Peptides for Hair Loss: What the Research Shows
Peptides for Hair Loss
4,000+ genes
The tripeptide GHK-Cu modulates the expression of over 4,000 human genes, including pathways involved in hair follicle cycling, collagen synthesis, and growth factor signaling.
Pickart et al., BioMed Research International, 2015
Pickart et al., BioMed Research International, 2015
If you only read one thing
Copper peptide (GHK-Cu) has the most research behind it — it modulates over 4,000 genes and enlarges hair follicles in lab studies. Thymosin beta-4 activates hair follicle stem cells in animal models. VIP protects against autoimmune hair loss. But here's the honest answer: most peptide hair loss research is done in petri dishes and mice, not on human scalps. No peptide has been proven in large clinical trials to work as well as minoxidil or finasteride. The biology is real and promising, but the evidence gap between lab and clinic is still wide.
Peptides for hair loss have gained widespread consumer attention, driven largely by marketing claims about copper peptides and growth factors. The reality is more nuanced than the advertising suggests. Several peptides have legitimate research behind them, including GHK-Cu (copper tripeptide), thymosin beta-4, biomimetic growth factor peptides, and Wnt pathway activators. Some have demonstrated effects on hair follicle cells in vitro and in animal models. Human clinical trial evidence remains thin for most peptide-based hair treatments, with the notable exception of GHK-Cu in topical formulations. This article surveys what the research actually shows for each peptide class, separating verified science from speculation. For deeper dives into individual topics, see our articles on Copper Peptides for Scalp Health: GHK-Cu and Hair Growth, Thymosin Beta-4 and Hair Follicle Regeneration, Biomimetic Peptides for Hair Restoration, and Wnt Signaling Peptides.
Key Takeaways
- The marketing is ahead of the science — no peptide has beaten minoxidil or finasteride in a real trial.
- Copper peptide turns on more than 4,000 genes, including ones tied to hair follicle cycling.
- Most peptide hair research is done on goats, mice, and cell cultures — not human scalps.
- Thymosin beta-4 works a different angle — it tells hair stem cells to move to where rebuilding is needed.
- Stress really does make hair fall out — a neuropeptide called substance P is the molecular middleman.
- Peptides are big molecules, so most of what you apply to the scalp never reaches the follicle.
- Your natural copper peptide levels drop over 50% between age 20 and 60 — tracking your follicle decline.
GHK-Cu: The Copper Tripeptide
GHK-Cu (glycyl-L-histidyl-L-lysine copper complex) is a naturally occurring tripeptide found in human plasma, saliva, and urine. Plasma levels decline from approximately 200 ng/mL at age 20 to 80 ng/mL by age 60. Loren Pickart first identified GHK in the 1970s while studying why liver tissue from young donors could stimulate old fibroblasts to behave like young ones. The active factor turned out to be a three-amino-acid peptide with strong affinity for copper ions.[1]
The peptide's relevance to hair growth stems from its broad biological activity. GHK-Cu stimulates collagen synthesis in fibroblasts, promotes angiogenesis, increases glycosaminoglycan production, and modulates the activity of matrix metalloproteinases that remodel extracellular matrix. These are all processes required for hair follicle cycling.
Pickart et al. (2015) used gene expression profiling to demonstrate that GHK modulates over 4,000 human genes at a concentration of 1 micromolar. Among the affected pathways were those governing Wnt signaling, TGF-beta regulation, notch signaling, and multiple growth factor systems directly involved in hair follicle biology.[3] Earlier work by Pickart and colleagues (2012) connected GHK-Cu to protection against oxidative stress and degenerative aging processes, both of which contribute to follicle miniaturization in androgenetic alopecia.[2]
The most relevant finding for hair loss is that GHK-Cu enlarges hair follicles. An in vitro study published in 2007 (Pyo et al.) showed that the tripeptide-copper complex increased hair follicle size and stimulated hair growth in organ culture. GHK-Cu also extended the anagen (growth) phase duration, meaning follicles remained in active growth longer before transitioning to catagen (regression). Combined with its angiogenic properties, which improve blood supply to the dermal papilla, GHK-Cu acts on multiple mechanisms relevant to hair health.
The evidence has limitations. The gene expression data, while impressive in scope, is derived from cell culture models. Whether topically applied GHK-Cu reaches the dermal papilla in sufficient concentrations to reproduce these gene expression changes in vivo is not firmly established. Delivery vehicle technology matters: a 2023 study developed ionic liquid-based microemulsions that improved GHK-Cu skin penetration approximately three-fold, and nanoliposomal co-delivery systems have shown enhanced efficacy.
Dou et al. (2020) reviewed GHK's potential as an anti-aging peptide, noting its effects on collagen remodeling, antioxidant defense, and stem cell activity, all relevant to the aging hair follicle.[4]
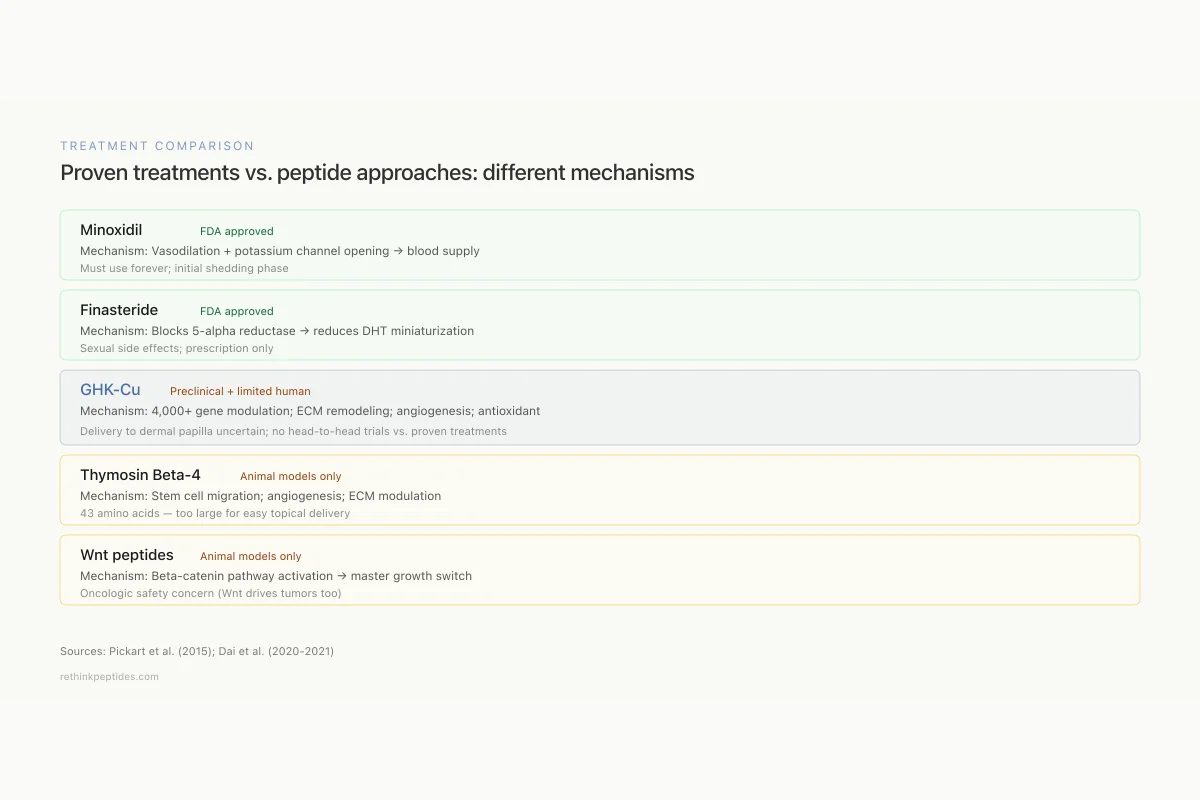
Treatment Comparison
Proven treatments vs. peptide approaches: different mechanisms, different targets
Minoxidil
FDA approvedMechanism: Vasodilation + potassium channel opening
Target: Blood supply to follicle
Must use forever; initial shedding phase
Finasteride
FDA approvedMechanism: Blocks 5-alpha reductase → reduces DHT
Target: Hormonal miniaturization
Sexual side effects; prescription only
GHK-Cu
Preclinical + limited humanMechanism: 4,000+ gene modulation; ECM remodeling; angiogenesis; antioxidant
Target: Follicle microenvironment (broad)
Delivery to dermal papilla uncertain
Thymosin Beta-4
Animal models onlyMechanism: Stem cell migration; angiogenesis; ECM modulation
Target: Follicle stem cell activation
43 amino acids — too large for easy topical delivery
Wnt peptides
Animal models onlyMechanism: Beta-catenin pathway activation → anagen entry
Target: Master growth switch
Oncologic safety concern (Wnt drives tumors too)
Sources: Pickart et al. (2015); Dai et al. (2020-2021)
 View as image
View as imageCompared to established hair loss treatments, GHK-Cu has a different mechanism profile. Minoxidil primarily works through vasodilation and potassium channel opening. Finasteride blocks 5-alpha reductase to reduce DHT. GHK-Cu addresses the follicle microenvironment more broadly: extracellular matrix quality, growth factor signaling, antioxidant defense, and vascular supply simultaneously. Whether this broader mechanism translates to equivalent or superior clinical results compared to proven treatments has not been established in head-to-head trials. The peptide's safety profile appears favorable based on its natural occurrence in human tissue, with concentrations declining naturally with age, which has led some researchers to frame hair loss partly as a consequence of declining endogenous GHK-Cu levels.
For detailed coverage of copper peptide mechanisms and evidence, see Copper Peptides for Scalp Health: GHK-Cu and Hair Growth and GHK-Cu: The Copper Peptide That Modulates Over 4,000 Genes.
Thymosin Beta-4: Hair Follicle Stem Cell Activation
Thymosin beta-4 (TB-4) is a 43-amino-acid peptide that plays central roles in cell migration, angiogenesis, and wound healing. Its connection to hair growth was first established when researchers observed that TB-4 promotes the migration of stem cells and progenitor cells to the base of the hair follicle, where they contribute to follicle regeneration.
Dai et al. (2020) identified thymosin beta-4 through transcriptomic analysis as a key gene differentially expressed during the anagen-to-telogen transition in secondary hair follicles. The study found that TB-4 promoted proliferation of secondary hair follicle dermal papilla cells in Albas cashmere goats, establishing it as an active regulator of follicle cycling rather than a passive bystander.[5]
A 2021 review by Dai et al. mapped the multiple potential roles of thymosin beta-4 in hair follicle growth and development. TB-4 promotes hair follicle stem cell migration to the bulge region, enhances angiogenesis around the dermal papilla, and modulates extracellular matrix composition in the follicle microenvironment. These actions collectively support the transition from telogen (resting) to anagen (growth phase).[6]
Padmanabhan et al. (2020) demonstrated that thymosin beta-4 is essential for adherens junction stability and epidermal planar cell polarity. Loss of TB-4 disrupted the structural organization of epidermal cells, suggesting that the peptide maintains the tissue architecture required for normal hair follicle function.[7]
The limitation of the TB-4 hair growth evidence is that it comes almost entirely from animal models and in vitro studies. Cashmere goat hair follicles, while biologically informative, differ from human scalp follicles in cycling duration and regulation. Human scalp follicles cycle over years while cashmere goat follicles cycle seasonally, meaning the molecular triggers for anagen entry may not be identical. The peptide's 43-amino-acid size also creates delivery challenges for topical application; larger peptides penetrate the stratum corneum poorly without specialized delivery vehicles.
TB-500, a synthetic fragment of thymosin beta-4, is available in the gray-market peptide space but has not been tested in controlled clinical trials for hair loss. The fragment retains the actin-sequestering and cell-migration-promoting activity of the full-length peptide, but whether it recapitulates the specific hair follicle effects observed with intact TB-4 is unverified. Some individuals self-administer TB-500 subcutaneously based on anecdotal reports of improved hair quality, but no controlled data support this application.
The mechanistic case for TB-4 in hair biology is strong: stem cell migration, angiogenesis, and ECM remodeling are all validated requirements for follicle regeneration. Translating this mechanism into a viable hair therapy requires solving the delivery problem and demonstrating efficacy in human follicles specifically. For the full animal evidence, see Thymosin Beta-4 and Hair Follicle Regeneration: Animal Model Evidence.
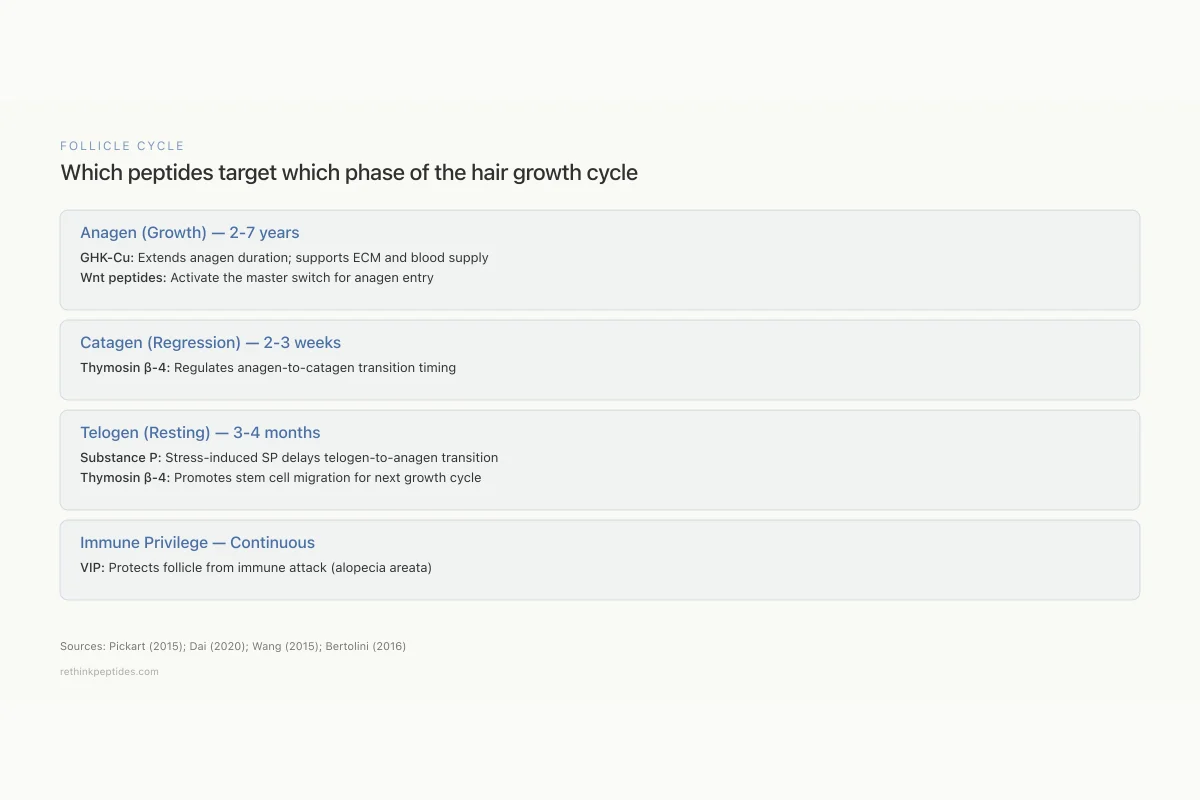
Follicle Cycle
Which peptides target which phase of the hair growth cycle
Anagen (Growth)
2-7 yearsCatagen (Regression)
2-3 weeksTelogen (Resting)
3-4 monthsImmune Privilege
ContinuousSources: Pickart (2015); Dai (2020); Wang (2015); Bertolini (2016)
 View as image
View as imageNeuropeptides and Hair: Substance P and VIP
Hair follicles are richly innervated, and neuropeptides released by sensory nerve endings directly influence follicle cycling, immune status, and stem cell activity. Two neuropeptides, substance P and vasoactive intestinal peptide (VIP), have particularly well-documented effects on hair biology.
Wang et al. (2015) demonstrated that substance P mediates psychological stress-induced hair growth delay in mice. Stress triggers oxidative stress and substance P release, which activates autophagy in hair follicle cells and delays the transition from telogen to anagen. This study provided a molecular mechanism for the long-observed clinical link between stress and hair loss.[8]
Substance P is a double-edged molecule in hair biology. While chronic stress-related substance P release inhibits growth, controlled substance P signaling can stimulate keratinocyte proliferation and promote anagen entry. The key appears to be dose and duration: acute neuropeptide signaling supports follicle function, while chronic activation drives pathological responses. This context-dependent effect is consistent with substance P's role in other tissues, where it functions as both a growth promoter (wound healing) and an inflammation driver (neurogenic inflammation) depending on the signaling environment.
The clinical relevance of the stress-hair loss connection is substantial. Telogen effluvium, the most common form of diffuse hair shedding, frequently follows periods of psychological or physiological stress. If substance P-mediated autophagy is the molecular mediator of this response, then blocking substance P signaling at the follicle could theoretically prevent stress-induced hair loss. NK1 receptor antagonists (already approved for chemotherapy nausea) have not been evaluated for this dermatological application, but the mechanistic rationale exists.
Bertolini et al. (2016) made a significant finding regarding VIP and alopecia areata. The study demonstrated that VIP provides protection against hair follicle immune privilege collapse, the process by which the immune system begins attacking the follicle in alopecia areata. VIP receptor-mediated signaling appeared to be defective in alopecia areata patients, suggesting that a neuropeptide deficit contributes to the autoimmune hair loss process.[9]
This finding has implications beyond basic science. If VIP signaling deficiency contributes to alopecia areata, then restoring VIP activity at the follicle level could represent a targeted therapeutic approach. VIP analogs and VIP receptor agonists exist but have not been specifically developed for dermatological applications.
Doche et al. (2020) extended the neuropeptide-hair connection to lichen planopilaris and frontal fibrosing alopecia, two scarring alopecia conditions. The study found evidence for neurogenic inflammation mediated by neuropeptide signaling in these conditions, with substance P and calcitonin gene-related peptide (CGRP) playing pathogenic roles.[10]
Wnt Signaling Peptides
The Wnt/beta-catenin signaling pathway is a master regulator of hair follicle development, cycling, and regeneration. Active Wnt signaling drives the transition from telogen to anagen, maintains dermal papilla cell identity, and supports hair follicle neogenesis (the formation of new follicles). Pathway inhibition causes hair follicle regression.
Several peptides have been designed to activate Wnt signaling specifically in hair follicle cells. PTD-DBM is a cell-penetrating peptide that disrupts the interaction between CXXC5 (a negative Wnt regulator) and Dishevelled, thereby removing the brake on Wnt pathway activation. In mouse models of androgenetic alopecia, topical PTD-DBM application enhanced hair regrowth.
An octapeptide that binds Lgr5 (leucine-rich repeat-containing G-protein coupled receptor 5, a Wnt target gene and stem cell marker) increased Wnt/beta-catenin signaling and promoted proliferation and differentiation of human primary hair cells, including dermal papilla cells, outer root sheath cells, germinal matrix cells, and hair follicle stem cells in a 2024 study by Lee et al.
AIMP1-derived peptide fragments, secreted by Wnt-treated hair follicle stem cells, activate dermal papilla cells through the MAPK pathway, leading to beta-catenin accumulation. This peptide promoted hair shaft elongation in cultured human hair follicles in a 2024 study, demonstrating that the hair follicle itself produces peptide signals that regulate its own growth cycle.
These Wnt-activating peptides are in early research stages. The challenge is specificity: Wnt signaling drives growth in many tissues, including tumors. Any hair loss therapy based on Wnt activation must demonstrate follicle-selective effects without systemic oncologic risk. Topical delivery helps limit systemic exposure, but penetration depth must be sufficient to reach the dermal papilla, which sits at the base of the follicle, several millimeters below the skin surface.
The biological rationale for Wnt-based approaches is among the strongest of any peptide hair therapy. Genetic studies have established that Wnt pathway mutations cause hairless phenotypes, and Wnt activation is both necessary and sufficient for hair follicle neogenesis in mouse models. The translation challenge is achieving targeted pathway activation in the follicle without off-target effects in surrounding tissue. This may ultimately require peptide-nanoparticle conjugates or microneedle delivery systems that deposit the peptide at the correct tissue depth. For the detailed science, see Wnt Signaling Peptides: Targeting the Hair Growth Pathway.
Biomimetic Peptides in Hair Products
The consumer hair care market has adopted biomimetic peptides, synthetic peptides that mimic the activity of natural growth factors. Octapeptide-2, a mimetic of thymosin beta-4, is marketed for hair growth based on its ability to promote stem cell migration. Decapeptide-10 and other proprietary sequences claim to activate dermal papilla cells and extend the anagen phase.
A randomized controlled trial evaluated a PRP-like cosmetic formulation containing biomimetic peptides for alopecia areata. The peptide-based treatment showed comparable efficacy to actual platelet-rich plasma in some measures, suggesting that synthetic peptide cocktails can reproduce at least some of the biological effects of growth factor-rich plasma.
The evidence quality for biomimetic hair peptides varies enormously. Some products cite the underlying biology of their target peptide (e.g., thymosin beta-4 stem cell migration) without demonstrating that the synthetic mimetic reproduces these effects at the concentrations present in the consumer product. Others have small clinical studies showing measurable hair count increases, though publication bias favors positive results and many studies are industry-funded.
The distinction between pharmaceutical-grade peptide research and consumer hair care marketing matters. The peptides discussed earlier in this article (GHK-Cu, thymosin beta-4, VIP, Wnt activators) have published research in peer-reviewed journals with defined mechanisms. Many consumer biomimetic peptide products reference these mechanisms without equivalent evidence for their specific formulation.
Concentration is a critical variable that marketing typically omits. GHK-Cu modulates over 4,000 genes at 1 micromolar concentration in cell culture.[3] Whether a topical serum delivers that concentration to the dermal papilla through the stratum corneum, epidermis, and dermis is a separate question from whether the peptide is present in the product. Peptide stability in commercial formulations is another concern: GHK-Cu requires copper chelation to maintain bioactivity, and formulation pH, temperature stability, and interactions with other ingredients all affect whether the peptide retains function by the time it reaches the consumer's scalp.
A 2022 nanoliposomal co-delivery study demonstrated that encapsulating bioactive peptides in lipid carriers significantly improved their efficacy for hair growth promotion, suggesting that delivery technology may be as important as the peptide itself. Products that combine the right peptide with an effective delivery system may perform better than those that list peptides as ingredients without addressing how they reach the target. For the full landscape, see Biomimetic Peptides for Hair Restoration: The New Frontier.
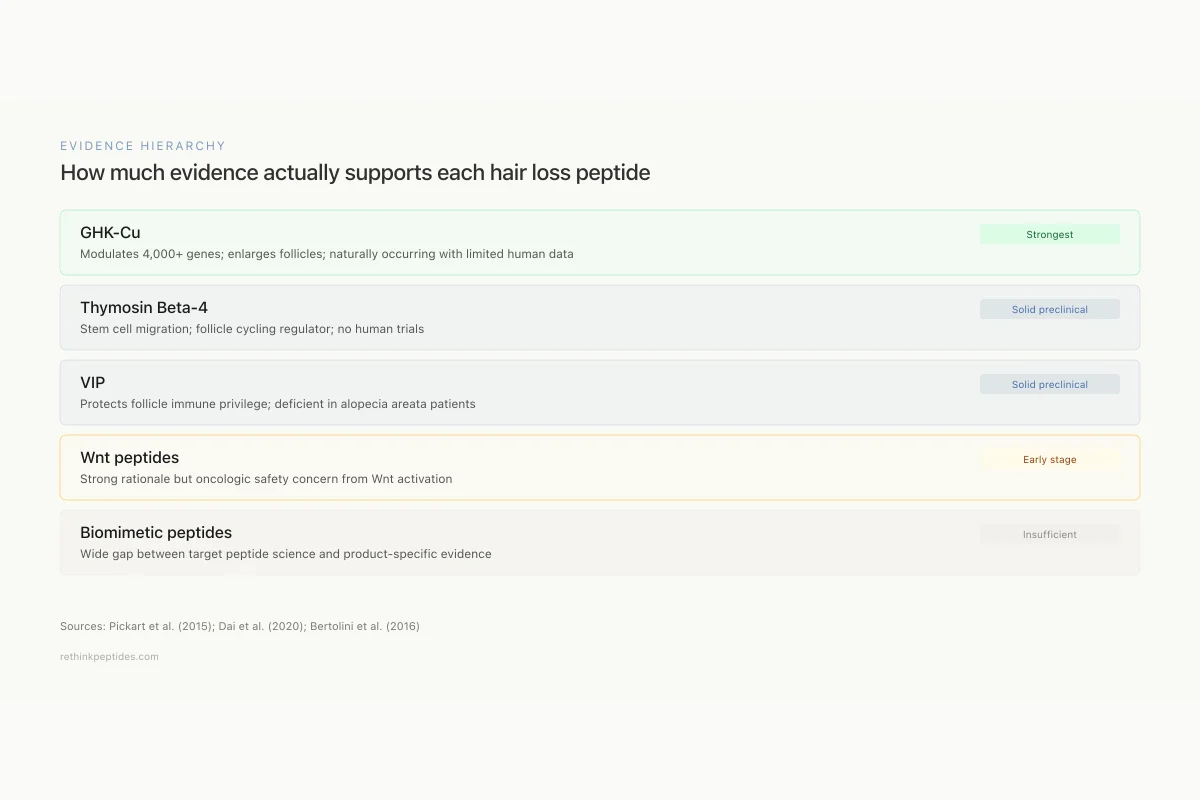
Evidence Hierarchy
How much evidence actually supports each hair loss peptide
GHK-Cu
StrongestModulates 4,000+ genes; enlarges follicles; naturally occurring
Thymosin Beta-4
Solid preclinicalStem cell migration; follicle cycling regulator; no human trials
VIP
Solid preclinicalProtects follicle immune privilege; deficient in AA patients
Wnt peptides
Early stageStrong rationale but oncologic safety concern from Wnt activation
Biomimetic peptides
InsufficientWide gap between target peptide science and product-specific evidence
Sources: Pickart et al. (2015); Dai et al. (2020); Bertolini et al. (2016)
 View as image
View as imageWhat the Evidence Actually Supports
Sorting through the research across all peptide classes reveals a clear evidence hierarchy for peptides for hair loss:
Strongest evidence: GHK-Cu has the broadest evidence base, with in vitro hair follicle enlargement, gene expression profiling showing modulation of relevant pathways, and some human data for topical formulations. The peptide is naturally occurring, has a well-characterized safety profile, and addresses multiple mechanisms (angiogenesis, growth factor signaling, ECM remodeling, antioxidant defense) simultaneously.[3]
Solid preclinical evidence: Thymosin beta-4 has clear mechanistic data for follicle stem cell activation and cycling regulation, supported by transcriptomic analysis and functional studies. The evidence is from animal models with no controlled human hair loss trials.[6] VIP has compelling data in alopecia areata specifically, with a defined mechanism (immune privilege protection) and evidence of defective VIP signaling in affected patients.[9]
Early-stage but promising: Wnt-activating peptides show strong biological rationale and positive results in animal models. The safety question regarding oncologic risk from Wnt activation remains open. AIMP1-derived peptides and Lgr5-binding octapeptides have recent in vitro data.
Insufficient evidence to evaluate: Most consumer biomimetic hair peptides lack peer-reviewed evidence specific to their formulations. The gap between the science of the target peptide and the efficacy of the commercial product is often wide.
The overall landscape is one of genuine biological promise with limited clinical validation. The hair follicle is a peptide-responsive organ: it expresses receptors for GHK-Cu, thymosin beta-4, VIP, substance P, and Wnt ligands. The question is whether topical delivery of these peptides achieves sufficient concentrations at the follicle level to produce clinically meaningful hair growth in humans. For most peptides in this space, that question remains unanswered.
The field would benefit from standardized clinical trial methodologies. Hair growth studies require 6-12 month treatment periods with phototrichogram measurements, global photography assessment by blinded evaluators, and quantitative hair count measurements. Most peptide hair studies use shorter timeframes and less rigorous outcome measures, making it difficult to compare results across compounds or against established treatments. Until peptide hair therapies are evaluated using the same rigor applied to minoxidil and finasteride trials, their place in the treatment hierarchy will remain uncertain.
The future of peptide-based hair treatments likely lies in combination approaches. GHK-Cu addressing the follicle microenvironment, Wnt-activating peptides driving stem cell activation, and VIP analogs maintaining immune privilege could theoretically work synergistically. Such combination strategies are common in dermatology (minoxidil plus finasteride, for example) but have not been explored for peptide-based hair therapies. The biological rationale for this approach is strong, and the key bottleneck remains rigorous clinical validation. For broader context on how peptides interact with skin biology, see Copper Peptides in Skincare: The Science Behind the Buzz.
The Bottom Line
Multiple peptides demonstrate biological activity relevant to hair growth, but the evidence base varies dramatically between compounds. GHK-Cu has the broadest data, spanning gene expression profiling, in vitro follicle enlargement, and limited human studies. Thymosin beta-4 has clear mechanistic support from animal models for follicle stem cell activation. VIP has specific relevance to alopecia areata through its role in maintaining follicle immune privilege. Wnt-activating peptides show promise in early research. For most peptide-based hair treatments, the gap between laboratory findings and validated clinical efficacy in humans remains the defining limitation.
Sources & References
- 1RPEP-01402·Pickart, Loren (2008). “GHK-Cu: The Complete Review of This Tissue Remodeling Tripeptide's Biology and Applications.” Journal of biomaterials science. Polymer edition.Study breakdown →PubMed →↩
- 2RPEP-02037·Pickart, Loren et al. (2012). “The human tripeptide GHK-Cu in prevention of oxidative stress and degenerative conditions of aging: implications for cognitive health..” Oxidative medicine and cellular longevity.Study breakdown →PubMed →↩
- 3RPEP-02768·Pickart, Loren et al. (2015). “GHK Peptide as a Natural Modulator of Multiple Cellular Pathways in Skin Regeneration..” BioMed research international.Study breakdown →PubMed →↩
- 4RPEP-04782·Dou, Yan et al. (2020). “The potential of GHK as an anti-aging peptide..” Aging pathobiology and therapeutics.Study breakdown →PubMed →↩
- 5RPEP-04748·Dai, Bai et al. (2020). “Thymosin Beta-4 Peptide Promotes Hair Follicle Cell Growth in Cashmere Goats.” International journal of molecular sciences.Study breakdown →PubMed →↩
- 6RPEP-05332·Dai, Bai et al. (2021). “Thymosin Beta-4 Promotes Hair Growth by Activating Hair Follicle Stem Cells.” Journal of cellular and molecular medicine.Study breakdown →PubMed →↩
- 7RPEP-05045·Padmanabhan, Krishnanand et al. (2020). “Thymosin Beta-4 Is Essential for Skin Cell Organization and Wound Closure.” Development (Cambridge.Study breakdown →PubMed →↩
- 8RPEP-02825·Wang, Lei et al. (2015). “How Stress Hormones and Oxidative Damage Stall Hair Growth Through Substance P.” Archives of dermatological research.Study breakdown →PubMed →↩
- 9RPEP-02874·Bertolini, M et al. (2016). “The Neuropeptide VIP Protects Hair Follicles From Immune Attack in Alopecia Areata.” The British journal of dermatology.Study breakdown →PubMed →↩
- 10RPEP-04775·Doche, Isabella et al. (2020). “Neuropeptides Substance P and CGRP Show Opposite Patterns in Two Types of Scarring Hair Loss.” Experimental dermatology.Study breakdown →PubMed →↩