Cerebrolysin for Stroke Recovery: The Evidence
Cerebrolysin
1,879 patients
A meta-analysis of nine randomized controlled trials found that cerebrolysin improved neurological outcomes at day 30 compared to placebo in acute ischemic stroke patients.
Bornstein et al., Neurological Sciences, 2018
Bornstein et al., Neurological Sciences, 2018
If you only read one thing
Cerebrolysin is a pig-brain-derived peptide mixture given by IV for 10-21 days after a stroke. It's used in over 50 countries but isn't FDA-approved in the US. Across nine randomized trials covering 1,879 patients, it modestly improved stroke recovery — about 1 in 8 treated patients gets a meaningful benefit they wouldn't have gotten otherwise. The effect is clearest in severe strokes and in motor function recovery during rehab. Safety looks fine, but the evidence isn't strong enough yet for US regulators.
Cerebrolysin is a mixture of low-molecular-weight neuropeptides and free amino acids derived from porcine brain tissue through a standardized biotechnological process. It has been used clinically for neurological conditions in over 50 countries for decades, primarily in Europe and Asia, though it has never received FDA approval in the United States. The interest in cerebrolysin for stroke recovery rests on a specific pharmacological rationale: the peptide mixture mimics the activity of endogenous neurotrophic factors like BDNF, GDNF, and NGF, which are critical for neuronal survival, plasticity, and repair after ischemic brain injury.
Brainin et al. described cerebrolysin as a "multi-target drug for recovery after stroke" in a 2018 review, noting that unlike single-target neuroprotective agents that have repeatedly failed in stroke trials, cerebrolysin's pleiotropic mechanism engages multiple recovery pathways simultaneously.[1] The evidence base includes nine randomized controlled trials, two large landmark studies (CASTA and CARS), and a meta-analysis pooling 1,879 patients. The results are positive but nuanced, with clearer benefits in specific patient subgroups than in unselected populations.
This article sits within a broader examination of cerebrolysin's clinical evidence. For its use in cognitive decline, see cerebrolysin for vascular dementia. For other neurological applications, see our articles on cerebrolysin and traumatic brain injury and how cerebrolysin works at the molecular level.
Key Takeaways
- Cerebrolysin is a peptide mixture made from pig brain tissue, used in over 50 countries to help people recover from strokes.
- It's not a take-at-home pill — it's a hospital IV given for 10 to 21 days, started within a few days of the stroke.
- Across nine trials of nearly 1,900 patients, treat 8 stroke patients and roughly 1 extra person walks away with a meaningful recovery.
- The benefit is biggest in severe strokes — one trial found mortality cut roughly in half compared to standard care alone.
- The drug works by imitating your brain's own repair signals, the same growth factors that keep neurons alive and rewire damaged circuits.
- Side effects look about the same as placebo — rare for a stroke drug, since most failed candidates got pulled for safety problems.
- It's not FDA-approved, and any "cerebrolysin" sold to US consumers comes through unregulated channels with no quality guarantees.
What Cerebrolysin Contains
Cerebrolysin is not a single peptide. It is a standardized preparation containing a defined mixture of neurotrophic peptides (approximately 25% by weight) and free amino acids (approximately 75%) derived from purified porcine brain proteins through controlled enzymatic proteolysis. The peptide fraction contains fragments that mimic the activity of several endogenous neurotrophic factors.[5]
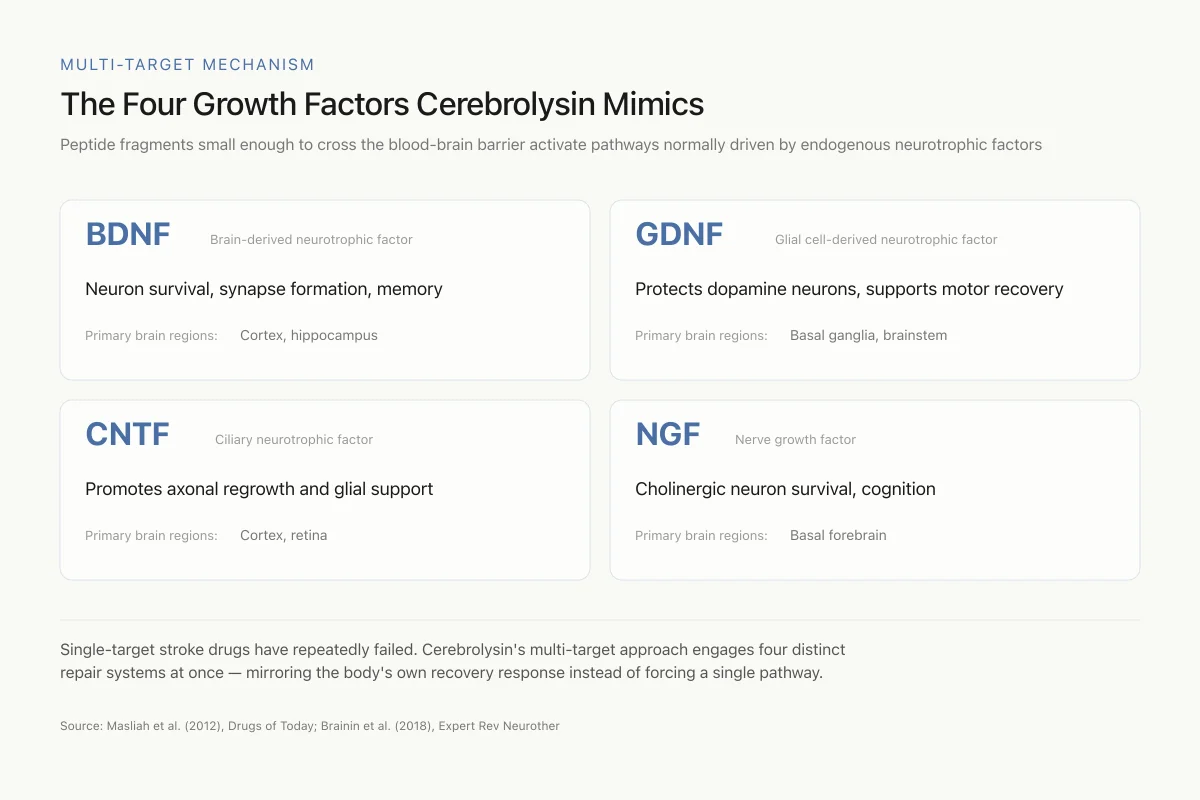
Masliah et al. characterized the pharmacological profile in 2012, demonstrating that cerebrolysin activates signaling pathways associated with BDNF (brain-derived neurotrophic factor), GDNF (glial cell-derived neurotrophic factor), CNTF (ciliary neurotrophic factor), and NGF (nerve growth factor).[5] These factors promote neuronal survival, axonal growth, synaptic formation, and neurogenesis. The peptide fragments in cerebrolysin are small enough to cross the blood-brain barrier, which is a critical advantage over administering full-length neurotrophic factor proteins, which cannot reach the brain through peripheral administration.
Multi-Target Mechanism
The Four Growth Factors Cerebrolysin Mimics
Peptide fragments small enough to cross the blood-brain barrier activate pathways normally driven by endogenous neurotrophic factors
Single-target stroke drugs have repeatedly failed. Cerebrolysin's multi-target approach engages four distinct repair systems at once — mirroring the body's own recovery response instead of trying to force a single pathway.
Source: Masliah et al. (2012), Drugs of Today; Brainin et al. (2018), Expert Rev Neurother
 View as image
View as imageStepanichev et al. showed in 2017 that cerebrolysin modulates the NGF system in aging rat brains, increasing both NGF protein levels and TrkA receptor expression in the hippocampus and cortex.[7] This is relevant to stroke recovery because neurotrophic factor levels decline with age, and older patients (who are most likely to have strokes) have a reduced endogenous capacity for neural repair.
The CASTA Trial: 1,070 Patients in Asia
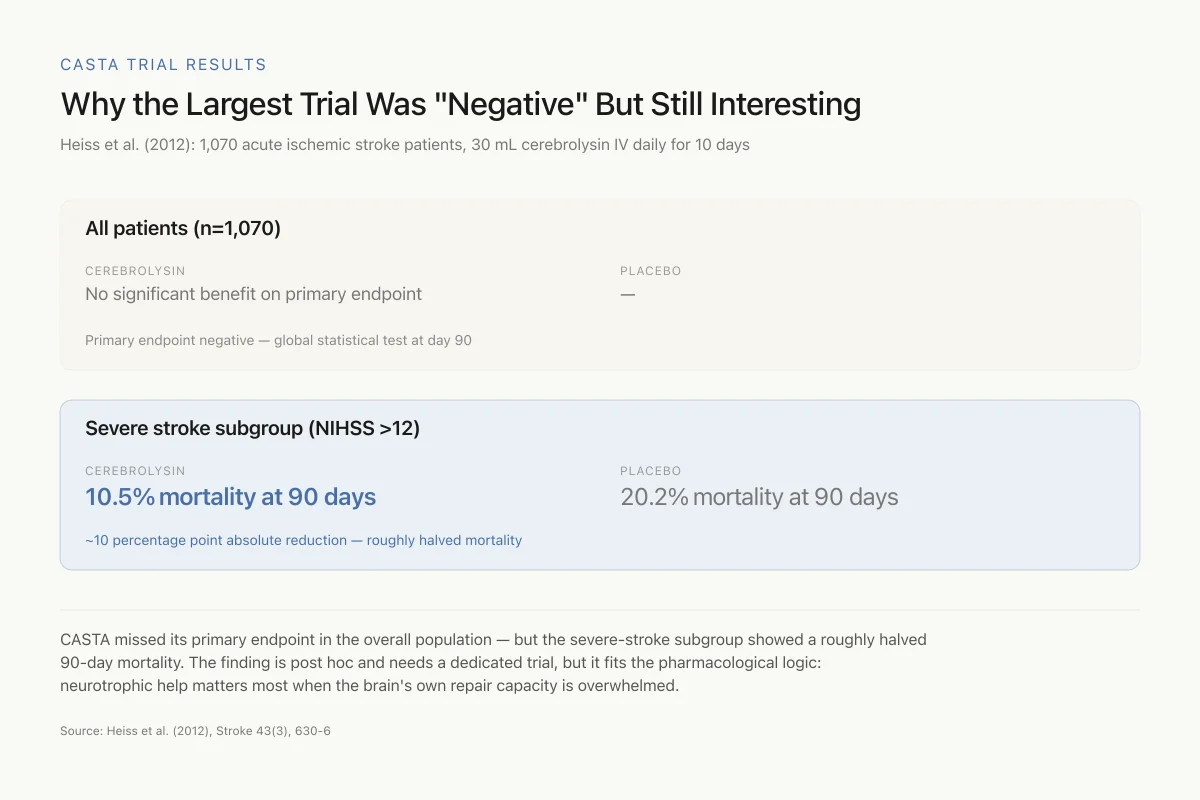
The Cerebrolysin Acute Stroke Treatment in Asia (CASTA) trial, published by Heiss et al. in 2012, was the largest single randomized trial of cerebrolysin in stroke.[3] It enrolled 1,070 patients with acute ischemic hemispheric stroke within 12 hours of symptom onset. Patients received either 30 mL cerebrolysin or placebo (saline) as a daily intravenous infusion for 10 days, in addition to standard care including aspirin.
The primary endpoint, a global statistical test combining four outcome measures at day 90, showed no statistically significant difference between cerebrolysin and placebo in the overall population. This result was widely interpreted as a negative trial.
However, a pre-specified subgroup analysis stratified by stroke severity revealed a different picture. In patients with severe strokes (NIHSS >12), cerebrolysin showed a strong trend toward benefit. The cumulative 90-day mortality in this subgroup was 10.5% with cerebrolysin compared to 20.2% with placebo.[3] The finding suggests that cerebrolysin's neurotrophic effects may be most relevant when the brain damage is substantial enough that endogenous repair mechanisms are overwhelmed.
CASTA Trial Results
Why the Largest Trial Was "Negative" But Still Interesting
Heiss et al. (2012): 1,070 acute ischemic stroke patients, 30 mL cerebrolysin IV daily for 10 days
CASTA missed its primary endpoint in the overall population — but the severe-stroke subgroup showed a roughly halved 90-day mortality. The finding is post hoc, which means it needs a dedicated trial before it changes practice, but it does fit the pharmacological logic: neurotrophic help matters most when the brain's own repair capacity is overwhelmed.
Source: Heiss et al. (2012), Stroke 43(3), 630-6
 View as image
View as imageThe CASTA trial was designed using the framework of a 2009 protocol paper by Hong et al., which established the rationale for testing cerebrolysin specifically in Asian stroke populations given the high incidence of ischemic stroke and the established clinical use of cerebrolysin across Asia.[4]
The CARS Trials: Motor Function Recovery
The Cerebrolysin and Recovery after Stroke (CARS) program consisted of two identical randomized, placebo-controlled, double-blind studies (CARS-1 and CARS-2) focused specifically on motor function recovery during early rehabilitation. Treatment with 30 mL cerebrolysin once daily for 21 days (longer than the CASTA 10-day protocol) was initiated 24 to 72 hours after stroke onset, and all patients participated in a standardized rehabilitation program.
The combined analysis showed cerebrolysin superiority over placebo on the primary outcome, the Action Research Arm Test (ARAT) score at day 90. The ARAT measures fine motor coordination, grasp, grip, and pinch functions of the affected upper limb. This is a clinically meaningful endpoint because upper extremity motor recovery is one of the most important determinants of post-stroke functional independence.[1]
The CARS trials differed from CASTA in two important ways: the treatment duration was longer (21 days vs. 10 days), and the endpoint focused specifically on motor function during rehabilitation rather than global neurological status. These design choices may explain why CARS showed clearer benefits than CASTA.
Meta-Analysis: Pooling Nine Trials
Bornstein et al. published a meta-analysis in 2018 pooling data from nine randomized controlled trials with a total of 1,879 patients, providing the most comprehensive assessment of cerebrolysin in acute ischemic stroke.[2]
The key findings:
Cerebrolysin showed statistically significant superiority over placebo on the NIHSS (National Institutes of Health Stroke Scale) at day 30 (p < 0.0001). The number needed to treat (NNT) for a clinically relevant neurological improvement was 7.7, meaning that for every 7 to 8 patients treated, one additional patient achieved meaningful recovery beyond what would have occurred with standard care alone.[2]
Safety was comparable to placebo across all nine trials. The incidence of adverse events, serious adverse events, and mortality did not differ between cerebrolysin and placebo groups. This is a notable finding because many previous neuroprotective drug candidates for stroke were abandoned due to safety concerns at effective doses.
The meta-analysis confirmed the pattern from CASTA: cerebrolysin's benefits were most pronounced in patients with moderate to severe strokes. Patients with mild strokes (low baseline NIHSS) showed less separation from placebo, likely because their endogenous recovery capacity was sufficient without pharmacological augmentation.
How Cerebrolysin Supports Stroke Recovery
Rejdak et al. reviewed the neurotrophic factor modulation by cerebrolysin in 2023, synthesizing evidence from stroke, dementia, and TBI studies.[6] The mechanisms relevant to stroke recovery include:
Neuroprotection in the acute phase. During the first hours to days after stroke, cerebrolysin's neurotrophic peptides activate survival signaling through the PI3K/AKT and GSK3-beta pathways, reducing apoptotic cell death in the ischemic penumbra (the brain tissue surrounding the stroke core that is damaged but potentially salvageable).
Neuroplasticity during recovery. In the weeks following stroke, cerebrolysin promotes axonal sprouting, dendritic remodeling, and synaptogenesis. These processes allow surviving neurons to form new connections that compensate for lost neural circuits. The CARS trial results, showing improved motor function at day 90, are consistent with this mechanism.
Neurogenesis. Cerebrolysin stimulates the proliferation and differentiation of neural progenitor cells in the subventricular zone, the brain's endogenous stem cell niche. New neurons migrate to damaged areas and integrate into existing circuits, contributing to long-term functional recovery.[5]
Anti-inflammatory effects. Cerebrolysin reduces microglial activation and the production of pro-inflammatory cytokines in the peri-infarct region. Post-stroke inflammation exacerbates tissue damage and impairs recovery; modulating this response supports a more favorable environment for neural repair.
Evidence Summary
Limitations and Open Questions
The cerebrolysin stroke evidence has genuine limitations that should be weighed alongside the positive findings.
The CASTA trial, the largest single study, was technically negative on its primary endpoint. The subgroup finding of mortality reduction in severe strokes was from a post hoc analysis, which carries a higher risk of spurious results. A prospective trial specifically enrolling severe stroke patients would be needed to confirm this signal.
The pooled meta-analysis was based on nine trials, but several were relatively small and conducted in different healthcare systems with different standards of stroke care. Heterogeneity in baseline stroke severity, time to treatment, concomitant therapies, and outcome measures complicates interpretation.
Cerebrolysin requires intravenous administration, which limits its use to hospital settings and the early post-stroke period. There is no oral formulation.
The drug is not approved by the FDA and is not part of major American or European stroke treatment guidelines, though it is included in treatment recommendations in several Asian and Eastern European countries. A 2023 Cochrane review of cerebrolysin for acute ischemic stroke concluded that while the drug appears safe, the certainty of evidence for efficacy remains low to moderate.
The mechanism of action, while supported by preclinical data, involves a complex mixture of peptides whose individual contributions to clinical effects are incompletely characterized. This makes it harder to optimize dosing, timing, and patient selection compared to single-molecule drugs.
Safety
ModerateUS availability is via gray-market channels
Concern
Because cerebrolysin is not FDA-approved, any product sold in the United States as 'cerebrolysin' comes through unregulated research-peptide vendors or personal import. These products are not subject to the quality, sterility, or purity standards that apply to the approved EVER Pharma product used in the clinical trials.
What the research says
The trial data applies to the standardized, pharmaceutical-grade preparation administered under hospital conditions. Extrapolating that safety and efficacy to gray-market peptides of unknown origin, purity, or concentration is not supported by the evidence.
Particularly relevant for: US-based consumers considering self-administration outside a clinical setting
What to do
If you're considering cerebrolysin for a medical indication, do so through a licensed physician in a country where it's approved, or as part of a clinical trial. Don't assume a research-peptide vial matches what was tested in CASTA or CARS.
FDA unapproved drug status; Cochrane 2023 stroke review notes pharmaceutical-grade product was used in all reviewed trials
The Bottom Line
Cerebrolysin demonstrates a consistent signal of benefit in stroke recovery across multiple randomized trials, particularly in patients with moderate to severe strokes. The meta-analysis of nine trials found a number needed to treat of 7.7 for clinically meaningful improvement, with safety comparable to placebo. The largest single trial (CASTA) was negative on its primary endpoint but showed suggestive mortality reduction in severe strokes. The CARS trials showed clear motor function benefits during rehabilitation. The drug remains unapproved in the US, and the evidence base, while positive, has limitations in trial design and certainty that preclude definitive conclusions.
Sources & References
- 1RPEP-03595·Brainin, Michael (2018). “Cerebrolysin: a multi-target drug for recovery after stroke..” Expert review of neurotherapeutics.Study breakdown →PubMed →↩
- 2RPEP-03592·Bornstein, Natan M et al. (2018). “Safety and efficacy of Cerebrolysin in early post-stroke recovery: a meta-analysis of nine randomized clinical trials..” Neurological sciences : official journal of the Italian Neurological Society and of the Italian Society of Clinical Neurophysiology.Study breakdown →PubMed →↩
- 3RPEP-01955·Heiss, Wolf-Dieter et al. (2012). “Cerebrolysin for Acute Stroke: Large Trial Shows No Overall Benefit but a Signal in Severe Cases.” Stroke.Study breakdown →PubMed →↩
- 4RPEP-01493·Hong, Z et al. (2009). “CASTA: A Major Trial Testing Whether Peptide Therapy Can Help Stroke Patients Recover.” International journal of stroke : official journal of the International Stroke Society.Study breakdown →PubMed →↩
- 5RPEP-02009·Masliah, E et al. (2012). “The pharmacology of neurotrophic treatment with Cerebrolysin: brain protection and repair to counteract pathologies of acute and chronic neurological disorders..” Drugs of today (Barcelona.Study breakdown →PubMed →↩
- 6RPEP-07313·Rejdak, Konrad et al. (2023). “How Cerebrolysin Mimics Your Brain's Own Growth Factors to Treat Dementia, Stroke, and Brain Injury.” Medicinal research reviews.Study breakdown →PubMed →↩
- 7RPEP-03482·Stepanichev, Mikhail et al. (2017). “Effects of cerebrolysin on nerve growth factor system in the aging rat brain..” Restorative neurology and neuroscience.Study breakdown →PubMed →↩