Amylin: The Pancreatic Peptide Behind Satiety
Amylin and Metabolic Peptides
100:1 Insulin to Amylin
Pancreatic beta cells co-secrete insulin and amylin in an approximate 100:1 ratio after every meal. Amylin slows gastric emptying, suppresses glucagon, and signals satiety to the brain. Pramlintide (Symlin) is the only approved amylin analog; cagrilintide is in late-stage trials for obesity.
Volcansek et al., Diabetes Therapy, 2025
Volcansek et al., Diabetes Therapy, 2025
If you only read one thing
Amylin is a hormone your pancreas makes every time you eat, right alongside insulin. It tells your brain you're full, slows down digestion, and stops your liver from dumping extra sugar into your blood. There's already one drug based on it (pramlintide, approved in 2005), but nobody uses it because the dosing is a pain. The exciting part: a once-weekly version called cagrilintide is in late-stage trials for weight loss, and combining it with semaglutide produces even bigger results than semaglutide alone.
Every time you eat, your pancreatic beta cells release two hormones: insulin, which lowers blood glucose by driving sugar into cells, and amylin, which controls how fast that glucose arrives. Amylin slows gastric emptying, suppresses post-meal glucagon secretion, and signals satiety to the brainstem. It is one of the most important metabolic peptides that most people have never heard of. A 2025 review in Diabetes Therapy described amylin as having "underappreciated" clinical potential despite being known since the late 1980s.[1] Pramlintide (brand name Symlin), the only approved amylin analog, has been available since 2005 but remains underused. Meanwhile, next-generation amylin analogs like cagrilintide are in Phase III trials for obesity, and the combination of amylin and GLP-1 receptor agonists represents what some researchers call the most promising frontier in weight-loss pharmacology.
Key Takeaways
- Every time you eat, your pancreas releases two hormones — insulin and a less-famous one called amylin — at the same time.
- If insulin is the key that unlocks the door for sugar to enter cells, amylin is the traffic controller slowing sugar down before it even arrives.
- Amylin also signals your brain that you're full — which is why drug companies are racing to turn it into the next weight loss medicine.
- People with type 1 diabetes who only take insulin are replacing half of what their pancreas normally produces. That's a quieter part of the disease most treatments ignore.
- The first amylin drug has been approved since 2005 but almost nobody uses it — it needs 3 daily injections with careful timing.
- A new once-weekly version (cagrilintide) is in late-stage trials for obesity. Early results show 10%+ weight loss in under 6 months.
- Combining amylin with semaglutide produces bigger weight loss than either alone — suggesting the next generation of obesity drugs will stack mechanisms, not replace them.
What Amylin Is
Amylin, formally known as islet amyloid polypeptide (IAPP), is a 37-amino-acid peptide hormone produced by pancreatic beta cells. It was identified in 1987 when researchers found that the amyloid deposits present in the pancreatic islets of patients with type 2 diabetes were composed of a previously unknown peptide. The peptide was simultaneously characterized by two groups: Westermark and colleagues in Sweden, who named it islet amyloid polypeptide, and Cooper and colleagues in New Zealand, who named it amylin.
The two names persist in the literature. IAPP emphasizes the peptide's role in islet amyloid formation (relevant to type 2 diabetes pathology), while amylin emphasizes its physiological function as a hormone. Both refer to the same molecule.
Amylin is stored in the same secretory granules as insulin within beta cells and is co-released in response to nutrient stimulation. The molar ratio of insulin to amylin in secretory granules is approximately 100:1. In healthy individuals, circulating amylin levels rise rapidly after meals, peak within 15-30 minutes, and return to baseline within 2-3 hours, mirroring the insulin secretion profile.
In type 1 diabetes, where beta cells are destroyed by autoimmune attack, amylin production drops to near zero alongside insulin. Patients treated with insulin alone are replacing only one of the two hormones their beta cells normally produce, leaving a gap in postprandial glucose regulation that insulin monotherapy cannot fully close. In type 2 diabetes, amylin levels may initially be elevated (reflecting beta cell compensation and hyperinsulinemia) but decline as beta cell function deteriorates over years. The progressive loss of amylin contributes to the postprandial glucose dysregulation characteristic of both diabetes types.
Amylin also has effects outside glucose metabolism. It influences bone remodeling through its shared receptor components with calcitonin, modulates renal function, and crosses the blood-brain barrier to affect central nervous system signaling. These pleiotropic effects are increasingly relevant as amylin analogs are being developed for indications beyond diabetes. For the specific case for amylin replacement in type 1 diabetes, see Why Amylin Replacement Makes Biological Sense for Type 1 Diabetes.
How Amylin Works
Amylin exerts its metabolic effects through three distinct mechanisms, all of which complement insulin's glucose-lowering action.
Slowing Gastric Emptying
Amylin delays the rate at which food leaves the stomach and enters the small intestine. This slowing reduces the rate of glucose appearance in the bloodstream after meals, preventing the sharp postprandial glucose spikes that are difficult for exogenous insulin to match. A 2025 review by Lutz described this as amylin's most pharmacologically reproducible effect.[2] The mechanism involves vagal nerve signaling from the area postrema in the brainstem, where amylin receptors are densely expressed.
Suppressing Glucagon
After meals, glucagon (a hormone that raises blood glucose by stimulating liver glucose output) is normally suppressed. In diabetes, this suppression fails, leading to inappropriate hepatic glucose production even when blood glucose is already elevated. Amylin restores this suppression. It reduces post-meal glucagon secretion from pancreatic alpha cells, an effect that insulin alone does not adequately achieve. The combination of insulin (driving glucose into cells) and amylin (preventing the liver from adding more glucose) provides tighter postprandial control than insulin alone.
Signaling Satiety
Amylin acts on the area postrema and nucleus of the solitary tract in the brainstem to reduce food intake. Hankir et al. (2025) mapped the central nervous system pathways through which amylin regulates energy homeostasis, identifying projections to the lateral parabrachial nucleus and ventral tegmental area as key mediators of amylin's anorexigenic effect.[3] The satiety effect involves the serotonin-histamine-dopaminergic system and is additive with GLP-1-mediated satiety, which acts through partially overlapping but distinct neural circuits.
The Amylin Receptor System
Amylin does not bind to a single, dedicated receptor. Instead, it acts on a family of heterodimeric receptors formed by the calcitonin receptor (CTR) complexed with one of three receptor activity-modifying proteins (RAMP1, RAMP2, or RAMP3). The combinations CTR/RAMP1, CTR/RAMP2, and CTR/RAMP3 form the AMY1, AMY2, and AMY3 receptors, respectively.
Cao et al. (2024) published a cryo-EM structure of the human AMY1 receptor, providing the first high-resolution view of how amylin and related peptides interact with this receptor complex.[4] The structure revealed that RAMP1 creates a binding pocket that distinguishes amylin from calcitonin, explaining the selectivity of amylin analogs for metabolic versus bone-related effects.
Rejili et al. (2025) reviewed amylin receptors as therapeutic targets in obesity, noting that the AMY1 and AMY3 subtypes are the primary mediators of amylin's appetite-suppressing effects in the brain, while AMY2 may play a greater role in glucose homeostasis.[5]
Receptor System
One Receptor, Three Flavors
Amylin binds a modular receptor — the "helper" (RAMP) swapped in determines what it does
AMY1
CTR + RAMP1
Primary Role
Appetite suppression (CNS)
Expressed In
Area postrema, NTS
Primary mediator of amylin's anorexigenic effect; drug target for obesity
AMY2
CTR + RAMP2
Primary Role
Glucose regulation
Expressed In
Pancreas, peripheral
May play a greater role in glucose homeostasis than in appetite
AMY3
CTR + RAMP3
Primary Role
Appetite suppression (CNS)
Expressed In
Brainstem, hypothalamus
Co-mediator of CNS effects with AMY1
The calcitonin receptor (CTR) is the core. Pair it with different receptor activity-modifying proteins (RAMPs), and you get three different receptor identities from a single gene product. This modularity is what allows amylin to do metabolic, neural, and bone-related work from one peptide.
Source: Cao et al. (2024), cryo-EM structure of AMY1; Rejili et al. (2025)
 View as image
View as imagePramlintide: The First Amylin Drug
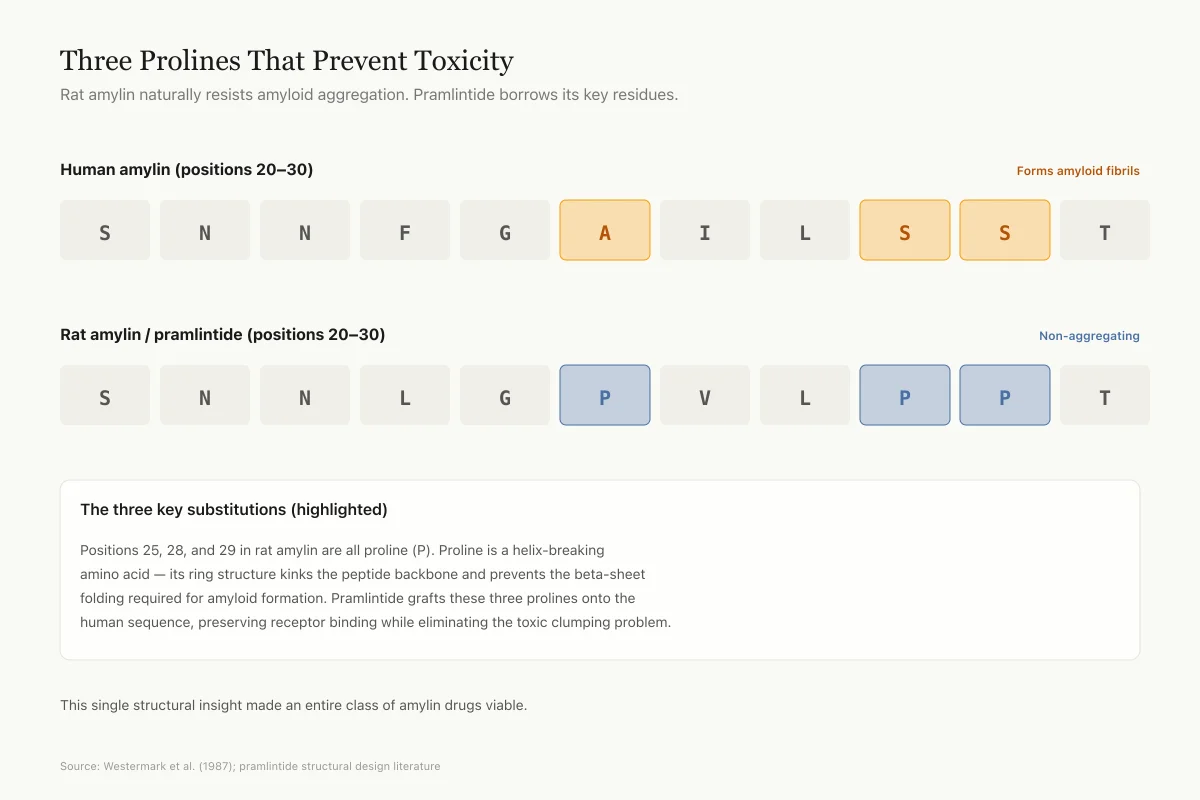
Pramlintide (Symlin) is a synthetic amylin analog that was approved by the FDA in 2005 as an adjunct to insulin therapy for both type 1 and type 2 diabetes. It differs from native human amylin by three proline substitutions at positions 25, 28, and 29, which are borrowed from rat amylin. These substitutions prevent the peptide from forming amyloid fibrils (a problem with native human amylin) while preserving receptor binding and biological activity.
In clinical trials, pramlintide added to insulin therapy reduced HbA1c by 0.3-0.6% and body weight by 1-2 kg compared to insulin alone. The weight loss, though modest, was significant because most insulin regimens cause weight gain. Pramlintide's main side effect is nausea, which typically resolves with dose titration.
Despite these benefits, pramlintide has been commercially underperforming since launch. It requires separate injections from insulin (it cannot be mixed in the same syringe), demands careful dose timing relative to meals, and carries a risk of hypoglycemia when combined with insulin. These practical barriers limited adoption. Erzen et al. (2024) described pramlintide as a "proof of concept" that demonstrated amylin replacement works but highlighted the need for longer-acting, easier-to-use formulations.[6] For a deeper look at pramlintide's role in postprandial glucose management, see Pramlintide for Postprandial Glucose: How It Complements Insulin.
Next-Generation Amylin Analogs
Cagrilintide
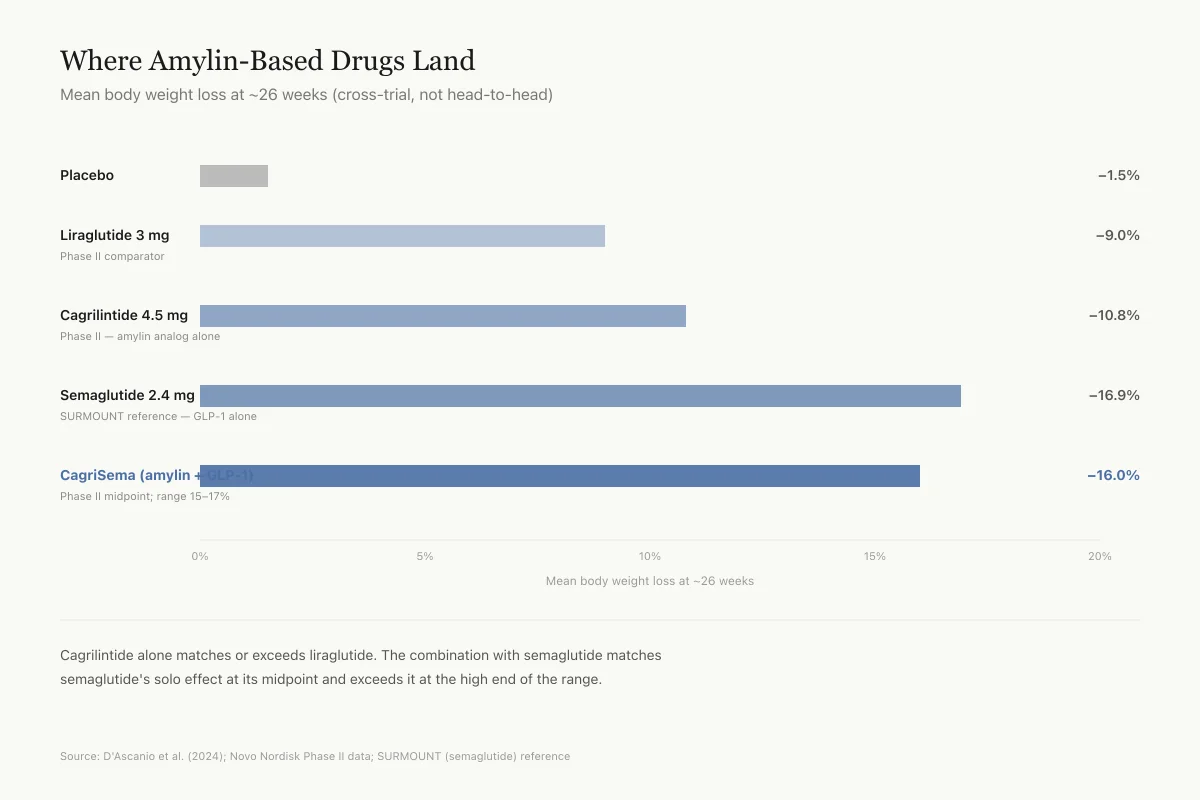
Cagrilintide is a long-acting amylin analog developed by Novo Nordisk. Unlike pramlintide, which requires injection before each meal, cagrilintide is administered once weekly. D'Ascanio et al. (2024) reviewed its Phase II clinical data, which showed 10.8% body weight loss at 26 weeks in patients with overweight or obesity, compared to 9.0% for liraglutide (a GLP-1 receptor agonist) in the same trial.[7]
Cao et al. (2025) published structural and dynamic data on how cagrilintide binds to the calcitonin receptor, showing that its acylated fatty acid side chain (the same albumin-binding technology used in semaglutide) extends its half-life to approximately 160 hours.[8]
Cagrilintide is being developed both as a standalone obesity treatment and in combination with semaglutide (the combination is called CagriSema). Phase III trials for CagriSema are ongoing, and Phase II data showed approximately 15-17% body weight loss, exceeding what either agent achieves alone.
Cross-Trial Weight Loss
Where Amylin-Based Drugs Land
Mean body weight loss at ~26 weeks (cross-trial, not head-to-head)
Phase II comparator
Phase II
SURMOUNT reference
Phase II midpoint (range 15–17%)
Cagrilintide alone matches or exceeds liraglutide. The combination with semaglutide (CagriSema) matches semaglutide's solo effect at its midpoint and exceeds it at the high end. Phase III trials are ongoing and will tell us whether these Phase II numbers hold up.
Source: D'Ascanio et al. (2024); Novo Nordisk Phase II data; SURMOUNT (semaglutide) reference
 View as image
View as imageOther Emerging Analogs
Bailey et al. (2026) reviewed the broader pipeline of long-acting amylin-related peptides for obesity and type 2 diabetes, including dual amylin/calcitonin receptor agonists (DACRAs) that activate both amylin and calcitonin signaling pathways.[9] Zhou et al. (2026) described a stapled dual amylin and calcitonin receptor agonist with enhanced proteolytic stability and prolonged duration of action in animal models.[10]
Chung et al. (2026) published a five-year perspective on amylin's emerging role in obesity treatment, noting that the field has shifted from viewing amylin as a diabetes drug to recognizing it as a primary obesity target, driven by the weight-loss efficacy data from cagrilintide trials.[11]
The GLP-1 and Amylin Combination
The combination of GLP-1 receptor agonism and amylin receptor agonism has emerged as a leading strategy in metabolic pharmacology. Both peptides reduce appetite, but they act through partially distinct neural pathways. GLP-1 primarily signals through the hypothalamus and vagal afferents, while amylin acts primarily through the area postrema. This complementarity produces additive or synergistic weight loss.
Wong et al. (2023) reviewed the clinical rationale for combining GLP-1 receptor agonists with amylin analogs, arguing that the two mechanisms address different components of energy balance: GLP-1 reduces hunger, while amylin enhances meal-specific satiety and slows nutrient absorption.[12]
Lee et al. (2025) reviewed the broader class of amylin peptide receptor activators being developed for obesity pharmacotherapy, concluding that the most promising candidates are those designed for combination with existing GLP-1 drugs.[13]
Walker et al. (2025) described amylin as presenting "emergent therapeutic opportunities" in a landscape where GLP-1 drugs have demonstrated that pharmacological weight loss is achievable and the question has shifted to how much additional efficacy can be gained.[14]
Amylin and the Brain
Amylin's effects extend beyond metabolic regulation into neuroprotection.
Alzheimer's Disease
Corrigan et al. (2022) reviewed amylin's paradoxical relationship with Alzheimer's disease.[15] Native amylin forms amyloid aggregates similar to the amyloid-beta plaques found in Alzheimer's brains, and IAPP amyloid has been detected in the brains of Alzheimer's patients. This cross-seeding between IAPP and amyloid-beta initially suggested that amylin might contribute to neurodegeneration.
The paradox is that amylin receptor activation appears neuroprotective. Corrigan et al. (2023) showed that amylin receptor agonism (not antagonism) reduced amyloid plaque burden, improved synaptic density, and enhanced cognitive performance in mouse models of Alzheimer's disease.[16] The proposed mechanism involves increased clearance of amyloid-beta through enhanced phagocytic activity of microglia. Pramlintide and other non-aggregating amylin analogs can activate the receptor without forming toxic amyloid, separating the beneficial signaling effect from the harmful aggregation effect.
Migraine
Moreno-Ajona et al. (2025) explored amylin and the amylin receptors as potential contributors to migraine pathophysiology, noting that amylin shares receptor components (CTR and RAMPs) with calcitonin gene-related peptide (CGRP), the peptide targeted by the newest class of migraine drugs.[17] Whether amylin receptor activation is pro-migraine or anti-migraine remains under investigation, but the receptor overlap with CGRP suggests amylin may play a role in headache biology.
Moghazy et al. (2024) compared liraglutide (GLP-1 agonist) and pramlintide (amylin analog) for neuroprotective effects, finding that both protected against cognitive decline in animal models through different but complementary mechanisms.[18]
The Amyloid Problem
Amylin has a dark side. In its native human form, the peptide is amyloidogenic: it aggregates into insoluble fibrils that deposit in and around pancreatic islets. These amyloid deposits are found in over 90% of type 2 diabetes patients at autopsy and contribute to beta cell death. The aggregation process involves misfolded amylin monomers assembling into oligomers (small aggregates), protofibrils, and mature fibrils. The oligomeric intermediates are believed to be the most toxic species, disrupting beta cell membranes and triggering apoptosis.
Rat amylin, by contrast, does not form amyloid. The three proline residues at positions 25, 28, and 29 in rat amylin prevent the peptide from adopting the beta-sheet conformation required for fibril formation. Proline is a helix-breaking amino acid that disrupts the regular secondary structure needed for amyloid stacking. This is why pramlintide incorporates rat prolines: it retains human amylin's receptor activity while eliminating its tendency to aggregate.
The Pramlintide Trick
Three Prolines That Prevent Toxicity
Rat amylin naturally resists amyloid aggregation. Pramlintide borrows its key residues.
The three key substitutions (highlighted)
Positions 25, 28, and 29 in rat amylin are all proline. Proline is a helix-breaking amino acid — its ring structure kinks the peptide backbone and prevents the beta-sheet folding required for amyloid formation. Pramlintide grafts these three prolines onto the human sequence, preserving receptor binding while eliminating the toxic clumping problem.
Source: Westermark et al. (1987) IAPP characterization; pramlintide structural design literature
 View as image
View as imageThe relationship between amylin aggregation and type 2 diabetes remains debated. It is unclear whether islet amyloid is a cause of beta cell death (toxic gain of function) or a consequence of beta cell dysfunction (impaired processing and clearance of excess amylin during hyperinsulinemic states). Both may be true at different disease stages. What is established is that the oligomeric intermediates, not the mature fibrils, are the primary toxic species. These soluble oligomers insert into beta cell membranes, form ion-permeable channels, and trigger calcium influx that leads to apoptosis.
Panou et al. (2024) reviewed the pipeline of non-aggregating amylin analogs for obesity treatment in patients without diabetes, noting that solving the aggregation problem was the critical step that made amylin-based drug development viable.[19] Modern drug design ensures that all clinical-stage amylin analogs incorporate structural modifications that eliminate fibril formation while preserving or enhancing receptor binding affinity.
Where Amylin Research Is Headed
Amylin has transitioned from a niche diabetes adjunct to a central player in obesity pharmacology. The key developments driving this shift are cagrilintide's Phase III program, the CagriSema combination trials, and the growing recognition that amylin's appetite-suppressing mechanism is distinct from and additive to GLP-1-based approaches.
The Alzheimer's connection adds a dimension that few metabolic peptides possess: the possibility that amylin receptor agonists could simultaneously address metabolic and neurodegenerative disease, conditions that share risk factors and often co-occur in aging populations.
The limitations are real. Pramlintide's commercial underperformance demonstrated that a biologically sound mechanism does not guarantee clinical adoption if the delivery is inconvenient. Cagrilintide's weekly dosing addresses this problem but has not yet demonstrated long-term safety and efficacy in Phase III data. The amyloid toxicity of native amylin means that any new analog must be rigorously tested for aggregation potential. Long-term safety monitoring for cardiovascular events, pancreatitis, and thyroid effects (concerns shared across the incretin class) will be required for regulatory approval.
The combination approach also raises questions about tolerability. Both GLP-1 agonists and amylin analogs cause nausea through partially overlapping brainstem mechanisms. Whether CagriSema's gastrointestinal side effects will be additive, and whether patients can tolerate the combined effect during dose escalation, remains to be answered by Phase III data.
There is also the question of what happens when patients stop treatment. Weight regain after discontinuation of GLP-1 agonists is well-documented. Whether amylin analogs, alone or in combination, produce more durable weight loss through sustained changes in appetite regulation is unknown. Aranas et al. (2025) reported that combined amylin and GLP-1 receptor agonism produced synergistic-like decreases in alcohol intake in animal models, suggesting that amylin's CNS effects may extend beyond satiety into broader behavioral modulation.
What amylin is no longer is overlooked. The convergence of obesity pharmacology, incretin biology, and neurodegeneration research has made amylin one of the most actively studied peptide hormones in current clinical development.
The Bottom Line
Amylin is a 37-amino-acid peptide co-secreted with insulin that slows gastric emptying, suppresses glucagon, and signals satiety. Pramlintide (approved 2005) proved the concept but saw limited adoption due to inconvenient dosing. Cagrilintide, a once-weekly amylin analog, showed 10.8% weight loss in Phase II trials and is in Phase III for obesity, with the combination CagriSema producing 15-17% weight loss. Amylin receptor agonists also show neuroprotective effects in Alzheimer's models, making amylin one of the most versatile peptide targets in current drug development.
Sources & References
- 1RPEP-13944·Volčanšek, Špela et al. (2025). “Amylin: From Mode of Action to Future Clinical Potential in Diabetes and Obesity..” Diabetes therapy : research.Study breakdown →PubMed →↩
- 2RPEP-12344·Lutz, Thomas A (2025). “How the Pancreatic Peptide Amylin Controls Your Meal Size and Food Preferences.” Neuropharmacology.Study breakdown →PubMed →↩
- 3RPEP-11279·Hankir, Mohammed K et al. (2025). “Central nervous system pathways targeted by amylin in the regulation of food intake..” Biochimie.Study breakdown →PubMed →↩
- 4RPEP-07928·Cao, Jianjun et al. (2024). “Cryo-EM Structure of the Human Amylin 1 Receptor in Complex with CGRP and Gs Protein..” Biochemistry.Study breakdown →PubMed →↩
- 5RPEP-13237·Rejili, Mokhtar et al. (2025). “Amylin receptors as therapeutic targets in obesity: Emerging peptide-based strategies..” Vascular pharmacology.Study breakdown →PubMed →↩
- 6RPEP-08165·Eržen, Stjepan et al. (2024). “Amylin, Another Important Neuroendocrine Hormone for the Treatment of Diabesity..” International journal of molecular sciences.Study breakdown →PubMed →↩
- 7RPEP-08044·D'Ascanio, Antonella M et al. (2024). “Cagrilintide: How an Amylin Analog Combined with Semaglutide Targets Obesity Through Dual Brain Pathways.” Cardiology in review.Study breakdown →PubMed →↩
- 8RPEP-10291·Cao, Jianjun et al. (2025). “Structural and dynamic features of cagrilintide binding to calcitonin and amylin receptors..” Nature communications.Study breakdown →PubMed →↩
- 9RPEP-14819·Bailey, Clifford J et al. (2026). “Next-Generation Long-Acting Amylin Drugs for Obesity and Type 2 Diabetes.” Peptides.Study breakdown →PubMed →↩
- 10RPEP-16611·Zhou, Yaqi et al. (2026). “New Long-Acting Peptide Combined With GLP-1 Drugs Achieves 41% Weight Loss in Obese Rats.” Bioconjugate chemistry.Study breakdown →PubMed →↩
- 11RPEP-15043·Chung, Chae Won et al. (2026). “Amylin: The Pancreatic Hormone Emerging as a Powerful Obesity and Diabetes Treatment.” Journal of obesity & metabolic syndrome.Study breakdown →PubMed →↩
- 12RPEP-07552·Wong, Gunther et al. (2023). “Combining GLP-1 Drugs with Pramlintide for Weight Loss in Type 1 Diabetes: Three Patient Cases.” JCEM case reports.Study breakdown →PubMed →↩
- 13RPEP-12024·Lee, Sangmin (2025). “Amylin: The Next Peptide Hormone Target for Obesity Drugs After GLP-1.” Current drug targets.Study breakdown →PubMed →↩
- 14RPEP-13964·Walker, Christopher S et al. (2025). “Amylin: emergent therapeutic opportunities in overweight, obesity and diabetes mellitus..” Nature reviews. Endocrinology.Study breakdown →PubMed →↩
- 15RPEP-06063·Corrigan, Rachel R et al. (2022). “Amylin's Paradox: The Diabetes Peptide That Both Harms and Protects the Alzheimer's Brain.” Current neuropharmacology.Study breakdown →PubMed →↩
- 16RPEP-06812·Corrigan, Rachel R et al. (2023). “Amylin Peptide Analog Pramlintide Improves Cognition in Alzheimer's Mice Through Peripheral Mechanisms.” Journal of Alzheimer's disease : JAD.Study breakdown →PubMed →↩
- 17RPEP-12633·Moreno-Ajona, David et al. (2025). “Amylin Peptide in Migraine: A New Therapeutic Target Beyond CGRP.” Cephalalgia : an international journal of headache.Study breakdown →PubMed →↩
- 18RPEP-08884·Moghazy, Hoda M et al. (2024). “Liraglutide versus pramlintide in protecting against cognitive function impairment through affecting PI3K/AKT/GSK-3β/TTBK1 pathway and decreasing Tau hyperphosphorylation in high-fat diet- streptozocin rat model..” Pflugers Archiv : European journal of physiology.Study breakdown →PubMed →↩
- 19RPEP-09024·Panou, Theodoros et al. (2024). “Amylin analogs for the treatment of obesity without diabetes: present and future..” Expert review of clinical pharmacology.Study breakdown →PubMed →↩