GLP-1 Agonists and Addictive Behavior
Peptides and Addiction
50-56% lower AUD risk
Semaglutide was associated with a 50-56% lower risk for both incidence and recurrence of alcohol use disorder compared to other anti-obesity medications in a real-world cohort study.
Wang et al., Nature Medicine, 2024
Wang et al., Nature Medicine, 2024
If you only read one thing
People taking Ozempic and similar weight-loss drugs started noticing they didn't want to drink alcohol anymore. Researchers investigated and found this isn't a fluke — a study of 800,000+ veterans showed these drugs were linked to lower rates of addiction across six substance types, including alcohol, nicotine, cocaine, and opioids. The first clinical trial confirmed it: semaglutide reduced drinking and cravings even in people who weren't trying to quit. The drugs work by turning down dopamine reward signals in the brain, which is the same circuit that drives all addictions.
Patients on semaglutide for weight loss or diabetes started reporting something their doctors did not expect: they had lost interest in alcohol. They were smoking less. Some described a general dampening of compulsive urges that extended beyond food. These anecdotal reports, which accumulated rapidly after semaglutide's widespread adoption in 2021-2023, prompted researchers to investigate whether GLP-1 receptor agonists (GLP-1 RAs) could genuinely reduce addictive behaviors. The evidence that has emerged since then is striking. A large cohort study of real-world patient data found that semaglutide was associated with a 50-56% lower risk for both incidence and recurrence of alcohol use disorder compared to other anti-obesity medications.[1] A BMJ study of over 800,000 U.S. veterans found that GLP-1 RA initiation was associated with reduced incidence of alcohol, cannabis, cocaine, nicotine, opioid, and other substance use disorders.[2] And in February 2025, the first randomized controlled trial of semaglutide for alcohol use disorder reported reduced alcohol consumption, reduced heavy drinking, and reduced cravings compared to placebo, published in JAMA Psychiatry. This article covers the full evidence landscape for GLP-1 agonists and addictive behavior, from molecular mechanisms through preclinical models to emerging clinical data. For related coverage, see our articles on orexin antagonists and drug-seeking behavior and peptide vaccines for nicotine and cocaine.
Key Takeaways
- Patients on Ozempic started telling their doctors they'd lost interest in drinking — nobody expected it.
- In a study of over 800,000 veterans, GLP-1 drugs lowered addiction rates across six different substances.
- Semaglutide users had a 50–56% lower risk of developing alcohol use disorder than people on other weight-loss drugs.
- It's not about feeling full — the drugs dial down the dopamine circuits that drive every addiction.
- The first controlled trial confirmed it: people drank less and craved less, even when they weren't trying to quit.
- GLP-1 receptors sit inside the brain's reward center — same reason cravings for nicotine and cocaine drop too.
- Treating addiction isn't an approved use yet — pivotal trials for alcohol use disorder are still underway.
How GLP-1 Receptors Ended Up in the Brain
GLP-1 (glucagon-like peptide-1) is a 30-amino-acid incretin hormone released by intestinal L-cells after eating. Its primary metabolic function is to stimulate insulin secretion, suppress glucagon, slow gastric emptying, and promote satiety. GLP-1 receptor agonists like semaglutide, liraglutide, and tirzepatide were developed to treat type 2 diabetes and obesity by amplifying these metabolic effects.
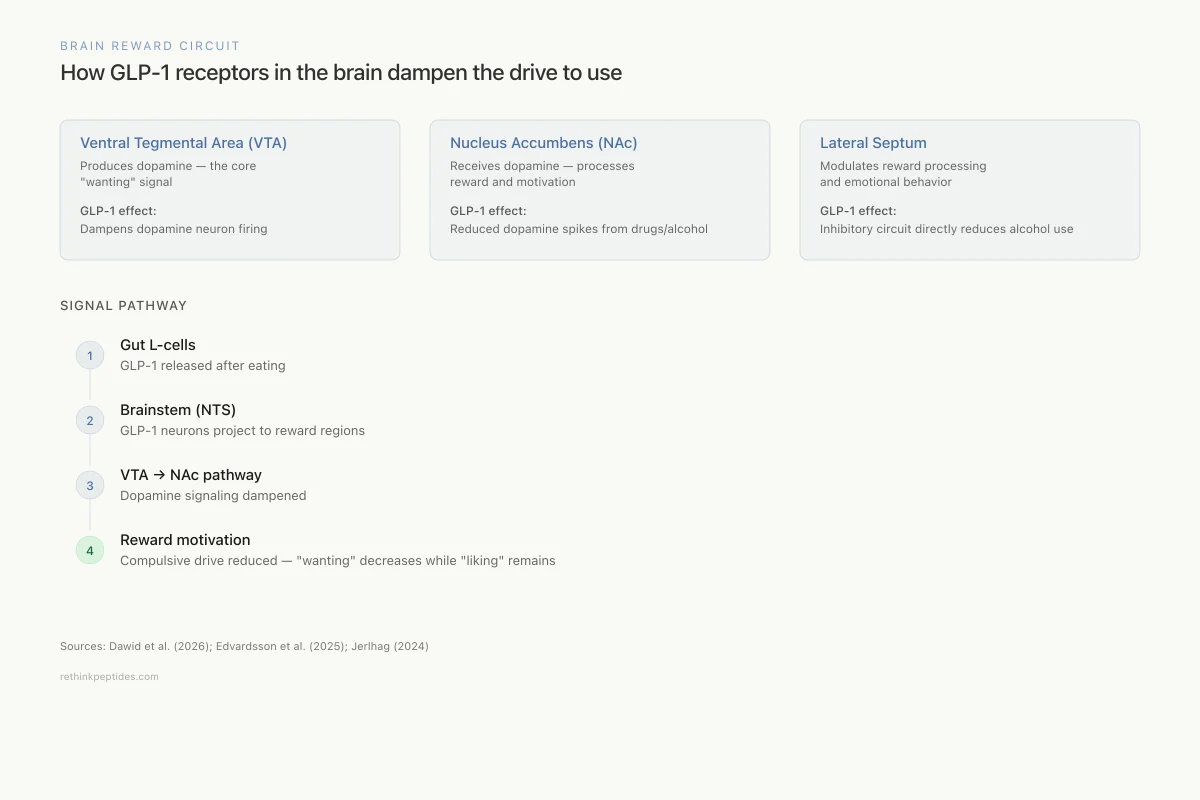
The GLP-1 receptor, however, is not confined to the pancreas and gut. It is expressed in multiple brain regions, including the ventral tegmental area (VTA), nucleus accumbens (NAc), lateral septum, hippocampus, amygdala, and hypothalamus. The VTA and NAc are the core components of the mesolimbic dopamine system, the brain's primary reward circuit. This anatomical fact, known for years but largely ignored in the diabetes literature, turned out to be critical: GLP-1 receptor activation in reward circuits modulates dopamine signaling and reduces the motivational drive toward rewarding stimuli, whether those stimuli are food, alcohol, or drugs.[3]
A 2025 study identified a specific inhibitory GLP-1 circuit in the lateral septum that modulates reward processing and alcohol consumption, providing direct neuroanatomical evidence that endogenous GLP-1 signaling in the brain regulates substance reward.[4]
Human post-mortem data adds a pathological dimension. Brain tissue from individuals with alcohol-related brain disease showed impaired expression of GLP-1, GIP, leptin, and ghrelin in the frontal lobe, with pancreatic polypeptide reduced in the cerebellar vermis and hypothalamus.[5] This suggests that chronic alcohol use disrupts the brain's incretin system, potentially creating a deficit that exogenous GLP-1 RAs could correct.
Brain Reward Circuit
How GLP-1 receptors in the brain dampen the drive to use
Ventral Tegmental Area (VTA)
Produces dopamine — the core "wanting" signal
GLP-1 effect:
GLP-1 receptors dampen dopamine neuron firing
Nucleus Accumbens (NAc)
Receives dopamine — processes reward and motivation
GLP-1 effect:
Reduced dopamine spikes from drugs, alcohol, and food
Lateral Septum
Modulates reward processing and emotional behavior
GLP-1 effect:
Inhibitory GLP-1 circuit directly reduces alcohol consumption
Signal Pathway
Gut L-cells
GLP-1 released after eating
Brainstem (NTS)
GLP-1 neurons project to reward regions
VTA → NAc pathway
Dopamine signaling dampened
Reward motivation
Compulsive drive reduced — "wanting" decreases
Sources: Dawid et al. (2026); Edvardsson et al. (2025); Jerlhag (2024)
 View as image
View as imageThe presence of GLP-1 receptors in reward circuits is not an accident of pharmacology. Endogenous GLP-1-producing neurons in the nucleus tractus solitarius (NTS) of the brainstem project to the VTA and other reward-related regions. This endogenous GLP-1 signaling pathway likely evolved to integrate metabolic status with reward-seeking behavior: when the body has adequate energy (high GLP-1), the drive to seek food diminishes. The "side effect" of reducing alcohol and drug cravings reflects the fact that substance addiction co-opts the same reward circuitry that GLP-1 was evolved to regulate.
The Alcohol Evidence
Epidemiological Data
The first large-scale signal came from real-world observational data. Wang and colleagues analyzed electronic health records and found that semaglutide was associated with a 50-56% lower risk for both incidence and recurrence of alcohol use disorder compared to other anti-obesity medications (non-GLP-1 RAs), after adjusting for confounders.[1]
The largest observational study to date, published in the BMJ in 2026, examined GLP-1 RA use among 817,309 U.S. veterans. GLP-1 RA initiation was associated with reduced incidence of alcohol use disorder along with cannabis, cocaine, nicotine, opioid, and other substance use disorders. The associations persisted after extensive adjustment and sensitivity analyses.[2]
Real-world observational data from the Kaiser Permanente system confirmed that GLP-1 RA users showed reduced alcohol consumption patterns compared to matched controls.[6]
The First Randomized Controlled Trial
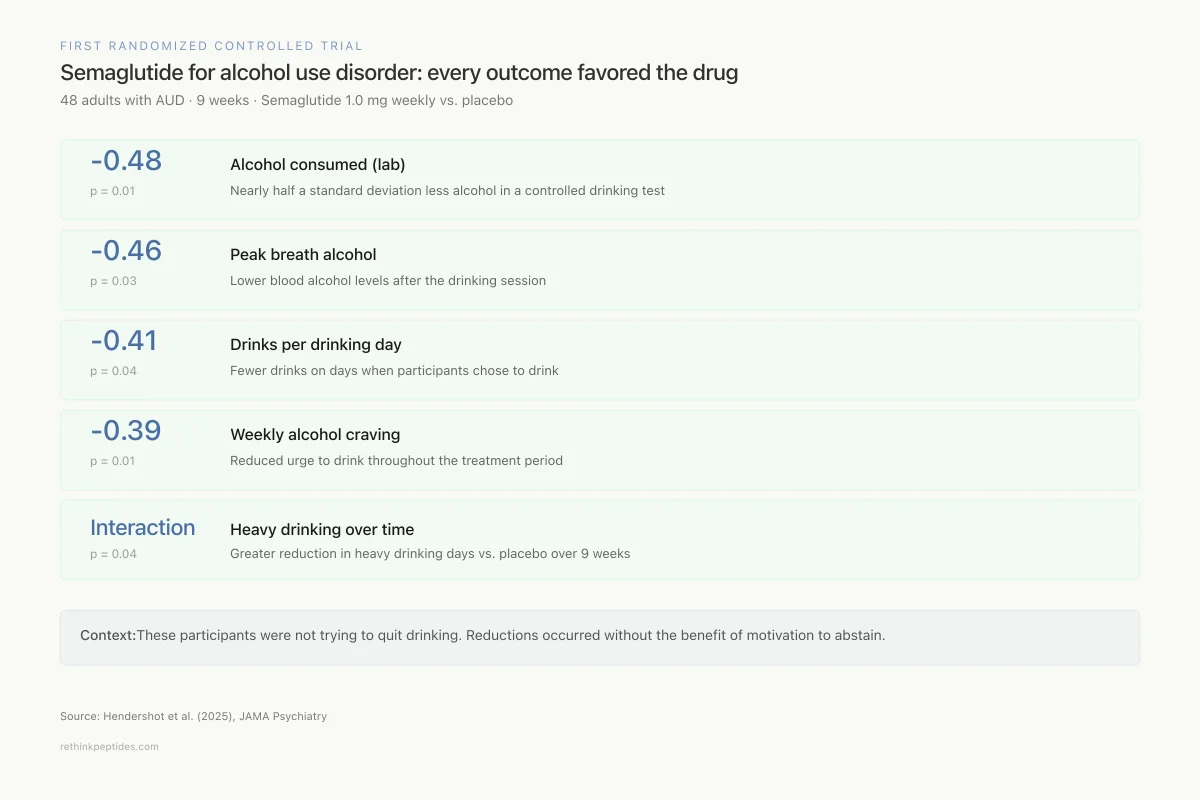
The Hendershot trial, published in JAMA Psychiatry in February 2025, was the first RCT of semaglutide specifically for alcohol use disorder. Forty-eight non-treatment-seeking adults with AUD received either semaglutide (titrated to 1.0 mg weekly) or placebo for 9 weeks.
Results showed medium-to-large effect sizes across multiple outcomes. In a post-treatment laboratory self-administration paradigm, semaglutide reduced grams of alcohol consumed (effect size -0.48, p=0.01) and peak breath alcohol concentration (-0.46, p=0.03). During the treatment period, semaglutide reduced drinks per drinking day (-0.41, p=0.04) and weekly alcohol craving (-0.39, p=0.01), and predicted greater reductions in heavy drinking over time compared to placebo (p=0.04). Participants who also smoked showed greater reductions in cigarettes per day.
First Randomized Controlled Trial
Semaglutide for alcohol use disorder: every outcome favored the drug
48 adults with AUD · 9 weeks · Semaglutide 1.0 mg weekly vs. placebo
-0.48
p = 0.01
Alcohol consumed (lab)
Nearly half a standard deviation less alcohol in a controlled drinking test
-0.46
p = 0.03
Peak breath alcohol
Lower blood alcohol levels after the drinking session
-0.41
p = 0.04
Drinks per drinking day
Fewer drinks on days when participants chose to drink
-0.39
p = 0.01
Weekly alcohol craving
Reduced urge to drink throughout the treatment period
Interaction
p = 0.04
Heavy drinking over time
Greater reduction in heavy drinking days compared to placebo over 9 weeks
Context: These are medium-to-large effect sizes in a small sample. The participants were not trying to quit drinking — these reductions happened without the added benefit of motivation to abstain.
Source: Hendershot et al. (2025), JAMA Psychiatry
 View as image
View as imageThese are phase 2 results in a small sample, but the consistency of effects across multiple endpoints is encouraging. The study was designed conservatively with non-treatment-seeking participants (people who were not actively trying to quit drinking), which means the reductions occurred without the benefit of motivation to abstain. Phase 3 trials evaluating semaglutide for AUD are underway, assessing efficacy, tolerability, and safety at a larger scale across multiple clinical sites.
The 9-week treatment duration is an important limitation. Alcohol use disorder is a chronic relapsing condition, and whether semaglutide's effects persist over months or years of treatment is unknown. Weight-loss trials of semaglutide have shown that effects reverse after discontinuation, raising the question of whether the same would apply to its anti-addiction effects.
Preclinical Alcohol Data
Animal models have provided mechanistic depth. Semaglutide reduced voluntary alcohol consumption in alcohol-preferring vervet monkeys, a primate model that closely recapitulates human drinking patterns.[7] Tirzepatide (a dual GIP/GLP-1 receptor agonist) attenuated alcohol reward-related dopamine release in the nucleus accumbens (p<0.001), dose-dependently reduced alcohol intake (p<0.001), and prevented both binge-like and relapse-like drinking in rodents.[8] Combining GLP-1 RA treatment with other pharmacotherapies produced synergistic-like decreases in alcohol intake in preclinical models.[9]
A systematic review and meta-analysis of GLP-1 RA effects on alcohol consumption confirmed consistent reductions across both preclinical and clinical datasets.[10]
The preclinical evidence base for alcohol is the most mature of any substance. Multiple research groups using different GLP-1 RAs (liraglutide, exendin-4, semaglutide, dulaglutide) across different species (rats, mice, vervet monkeys) and different drinking paradigms (voluntary two-bottle choice, operant self-administration, binge access, relapse after abstinence) have reported consistent reductions in alcohol consumption. The effect appears to generalize across the full spectrum of alcohol-related behaviors rather than being specific to one aspect of drinking.
The vervet monkey data is particularly relevant because vervet monkeys naturally self-select into drinkers and non-drinkers when given access to alcohol, creating a primate model that closely mirrors the heterogeneity of human drinking behavior. Semaglutide reduced alcohol consumption in heavy-drinking monkeys without causing apparent distress or withdrawal symptoms, suggesting that the mechanism involves reduced motivation rather than aversion.[7]
Beyond Alcohol: Nicotine, Cocaine, and Opioids
Nicotine
Preclinical studies consistently demonstrate that GLP-1 RAs reduce nicotine self-administration, attenuate nicotine-conditioned place preference, and suppress reinstatement of nicotine seeking in rodent models.[11] The clinical data is emerging: a 2026 meta-analysis of GLP-1 RA effects on cigarette smoking found reductions in smoking quantity among GLP-1 RA users, though the data is primarily observational or drawn from subgroup analyses of weight-loss trials rather than addiction-focused studies.[12] GLP-1 RAs for smoking cessation are being reviewed with consideration of their effects on weight gain, which is a common barrier to quitting.
Cocaine
Semaglutide suppressed cocaine taking, cocaine-seeking behavior, and cocaine-evoked dopamine levels in the nucleus accumbens of rats, providing strong preclinical evidence for GLP-1 drugs in psychostimulant addiction.[13] No clinical trials of GLP-1 RAs for cocaine use disorder have been completed, but the preclinical signal is robust enough to warrant investigation.
Opioids
The evidence for GLP-1 RAs in opioid use disorder is the least developed but potentially the most consequential given the ongoing opioid overdose crisis. Epidemiological data from the BMJ veterans study showed reduced incidence of opioid use disorder among GLP-1 RA users, but preclinical mechanistic studies are limited.[2] The potential therapeutic role of GLP-1 RAs in opioid use disorder management has been reviewed but remains speculative pending dedicated trials.
The interaction between GLP-1 signaling and the endogenous opioid system is complex. Opioid drugs directly activate mu opioid receptors, producing a qualitatively different reward signal than the dopamine-mediated reward that GLP-1 RAs appear to modulate. Whether GLP-1 RAs can reduce opioid reward through their downstream effects on dopamine, or whether the direct mu receptor activation bypasses the GLP-1-sensitive circuit entirely, is unclear. Preclinical studies specifically examining GLP-1 RA effects on opioid self-administration and opioid-evoked dopamine release are needed before clinical translation can proceed.
The possibility that GLP-1 RAs could complement existing opioid use disorder treatments (buprenorphine, methadone, naltrexone) rather than replacing them deserves investigation. Combination approaches that address both the opioid receptor-mediated and dopamine-mediated components of opioid addiction could theoretically improve outcomes beyond what either mechanism achieves alone.
The cross-substance breadth of the GLP-1 RA effect is notable. Rather than targeting a substance-specific receptor (as naltrexone targets opioid receptors or varenicline targets nicotinic receptors), GLP-1 RAs appear to modulate the shared downstream reward circuitry that drives compulsive use across substance classes. This positions them as potential "broad-spectrum" anti-addiction agents, a category that essentially did not exist before these discoveries.
Cross-Substance Evidence
GLP-1 agonist evidence strength across six addiction types
Epidemiological
RCT
Preclinical
Key Finding
Alcohol
50-56% lower AUD risk; RCT showed reduced consumption & cravings
Nicotine
Reduced self-administration in rodents; observational smoking reductions
Cocaine
Suppressed cocaine taking, seeking, and dopamine release in rats
Opioids
BMJ veterans study showed reduced OUD incidence; no mechanistic studies yet
Cannabis
BMJ veterans study showed reduced CUD incidence; limited preclinical data
Behavioral
Anecdotal reports of reduced gambling and compulsive behavior
Alcohol
50-56% lower AUD risk; RCT showed reduced consumption & cravings
Nicotine
Reduced self-administration in rodents; observational smoking reductions
Cocaine
Suppressed cocaine taking, seeking, and dopamine release in rats
Opioids
BMJ veterans study showed reduced OUD incidence; no mechanistic studies yet
Cannabis
BMJ veterans study showed reduced CUD incidence; limited preclinical data
Behavioral
Anecdotal reports of reduced gambling and compulsive behavior
Sources: Wang et al. (2024); Cai et al. (2026); Hendershot et al. (2025); Aranäs et al. (2025)
 View as image
View as imageGambling and Behavioral Addictions
Anecdotal reports and early observational data suggest GLP-1 RA effects may extend beyond substance use to behavioral addictions. Patients have reported reduced compulsive gambling, shopping, and sexual behavior while on semaglutide. If confirmed, this would strengthen the hypothesis that GLP-1 RAs dampen general reward-seeking motivation rather than interfering with substance-specific pharmacology. No controlled studies of GLP-1 RAs for behavioral addictions have been published as of early 2026.
Comparison to Existing Addiction Treatments
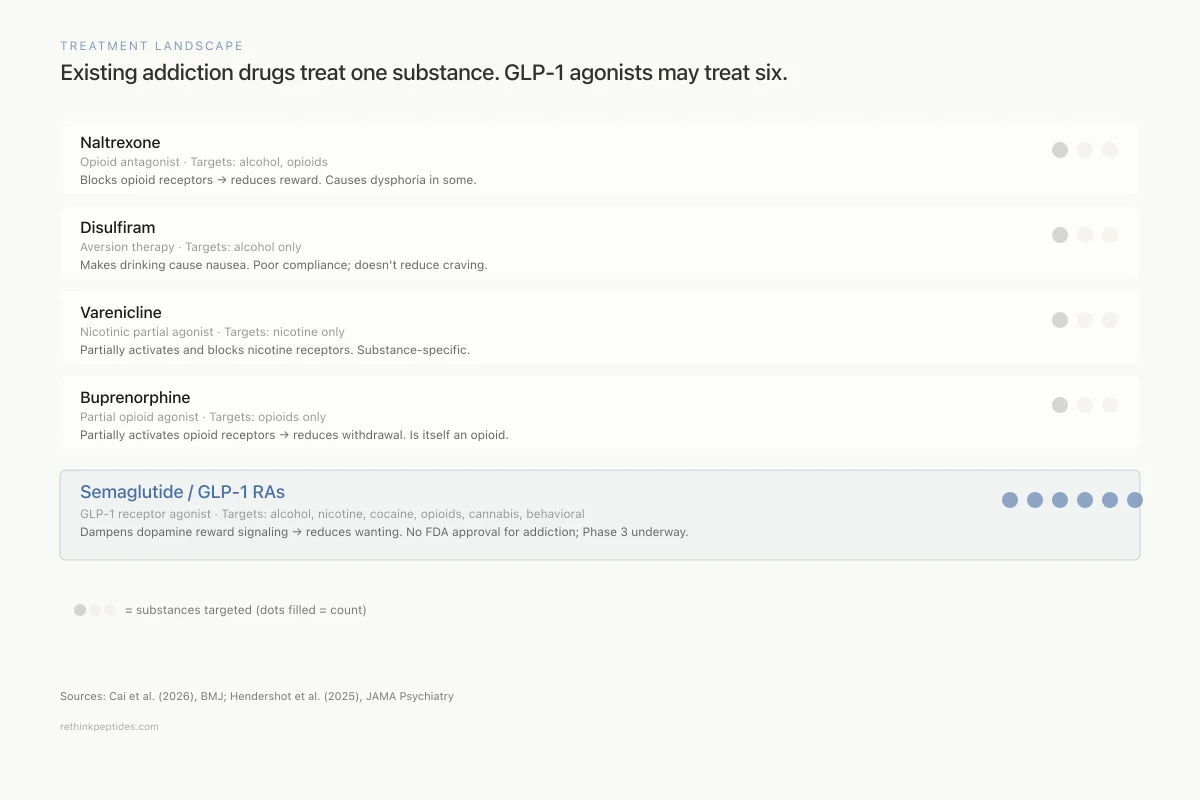
Current pharmacological treatments for addiction are substance-specific and modestly effective. Naltrexone (opioid antagonist) reduces alcohol and opioid use but has limited efficacy and causes dysphoria in some patients. Disulfiram produces aversion to alcohol through acetaldehyde accumulation but compliance is poor. Acamprosate modulates glutamate signaling in alcohol use disorder with modest effect sizes. Buprenorphine and methadone are effective for opioid use disorder but are themselves opioids with abuse potential. Varenicline is effective for smoking cessation but does not address other substances.
Treatment Landscape
Existing addiction drugs treat one substance. GLP-1 agonists may treat six.
Naltrexone
Opioid antagonist
Targets: Alcohol, opioids
Blocks opioid receptors → reduces reward
Causes dysphoria in some; modest efficacy
Disulfiram
Aversion therapy
Targets: Alcohol only
Makes drinking cause nausea via acetaldehyde buildup
Poor compliance; doesn't reduce craving
Varenicline
Nicotinic partial agonist
Targets: Nicotine only
Partially activates and blocks nicotine receptors
Substance-specific; doesn't address other addictions
Buprenorphine
Partial opioid agonist
Targets: Opioids only
Partially activates opioid receptors → reduces withdrawal
Is itself an opioid with abuse potential
Semaglutide / GLP-1 RAs
GLP-1 receptor agonist
Targets: Alcohol, nicotine, cocaine, opioids, cannabis, behavioral
Dampens dopamine reward signaling → reduces wanting
No FDA approval for addiction; $1,000+/month; Phase 3 underway
Source: Cai et al. (2026), BMJ; Hendershot et al. (2025), JAMA Psychiatry
 View as image
View as imageGLP-1 RAs represent a fundamentally different approach: rather than blocking a specific substance's receptor or creating an aversive response, they modulate the reward circuit itself. The potential advantage is breadth across substances and a mechanism that reduces wanting rather than producing aversion or withdrawal. The potential disadvantage is that dampening reward broadly could affect motivation for beneficial activities, though clinical reports suggest the effect is preferentially directed at compulsive rather than healthy reward-seeking.
Mechanism: How GLP-1 RAs Reduce Reward
The mechanism by which GLP-1 RAs reduce addictive behavior centers on their modulation of mesolimbic dopamine signaling.[3]
GLP-1 receptors on VTA neurons regulate dopamine neuron excitability. When GLP-1 RAs activate these receptors, they blunt dopamine release in the nucleus accumbens in response to rewarding stimuli. The result is not anhedonia (complete loss of pleasure) but rather a reduction in the supraphysiological dopamine spikes that drive compulsive reward-seeking. Individuals on semaglutide describe not that food or alcohol becomes unpleasant, but that the compulsive drive toward these rewards diminishes.
The ghrelin system provides a complementary perspective. Ghrelin, the "hunger hormone," promotes both feeding and alcohol consumption through opposing effects on the same reward circuits that GLP-1 modulates. GLP-1 and ghrelin can be conceptualized as a push-pull system: ghrelin drives reward seeking, GLP-1 dampens it.[14] This framework connects metabolic signaling to addiction neuroscience through shared peptide-mediated reward regulation.
The lateral septum GLP-1 circuit provides additional specificity. Edvardsson and colleagues identified GLP-1 receptor-expressing neurons in the lateral septum that, when activated, reduced alcohol consumption and modulated reward processing. This circuit is distinct from the hypothalamic feeding circuits traditionally associated with GLP-1 signaling, suggesting that the anti-addiction effects operate through dedicated reward-regulatory pathways rather than being a secondary consequence of appetite suppression.[4]
Functional neuroimaging data in humans is limited. A 2026 systematic review found only 11 fMRI studies examining GLP-1 RA effects on brain reward responses. Limited evidence suggests acute GLP-1 RA administration may reduce food cue reactivity in reward regions, but data on drug and alcohol cue reactivity is sparse.[15]
The connection between GLP-1 RA effects on reward and the endogenous opioid peptide system is an emerging area of investigation. The dynorphin/kappa opioid receptor system drives the dysphoric "dark side" of addiction during withdrawal, while beta-endorphin drives the euphoric reinforcement during drug use. GLP-1 RAs may modulate both sides of this equation by dampening the dopamine signal that triggers compensatory dynorphin release. If GLP-1 RAs reduce the magnitude of drug-induced dopamine spikes, the subsequent opponent-process dynorphin response would also be reduced, potentially attenuating the negative emotional state that drives compulsive re-dosing. This hypothesis remains speculative but connects two of the most active areas of peptide addiction research.
The distinction between "liking" and "wanting" in addiction neuroscience may explain why GLP-1 RAs reduce compulsive use without eliminating pleasure entirely. The mesolimbic dopamine system primarily encodes incentive salience (wanting) rather than hedonic value (liking). By dampening dopamine-mediated wanting, GLP-1 RAs may reduce the compulsive drive to seek substances without making the experience of consuming them unpleasant when it does occur.
Tirzepatide and Multi-Receptor Agonism
Tirzepatide, a dual GIP/GLP-1 receptor agonist, has shown addiction-relevant effects that may exceed those of GLP-1-selective agonists. In the Edvardsson 2026 study, tirzepatide attenuated alcohol reward-related dopamine release, dose-dependently reduced alcohol intake, and prevented relapse-like drinking in rats.[8] Whether dual GIP/GLP-1 agonism provides superior anti-addiction effects compared to GLP-1 RA alone is an open question with direct clinical implications, since tirzepatide is already approved for diabetes and obesity.
GIP receptors are also expressed in brain reward regions, and GIP agonism may contribute independently to reward modulation. The multi-target incretin therapeutic approach, including dual and triple agonists (GLP-1/GIP/glucagon), represents an emerging strategy where addiction-relevant effects may be a differentiating feature between compounds in the same class.
A 2026 analysis of relative efficacy found differences between GLP-1 and GLP-1/GIP receptor agonists in prevention of substance-related outcomes, though the available data does not yet definitively establish whether dual agonism is superior for addiction-related endpoints. The rapid expansion of the incretin agonist drug class means that multiple compounds with different receptor selectivity profiles will be available for addiction-focused clinical trials in the coming years.
From Metabolic Side Effect to Primary Indication
The trajectory of GLP-1 RAs in addiction research mirrors an earlier pattern in medicine: drugs developed for one indication proving transformative for another. Sildenafil was developed for angina before becoming a treatment for erectile dysfunction. Minoxidil was an antihypertensive before becoming a hair growth treatment. The GLP-1 RA story may represent the largest such repurposing in modern pharmacology, given the scale of the addiction crisis and the potential patient population.
The transition from serendipitous observation to dedicated clinical development has been remarkably fast. The first anecdotal reports emerged around 2021-2022, the first large observational studies were published in 2024, the first RCT results appeared in early 2025, and phase 3 trials were initiated by 2025-2026. This timeline, spanning roughly four years from observation to pivotal trials, is accelerated compared to traditional drug development timelines for psychiatric indications.
Evidence Gaps and Open Questions
The field has moved fast, but critical gaps remain. The semaglutide AUD RCT involved only 48 participants over 9 weeks. Whether effects persist with long-term treatment, what happens when the drug is discontinued, and whether tolerance develops are unanswered. Phase 3 trials will address some of these questions.
The mechanism by which peripheral GLP-1 RAs access brain reward circuits is debated. Semaglutide crosses the blood-brain barrier poorly, yet produces robust behavioral effects on reward. One hypothesis is that peripheral GLP-1 RA signaling is transmitted centrally through vagal afferents. Another is that even low brain penetration is sufficient to activate central GLP-1 receptors. A third possibility is that the drugs primarily act through peripheral metabolic changes that indirectly affect brain reward circuits.
Selection bias in observational studies is a concern. Patients who choose GLP-1 RAs may differ from those who choose other treatments in ways that independently affect addiction risk. The BMJ veterans study attempted to address this through active comparator designs and propensity score matching, but residual confounding cannot be excluded.
The potential for GLP-1 RAs to be marketed or prescribed specifically for addiction raises regulatory and ethical questions. None of these drugs are approved for substance use disorders, and off-label prescribing for addiction is already occurring. Whether the evidence base justifies this practice is a matter of ongoing debate in addiction psychiatry.[16]
Access and equity present additional challenges. GLP-1 RAs are expensive (often exceeding $1,000/month without insurance) and subject to supply constraints. If these drugs prove effective for addiction, ensuring equitable access for populations most affected by substance use disorders, who often lack adequate insurance coverage, would be a substantial policy challenge. The populations with the highest rates of substance use disorders overlap with those who have the least access to specialty pharmacotherapy.
The question of whether GLP-1 RAs work through weight loss rather than direct neural effects on reward needs resolution. Many patients with AUD are overweight, and weight loss itself can improve mood, self-efficacy, and health behaviors. Distinguishing the direct anti-addiction effect from indirect benefits of metabolic improvement requires studies in normal-weight individuals with substance use disorders, which have not been conducted.
Finally, long-term safety of GLP-1 RAs in addiction populations is unknown. The gastrointestinal side effects (nausea, vomiting, diarrhea) that are common with these drugs may interact unpredictably with the gastrointestinal complications of heavy alcohol use (gastritis, pancreatitis, liver disease). Dedicated safety monitoring in addiction populations will be essential as clinical use expands.
The Bottom Line
GLP-1 receptor agonists reduce addictive behaviors across multiple substance classes through modulation of brain reward circuitry. Epidemiological data shows 50-56% reduced alcohol use disorder risk with semaglutide, and the first RCT confirmed reduced consumption and cravings. Preclinical evidence extends to nicotine, cocaine, and opioids. Phase 3 trials are underway for alcohol use disorder, and the dual GIP/GLP-1 agonist tirzepatide may offer additional efficacy. The mechanism centers on dampened mesolimbic dopamine signaling through GLP-1 receptors in the VTA, nucleus accumbens, and lateral septum.
Sources & References
- 1RPEP-09493·Wang, William et al. (2024). “Semaglutide Linked to 50-56% Lower Risk of Alcohol Use Disorder in Large Real-World Study.” Nature communications.Study breakdown →PubMed →↩
- 2RPEP-14921·Cai, Miao et al. (2026). “GLP-1 Drugs Linked to Reduced Substance Use Disorders in US Veterans.” BMJ (Clinical research ed.).Study breakdown →PubMed →↩
- 3RPEP-15083·Dawid, Rosiejka et al. (2026). “GLP-1 Drugs Show Promise for Treating Alcohol, Nicotine, and Other Addictions.” Journal of clinical medicine.Study breakdown →PubMed →↩
- 4RPEP-10811·Edvardsson, Christian E et al. (2025). “An inhibitory GLP-1 circuit in the lateral septum modulates reward processing and alcohol intake in rodents..” EBioMedicine.Study breakdown →PubMed →↩
- 5RPEP-15086·de la Monte, Suzanne M et al. (2026). “Alcohol Damages the Brain's Own GLP-1 System, Suggesting Incretin Drugs Could Help.” Biomolecules.Study breakdown →PubMed →↩
- 6RPEP-15804·Noemi Torres, Isabel et al. (2026). “GLP-1 Weight Loss Drugs Show Promise for Treating Alcohol and Nicotine Addiction.” Substance use & addiction journal.Study breakdown →PubMed →↩
- 7RPEP-10953·Fink-Jensen, Anders et al. (2025). “Semaglutide Significantly Reduces Alcohol Drinking in Primates That Prefer Alcohol.” Psychopharmacology.Study breakdown →PubMed →↩
- 8RPEP-15136·Edvardsson, Christian E et al. (2026). “Tirzepatide Reduces Alcohol Drinking and Relapse in Rodents Through Brain Reward Circuits.” EBioMedicine.Study breakdown →PubMed →↩
- 9RPEP-09967·Aranäs, Cajsa et al. (2025). “Combining GLP-1 Drugs With Other Therapies Synergistically Reduces Alcohol Intake.” British journal of pharmacology.Study breakdown →PubMed →↩
- 10RPEP-15777·Nasrollahizadeh, Amir et al. (2026). “Repurposing GLP-1 receptor agonists for alcohol use disorder: a systematic review and meta-analysis..” Diabetology & metabolic syndrome.Study breakdown →PubMed →↩
- 11RPEP-08369·Herman, Rae J et al. (2024). “Targeting GLP-1 receptors to reduce nicotine use disorder: Preclinical and clinical evidence..” Physiology & behavior.Study breakdown →PubMed →↩
- 12RPEP-14973·Chen, Da et al. (2026). “Clinical Trial Protocol: Can GLP-1 Drugs Help People with Diabetes Quit Smoking?.” Frontiers in clinical diabetes and healthcare.Study breakdown →PubMed →↩
- 13RPEP-09966·Aranäs, Cajsa et al. (2025). “Semaglutide Suppresses Cocaine Use and Dopamine Reward Signaling.” European neuropsychopharmacology : the journal of the European College of Neuropsychopharmacology.Study breakdown →PubMed →↩
- 14RPEP-08473·Jerlhag, Elisabet (2024). “Ghrelin system and GLP-1 as potential treatment targets for alcohol use disorder..” International review of neurobiology.Study breakdown →PubMed →↩
- 15RPEP-15075·Dang, Vincent et al. (2026). “Do GLP-1 Drugs Change How the Brain Responds to Food and Reward Cues?.” bioRxiv : the preprint server for biology.Study breakdown →PubMed →↩
- 16RPEP-15153·Farokhnia, Mehdi et al. (2026). “GLP-1 Therapies for Addiction: Where the Evidence Stands and What's Needed.” JAMA psychiatry.Study breakdown →PubMed →↩