Cagrilintide: The Amylin Half of CagriSema
Cagrilintide and CagriSema
20.4% Mean Weight Loss
In the REDEFINE 1 trial, cagrilintide 2.4 mg combined with semaglutide 2.4 mg produced a mean body weight reduction of 20.4% at 68 weeks in adults without diabetes, with 60% of participants losing at least 20% of their body weight.
Garvey et al., NEJM, 2025
Garvey et al., NEJM, 2025
If you only read one thing
Cagrilintide is a weekly injection of a redesigned version of amylin, a natural appetite hormone your pancreas makes after meals. Novo Nordisk rebuilt it so it sticks to albumin in your blood and lasts about a week instead of 13 minutes. On its own it caused about 11% weight loss in a phase 2 trial. Paired with semaglutide as CagriSema, it hit 20.4% at 68 weeks. Novo Nordisk filed with the FDA in December 2025 and expects a decision in late 2026.
Cagrilintide is a synthetic long-acting analog of the peptide hormone amylin, developed by Novo Nordisk specifically for obesity treatment. Amylin is a 37-amino-acid hormone co-secreted with insulin from pancreatic beta cells after meals. It signals satiety through receptors in the area postrema and nucleus of the solitary tract in the hindbrain.[1] The problem with native amylin as a drug is its half-life: roughly 13 minutes. Pramlintide, the only FDA-approved amylin analog (marketed as Symlin for diabetes), requires three injections per day. Cagrilintide was engineered to solve that problem. Through lipidation and specific amino acid substitutions, Novo Nordisk extended the half-life to approximately 8 days, enabling once-weekly subcutaneous injection.[2] The result is the amylin half of CagriSema, the combination therapy that filed for FDA approval in December 2025. For an overview of how amylin works in the body, see Amylin: The Pancreatic Peptide You've Never Heard Of.
Key Takeaways
- Cagrilintide is a long-acting amylin analog with an approximately 8-day half-life, engineered through lipidation and amino acid substitutions for once-weekly dosing (Kruse et al., Journal of Medicinal Chemistry, 2021)
- As monotherapy in a phase 2 trial, cagrilintide 4.5 mg produced 10.8% mean weight loss at 26 weeks versus 3.0% for placebo (Lau et al., Lancet, 2021)
- Combined with semaglutide 2.4 mg in REDEFINE 1, the combination produced 20.4% mean weight loss at 68 weeks; 60% of participants lost at least 20% of body weight (Garvey et al., NEJM, 2025)
- In adults with type 2 diabetes (REDEFINE 2), CagriSema produced 13.7% weight loss versus 3.4% for placebo at 68 weeks (Davies et al., NEJM, 2025)
- Novo Nordisk filed an NDA for CagriSema in December 2025; the FDA decision is expected in late 2026
- Cagrilintide activates amylin receptor subtypes 1 and 3 in the area postrema, reducing food intake through both homeostatic and hedonic pathways distinct from GLP-1
Why Amylin Matters for Obesity
Amylin is one of the body's natural satiety signals. When you eat, pancreatic beta cells release amylin alongside insulin. Amylin travels to the hindbrain, where it binds to heterodimeric receptors composed of a calcitonin receptor (CTR) core paired with receptor activity-modifying proteins (RAMP1 or RAMP3).[3] This activates neurons in the area postrema, a brain region outside the blood-brain barrier that monitors circulating hormones and metabolites.
The downstream effects include reduced food intake, delayed gastric emptying, and suppression of post-meal glucagon release. A 2015 review cataloged amylin's pharmacological effects across multiple systems: beyond satiety, amylin influences bone metabolism, cardiovascular function, and renal physiology.[1]
Amylin's satiety effects operate through both homeostatic and hedonic pathways. In homeostatic regulation, amylin acts directly on area postrema neurons to reduce meal size. In hedonic regulation, amylin signaling modulates the reward value of food through connections to the lateral parabrachial nucleus and ventral tegmental area. A 2020 study demonstrated that amylin and its analog pramlintide modulate gamma oscillatory activity in the brain, suggesting direct effects on neural processing of food-related stimuli.[12] This dual action on both "need to eat" and "want to eat" circuits distinguishes amylin from hormones that affect only one pathway.
What makes amylin pharmacologically interesting for obesity is that it operates through pathways distinct from GLP-1. Amylin and GLP-1 receptors are expressed on different populations of neurons in the area postrema. Both reduce food intake, but through separate circuits. This is the pharmacological rationale for combining cagrilintide (amylin pathway) with semaglutide (GLP-1 pathway): two distinct satiety signals, additive effects. For a deeper dive into what happens when these two mechanisms are combined, see CagriSema: What Happens When You Combine Semaglutide with an Amylin Analog.
The concept of dual-mechanism appetite suppression is broader than any single drug combination. For the theoretical framework behind combining peptide pathways, see Combination Peptide Therapy: Why Two Mechanisms May Beat One.
How Cagrilintide Was Engineered
The challenge with amylin-based drugs has always been pharmacokinetics. Native human amylin has a plasma half-life of approximately 13 minutes. It also forms amyloid fibrils, aggregating into insoluble structures that can deposit in pancreatic tissue. This aggregation tendency is linked to the pathophysiology of type 2 diabetes and makes native amylin unsuitable as a drug.
Pramlintide, approved in 2005, solved the aggregation problem by substituting three proline residues (positions 25, 28, and 29) based on rat amylin, which does not form fibrils. Pramlintide suppresses appetite and improves glycemic control, but its 20-to-45-minute half-life requires injections with every meal.
Kruse and colleagues at Novo Nordisk described the development of cagrilintide in a 2021 Journal of Medicinal Chemistry paper.[2] The engineering strategy involved multiple modifications to human amylin:
Anti-aggregation mutations. Like pramlintide, cagrilintide carries the 25P/28P/29P substitutions to prevent fibril formation.
Helix stabilization. Mutations at positions 14 (glutamate) and 17 (arginine) were designed to form a salt bridge that stabilizes the central alpha-helix, improving receptor binding potency.
C-terminal modification. A proline was added at the C-terminus to improve potency at the calcitonin receptor specifically, which is relevant because the amylin receptors are built on a CTR scaffold.
Lipidation for albumin binding. The defining modification: a C20 fatty acid chain linked to the N-terminus. This fatty acid binds to serum albumin in the bloodstream, creating a slow-release depot effect. Albumin-bound cagrilintide circulates with albumin's own long half-life, releasing the active peptide gradually over days. This is the same pharmacokinetic strategy Novo Nordisk used for semaglutide (C18 fatty acid, once-weekly GLP-1) and insulin degludec.
The result: a half-life of 159 to 195 hours (approximately 7 to 8 days) in humans, supporting once-weekly subcutaneous injection. A 2022 review of long-acting amylin analogues noted that cagrilintide's extended pharmacokinetic profile represented a generational advance over pramlintide for obesity applications.[4]
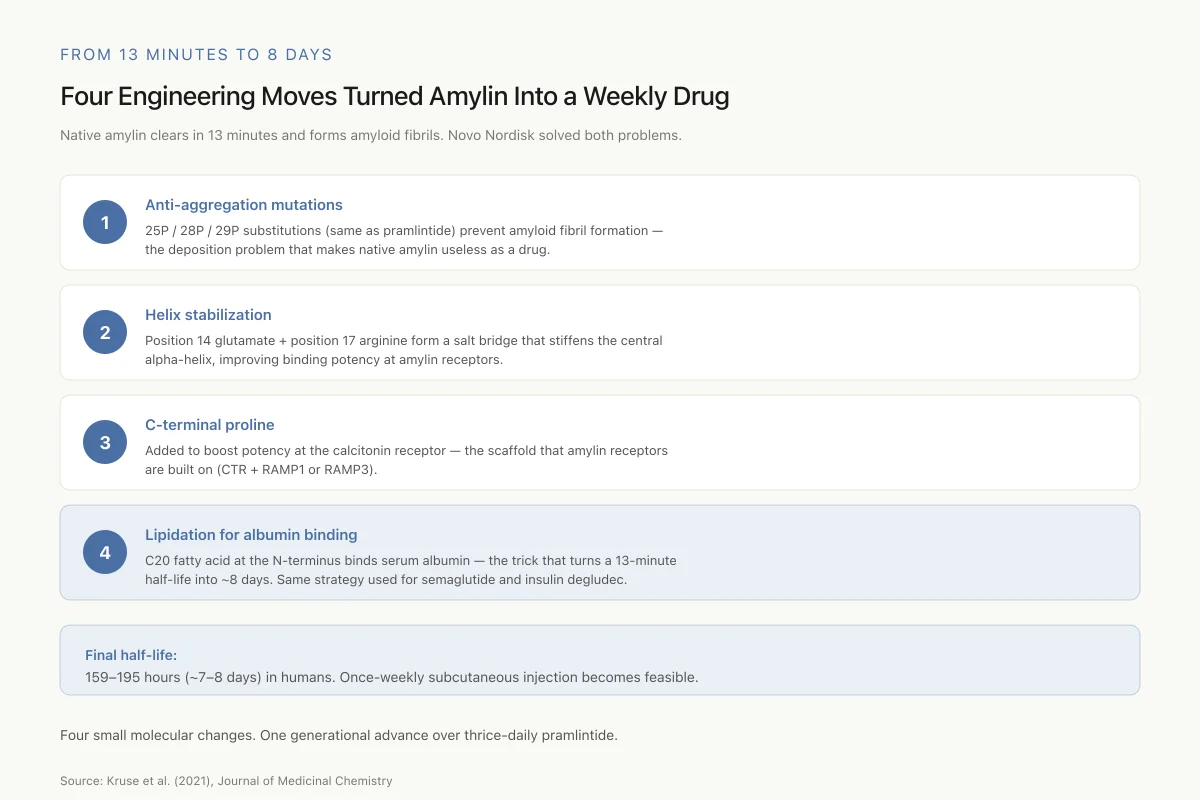
From 13 Minutes to 8 Days
Four Engineering Moves Turned Amylin Into a Weekly Drug
Native amylin clears in 13 minutes and forms amyloid fibrils. Novo Nordisk solved both problems.
Anti-aggregation mutations
25P / 28P / 29P substitutions (same as pramlintide) prevent amyloid fibril formation
Helix stabilization
Position 14 glutamate + position 17 arginine form a salt bridge that stiffens the central alpha-helix, improving receptor binding
C-terminal proline
Added to boost potency at the calcitonin receptor, the scaffold on which amylin receptors are built
Lipidation for albumin binding
C20 fatty acid at the N-terminus binds serum albumin — the trick that turns a 13-minute half-life into ~8 days
Final half-life: 159–195 hours (~7–8 days). Same albumin-binding trick used for semaglutide and insulin degludec.
Source: Kruse et al. (2021), J Med Chem
 View as image
View as imagePhase 2 Monotherapy Results
Before CagriSema, cagrilintide was tested alone. In a phase 2 dose-finding trial published in The Lancet in 2021, Lau and colleagues randomized 706 adults with overweight or obesity (without diabetes) across 57 sites in 10 countries to once-weekly cagrilintide at doses of 0.3, 0.6, 1.2, 2.4, or 4.5 mg, placebo, or liraglutide 3.0 mg daily (as an active comparator).[5]
At 26 weeks, the results by dose:
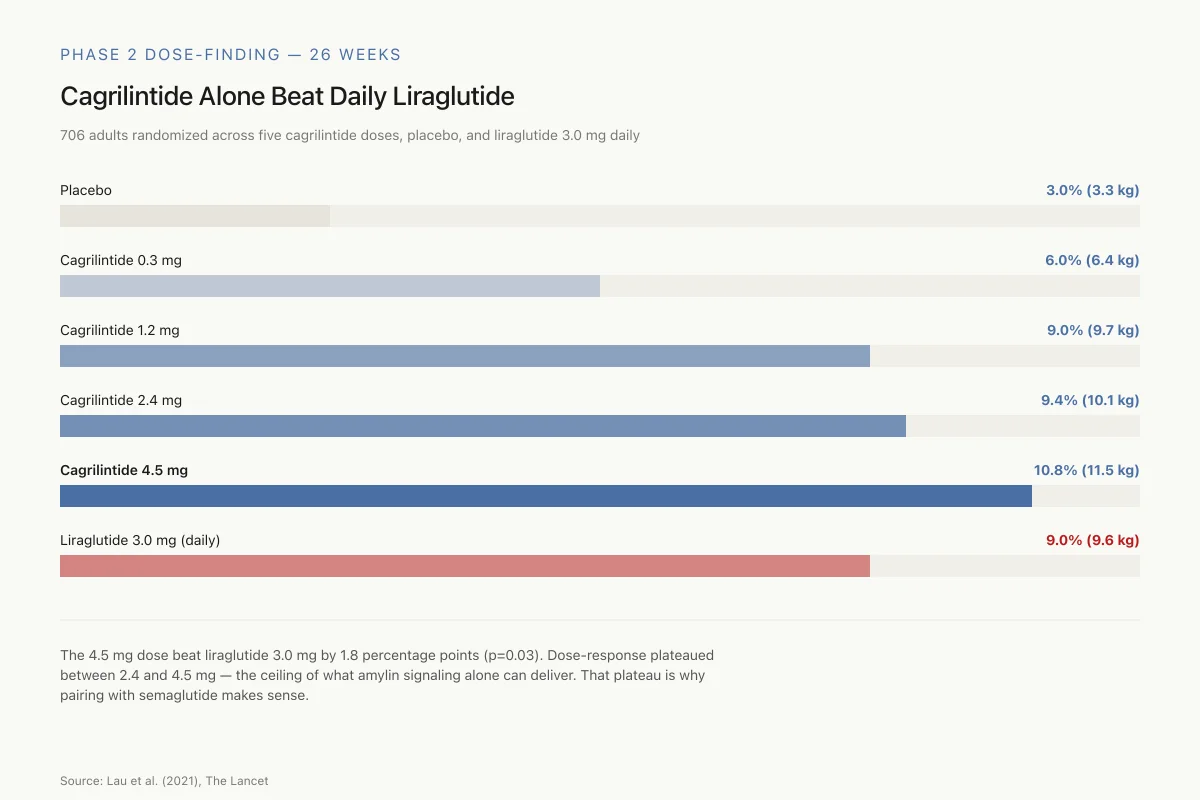
- Placebo: 3.0% weight loss (3.3 kg)
- Cagrilintide 0.3 mg: 6.0% (6.4 kg)
- Cagrilintide 1.2 mg: 9.0% (9.7 kg)
- Cagrilintide 2.4 mg: 9.4% (10.1 kg)
- Cagrilintide 4.5 mg: 10.8% (11.5 kg)
- Liraglutide 3.0 mg: 9.0% (9.6 kg)
The highest cagrilintide dose (4.5 mg) produced statistically greater weight loss than liraglutide 3.0 mg (estimated treatment difference 1.8%, p=0.03). Gastrointestinal adverse events occurred in 41% to 63% of cagrilintide groups versus 32% for placebo, primarily nausea (20% to 47% versus 18%). Most gastrointestinal events were transient and mild to moderate.
These monotherapy results established that cagrilintide alone could produce weight loss comparable to a first-generation GLP-1 agonist. The dose-response curve showed a clear plateau between 2.4 mg and 4.5 mg, suggesting diminishing returns at higher doses through the amylin pathway alone. Body composition analysis was particularly interesting: cagrilintide reduced relative fat mass while maintaining relative lean mass, a finding that differentiates it from some caloric restriction approaches that reduce both.
Phase 2 Dose-Finding — 26 Weeks
Cagrilintide Alone Beat Daily Liraglutide
706 adults with overweight/obesity, randomized across five cagrilintide doses, placebo, and liraglutide 3.0 mg daily.
The 4.5 mg dose beat liraglutide by 1.8 percentage points (p=0.03). Dose-response plateaued between 2.4 and 4.5 mg — the signal amylin alone could give. That plateau is why pairing with semaglutide makes sense.
Source: Lau et al. (2021), The Lancet
 View as image
View as imageThe trial also included a 26-week weight-maintenance period after treatment discontinuation. As with virtually all anti-obesity pharmaceuticals, weight regain occurred when the drug was stopped, reinforcing the chronic disease model that frames modern obesity pharmacotherapy.
The question was whether combining cagrilintide with a more potent GLP-1 agonist would produce additive effects. A phase 1b concomitant-dosing study confirmed that cagrilintide and semaglutide could be safely co-administered, with pharmacokinetic profiles consistent with each drug acting independently. This cleared the path for the phase 3 REDEFINE program.
The REDEFINE Trials: Cagrilintide Plus Semaglutide
The phase 3 REDEFINE program tested the fixed-dose combination of cagrilintide 2.4 mg plus semaglutide 2.4 mg (CagriSema) in two pivotal trials.
REDEFINE 1: Adults Without Diabetes
Published in the New England Journal of Medicine in June 2025, REDEFINE 1 randomized 3,417 adults without diabetes who had a BMI of 30 or higher (or 27 or higher with at least one obesity-related complication) to CagriSema, cagrilintide 2.4 mg alone, semaglutide 2.4 mg alone, or placebo.[6]
At 68 weeks:
- CagriSema: 20.4% mean body weight reduction
- Semaglutide 2.4 mg alone: 14.9%
- Cagrilintide 2.4 mg alone: 11.5%
- Placebo: 3.0%
The combination outperformed both components individually. Sixty percent of CagriSema participants achieved at least 20% weight loss. Twenty-three percent lost 30% or more. These are among the highest weight loss figures reported for any pharmaceutical intervention in a phase 3 obesity trial. The combination also improved systolic blood pressure, waist circumference, lipid profiles, and glycemic markers. Among participants with prediabetes at baseline, 88% returned to normoglycemia.
Gastrointestinal adverse events were reported by 72.5% of the CagriSema group versus 34.4% for placebo, consistent with the known side effect profiles of both drug classes. Most events were transient and mild to moderate in severity.
REDEFINE 2: Adults With Type 2 Diabetes
The companion trial, also published in the NEJM in 2025, enrolled 1,206 adults with type 2 diabetes and overweight or obesity.[7]
At 68 weeks:
- CagriSema: 13.7% mean body weight reduction
- Placebo: 3.4%
Weight loss in the type 2 diabetes population was lower than in REDEFINE 1, consistent with prior observations that people with type 2 diabetes lose less weight on GLP-1 and amylin agonists than those without diabetes. The trial also showed improvements in HbA1c and cardiometabolic risk factors. A subsequent analysis found CagriSema significantly reduced blood pressure compared to placebo across multiple measures.[8]
For a detailed breakdown of how CagriSema performed against semaglutide alone across both trials, see REDEFINE Trial Results: How CagriSema Performed Against Semaglutide Alone.
The 25% Target and What It Means
CagriSema generated headlines in late 2024 when Novo Nordisk disclosed that the combination did not meet the company's internal target of 25% mean weight loss. The stock dropped sharply on the news.
Context matters. The 25% target was a company projection, not a pre-specified primary endpoint. The REDEFINE 1 primary endpoints were superiority over placebo in percent weight change and proportion of participants achieving at least 5% weight loss. CagriSema met both. The 20.4% result also numerically exceeded semaglutide 2.4 mg alone by 5.5 percentage points and cagrilintide alone by 8.9 percentage points.
The "miss" was relative to investor expectations, not clinical performance. No anti-obesity medication had previously demonstrated 20% mean weight loss in a phase 3 trial with both active-comparator arms. A meta-analysis comparing CagriSema with semaglutide monotherapy confirmed statistically superior weight reduction for the combination.[9]
Mechanism of Action: How Two Pathways Combine
The pharmacological rationale for CagriSema rests on the complementary but non-overlapping mechanisms of amylin and GLP-1 signaling.
Cagrilintide (amylin pathway): Activates amylin receptor subtypes 1 (AMY1) and 3 (AMY3) in the area postrema. A 2025 study using receptor-specific knockout models demonstrated that cagrilintide's food intake reduction and body weight effects were mediated specifically through AMY1 and AMY3, not through calcitonin receptors alone.[10] The downstream effects include reduced meal size, delayed gastric emptying, and suppression of post-prandial glucagon.
Semaglutide (GLP-1 pathway): Activates GLP-1 receptors in the hypothalamus, area postrema, and gut. Effects include appetite suppression through both homeostatic and hedonic reward pathways, enhanced insulin secretion, delayed gastric emptying, and glucagon suppression.
The pathways converge on appetite suppression but through different receptor systems on different neuronal populations. This is distinct from combining two drugs that hit the same receptor (which tends to produce diminishing returns). A 2016 review of amylin's structure-function relationships noted that the amylin signaling system evolved independently from the incretin system, creating genuinely separate regulatory axes for energy balance.[3]
The safety of this dual-receptor approach was also evaluated. A thorough QT study demonstrated that cagrilintide at therapeutic and supratherapeutic doses did not prolong the QTc interval, addressing a key cardiac safety concern for novel peptide therapeutics.[11]
Safety Profile: What the Data Shows
The gastrointestinal side effect profile of cagrilintide, both alone and in combination, follows the pattern established by other gut peptide therapeutics. Nausea is the most common adverse event, occurring dose-dependently. In the phase 2 monotherapy trial, nausea rates ranged from 20% at the lowest dose (0.3 mg) to 47% at the highest (4.5 mg), compared to 18% for placebo.[5] In REDEFINE 1, the combination produced gastrointestinal events in 72.5% of participants, consistent with layering two peptide agonists that both slow gastric motility.
Several specific safety questions have been addressed:
Cardiac safety. The thorough QT study found no clinically relevant QTc prolongation at therapeutic (2.4 mg) or supratherapeutic (4.5 mg) cagrilintide doses.[11] This was a prerequisite for regulatory submission, as QT prolongation has derailed other peptide drug candidates.
Pancreatitis risk. GLP-1 agonists carry a class-wide concern for pancreatitis. In the REDEFINE trials, pancreatitis events were rare and occurred at similar rates across treatment groups, though the trials were not powered to detect rare events.
Weight regain after discontinuation. No published data yet addresses whether weight regain after stopping CagriSema differs from the well-documented rebound seen with semaglutide discontinuation. This is a critical open question for any obesity therapeutic positioned for chronic use.
Lean mass preservation. In the phase 2 monotherapy trial, cagrilintide reduced relative fat mass while maintaining relative lean mass. Whether this lean mass preservation holds in the longer 68-week REDEFINE data has been reported in secondary analyses but warrants attention. A 2024 review noted cagrilintide's preferential fat loss as a potential differentiator from GLP-1 monotherapy.[12]
Safety
ModerateStacked gastrointestinal side effects
Concern
REDEFINE 1 reported GI events in 72.5% of CagriSema recipients vs. 34.4% for placebo. Layering two peptide agonists that both slow gastric motility compounds the nausea, vomiting, and constipation profile of each.
What the research says
Most GI events were transient and mild to moderate. Slow titration protocols (standard for GLP-1 agonists) are expected at launch. QT study ruled out cardiac prolongation.
Particularly relevant for: Anyone starting CagriSema, especially with history of gastroparesis or significant reflux
What to do
Expect a titration schedule similar to Wegovy/Ozempic. Report severe or persistent GI symptoms early — dose can be held or stepped back.
Garvey et al. 2025; Lau et al. 2021; Gabe 2024 QT study
The Competitive Landscape
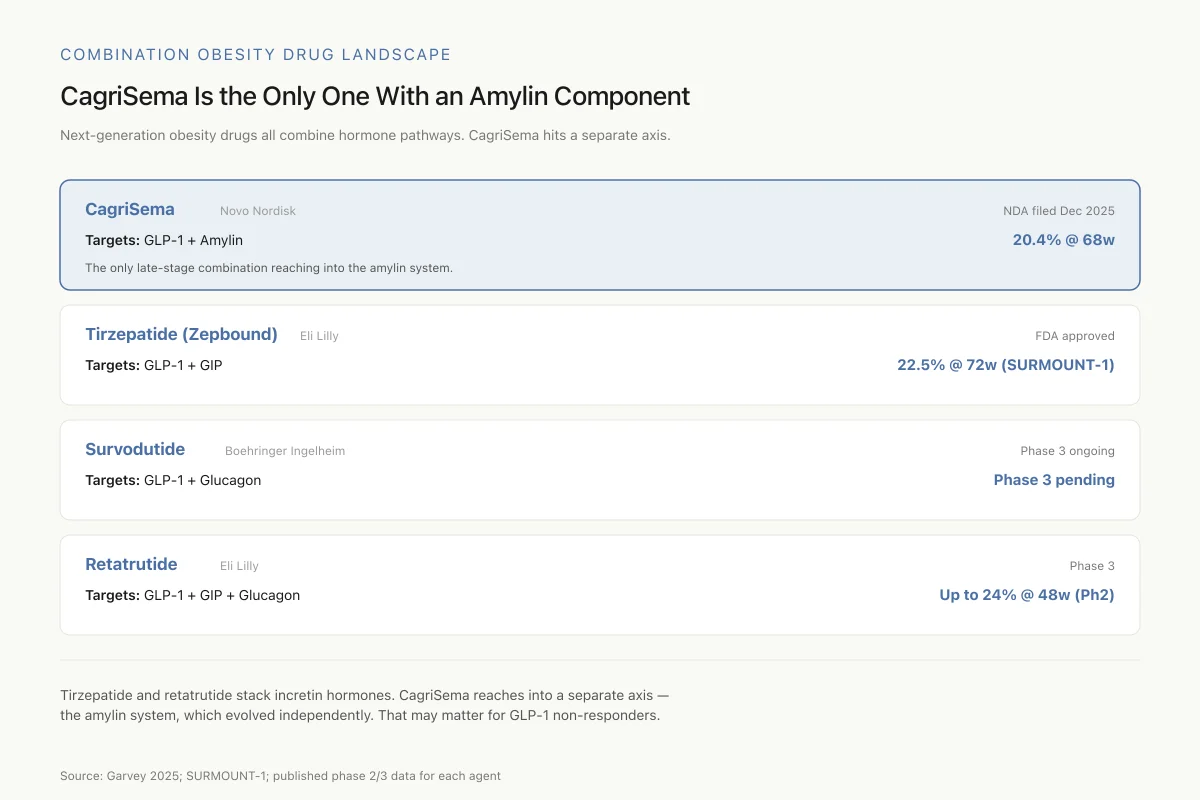
Combination Obesity Drug Landscape
CagriSema Is the Only One With an Amylin Component
The next generation of obesity drugs all combine hormone pathways. CagriSema’s differentiator isn’t weight loss — it’s which receptors it hits.
Tirzepatide and retatrutide stack incretin hormones on top of each other. CagriSema reaches into a separate axis — the amylin system, which evolved independently from incretins. That may matter for non-responders to GLP-1 alone.
Source: Garvey 2025; SURMOUNT-1; published phase 2/3 data for each agent
 View as image
View as imageCagriSema enters a market that has transformed since semaglutide's approval. The first-generation GLP-1 agonists (liraglutide, semaglutide) established that peptide-based weight management could produce clinically meaningful results. The next generation is defined by combination strategies:
Tirzepatide (Eli Lilly): Dual GLP-1/GIP receptor agonist. Already approved as Mounjaro (diabetes) and Zepbound (obesity). SURMOUNT-1 showed 22.5% mean weight loss at 72 weeks. This is CagriSema's primary competitor, and the head-to-head comparison is striking: CagriSema achieved 20.4% at 68 weeks using a different combination strategy (GLP-1 + amylin versus GLP-1 + GIP).
Survodutide (Boehringer Ingelheim): Dual GLP-1/glucagon agonist. Phase 3 ongoing. Targets a third distinct combination approach.
Retatrutide (Eli Lilly): Triple GLP-1/GIP/glucagon agonist. Phase 2 showed up to 24% weight loss at 48 weeks. Phase 3 results pending.
Each combination hits overlapping but distinct receptor systems. The amylin pathway that cagrilintide targets is unique among approved or late-stage candidates. No other combination in phase 3 includes an amylin component. This gives CagriSema a differentiated mechanism regardless of how the weight loss numbers compare.
Where Cagrilintide Stands in 2026
Novo Nordisk submitted the NDA for CagriSema to the FDA in December 2025. The application covers the fixed-dose combination of cagrilintide 2.4 mg and semaglutide 2.4 mg for chronic weight management in adults with obesity or overweight with at least one weight-related comorbidity. An FDA decision is expected in late 2026, with potential commercial launch by early 2027.
If approved, CagriSema would be the first combination GLP-1 plus amylin analog on the market. For the broader context of why combination peptide strategies are becoming the standard in metabolic medicine, see Combination Peptide Therapy: Why Two Mechanisms May Beat One.
Cagrilintide itself is not available as a standalone product. All clinical development has been through CagriSema. There are no plans for regulatory filing of cagrilintide monotherapy, despite the positive phase 2 results. The strategic decision reflects Novo Nordisk's view that the combination's efficacy profile is the competitive product, not the individual component.
Several questions remain open. Long-term cardiovascular outcomes data for CagriSema are not yet available. The duration of effect after discontinuation is unknown. Whether CagriSema's amylin component offers benefits beyond weight loss, such as beta-cell preservation or reduced amyloid deposition, remains theoretical. The existing amylin literature suggests these possibilities, but CagriSema's clinical program was not designed to test them.
The amylin system remains one of the less explored peptide pathways in metabolic medicine, despite decades of basic research. For the broader biology of amylin beyond obesity, including its roles in bone metabolism, cardiovascular regulation, and the Alzheimer's connection, see Amylin (IAPP): The Third Pancreatic Peptide Hormone.
The Bottom Line
Cagrilintide is a long-acting amylin analog engineered through lipidation for once-weekly dosing. As monotherapy, it produced up to 10.8% weight loss in a phase 2 trial. Combined with semaglutide as CagriSema, it produced 20.4% mean weight loss in the REDEFINE 1 trial with 60% of participants losing at least 20% of their body weight. Novo Nordisk filed for FDA approval in December 2025. If approved, CagriSema would be the first drug combining amylin and GLP-1 receptor agonism for obesity.
Sources & References
- 1RPEP-02659·Hay, Debbie L et al. (2015). “Amylin: The Pancreatic Peptide That Regulates Blood Sugar, Appetite, and More.” Pharmacological reviews.Study breakdown →PubMed →↩
- 2RPEP-05514·Kruse, Thomas et al. (2021). “How Scientists Designed Cagrilintide, a Long-Acting Amylin Peptide for Weight Loss.” Journal of medicinal chemistry.Study breakdown →PubMed →↩
- 3RPEP-02881·Bower, Rebekah L et al. (2016). “Amylin: The Diabetic Hormone Peptide That's Hard to Work With But Worth Improving.” British journal of pharmacology.Study breakdown →PubMed →↩
- 4RPEP-06361·Mathiesen, David S et al. (2022). “Long-acting amylin analogues for the management of obesity..” Current opinion in endocrinology.Study breakdown →PubMed →↩
- 5RPEP-05529·Lau, David C W et al. (2021). “Once-weekly cagrilintide for weight management in people with overweight and obesity: a multicentre, randomised, double-blind, placebo-controlled and active-controlled, dose-finding phase 2 trial..” Lancet (London.Study breakdown →PubMed →↩
- 6RPEP-11062·Garvey, W Timothy et al. (2025). “Coadministered Cagrilintide and Semaglutide in Adults with Overweight or Obesity..” The New England journal of medicine.Study breakdown →PubMed →↩
- 7RPEP-10633·Davies, Melanie J et al. (2025). “Cagrilintide-Semaglutide in Adults with Overweight or Obesity and Type 2 Diabetes..” The New England journal of medicine.Study breakdown →PubMed →↩
- 8RPEP-16312·Verma, Subodh et al. (2026). “CagriSema Drops Blood Pressure by 11 mmHg in Overweight Adults — Even in Resistant Hypertension.” Hypertension (Dallas.Study breakdown →PubMed →↩
- 9RPEP-15178·Gadelmawla, Ahmed Farid et al. (2026). “CagriSema (Amylin + GLP-1) Achieves 11 kg More Weight Loss Than Semaglutide Alone.” The American journal of cardiology.Study breakdown →PubMed →↩
- 10RPEP-11548·Jacobsen, Julie Mie et al. (2025). “CagriSema drives weight loss in rats by reducing energy intake and preserving energy expenditure..” Nature metabolism.Study breakdown →PubMed →↩
- 11RPEP-08227·Gabe, Maria B N et al. (2024). “Cagrilintide is not associated with clinically relevant QTc prolongation: A thorough QT study in healthy participants..” Diabetes.Study breakdown →PubMed →↩
- 12RPEP-08044·D'Ascanio, Antonella M et al. (2024). “Cagrilintide: How an Amylin Analog Combined with Semaglutide Targets Obesity Through Dual Brain Pathways.” Cardiology in review.Study breakdown →PubMed →↩