Amylin (IAPP): The Third Pancreatic Peptide Hormone
Pancreatic Peptide Hormones
22.7% Weight Loss
In the REDEFINE 1 trial, CagriSema (cagrilintide plus semaglutide) produced 22.7% mean weight loss at 68 weeks in adults with overweight or obesity, exceeding semaglutide alone by approximately 7 percentage points.
Garvey et al., New England Journal of Medicine, 2025
Garvey et al., New England Journal of Medicine, 2025
If you only read one thing
Amylin is a hormone your pancreas releases alongside insulin every time you eat. It slows digestion, tells your liver to stop making sugar, and signals your brain that you're full. The first amylin drug (pramlintide) flopped because it needed three shots a day. But a new long-acting version called cagrilintide, combined with semaglutide in a single weekly injection called CagriSema, just produced 22.7% weight loss in a major trial — beating semaglutide alone by about 7 percentage points.
Key Takeaways
- Amylin is a third pancreatic hormone your body releases alongside insulin every time you eat. Most people have never heard of it.
- It does three jobs at once: slows digestion, tells your liver to stop making sugar, and signals your brain that you're full.
- The first amylin drug flopped in 2005 because it needed three injections a day. A new once-weekly version is rewriting the story.
- Combining the new amylin drug with semaglutide in one weekly shot produced 22.7% weight loss — meaningfully more than semaglutide alone.
- In people with type 2 diabetes, the same combination more than doubled the weight loss — 15.6% versus 6% on semaglutide alone.
- Amylin works through a completely different receptor system than Ozempic, which is why combining them produces more than either does alone.
- Early research also suggests amylin is neuroprotective — in a mouse model of Alzheimer's, it cut amyloid plaque buildup by nearly half.
What Amylin Does: Three Jobs in One Peptide
Amylin performs three distinct physiological functions that together regulate the rate at which nutrients enter the bloodstream after a meal.[3]
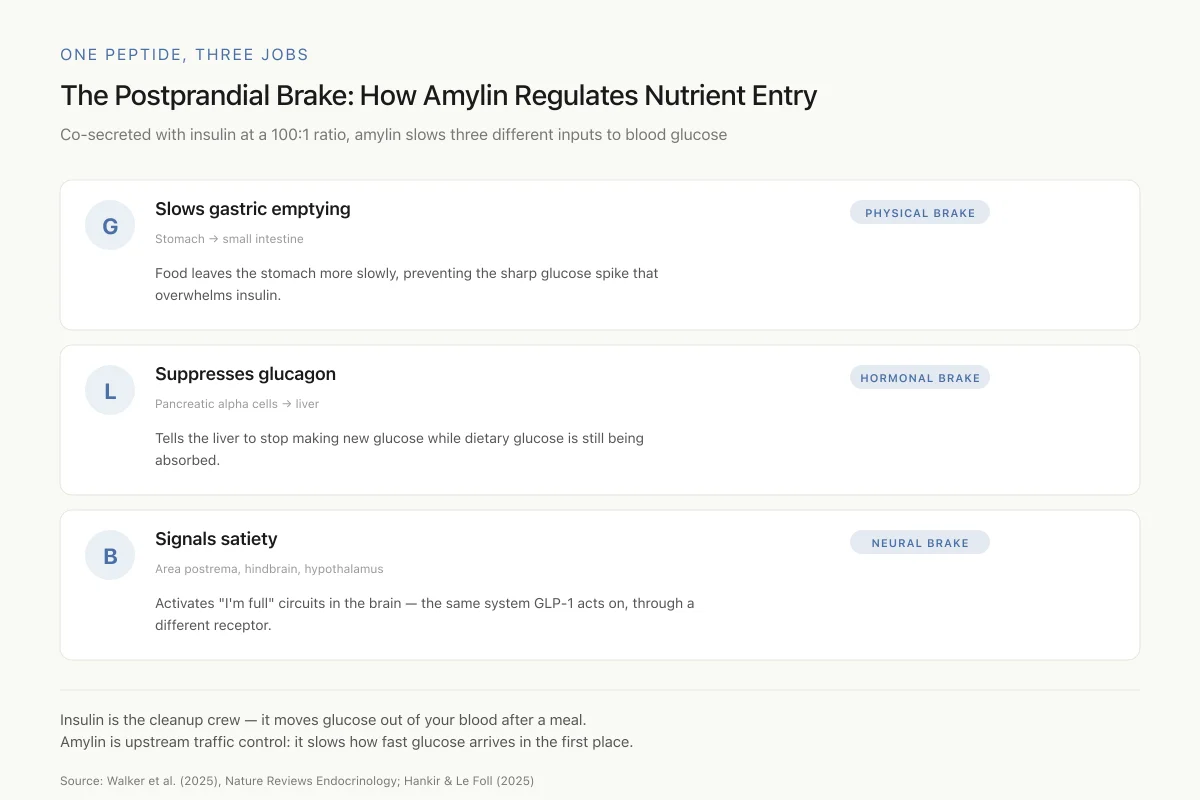
Gastric emptying. Amylin slows the rate at which food leaves the stomach and enters the small intestine. This reduces the speed of glucose absorption, preventing the sharp postprandial glucose spikes that challenge insulin's ability to maintain homeostasis. The effect is dose-dependent and mediated through vagal afferent pathways.
Glucagon suppression. After a meal, amylin suppresses the secretion of glucagon from pancreatic alpha cells. Glucagon normally stimulates hepatic glucose production, so suppressing it during meals prevents the liver from releasing glucose at the same time dietary glucose is being absorbed. This complements insulin's action: insulin promotes glucose uptake into cells while amylin reduces the amount of new glucose entering the bloodstream.
Satiety signaling. Amylin acts on the area postrema and nucleus of the solitary tract in the caudal hindbrain to promote meal-ending satiety. Hankir and Le Foll documented in 2025 that amylin's central nervous system effects extend beyond these traditionally recognized brainstem targets. Their review identified amylin-responsive neurons in the lateral parabrachial nucleus, the ventral tegmental area, and the hypothalamus, suggesting a distributed network of satiety circuits rather than a single brainstem relay.[4]
One Peptide, Three Jobs
The Postprandial Brake: How Amylin Regulates Nutrient Entry
Co-secreted with insulin at a 100:1 ratio, amylin slows three different inputs to blood glucose after every meal
Insulin is the cleanup crew — it moves glucose out of your blood after a meal. Amylin is upstream traffic control: it slows how fast glucose arrives in the first place, and tells you to stop eating before the crew gets overwhelmed.
Source: Walker et al. (2025), Nature Reviews Endocrinology; Hankir & Le Foll (2025)
 View as image
View as imageTogether, these three functions make amylin a postprandial brake, a hormone that controls the speed and magnitude of nutrient absorption after eating. The net effect is a smoother glucose profile, a reduced insulin requirement per meal, and earlier meal termination.
These functions explain why amylin deficiency matters. In type 1 diabetes, beta cell destruction eliminates both insulin and amylin production. In advanced type 2 diabetes, beta cell failure reduces amylin secretion. The result in both cases is faster gastric emptying, unopposed postprandial glucagon secretion, and reduced satiety signaling, all of which contribute to glucose instability and weight gain.
The Amyloid Problem: When Amylin Misfolds
Human amylin has a structural property that makes it both biologically interesting and medically dangerous: it aggregates. The 20-29 amino acid segment of human amylin is amyloidogenic, meaning it spontaneously forms beta-sheet-rich fibrils that accumulate as insoluble deposits in pancreatic islets.[1]
These islet amyloid deposits are found in approximately 90% of people with type 2 diabetes at autopsy. The aggregation process is toxic to beta cells through multiple mechanisms: membrane disruption by amylin oligomers, endoplasmic reticulum stress from misfolded protein accumulation, mitochondrial dysfunction, and activation of the NLRP3 inflammasome. The toxic species appear to be the intermediate oligomers rather than the mature fibrils, which are relatively inert.
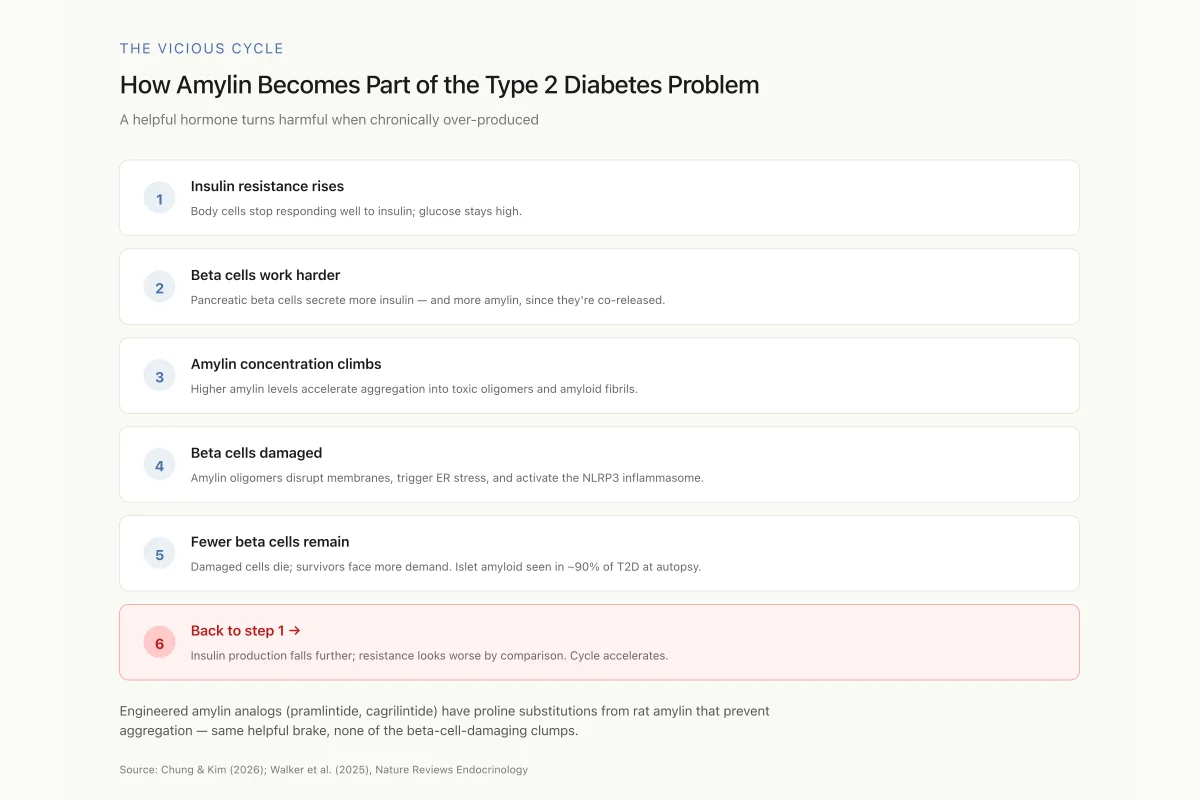
This creates a vicious cycle. Insulin resistance increases demand on beta cells, which respond by secreting more insulin and more amylin. Higher amylin concentrations accelerate aggregation, which damages beta cells, which further reduces their capacity to meet insulin demand. Whether amyloid deposition is a cause or a consequence of beta cell failure in type 2 diabetes remains debated, but the evidence for direct toxicity of amylin oligomers to beta cells is substantial.
The Vicious Cycle
How Amylin Becomes Part of the Type 2 Diabetes Problem
A helpful hormone turns harmful when chronically over-produced
This is why engineered amylin analogs matter: pramlintide and cagrilintide have proline substitutions (borrowed from rat amylin) that prevent aggregation while keeping full receptor activity. You get the helpful postprandial brake without the beta-cell-damaging clumps.
Source: Chung & Kim (2026); Walker et al. (2025), Nature Reviews Endocrinology
 View as image
View as imageThe amyloidogenic property of human amylin is species-specific. Rat and mouse amylin have proline substitutions in the 20-29 region that prevent aggregation, which is why rodents do not develop islet amyloid. This species difference has important implications for drug design: the first successful amylin analog, pramlintide, was engineered with three proline substitutions from rat amylin to prevent aggregation while preserving receptor activity.
The aggregation process also has implications for studying amylin in the laboratory. Synthetic human amylin is notoriously difficult to work with because it begins aggregating immediately upon dissolution. Researchers must prepare fresh solutions and work quickly to study the monomeric form. The kinetics of aggregation depend on concentration, pH, temperature, and the presence of lipid membranes, making in vitro studies sensitive to experimental conditions. Walker and colleagues in their 2025 Nature Reviews Endocrinology article emphasized that the aggregation-prone nature of native human amylin was a primary driver for developing stabilized analogs, not only for therapeutic use but for basic research tools.[3]
The Amylin Receptor: More Complex Than Expected
Amylin does not have a single dedicated receptor. Instead, it acts through a family of receptors formed by the heterodimerization of the calcitonin receptor (CTR) with receptor activity-modifying proteins (RAMPs). The combination of CTR with RAMP1 forms the amylin 1 receptor (AMY1R), CTR with RAMP2 forms AMY2R, and CTR with RAMP3 forms AMY3R.[5]
This receptor complexity has practical consequences. Each AMY receptor subtype has a different tissue distribution, different signaling properties, and different affinities for amylin and related peptides. Calcitonin gene-related peptide (CGRP), a neuropeptide involved in migraine, can activate AMY1R, which is one reason amylin receptor pharmacology intersects with migraine research.
Cao and colleagues resolved the cryo-EM structure of the human AMY1R in complex with CGRP and Gs protein at 3.3-angstrom resolution in 2024. This structure revealed the molecular basis for CGRP cross-reactivity at amylin receptors and identified key differences from the CGRP receptor that could guide the development of receptor-selective drugs.[6]
Lee's 2025 review of amylin peptide receptor activators characterized the pharmacological profiles of existing and developmental amylin agonists across these receptor subtypes. Some compounds, like cagrilintide, are non-selective agonists of the entire calcitonin receptor family, while others show selectivity for specific AMY receptor subtypes. Whether receptor selectivity matters for clinical outcomes is an active area of investigation.[5]
Pramlintide: The First Amylin Drug
Pramlintide (Symlin), approved by the FDA in 2005, is a synthetic analog of human amylin with three proline substitutions (positions 25, 28, and 29) that prevent aggregation. It is approved as an adjunct to mealtime insulin in patients with type 1 or type 2 diabetes who have not achieved adequate glycemic control.
The development of pramlintide illustrates a recurring challenge in peptide pharmacology: turning a biological insight into a practical medicine. Amylin's biology was well-characterized by the mid-1990s, but the native peptide could not be used therapeutically because it aggregated in solution and had an impractically short half-life. The solution was to borrow from rat amylin, which has three proline residues in the amyloidogenic region that act as "structural breakers," preventing the beta-sheet stacking that leads to fibril formation. Grafting these prolines onto the human sequence produced a molecule that retained full receptor activity while remaining stable in solution.
Pramlintide replicates all three of amylin's physiological functions: it slows gastric emptying, suppresses postprandial glucagon, and promotes satiety. Clinical trials demonstrated modest improvements in HbA1c (approximately 0.3-0.6% reduction) and weight loss (approximately 1-2 kg) compared to insulin alone.[7]
Despite its physiological logic, pramlintide achieved limited commercial success for two reasons. First, its half-life of 20 to 45 minutes requires subcutaneous injection before every meal, adding three daily injections on top of an existing insulin regimen. Second, the magnitude of benefit on glycemic control and weight was modest compared to the injection burden. The weight loss, while statistically meaningful, was not clinically transformative in an era when patients and physicians had begun to expect the larger effects seen with bariatric surgery. Pramlintide demonstrated proof of concept that amylin replacement is therapeutically useful, but its pharmacokinetics made it impractical for most patients.
The pramlintide experience shaped the subsequent development strategy. Bailey and colleagues noted that the key lesson from pramlintide was not that amylin agonism lacked clinical value but that the delivery method could not match the biology. A peptide that regulates meal-related physiology but must be injected three times daily at every meal creates a compliance barrier that undermines its therapeutic potential. This realization drove the development of long-acting amylin analogs that could decouple the pharmacological effect from the meal-by-meal dosing requirement.[7]
Cagrilintide: The Long-Acting Revolution
Cagrilintide is a long-acting acylated amylin analog developed by Novo Nordisk. Through fatty acid acylation and amino acid substitutions, cagrilintide achieves a half-life sufficient for once-weekly subcutaneous administration, solving the central pharmacokinetic limitation of pramlintide.[8]
Cagrilintide monotherapy
D'Ascanio and colleagues reviewed cagrilintide's development as an obesity treatment in 2024. In a Phase 2 dose-ranging trial, cagrilintide monotherapy at 4.5 mg weekly produced 10.8% weight loss (7.8% placebo-subtracted) at 26 weeks in adults with overweight or obesity without diabetes.[8] Dutta and colleagues' meta-analysis of cagrilintide trials confirmed the weight loss effect and characterized the safety profile, with nausea being the most common adverse event, occurring in approximately 25 to 40% of patients depending on dose. The gastrointestinal side effect profile was comparable to GLP-1 agonists, reflecting the shared effect on gastric motility. Importantly, hypoglycemia was rare in non-diabetic patients, consistent with amylin's glucose-dependent mechanism of action.[9]
CagriSema: the combination that changed the field
The clinical significance of cagrilintide emerged fully when combined with semaglutide (a GLP-1 receptor agonist) in a co-formulated injection called CagriSema.
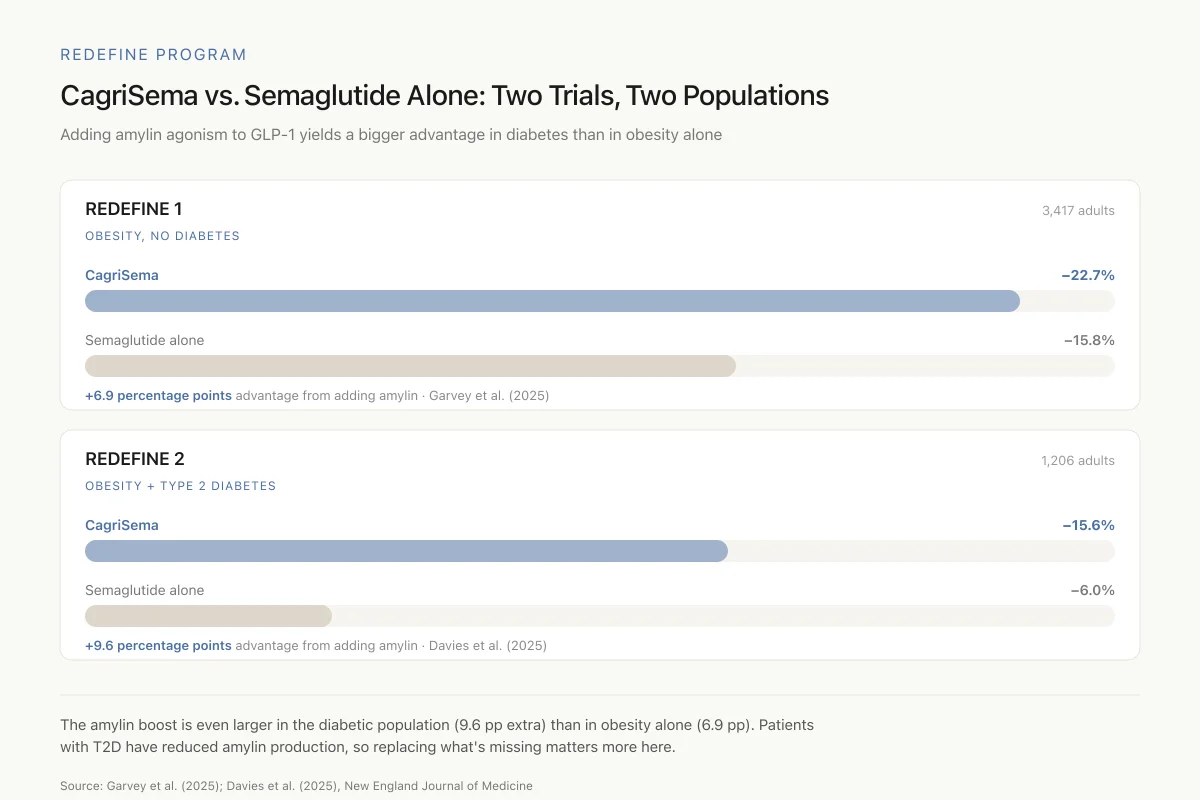
REDEFINE 1 (obesity without diabetes): Garvey and colleagues published the REDEFINE 1 results in the New England Journal of Medicine in 2025. In 3,417 adults with overweight or obesity, CagriSema produced 22.7% mean weight loss at 68 weeks versus 15.8% for semaglutide 2.4 mg alone and 8.6% for cagrilintide 2.4 mg alone. The approximately 7 percentage point advantage over semaglutide alone was maintained across subgroups and represented the largest weight loss achieved by any peptide therapy in a Phase 3 trial.[2]
REDEFINE Program
CagriSema vs. Semaglutide Alone: Two Trials, Two Populations
Adding amylin agonism to GLP-1 yields a bigger advantage in diabetes than in obesity alone
The amylin boost is even larger in the diabetic population (9.6 extra percentage points) than in obesity alone (6.9 points). Patients with type 2 diabetes typically have reduced amylin production, so replacing what's missing appears to matter more in this group.
Source: Garvey et al. (2025); Davies et al. (2025), New England Journal of Medicine
 View as image
View as imageREDEFINE 2 (type 2 diabetes): Davies and colleagues reported the REDEFINE 2 results, also in the New England Journal of Medicine, showing that CagriSema produced 15.6% weight loss at 68 weeks in adults with overweight or obesity and type 2 diabetes, compared to 6.0% for semaglutide alone. The trial also demonstrated superior HbA1c reduction with CagriSema (approximately 2.2 percentage points versus 1.8 for semaglutide alone) and included a continuous glucose monitoring subgroup analysis showing improved time in range.[10]
The REDEFINE results established a pharmacological principle: amylin agonism and GLP-1 agonism activate complementary satiety pathways, and the combination produces effects greater than either agent alone. Amylin acts primarily through area postrema and hindbrain circuits, while GLP-1 acts through hypothalamic and reward pathways. The overlapping but distinct mechanisms explain the additive weight loss.
The safety profile of CagriSema in both REDEFINE trials was consistent with the known adverse event profiles of its individual components. Gastrointestinal adverse events (nausea, vomiting, diarrhea) were the most common, occurring at rates slightly higher than semaglutide alone. The incidence of serious adverse events was low and comparable across treatment groups. Injection site reactions were mild and infrequent. The key safety question that Phase 3 data could not answer is whether the greater weight loss with CagriSema translates to greater cardiovascular benefit compared to semaglutide alone, a question the ongoing SELECT-2 cardiovascular outcome trial is designed to address.
The clinical significance of the approximately 7 percentage point improvement over semaglutide alone extends beyond the weight loss number itself. At the population level, a higher proportion of CagriSema-treated patients achieved clinically meaningful weight loss thresholds: more patients crossed the 15%, 20%, and 25% weight loss marks that are associated with improvements in obesity-related comorbidities including obstructive sleep apnea, nonalcoholic steatohepatitis, and osteoarthritis symptom reduction.
The Next Generation of Amylin Analogs
The success of cagrilintide has accelerated development of additional amylin-based therapeutics. Bailey and colleagues reviewed the landscape of long-acting amylin-related peptides in 2026, identifying several distinct approaches to extending amylin's pharmacological activity.[7]
Petrelintide is a potent, stable, long-acting human amylin analog using a different chemical strategy than cagrilintide's acylation approach. Its development aims to provide an alternative long-acting amylin agonist with potentially different receptor selectivity profiles.
Amycretin is a unimolecular dual agonist that activates both GLP-1 and amylin receptors from a single peptide molecule, eliminating the need for co-formulation. Preliminary data from a Phase 1b/2a trial showed that oral amycretin produced 13.1% weight loss at 12 weeks, a rate that, if sustained, would exceed injectable CagriSema. However, longer-duration data is needed. The oral bioavailability of amycretin, if confirmed in larger trials, would represent a meaningful advance over injectable formulations, potentially expanding the patient population willing to initiate therapy. The compound uses semaglutide-like oral absorption technology to achieve systemic peptide delivery from a tablet, a technical achievement given amylin's sensitivity to proteolytic degradation in the gastrointestinal tract.
Dual amylin and calcitonin receptor agonists (DACRAs) represent another approach, exploiting the structural relationship between the amylin and calcitonin receptor systems to achieve enhanced metabolic effects. Volcansek and colleagues characterized these as part of the emerging "precision diabetology" approach, where the specific hormonal imbalance in each patient guides the choice of therapy.[11]
Chung and Kim's 2026 five-year retrospective review noted that the amylin field has evolved from treating amylin as a minor adjunct to insulin therapy to recognizing it as a central component of metabolic pharmacology. The term "diabesity" has entered the clinical vocabulary, and amylin agonists are positioned as treatments for the combined metabolic dysfunction rather than for diabetes or obesity alone.[1]
Beyond Metabolism: Amylin and the Brain
Amylin's effects extend beyond glucose regulation and appetite. The presence of amylin receptors in brain regions associated with cognition, particularly the hippocampus, has led to investigation of amylin in neurodegenerative disease.
Corrigan and colleagues studied amylin receptor activation in APP/PS1 transgenic mice, a model of Alzheimer's disease. Their 2023 study found that amylin receptor agonism (not antagonism, as some had proposed) reduced amyloid plaque burden by 47% and improved spatial memory performance. The mechanism appeared to involve enhanced microglial clearance of amyloid-beta and reduced neuroinflammation.[12]
The Alzheimer's connection raises a question about the relationship between amylin aggregation in the pancreas and amyloid-beta aggregation in the brain. Both are amyloidogenic peptides. Both form toxic oligomeric intermediates. Epidemiological data showing elevated dementia risk in type 2 diabetes patients has led some researchers to hypothesize that amylin and amyloid-beta may co-aggregate or that systemic amylin amyloidosis may contribute to brain pathology. The Corrigan study suggests that pharmacological amylin receptor activation, as opposed to native amylin aggregation, may be neuroprotective, a distinction that could have therapeutic relevance.
This distinction between endogenous amylin (which can aggregate and cause damage) and exogenous amylin receptor agonists (which are engineered not to aggregate and may be protective) is critical. Non-aggregating amylin analogs like pramlintide and cagrilintide activate the same receptors as native amylin but cannot form the toxic oligomeric species. If the neuroprotective effects observed in mouse models translate to humans, the widespread use of amylin analogs for metabolic indications could provide an incidental window into their neurological effects through post-marketing surveillance data. Several research groups have proposed repurposing metabolic amylin agonists for Alzheimer's trials, though no such trial has been initiated as of early 2026.
What the Evidence Does Not Yet Show
The CagriSema REDEFINE trials are impressive but leave several questions unanswered. Long-term safety beyond 68 weeks has not been established. The cardiovascular outcome trial (SELECT-2) is ongoing but results are not yet available. Whether the weight loss observed with CagriSema is maintained after discontinuation, a known limitation of GLP-1 agonist monotherapy, has not been studied.
The weight regain question is particularly relevant for amylin-based therapies. Because amylin's satiety effects are pharmacological rather than structural (unlike bariatric surgery, which physically restricts intake), stopping the drug removes the satiety signal. If CagriSema produces greater initial weight loss than semaglutide alone but the same proportional regain after discontinuation, the net long-term benefit may be proportionally larger. Alternatively, if the dual-pathway satiety suppression produces a stronger rebound effect, the maintenance picture could be less favorable. These questions can only be answered by controlled discontinuation studies, which have not yet been reported for CagriSema.
The amylin-Alzheimer's connection is entirely preclinical. No human clinical trial has tested an amylin analog for cognitive outcomes. The neuroprotective effects seen in APP/PS1 mice may not translate to human Alzheimer's disease, which has a different pathological complexity.
The receptor pharmacology raises a practical question for drug development: amylin acts on calcitonin receptors, calcitonin gene-related peptide pathways, and amylin-specific receptor subtypes. Non-selective agonism (as with cagrilintide) activates all of these, which may contribute to both therapeutic effects and side effects. The observation that CGRP, a migraine-associated peptide, can activate amylin receptors raises the question of whether amylin agonists could have effects on headache physiology, either beneficial or adverse. Whether receptor-selective amylin analogs would offer a better therapeutic index remains unknown.
The competitive landscape is evolving rapidly. With CagriSema poised for regulatory submission, tirzepatide (a dual GIP/GLP-1 agonist) already approved for obesity, and retatrutide (a triple agonist targeting GLP-1, GIP, and glucagon receptors) in Phase 3 trials, the metabolic peptide field is moving toward multi-receptor strategies. Amylin agonism adds a fourth receptor system to this mix, and the question is whether dual, triple, or quadruple receptor targeting will ultimately provide the best balance of efficacy, safety, and tolerability for different patient populations.
For how amylin relates to other insulin-associated peptides, see C-Peptide: The Overlooked Insulin Byproduct That's Actually a Biomarker.
The Bottom Line
Amylin is a pancreatic peptide hormone that regulates gastric emptying, glucagon secretion, and appetite. Long-acting analogs, particularly cagrilintide combined with semaglutide (CagriSema), have produced 22.7% weight loss in Phase 3 trials. The amylin field has expanded from a minor adjunct to insulin therapy to a central pillar of metabolic pharmacology, with additional applications under investigation in neurodegenerative disease.
Sources & References
- 1RPEP-15043·Chung, Chae Won et al. (2026). “Amylin: The Pancreatic Hormone Emerging as a Powerful Obesity and Diabetes Treatment.” Journal of obesity & metabolic syndrome.Study breakdown →PubMed →↩
- 2RPEP-11062·Garvey, W Timothy et al. (2025). “Coadministered Cagrilintide and Semaglutide in Adults with Overweight or Obesity..” The New England journal of medicine.Study breakdown →PubMed →↩
- 3RPEP-13964·Walker, Christopher S et al. (2025). “Amylin: emergent therapeutic opportunities in overweight, obesity and diabetes mellitus..” Nature reviews. Endocrinology.Study breakdown →PubMed →↩
- 4RPEP-11279·Hankir, Mohammed K et al. (2025). “Central nervous system pathways targeted by amylin in the regulation of food intake..” Biochimie.Study breakdown →PubMed →↩
- 5RPEP-12024·Lee, Sangmin (2025). “Amylin: The Next Peptide Hormone Target for Obesity Drugs After GLP-1.” Current drug targets.Study breakdown →PubMed →↩
- 6RPEP-07928·Cao, Jianjun et al. (2024). “Cryo-EM Structure of the Human Amylin 1 Receptor in Complex with CGRP and Gs Protein..” Biochemistry.Study breakdown →PubMed →↩
- 7RPEP-14819·Bailey, Clifford J et al. (2026). “Next-Generation Long-Acting Amylin Drugs for Obesity and Type 2 Diabetes.” Peptides.Study breakdown →PubMed →↩
- 8RPEP-08044·D'Ascanio, Antonella M et al. (2024). “Cagrilintide: How an Amylin Analog Combined with Semaglutide Targets Obesity Through Dual Brain Pathways.” Cardiology in review.Study breakdown →PubMed →↩
- 9RPEP-08129·Dutta, Deep et al. (2024). “CagriSema Combination Outperforms Semaglutide Alone for Weight Loss in Meta-Analysis.” Indian journal of endocrinology and metabolism.Study breakdown →PubMed →↩
- 10RPEP-10633·Davies, Melanie J et al. (2025). “Cagrilintide-Semaglutide in Adults with Overweight or Obesity and Type 2 Diabetes..” The New England journal of medicine.Study breakdown →PubMed →↩
- 11RPEP-13944·Volčanšek, Špela et al. (2025). “Amylin: From Mode of Action to Future Clinical Potential in Diabetes and Obesity..” Diabetes therapy : research.Study breakdown →PubMed →↩
- 12RPEP-06812·Corrigan, Rachel R et al. (2023). “Amylin Peptide Analog Pramlintide Improves Cognition in Alzheimer's Mice Through Peripheral Mechanisms.” Journal of Alzheimer's disease : JAD.Study breakdown →PubMed →↩