Orforglipron: The Oral GLP-1 That Changes Everything
Next-Generation Obesity Peptides
11.2% weight loss
In the ATTAIN-1 Phase 3 trial, orforglipron 36 mg produced 11.2% mean weight loss at 72 weeks in adults with obesity, approaching injectable GLP-1 efficacy in a once-daily pill.
Wharton et al., NEJM, 2025
Wharton et al., NEJM, 2025
If you only read one thing
Every weight loss drug in the GLP-1 class — Ozempic, Wegovy, Mounjaro — is a peptide you inject. Orforglipron is the first non-peptide version: a simple daily pill, no injection, no fasting, taken with or without food. In a trial of over 3,000 people, it produced 11.2% weight loss. That's less than the best injectables, but it's a pill that's cheaper to make, doesn't need refrigeration, and could reach millions of people who can't access injections.
Every GLP-1 receptor agonist approved for obesity or diabetes is a peptide that must be injected. Semaglutide (Ozempic, Wegovy) is injected weekly. Tirzepatide (Mounjaro, Zepbound) is injected weekly. Liraglutide (Saxenda) is injected daily. Even oral semaglutide (Rybelsus) is a peptide that requires fasting 30 minutes before and after dosing to survive the gastrointestinal tract. Orforglipron is none of these things. It is a non-peptide small molecule that activates the GLP-1 receptor from an entirely different binding site, can be taken as a daily pill with or without food, and produced 11.2% mean weight loss in its Phase 3 obesity trial.[1]
Developed by Eli Lilly under the designation LY3502970, orforglipron represents the first non-peptide oral GLP-1 agonist to complete Phase 3 trials with positive results in both obesity (ATTAIN-1) and type 2 diabetes (ATTAIN-2). If approved, it would fundamentally alter the economics and accessibility of GLP-1 therapy: small molecules are cheaper to manufacture than peptides, do not require cold-chain storage, and can be taken as a simple pill. This article covers the pharmacology, the clinical data, and the implications for the peptide obesity landscape. For how existing GLP-1 drugs affect the heart, see GLP-1 Drugs and Heart Disease: What the Cardiovascular Trials Show. For the dual-agonist approach, see Pemvidutide: A Dual Agonist Targeting Both Weight and Liver Disease. For the competitor, see Danuglipron: Pfizer's Oral GLP-1 and Its Rocky Development Path. For the full pipeline, see Every Obesity Peptide in the Pipeline: A 2026 Tracker.
Key Takeaways
- Every weight-loss drug in the Ozempic class is an injection — orforglipron is the first that's just a pill.
- Phase 3 trials showed about 11% weight loss at 72 weeks, compared to roughly 2% on placebo.
- Nearly 40% of patients on the top dose lost at least 15% of their body weight.
- It's less effective than Wegovy or Zepbound, but cheaper to make and needs no refrigeration.
- Unlike oral semaglutide, orforglipron doesn't require fasting — you can take it with or without food.
- It's a small molecule, not a peptide, and it binds the GLP-1 receptor from a different angle entirely.
- A cheap shelf-stable pill could finally reach the billion people with obesity who can't access injectables.
Why a non-peptide GLP-1 matters
The GLP-1 receptor agonist market is projected to exceed $100 billion annually by 2030. Every product in this market is a peptide: a chain of amino acids that must be synthesized through expensive fermentation or solid-phase peptide synthesis, stored under refrigeration, and delivered by injection (or, in the case of oral semaglutide, protected from gastric degradation by a specialized formulation that requires strict fasting protocols).
These constraints create real barriers. Injectable delivery deters a substantial fraction of patients who could benefit from GLP-1 therapy. Cold-chain requirements increase distribution costs and limit availability in low-resource settings. Manufacturing cost contributes to prices that, even with insurance, leave many patients unable to access treatment. The cost-effectiveness debate around GLP-1s revolves in part around these structural constraints.
A non-peptide oral GLP-1 agonist eliminates all three barriers simultaneously. Small molecules are synthesized through conventional chemical processes at a fraction of peptide manufacturing cost. They are shelf-stable at room temperature. They are taken as pills. Orforglipron is the first compound to demonstrate that this approach can produce clinically meaningful weight loss and glycemic control in Phase 3 trials.
Pharmacology: a different way to activate GLP-1R
Sloop et al. (2024) published the structural and pharmacological basis for orforglipron's mechanism in Science Translational Medicine. The key finding: orforglipron does not bind where peptide GLP-1 agonists bind.[2]
Endogenous GLP-1 and peptide analogs (semaglutide, tirzepatide, liraglutide) bind to the extracellular domain and orthosteric pocket of the GLP-1 receptor, inserting their N-terminal portions into the transmembrane core. Orforglipron binds within a transmembrane pocket formed by TM1, TM2, TM3, TM7, and ECL2, stabilizing an active receptor conformation from this allosteric site. This is an entirely different molecular interaction than peptide agonism.
The pharmacological profile is also distinct. Orforglipron is a partial agonist for cAMP production (the primary intracellular signal that mediates GLP-1R effects on insulin secretion and appetite) but has minimal effect on beta-arrestin recruitment. Peptide GLP-1 agonists are full agonists that activate both cAMP and beta-arrestin pathways. Beta-arrestin recruitment drives receptor internalization and desensitization. Orforglipron's lack of beta-arrestin engagement may explain why low receptor occupancy is sufficient for full biological effect: the receptor remains on the cell surface, available for continued signaling, rather than being internalized after activation.[2]
This biased agonism (cAMP-preferring, beta-arrestin-sparing) has implications for both efficacy and tolerability. The GI side effects of GLP-1 agonists (nausea, vomiting, diarrhea) may be partly mediated by beta-arrestin pathways. A compound that achieves full metabolic efficacy through cAMP signaling alone, while minimizing beta-arrestin-mediated effects, could theoretically have a better tolerability profile. Whether this theoretical advantage translates to clinical practice is still being evaluated in the Phase 3 data.
Pratt et al. (2023) published the Phase 1a first-in-human data in a blinded, placebo-controlled, single- and multiple-ascending-dose study in healthy participants. Orforglipron was rapidly absorbed with a half-life of approximately 30 hours, supporting once-daily dosing. Plasma levels were not meaningfully affected by food, eliminating the fasting requirement that limits oral semaglutide. The compound showed dose-proportional pharmacokinetics across the tested range, and GLP-1R-mediated effects (glucose-dependent insulin secretion, gastric emptying delay) were observed at pharmacologically active doses.[3]
The 30-hour half-life is worth noting in the context of the GLP-1 class. Injectable semaglutide has a half-life of approximately 7 days (enabling weekly dosing). Liraglutide has a half-life of approximately 13 hours (requiring daily injection). Orforglipron's 30-hour half-life is long enough for once-daily dosing but short enough that a missed dose does not produce prolonged drug exposure, a potential safety advantage: if a patient experiences a severe adverse event, the drug clears the system within days rather than weeks.
Phase 2: establishing the dose-response
Wharton et al. (2023) published the Phase 2 obesity trial in the New England Journal of Medicine. A total of 272 adults with obesity (BMI 30 or higher, or 27 with comorbidities) received orforglipron at doses of 12, 24, 36, or 45 mg daily, or placebo, for 36 weeks.[4]
The results showed dose-dependent weight loss: -8.6% at 12 mg, -12.6% at 24 mg, -12.4% at 36 mg, and -14.7% at 45 mg, versus -2.0% for placebo. At the highest dose, 46% of participants lost at least 15% of their body weight. Improvements in fasting glucose, HbA1c, insulin, triglycerides, and cholesterol accompanied the weight loss.
Frias et al. (2023) published the parallel Phase 2 trial in type 2 diabetes, showing that orforglipron produced significant HbA1c reductions (up to -1.67 percentage points) and weight loss in patients already on metformin. The glycemic effects were comparable to injectable semaglutide at similar timepoints.[5]
The Phase 2 data established the therapeutic window: the 36 mg dose was selected as the highest dose for Phase 3 trials, balancing efficacy against the GI side effect burden that increased at 45 mg. The Phase 2 results also confirmed that orforglipron's effects extend beyond weight loss to include improvements in multiple cardiometabolic risk factors, including waist circumference, blood pressure, lipid profile, and inflammatory markers, a pattern consistent with the established benefits of the GLP-1 receptor agonist class as a whole.
Phase 3: ATTAIN-1 and ATTAIN-2
ATTAIN-1: obesity without diabetes
Wharton et al. (2025) published the ATTAIN-1 results in the New England Journal of Medicine. This was the definitive obesity trial: 3,127 adults with BMI 30 or higher (or 27 with comorbidities) randomized to orforglipron 6, 12, or 36 mg daily, or placebo, for 72 weeks.[1]
At 72 weeks, mean weight loss was -7.5% with 6 mg, -8.4% with 12 mg, and -11.2% with 36 mg, versus -2.1% with placebo. At the 36 mg dose, 59.6% of participants lost at least 10% of their body weight, and 39.6% lost at least 15%. Waist circumference, systolic blood pressure, triglycerides, and non-HDL cholesterol all improved significantly compared to placebo.
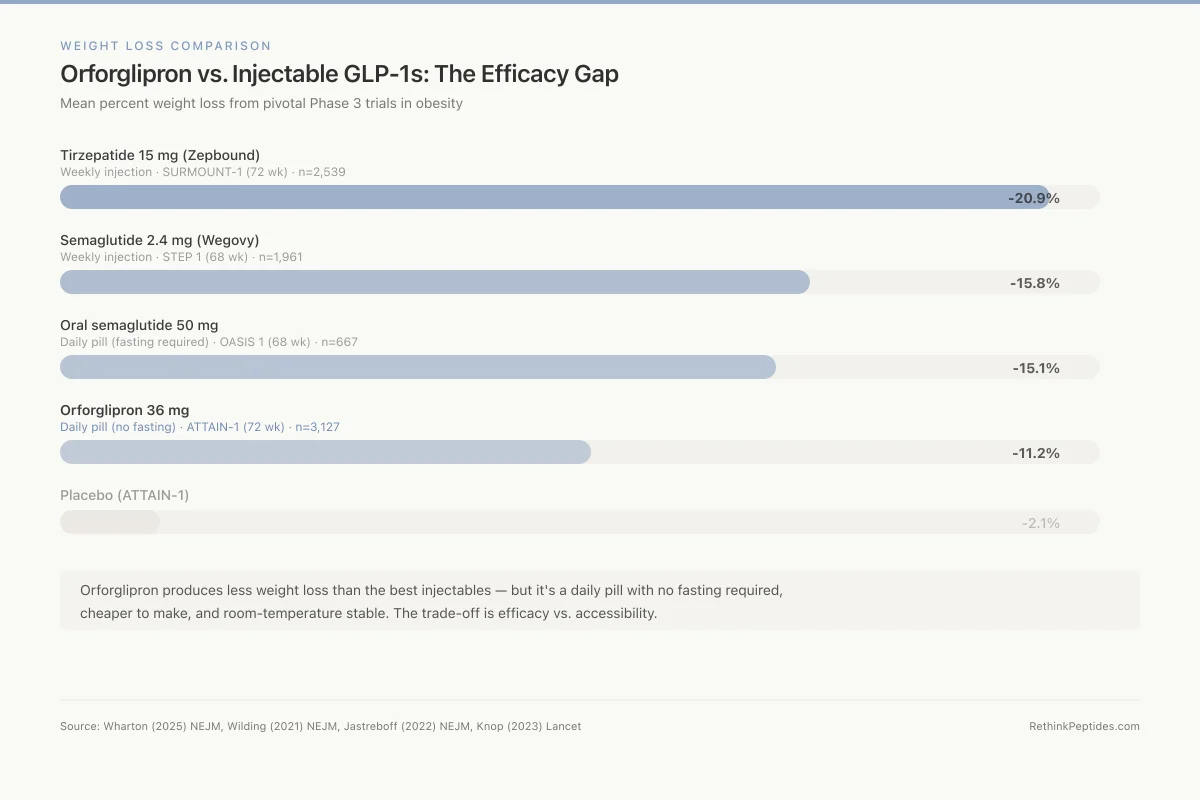
The 11.2% weight loss at 36 mg is clinically meaningful but less than the ~15-17% seen with injectable semaglutide 2.4 mg (Wegovy) or the ~20-22% seen with tirzepatide (Zepbound) in their respective Phase 3 trials. This efficacy gap is the central tension in orforglipron's commercial positioning: it is less effective than the best injectables but far more convenient.
Weight Loss Comparison
Orforglipron vs. Injectable GLP-1s: The Efficacy Gap
Mean percent weight loss from pivotal Phase 3 trials in obesity
ATTAIN-1 (72 wk), n=3,127
STEP 1 (68 wk), n=1,961
SURMOUNT-1 (72 wk), n=2,539
OASIS 1 (68 wk), n=667
ATTAIN-1 (72 wk), n=3,127
Orforglipron produces less weight loss than the best injectables — but it's a daily pill with no fasting required, cheaper to make, and room-temperature stable. The trade-off is efficacy vs. accessibility.
Source: Wharton (2025) NEJM, Wilding (2021) NEJM, Jastreboff (2022) NEJM, Knop (2023) Lancet
 View as image
View as imageThe dose-response curve from ATTAIN-1 is informative. The 6 mg dose produced 7.5% weight loss, the 12 mg dose 8.4%, and the 36 mg dose 11.2%. The curve has not clearly plateaued, and the Phase 2 data showed 14.7% at 45 mg. This suggests that higher doses might close the gap with injectable GLP-1s, but the Phase 2 45 mg arm also showed higher rates of GI adverse events, likely contributing to Lilly's decision to cap Phase 3 dosing at 36 mg. The trade-off between maximum efficacy and tolerable side effects is a recurring challenge in GLP-1 drug development: every agent in this class shows dose-dependent efficacy and dose-dependent nausea.
ATTAIN-2: obesity with type 2 diabetes
Horn et al. (2026) published the ATTAIN-2 results in the Lancet. In adults with obesity and type 2 diabetes, orforglipron produced up to 9.6% weight loss at the highest dose, with significant HbA1c reductions. The weight loss was less than in ATTAIN-1, consistent with the well-established finding that patients with type 2 diabetes lose less weight on GLP-1 agonists than those without diabetes.[6]
Rosenstock et al. (2025) published additional data on orforglipron in early type 2 diabetes, showing that the compound improved markers of metabolic function beyond glucose control, including liver fat reduction and inflammatory markers.[7]
Safety profile
The adverse event profile in ATTAIN-1 and ATTAIN-2 was consistent with the GLP-1 receptor agonist class: nausea, diarrhea, vomiting, and constipation were the most common side effects, occurring at higher rates with higher doses. Most GI events were mild to moderate and occurred during the dose-titration phase, diminishing over time. Discontinuation rates due to adverse events were higher in the orforglipron groups than placebo but within the range seen with injectable GLP-1 agonists.
Wharton et al. (2025) specifically analyzed cardiovascular safety biomarkers and found that orforglipron produced improvements in multiple cardiometabolic parameters alongside weight loss. A dedicated cardiovascular outcomes trial is planned but has not yet reported.[8]
The disruption argument
Orforglipron's significance extends beyond its clinical data. It represents a potential structural shift in the GLP-1 market for several reasons.
Manufacturing cost. Peptide synthesis (whether by fermentation for recombinant peptides or solid-phase synthesis for synthetic peptides) is inherently expensive. Small molecule synthesis uses conventional pharmaceutical chemistry at dramatically lower cost per dose. If orforglipron is priced significantly below injectable GLP-1s, it could expand the addressable market to patients and health systems currently priced out.
Supply chain simplicity. Injectable GLP-1s require cold-chain storage, specialized syringes or autoinjectors, and healthcare professional training for administration. Orforglipron is a room-temperature pill. This simplifies distribution in every setting from urban pharmacies to rural clinics to global health programs.
Patient adherence. Injection phobia affects an estimated 20-30% of patients prescribed injectable medications. Even among those who accept injections, adherence declines over time. A once-daily pill taken with or without food removes the injection barrier entirely. The ATTAIN-Maintain sub-study showed that patients who switched from injectable GLP-1s to oral orforglipron maintained their weight loss, suggesting the transition is pharmacologically feasible.
Access Comparison
Injectable Semaglutide vs. Oral Semaglutide vs. Orforglipron
| Injectable semaglutide | Oral semaglutide | Orforglipron | |
|---|---|---|---|
| Delivery | Weekly subcutaneous injection via autoinjector pen | Daily pill — must fast 30 min before and after, water only | Daily pill — with or without food, no restrictions |
| Storage | Refrigerated (2–8°C) before first use; limited room temp window | Room temperature | Room temperature |
| Manufacturing | Peptide synthesis (fermentation or solid-phase) — expensive, complex | Peptide synthesis + SNAC permeation enhancer formulation | Conventional small-molecule chemistry — dramatically cheaper |
| Bioavailability | ~100% (injected) | ~1% (99% destroyed in gut) | High oral bioavailability, dose-proportional |
| Half-life | ~7 days (weekly dosing) | ~7 days (but daily dosing due to low absorption) | ~30 hours (daily dosing, clears in days if stopped) |
Source: Pratt (2023), Sloop (2024), product prescribing information
Competition dynamics. Pfizer's oral GLP-1 candidate, danuglipron, has had development setbacks, including a reformulation that delayed its timeline. If orforglipron reaches market first with Phase 3 data in both obesity and diabetes, it would establish the oral non-peptide GLP-1 category and set the benchmark that all future oral GLP-1s must beat. The head-to-head trial comparing orforglipron to oral semaglutide (Rosenstock et al., 2026) will directly address how the small molecule compares to the peptide in oral form.[9]
Global health implications. The WHO estimates that over 1 billion people worldwide have obesity. In low- and middle-income countries, where the obesity burden is growing fastest, injectable GLP-1 therapy is essentially unavailable due to cost and cold-chain requirements. A room-temperature oral pill manufactured through conventional pharmaceutical chemistry could be the first GLP-1 therapy realistically deployable at global scale. This is not a hypothetical: the trajectory from expensive injectable to affordable oral formulation has played out before in HIV treatment (from injectable enfuvirtide to oral antiretrovirals) and hepatitis C (from injectable interferon to oral direct-acting antivirals). Each transition dramatically expanded treatment access.
The peptide industry question. Orforglipron's success raises a fundamental question for the peptide therapeutics field: if a small molecule can replicate the effect of a peptide drug at lower cost with simpler delivery, does the peptide lose its therapeutic niche? This question has different answers depending on the target. For GLP-1R mono-agonism, orforglipron may eventually make injectable GLP-1 peptides obsolete for many patients. For multi-receptor agonism (GLP-1/GIP dual agonism as in tirzepatide, or GLP-1/GIP/glucagon triple agonism as in retatrutide), developing small molecules that simultaneously engage multiple peptide receptors with the right potency ratios is substantially more difficult. The most advanced injectable GLP-1s may retain their advantage through multi-receptor pharmacology that small molecules cannot easily replicate.
Orforglipron versus oral semaglutide
The most relevant comparison for orforglipron is not injectable semaglutide but oral semaglutide (Rybelsus), since both are pills. Oral semaglutide is a peptide formulated with a permeation enhancer (SNAC) that allows absorption through the gastric mucosa. It requires strict fasting: the pill must be taken on an empty stomach with no more than 120 mL of water, and the patient must wait 30 minutes before eating, drinking, or taking other medications.
These restrictions are clinically significant. Real-world adherence to oral semaglutide is lower than to injectable semaglutide, partly because of the fasting burden. Patients who eat breakfast before their morning medication, or who take other pills with water in the morning, cannot easily accommodate oral semaglutide's requirements. Orforglipron has no such restrictions.
Rosenstock et al. (2026) conducted a direct head-to-head comparison of oral orforglipron versus oral semaglutide in adults with type 2 diabetes.[9] The results of this trial will define the competitive positioning between the two oral approaches. If orforglipron matches or approaches oral semaglutide's efficacy while being simpler to take, the convenience advantage alone could drive market share. If oral semaglutide proves meaningfully more effective, the choice between the two becomes an efficacy-versus-convenience trade-off.
The bioavailability difference is also relevant. Oral semaglutide has approximately 1% bioavailability (99% of the swallowed dose is destroyed in the GI tract), which is why it requires the SNAC enhancer and fasting. Orforglipron, as a small molecule, has substantially higher oral bioavailability without requiring any special formulation or dosing conditions. This means a lower manufactured dose per pill and more predictable drug exposure across patients, both of which are manufacturing and clinical advantages.
What the evidence does and does not show
The clinical evidence for orforglipron is strong and growing. Two Phase 3 trials with thousands of patients demonstrate clinically meaningful weight loss and glycemic control. The pharmacological mechanism is well-characterized at the structural level. The safety profile is consistent with the GLP-1 class without novel safety signals.
The efficacy gap relative to injectable GLP-1s is real and important. At 11.2% weight loss (36 mg, 72 weeks), orforglipron produces less weight loss than semaglutide 2.4 mg (~15%) or tirzepatide at optimal doses (~20%). For patients whose primary goal is maximum weight loss, injectable options remain superior. Whether orforglipron's convenience advantage compensates for this efficacy difference is ultimately a patient-level decision that depends on individual priorities, injection tolerance, and treatment goals.
The long-term safety data is limited. The longest controlled data is 72 weeks. Cardiovascular outcomes, cancer risk, and effects on bone density and muscle mass during chronic use have not been assessed in dedicated trials. The sarcopenia risk with GLP-1 weight loss applies to orforglipron as it does to all GLP-1 agonists, and whether the partial agonist pharmacology changes this risk profile is unknown. The cardiovascular outcome question is particularly important: injectable semaglutide has demonstrated cardiovascular benefit in the SELECT trial. Whether orforglipron, with its lower weight loss magnitude and different pharmacological profile, would produce similar cardiovascular protection is uncertain and cannot be assumed from class effects alone.
There is also a question about the population that orforglipron would serve. If it is positioned as a first-line treatment for patients with less severe obesity (BMI 30-35 without comorbidities), it might fill a gap that injectable GLP-1s currently do not serve well because the injection barrier discourages uptake. If positioned as a second-line option for patients who cannot tolerate or prefer not to use injections, it competes directly with oral semaglutide. If positioned as a cost-effective alternative at scale, it could expand the total market rather than cannibalize existing GLP-1 prescriptions. Lilly's pricing and marketing strategy will determine which of these roles orforglipron ultimately fills.
The comprehensive review by Kansakar et al. (2026) noted that orforglipron's biased agonism (cAMP-preferring) distinguishes it pharmacologically from all existing GLP-1 therapies, which are full agonists. Whether this translates to clinically meaningful differences in efficacy or safety beyond what the trials have shown remains to be determined in longer follow-up and real-world use.[10]
Panou et al. (2025) published an overview positioning orforglipron within the evolving GLP-1 landscape, noting that the compound's greatest impact may not be in replacing injectable GLP-1s for existing patients but in expanding treatment access to the hundreds of millions of people with obesity or diabetes who currently receive no pharmacotherapy at all.[11] For the broader pipeline of next-generation obesity compounds, see Beyond Triple Agonists: Are Quadruple Receptor Targets Coming? and Every Obesity Peptide in the Pipeline: A 2026 Tracker.
The Bottom Line
Orforglipron is the first non-peptide oral GLP-1 receptor agonist to complete Phase 3 trials with positive results. It binds the GLP-1 receptor at an allosteric transmembrane site distinct from peptide agonists, acting as a cAMP-biased partial agonist. Phase 3 data show 11.2% weight loss at 72 weeks in obesity (ATTAIN-1) and up to 9.6% in obesity with type 2 diabetes (ATTAIN-2), with a safety profile consistent with the GLP-1 class. As a small molecule, it could dramatically reduce manufacturing costs, eliminate cold-chain requirements, and remove injection barriers, potentially expanding GLP-1 therapy access to hundreds of millions of untreated patients worldwide.
Sources & References
- 1RPEP-14142·Wharton, Sean et al. (2025). “Orforglipron, an Oral Small-Molecule GLP-1 Receptor Agonist for Obesity Treatment..” The New England journal of medicine.Study breakdown →PubMed →↩
- 2RPEP-09288·Sloop, Kyle W et al. (2024). “Orforglipron: How a Pill Can Activate the Same GLP-1 Receptor as Injectable Peptides — The Pharmacology Behind It.” Science translational medicine.Study breakdown →PubMed →↩
- 3RPEP-07284·Pratt, Edward et al. (2023). “Oral GLP-1 Drug Orforglipron Shows Promising Weight Loss and Safety in First Human Trial.” Diabetes.Study breakdown →PubMed →↩
- 4RPEP-07549·Wharton, Sean et al. (2023). “Orforglipron: The Daily Weight Loss Pill That Produced Up to 14.7% Body Weight Reduction in 36 Weeks.” The New England journal of medicine.Study breakdown →PubMed →↩
- 5RPEP-06884·Frias, Juan P et al. (2023). “Oral GLP-1 Pill Orforglipron Cuts Blood Sugar and Weight More Than Injectable Dulaglutide in Phase 2 Trial.” Lancet (London.Study breakdown →PubMed →↩
- 6RPEP-15308·Horn, Deborah B et al. (2026). “Orforglipron Phase 3: Oral Non-Peptide GLP-1 Drug Achieves 7.9% Weight Loss in Diabetic Obesity.” Lancet (London.Study breakdown →PubMed →↩
- 7RPEP-13312·Rosenstock, Julio et al. (2025). “Orforglipron, an Oral Small-Molecule GLP-1 Receptor Agonist, in Early Type 2 Diabetes..” The New England journal of medicine.Study breakdown →PubMed →↩
- 8RPEP-14143·Wharton, Sean et al. (2025). “Oral GLP-1 Drug Orforglipron Improves Heart Disease Risk Markers in Diabetes and Obesity.” Cardiovascular diabetology.Study breakdown →PubMed →↩
- 9RPEP-16015·Rosenstock, Julio et al. (2026). “Oral Orforglipron Beat Oral Semaglutide at Lowering Blood Sugar in a 1,698-Person Diabetes Trial.” Lancet (London.Study breakdown →PubMed →↩
- 10RPEP-15411·Kansakar, Urna et al. (2026). “Orforglipron Comprehensive Review: The First Non-Peptide Oral GLP-1 Drug Approaching Approval.” International journal of molecular sciences.Study breakdown →PubMed →↩
- 11RPEP-12945·Panou, Theodoros et al. (2025). “Orforglipron: a promising oral GLP-1 drug for type 2 diabetes and obesity without needing injections.” Expert review of clinical pharmacology.Study breakdown →PubMed →↩