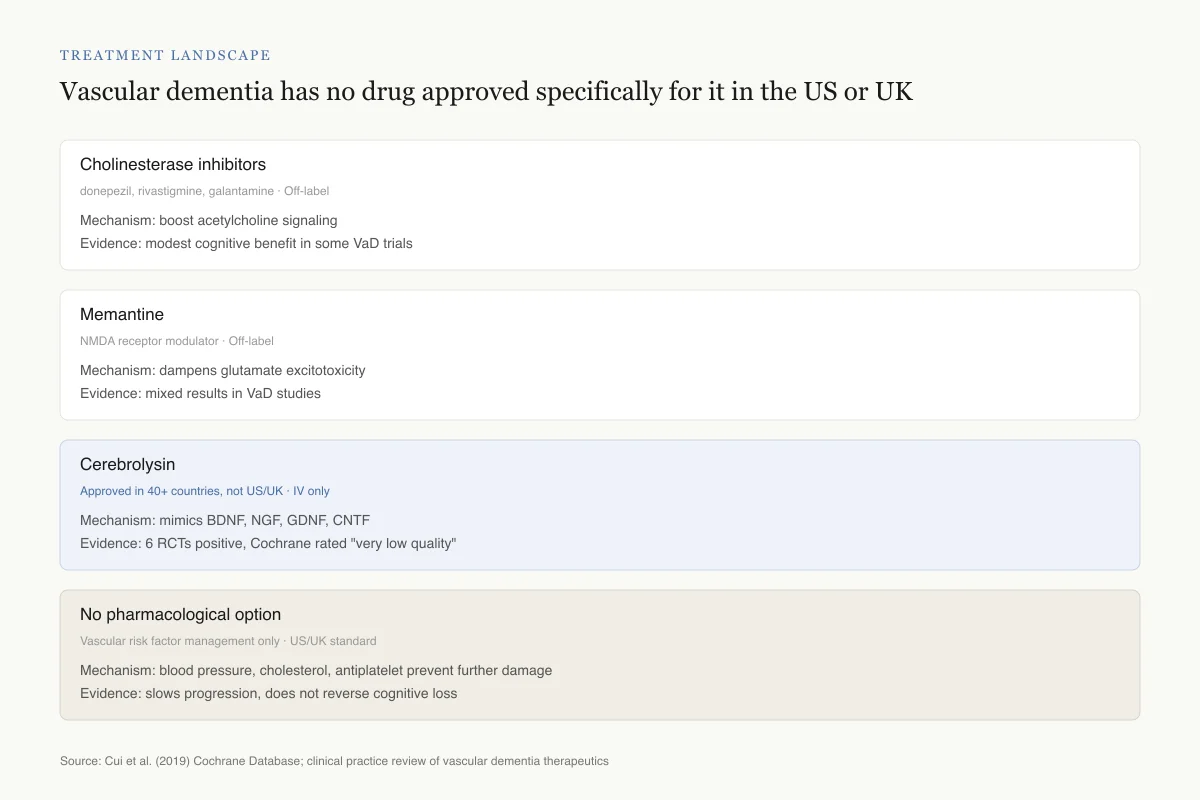
Treatment landscape
Vascular dementia has no drug approved specifically for it in the US or UK
Clinicians choose among off-label options. Cerebrolysin is the only agent with trials designed in vascular dementia populations — and it is unavailable in the two largest English-speaking markets.
Cholinesterase inhibitors
donepezil, rivastigmine, galantamine
Status
Off-label
How it works
Boost acetylcholine signaling
Evidence
Modest cognitive benefit in some VaD trials
Approved for Alzheimer's, not vascular dementia
Memantine
NMDA receptor modulator
Status
Off-label
How it works
Dampens glutamate excitotoxicity
Evidence
Mixed results in VaD studies
Approved for moderate-to-severe Alzheimer's
Cerebrolysin
80% amino acids + 20% neuropeptides
Status
Approved in 40+ countries, not US/UK
How it works
Mimics BDNF, NGF, GDNF, CNTF
Evidence
6 RCTs positive but rated "very low quality"
Approved for dementia where available; IV only
No pharmacological option
Vascular risk factor management only
Status
Current standard in the US and UK
How it works
Secondary stroke prevention
Evidence
Blood pressure, cholesterol, antiplatelet therapy prevent further damage but do not reverse cognitive loss
Default for patients without access to approved therapies
No head-to-head trial has compared cerebrolysin with cholinesterase inhibitors or memantine in vascular dementia. That comparison would settle a lot.
Source: Cui et al. (2019) Cochrane Database; clinical practice review of vascular dementia therapeutics
 View as image
View as imageVascular dementia is the second most common form of dementia after Alzheimer's disease, accounting for approximately 15-20% of all dementia cases. Unlike Alzheimer's, which has several approved pharmacological treatments (cholinesterase inhibitors, memantine, and newer anti-amyloid antibodies), vascular dementia has no drug specifically approved for it in most regulatory jurisdictions.
The lack of approved treatments reflects several challenges. Vascular dementia is heterogeneous: it results from various cerebrovascular pathologies including large vessel strokes, small vessel disease, hemorrhages, and mixed pathology. This heterogeneity makes clinical trial design difficult, as patients with different underlying vascular pathologies may respond differently to treatment. Diagnostic criteria have varied over time, and the boundary between vascular dementia and mixed dementia (with concurrent Alzheimer's pathology) is often unclear.
In practice, clinicians often prescribe cholinesterase inhibitors off-label for vascular dementia based on evidence of modest benefit in some trials. Memantine has also been studied with mixed results. Neither class has regulatory approval specifically for vascular dementia.
Cerebrolysin, in the countries where it is approved, offers a neurotrophic mechanism distinct from cholinergic enhancement. Rather than boosting neurotransmitter levels (the approach of cholinesterase inhibitors), cerebrolysin aims to protect and repair the neurons themselves. This mechanistic distinction is theoretically appealing for vascular dementia, where the primary pathology is neuronal loss from vascular damage rather than neurotransmitter depletion. No head-to-head trial has compared cerebrolysin to cholinesterase inhibitors in vascular dementia, which is a significant evidence gap. The neurotrophic approach also overlaps conceptually with emerging interest in other neuroprotective peptides and growth factors for neurodegeneration.
The treatment landscape for vascular dementia represents an area where a peptide-based neurotrophic therapy could fill a genuine clinical need, if the evidence quality could be elevated through larger, independently funded, multicenter trials meeting US and European regulatory standards.
The other vascular dementia trials
Beyond the Guekht 2011 study, five additional RCTs contributed to the Cochrane reviews. While smaller and less well-designed, they provide supporting data.
Earlier trials from the 1990s and 2000s used varying doses (10-30 mL IV daily), treatment durations (4-24 weeks), and cognitive assessment tools, which complicated meta-analysis. Most were conducted in Eastern European clinical settings. The Cochrane reviewers noted that allocation concealment was unclear in several trials, and outcome reporting was incomplete in some.
Despite these methodological weaknesses, a consistent pattern emerged: every trial that measured cognitive outcomes reported numerical improvement favoring cerebrolysin over placebo. The effect sizes varied, with the Guekht 2011 study showing the largest treatment difference. No trial reported cerebrolysin performing worse than placebo on any primary outcome measure.
This consistency of direction across six independent trials is worth noting, even with the quality limitations. In pharmaceutical development, consistent positive signal across multiple studies, even imperfect ones, typically triggers investment in larger, more rigorous confirmatory trials. For cerebrolysin in vascular dementia, that confirmatory trial has not materialized. The reasons likely include the manufacturer's business strategy (cerebrolysin is already marketed in 40+ countries without US/UK approval), the high cost of large phase III dementia trials, and the relatively small vascular dementia market compared to Alzheimer's disease.
A 2024 network meta-analysis that included cerebrolysin data from vascular dementia trials concluded that evidence on cognitive efficacy "remains limited" but did not contradict the positive direction of effect.[3]
Cerebrolysin's neuroprotective mechanisms in vascular injury
The biological plausibility of cerebrolysin in vascular dementia rests on its documented neuroprotective and neurorestorative properties. Vascular dementia involves ongoing neuronal damage from compromised cerebral blood flow, making neuroprotection a rational therapeutic target.
Formichi et al. (2012) demonstrated that cerebrolysin reduced oxidative stress markers in neuronal cell cultures exposed to hypoxic conditions, a laboratory model of the ischemic environment in vascular dementia.[4] Oxidative stress is a key mediator of neuronal death in both acute stroke and chronic cerebrovascular disease, and its reduction could preserve neuronal populations that would otherwise be lost.
Hartwig et al. (2014) showed that cerebrolysin protected PC12 cells (a neuronal cell model) from various toxicity challenges, demonstrating direct cytoprotective effects independent of neurotrophic factor signaling. This suggests cerebrolysin may have multiple protective mechanisms operating simultaneously: neurotrophic factor modulation, anti-oxidant effects, and direct cellular protection.
The convergence of neurotrophic, anti-oxidant, and cytoprotective properties provides biological rationale for why a complex peptide mixture might outperform single-mechanism drugs in a heterogeneous condition like vascular dementia, where multiple pathological processes contribute to cognitive decline simultaneously.
Cerebrolysin in related neurological conditions
The vascular dementia evidence exists within a broader context of cerebrolysin research across neurological conditions, which informs the plausibility of its effects.
Stroke recovery
Cerebrolysin has been studied in multiple stroke recovery trials, with the largest evidence base in acute ischemic stroke. The stroke and vascular dementia applications share a common rationale: both conditions involve vascular damage to brain tissue, and both could theoretically benefit from neurotrophic support that promotes neuronal survival and circuit repair. Stroke is also a major risk factor for subsequent vascular dementia, creating a potential preventive application.
Alzheimer's disease
Cerebrolysin has been tested in Alzheimer's disease trials, where its anti-amyloid and synaptic protective effects are relevant. Some vascular dementia patients have concurrent Alzheimer's pathology, making the Alzheimer's evidence partially applicable to mixed dementia cases.
Traumatic brain injury
Cerebrolysin has been investigated for TBI, where its neuroprotective and neurorestorative properties are tested in a different injury context. The TBI evidence contributes to understanding cerebrolysin's general neurotrophic activity profile.
Rockenstein et al. (2011) demonstrated that cerebrolysin's effects varied by brain region in animal models, with differential effects on neurotrophic factor levels in cortex, hippocampus, and striatum. This regional specificity is relevant because vascular dementia often produces patchy, region-specific damage depending on which blood vessels are affected.[8]
Limitations and open questions
The cerebrolysin vascular dementia evidence has several constraints that explain why it remains "overlooked" in some clinical guidelines despite consistent positive trial results.
Evidence quality. The Cochrane "very low quality" rating means that confidence in the effect estimate is limited, and the true effect may be substantially different from the estimated effect. This rating does not mean the studies showed no benefit; it means the methodological quality was insufficient for definitive conclusions.
Administration route. Cerebrolysin requires intravenous administration, typically daily for treatment cycles of several weeks. This is impractical compared to oral medications. The IV requirement limits its use in outpatient settings and increases cost and logistical burden.
Geographic research concentration. Much of the cerebrolysin research has been conducted in Europe and Asia, with limited participation from US or UK research institutions. This geographic concentration, combined with the manufacturer's involvement in many studies, raises questions about publication bias and generalizability.
No active comparator trials. All vascular dementia trials compared cerebrolysin to placebo. No trial has compared it to cholinesterase inhibitors, memantine, or any other active treatment. Without active comparator data, it is impossible to know whether cerebrolysin is better than, equivalent to, or worse than existing off-label treatments.
Regulatory status. Cerebrolysin's non-approval in the US and UK means it has not undergone the most stringent regulatory review processes, which require large, well-designed Phase III trials. The absence of FDA or MHRA review is itself a limitation on evidence evaluation.
Complex composition. As a mixture of peptides and amino acids, cerebrolysin's exact active components remain unidentified. Seidl et al. (2024) showed significant differences between cerebrolysin and competing preparations, underscoring that the biological activity depends on the specific manufacturing process.[7] This complexity complicates both regulatory evaluation and mechanistic understanding.
Effect durability. Most trials assessed outcomes during or shortly after treatment cycles. Whether cerebrolysin's cognitive benefits persist long-term, or require ongoing treatment, is not established.
Clinical significance versus statistical significance. The Cochrane reviewers specifically questioned whether the observed cognitive improvements, even if real, are "important to people living with dementia." A 6-point difference on ADAS-cog+ is statistically significant but may not translate into observable improvements in daily function, caregiver burden, or quality of life. The clinical significance threshold for vascular dementia has never been formally established, unlike in Alzheimer's trials where 4-point ADAS-cog changes are considered clinically meaningful.
Patient selection uncertainty. The six trials enrolled patients with varying severities of vascular dementia and used different diagnostic criteria across different eras. Which patients benefit most from cerebrolysin, whether those with small vessel disease versus large vessel strokes, mild versus moderate impairment, or early versus late-stage disease, cannot be determined from the current evidence. Subgroup analyses were limited by small sample sizes.
Manufacturer involvement. EVER Neuro Pharma has funded or co-funded most cerebrolysin research. While industry funding does not automatically invalidate results, the absence of fully independent academic trials, combined with the consistent positive outcomes, warrants consideration of potential bias. The Cochrane reviewers noted this concern in their assessment of reporting quality.
Safety
ModerateCerebrolysin is not approved in the US or UK
Concern
The FDA and MHRA have not reviewed cerebrolysin. Anyone obtaining it outside an approved jurisdiction is using a product without that regulatory oversight. Gray-market peptide sources may ship material that is not pharmaceutical grade.
What the research says
In countries where cerebrolysin is licensed, it is given under medical supervision with established dosing protocols. The published safety record in trials has been favorable, with adverse event rates similar to placebo, but IV administration itself carries its own risks and should be clinical, not self-directed.
Particularly relevant for: Anyone considering cerebrolysin outside its approved regions
What to do
Work with a clinician licensed in a jurisdiction where cerebrolysin is approved. Never attempt self-administered IV peptide therapy. Discuss all dementia treatment options with a neurologist before making choices.
Review of EVER Neuro Pharma regulatory status and published safety data
The Bottom Line
Cerebrolysin consistently improved cognitive function in vascular dementia across six randomized controlled trials, with the largest study showing a 10.6-point ADAS-cog+ improvement versus 4.4 for placebo. Two Cochrane reviews confirmed the beneficial direction of effect but rated the evidence as "very low quality" due to methodological limitations. Cerebrolysin's complex neurotrophic mechanism, which mimics BDNF, NGF, and other growth factors, provides biological plausibility for the clinical findings. The evidence is sufficient to generate interest but insufficient to establish cerebrolysin as a standard treatment for vascular dementia.
Sources & References
- 1RPEP-01768·Guekht, Alla B et al. (2011). “Cerebrolysin Improved Cognition in Vascular Dementia: A Randomized Controlled Trial.” Journal of stroke and cerebrovascular diseases : the official journal of National Stroke Association.Study breakdown →PubMed →↩
- 2RPEP-02148·Chen, Ning et al. (2013). “Cerebrolysin for vascular dementia..” The Cochrane database of systematic reviews.Study breakdown →PubMed →↩
- 3RPEP-04131·Cui, Shuhui et al. (2019). “Cerebrolysin for Vascular Dementia: A Cochrane Review Finds Hints of Benefit But Very Weak Evidence.” The Cochrane database of systematic reviews.Study breakdown →PubMed →↩
- 4RPEP-02009·Masliah, E et al. (2012). “The pharmacology of neurotrophic treatment with Cerebrolysin: brain protection and repair to counteract pathologies of acute and chronic neurological disorders..” Drugs of today (Barcelona.Study breakdown →PubMed →↩
- 5RPEP-07313·Rejdak, Konrad et al. (2023). “How Cerebrolysin Mimics Your Brain's Own Growth Factors to Treat Dementia, Stroke, and Brain Injury.” Medicinal research reviews.Study breakdown →PubMed →↩
- 6RPEP-03482·Stepanichev, Mikhail et al. (2017). “Effects of cerebrolysin on nerve growth factor system in the aging rat brain..” Restorative neurology and neuroscience.Study breakdown →PubMed →↩
- 7RPEP-09234·Seidl, Lisa-Franziska et al. (2024). “Comparing the biological activity and composition of Cerebrolysin with other peptide preparations..” Journal of medicine and life.Study breakdown →PubMed →↩
- 8RPEP-01843·Rockenstein, Edward et al. (2011). “Regional comparison of the neurogenic effects of CNTF-derived peptides and cerebrolysin in AβPP transgenic mice..” Journal of Alzheimer's disease : JAD.Study breakdown →PubMed →↩