Cyclotides: Ultra-Stable Plant Peptide Scaffolds
Cyclic & Macrocyclic Peptides
280+ sequences
Over 280 cyclotide sequences have been isolated from plants, representing the largest known family of circular proteins in nature.
de Veer et al., Chemical Reviews, 2019
de Veer et al., Chemical Reviews, 2019
If you only read one thing
Cyclotides are small circular peptides made by certain plants. Their shape — a closed loop reinforced by three locking chemical bridges — makes them nearly indestructible. They survive boiling, stomach acid, and the digestive enzymes that destroy normal peptides. That matters because almost all peptide medicines today have to be injected. Researchers are using cyclotides as a template to design future oral peptide drugs. No cyclotide medication has been approved yet — everything is still in the lab or early animal testing.
Most peptides are fragile. Stomach acid degrades them. Proteolytic enzymes shred them. Heat denatures them. These vulnerabilities explain why most peptide drugs require injection rather than oral administration, and why peptide therapeutics have historically been limited to targets accessible from the bloodstream. Cyclotides break every one of these rules.
Cyclotides are a family of small plant peptides, approximately 28-37 amino acids in size, that possess two structural features making them extraordinarily resistant to degradation: a head-to-tail cyclized backbone (the peptide chain forms a complete circle with no free ends) and a cyclic cystine knot (CCK) motif formed by three interlocking disulfide bonds. This combination creates what is arguably the most stable peptide scaffold found in nature.[1]
The first cyclotide, kalata B1, was discovered in the 1970s in the plant Oldenlandia affinis, used in traditional Congolese medicine to accelerate childbirth. The active compound turned out to be a circular peptide that survived boiling in water and passage through the gastrointestinal tract while retaining its uterotonic activity. That discovery launched a field that now encompasses over 280 characterized cyclotide sequences across multiple plant families and a growing pipeline of engineered cyclotide-based drug candidates. A 2025 review described cyclotides as "ideal scaffolds for the design of novel edible drugs," reflecting the field's trajectory from natural product curiosity to pharmaceutical platform.[2]
Key Takeaways
- Cyclotides are ~30-amino-acid plant peptides with a circular backbone and three interlocking disulfide bonds forming a cyclic cystine knot (CCK) motif[1]
- The CCK structure makes cyclotides resistant to heat, acid, proteolytic enzymes, and chemical degradation, surviving conditions that destroy most peptides
- Over 280 cyclotide sequences have been isolated from plants in the Violaceae, Rubiaceae, Cucurbitaceae, Fabaceae, and Solanaceae families[2]
- Native cyclotides show antimicrobial, insecticidal, anti-HIV, and cytotoxic activities, serving as plant defense molecules
- Engineered cyclotide scaffolds have been used to create oral peptide drug candidates targeting GPCRs, intracellular protein-protein interactions, and cancer pathways[3]
- Kalata B1-based grafted cyclotides demonstrated measurable oral bioavailability in pharmacokinetic studies, approaching levels seen with some clinical peptide drugs[4]
The Cyclic Cystine Knot: Why Cyclotides Are So Stable
The structural feature that defines cyclotides and explains their exceptional stability is the cyclic cystine knot. Understanding this motif explains why cyclotides can do things other peptides cannot.
A typical cyclotide contains six conserved cysteine residues that form three disulfide bonds. Two of these disulfide bonds create a ring through which the third disulfide bond threads, forming a topological knot. This arrangement is embedded within a circular (head-to-tail cyclized) peptide backbone, creating a structure with no free N- or C-terminal ends for exopeptidases to attack.
The result is a molecule that is:
- Thermostable: cyclotides retain structure and activity after boiling
- Acid-stable: they survive gastric acid conditions (pH 1-2)
- Protease-resistant: the compact knotted structure shields the peptide backbone from enzymatic cleavage
- Chemically stable: they resist oxidation and reduction under physiological conditions
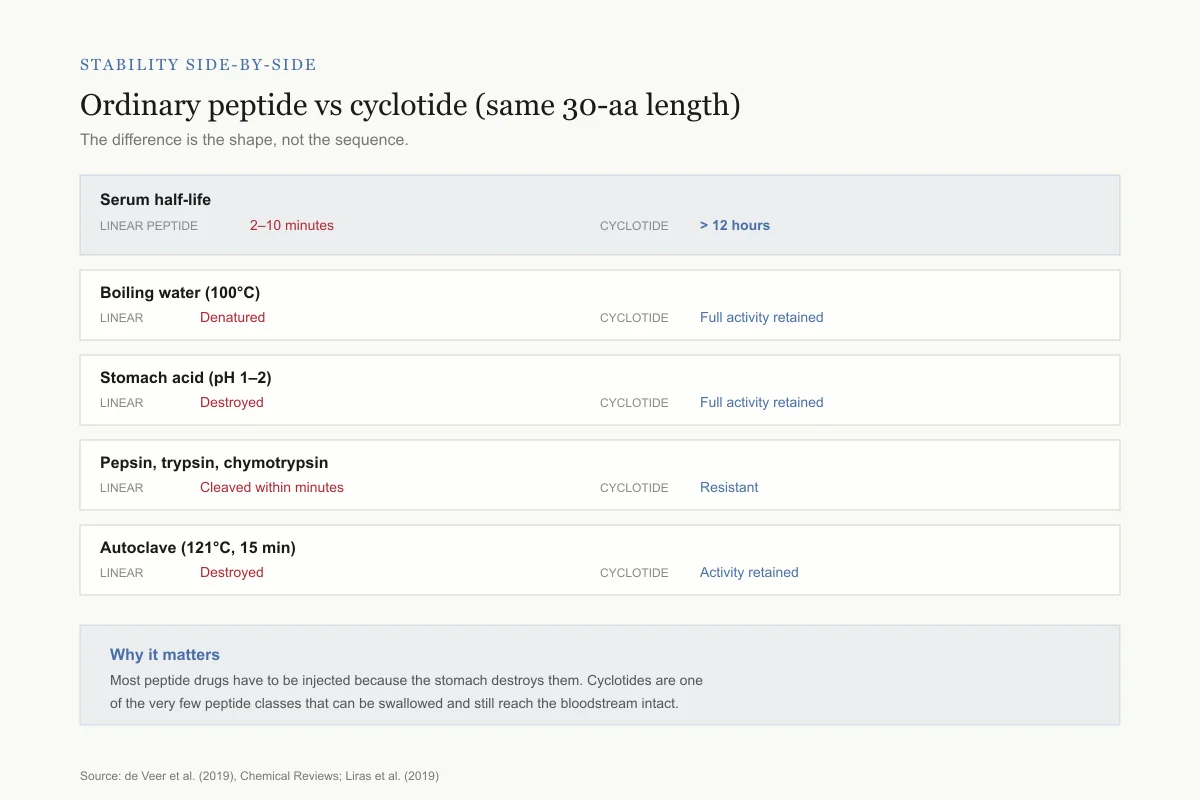
This stability profile is unmatched among peptides of similar size. Linear peptides of 30 amino acids have half-lives measured in minutes in biological fluids. Cyclotides persist for hours to days. To quantify this difference: the serum half-life of a typical linear 30-amino-acid peptide is 2-10 minutes. The serum half-life of kalata B1 exceeds 12 hours. That represents a stability improvement of roughly 100-fold, achieved entirely through structural engineering rather than chemical modification.
The stability extends to extreme conditions. Cyclotides have been shown to retain full biological activity after autoclaving (121 degrees Celsius, 15 minutes), after incubation in simulated gastric fluid (pepsin at pH 1.2 for 2 hours), and after treatment with multiple serum proteases including trypsin, chymotrypsin, and thermolysin. In comparative studies, linear analogs of cyclotides (with the same amino acid sequence but a linear rather than circular backbone) lost activity under the same conditions, confirming that the cyclic topology itself, not just the amino acid sequence, confers stability.
The practical consequence is transformative for drug design: if you can engineer a therapeutic function into a cyclotide framework, that function will survive the journey through the gastrointestinal tract, a challenge that has stymied oral peptide drug development for decades. The majority of the approximately 80 FDA-approved peptide drugs require injection. Cyclotides offer a structural solution to this delivery limitation.
It is worth noting that the CCK motif is not unique to cyclotides. Related structural motifs are found in cone snail venoms (conotoxins), spider venoms (knottins), and even in some mammalian defensins. The cyclotide-specific addition is the head-to-tail backbone cyclization, which adds exopeptidase resistance on top of the protease resistance conferred by the cystine knot alone.
For more on why cyclic peptide structures confer stability, see Cyclic Peptides: Nature's Most Stable Peptide Design. For how disulfide bridges contribute to peptide stability, see How Disulfide Bridges Lock Peptides into Stable Shapes.
Stability side-by-side
Ordinary 30-aa peptide vs a cyclotide of the same length
Same amino acid count, wildly different survival. The difference is the shape, not the sequence.
Serum half-life
Linear peptide
2–10 minutes
Cyclotide
> 12 hours
Boiling water (100°C)
Linear peptide
Denatured
Cyclotide
Full activity retained
Stomach acid (pH 1–2)
Linear peptide
Destroyed
Cyclotide
Full activity retained
Pepsin, trypsin, chymotrypsin
Linear peptide
Cleaved within minutes
Cyclotide
Resistant
Autoclave (121°C, 15 min)
Linear peptide
Destroyed
Cyclotide
Activity retained
Why it matters: Most peptide drugs have to be injected because the stomach destroys them. Cyclotides are one of the very few peptide classes that can be swallowed and still reach the bloodstream intact.
Source: de Veer et al. (2019), Chemical Reviews; Liras et al. (2019)
 View as image
View as imageWhere Cyclotides Come From
Cyclotides have been identified in at least five plant families: Violaceae (violets), Rubiaceae (coffee family), Cucurbitaceae (squash family), Fabaceae (legumes), and Solanaceae (nightshades). The Violaceae family is the richest source, with a single violet species potentially containing dozens of different cyclotides.[2]
In plants, cyclotides function primarily as defense molecules. Their insecticidal activity was among the first biological functions characterized. They disrupt the membranes of insect gut cells, causing growth inhibition and mortality in caterpillars and other herbivorous insects at concentrations found in plant tissues. This membrane-disrupting activity is mediated by a hydrophobic patch on the cyclotide surface that inserts into lipid bilayers.
Additional natural bioactivities include antimicrobial effects (activity against both gram-positive and gram-negative bacteria), anthelmintic activity (effective against intestinal parasites), and cytotoxic activity (killing mammalian cells at higher concentrations). A 2026 study reported the discovery and isolation of a new cyclotide from Spigelia anthelmia with bactericidal activity, demonstrating that new natural cyclotides continue to be identified.[5]
The diversity of cyclotides within a single plant species is notable. A single violet plant may express 50-100 different cyclotide sequences simultaneously. This natural combinatorial library likely evolved to provide broad-spectrum defense against multiple herbivores and pathogens, with each variant optimized for different target organisms or conditions. The evolutionary success of this strategy is evident: cyclotide-producing plant families span tropical and temperate regions across multiple continents, and cyclotides are estimated to be among the most abundant circular proteins in the plant kingdom, present at concentrations up to 2 g/kg of plant tissue.
The biosynthesis of cyclotides involves expression as linear precursor proteins that are enzymatically processed and cyclized. The cyclization step is catalyzed by asparaginyl endopeptidase (AEP), which cleaves the precursor and simultaneously ligates the N- and C-terminal ends to form the circular backbone. Understanding this biosynthetic pathway has enabled recombinant production of cyclotides in plant expression systems, potentially reducing manufacturing costs compared to chemical synthesis.
For more on antimicrobial peptides from natural sources, see Antimicrobial Peptides as Alternatives to Antibiotics: Can They Solve Resistance?.
Cyclotides as Drug Design Scaffolds
The property that has generated the most pharmaceutical interest in cyclotides is not their natural bioactivity but their structural tolerance. Cyclotides can accept substantial amino acid substitutions in the loops between their cysteine residues while retaining the CCK fold and its associated stability. This makes them ideal scaffolds for "molecular grafting," the process of inserting a bioactive peptide sequence into the cyclotide framework to create a stable, orally bioavailable therapeutic.
The Grafting Approach
Craik and colleagues pioneered the concept of using cyclotides as drug scaffolds in the early 2010s.[6] The approach works as follows: identify a peptide sequence with a desired biological activity (e.g., binding to a specific receptor), then replace one of the cyclotide's inter-cysteine loops with that sequence. The resulting grafted cyclotide retains the stability of the parent scaffold while acquiring the biological activity of the inserted sequence.
This has been demonstrated for multiple therapeutic targets:
GPCR ligands. Muratspahic and colleagues showed that cyclotide scaffolds can be engineered to contain peptide sequences that bind G protein-coupled receptors, creating stable, potentially orally bioavailable receptor agonists or antagonists. Grafted cyclotides targeting melanocortin and bradykinin receptors have been reported.[3]
Intracellular targets. One of cyclotides' most remarkable properties is their ability to penetrate cell membranes. Philippe and colleagues demonstrated a grafted cyclotide conjugate that delivered a p53-reactivating peptide into cancer cells, restoring tumor suppressor function. This represents a significant advance because most peptide therapeutics cannot reach intracellular targets.[7]
Protease inhibitors. An ultrapotent cyclic peptide inhibitor of human beta-Factor XIIa was developed using a cyclotide scaffold, achieving picomolar-range inhibition with the stability to survive in vivo conditions. Factor XIIa is a serine protease involved in blood coagulation, and its inhibition has therapeutic relevance for thrombotic disorders.
Anti-HIV activity. Several native cyclotides, particularly those from the Möbius subfamily, have demonstrated anti-HIV activity in vitro. The mechanism involves disruption of the viral envelope through the same membrane-interactive hydrophobic patch that mediates insecticidal activity. While the concentrations required for anti-HIV activity are too high for direct therapeutic use, the activity demonstrates that the cyclotide scaffold can interact with lipid membranes of diverse composition.
Cancer applications. Beyond the p53-reactivating grafted cyclotide, researchers have explored cyclotides as cytotoxic agents that selectively target cancer cell membranes. Cancer cells typically have altered membrane composition (increased phosphatidylserine exposure on the outer leaflet) compared to healthy cells, and some cyclotides show preferential interaction with these altered membranes. Grafted cyclotides carrying chemotherapy-sensitizing peptide sequences represent another approach, using the cyclotide as a delivery vehicle to enhance existing cancer therapies rather than as a standalone cytotoxic agent.
Chronic pain. Grafted cyclotides targeting bradykinin B1 receptors have been developed for chronic inflammatory pain, an application where oral bioavailability would be a significant advantage over injectable peptide analgesics. The bradykinin-targeting grafted cyclotides retained receptor binding affinity while gaining the stability and oral activity of the cyclotide scaffold.
Chemical Synthesis of Grafted Cyclotides
The practical challenge of making grafted cyclotides was addressed by Koehbach and colleagues, who developed a "plug and play" chemical synthesis approach allowing modular insertion of different bioactive sequences into the cyclotide framework. This method streamlined the production of diverse grafted cyclotides for screening and optimization.[8]
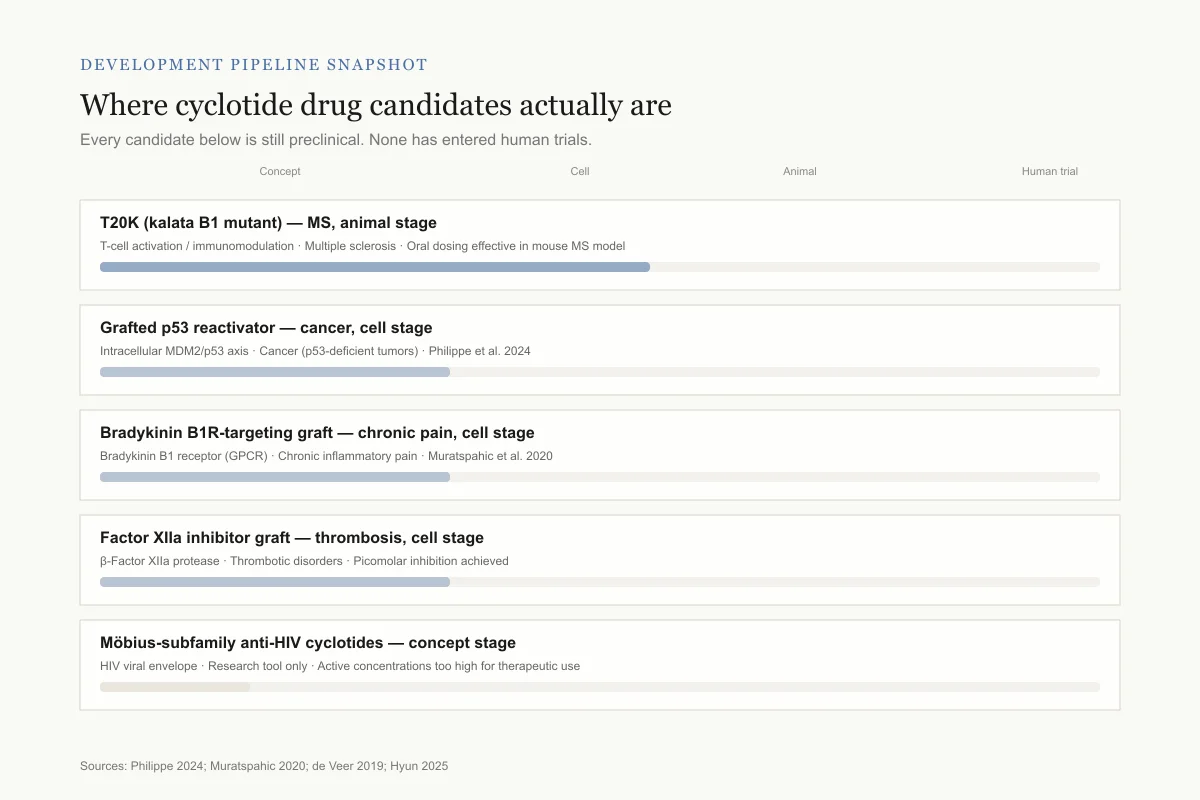
Development pipeline snapshot
Where cyclotide drug candidates actually are
Every candidate below is still preclinical. No cyclotide has entered human trials.
T20K (kalata B1 mutant)
Animal studiesTarget: T-cell activation / immunomodulation · Indication: Multiple sclerosis
Oral dosing effective in mouse MS model
Grafted cyclotide — p53 reactivator
Cell studiesTarget: Intracellular MDM2/p53 axis · Indication: Cancer (p53-deficient tumors)
Philippe et al. 2024
Bradykinin B1R-targeting graft
Cell studiesTarget: Bradykinin B1 receptor (GPCR) · Indication: Chronic inflammatory pain
Muratspahic et al. 2020
Factor XIIa inhibitor graft
Cell studiesTarget: Human β-Factor XIIa protease · Indication: Thrombotic disorders
Picomolar inhibition achieved
Native Möbius-subfamily cyclotides
ConceptTarget: HIV viral envelope · Indication: Anti-HIV (research tool only)
Active concentrations too high for therapeutic use
Biggest unknown: manufacturing. Making a correctly folded, triple-disulfide cyclic peptide at pharmaceutical scale is technically difficult and expensive — which is part of why no candidate has broken through to the clinic yet.
Sources: Philippe 2024; Muratspahic 2020; de Veer 2019; Hyun 2025
 View as image
View as imageOral Bioavailability: The Key Advantage
The most significant practical advantage of cyclotide-based therapeutics over conventional peptide drugs is oral bioavailability. The vast majority of peptide drugs on the market require subcutaneous or intravenous injection because oral administration results in complete degradation in the gastrointestinal tract. Cyclotides resist this degradation.
Liras and colleagues analyzed the permeability of cyclic peptide macrocycles including cyclotides, finding that the cyclic cystine knot structure conferred membrane permeability alongside protease resistance, two properties that are typically difficult to achieve simultaneously in peptide drug design.[4]
Pharmacokinetic studies of kalata B1 and grafted cyclotides demonstrated measurable oral bioavailability at levels comparable to several clinically used oral peptide drugs, including cyclosporine. While absolute oral bioavailability remains lower than most small molecule drugs, the fact that any intact peptide reaches systemic circulation after oral dosing is remarkable for a molecule of this size.
The most advanced example is T20K, a mutant of kalata B1, which demonstrated oral efficacy in a mouse model of multiple sclerosis. Oral dosing at 20 mg/kg impeded disease progression without observable adverse effects. T20K's immunomodulatory mechanism involves modulation of T-cell activation, and its ability to achieve therapeutic effects through oral dosing represents a proof of concept for cyclotide-based oral peptide therapeutics.
Wang and colleagues characterized the biodistribution of the cyclotide MCoTI-II after systemic administration, showing tissue-specific distribution patterns relevant to understanding how cyclotide-based drugs would reach their targets in vivo. MCoTI-II accumulated preferentially in the kidney and liver, with lower distribution to brain, heart, and lung tissue. The biodistribution profile has implications for drug design: cyclotide-based therapeutics targeting liver or kidney diseases may benefit from natural accumulation, while targeting the central nervous system would require additional engineering to cross the blood-brain barrier.[9]
The cell-penetrating property of cyclotides is particularly important for the growing field of intracellular peptide therapeutics. Most peptide drugs act on extracellular targets (cell surface receptors, circulating proteins) because they cannot cross cell membranes. Cyclotides' ability to penetrate membranes through their hydrophobic patch opens the possibility of targeting intracellular protein-protein interactions, a drug target class that has historically been considered "undruggable" by peptide approaches. The p53-reactivating grafted cyclotide reported by Philippe and colleagues is a direct example of this capability.[7]
Cyclotides as Research Tools
Beyond drug development, cyclotides serve as valuable tools in chemical biology. De Veer and colleagues reviewed cyclotides' utility as tools for probing biological systems, including their use as molecular probes for studying membrane dynamics, protease specificity, and protein-protein interactions. Their stability in complex biological matrices makes them suitable for experiments where conventional peptide probes would be degraded.[10]
Cyclotides have also been explored for agricultural applications. Their natural insecticidal activity could potentially be harnessed for crop protection, either through direct application or by engineering crop plants to produce cyclotides as endogenous pest resistance factors. The fact that cyclotides are already present in food plants (some Cucurbitaceae species) provides a baseline safety argument, though regulatory approval for transgenic cyclotide-producing crops would require extensive safety testing.
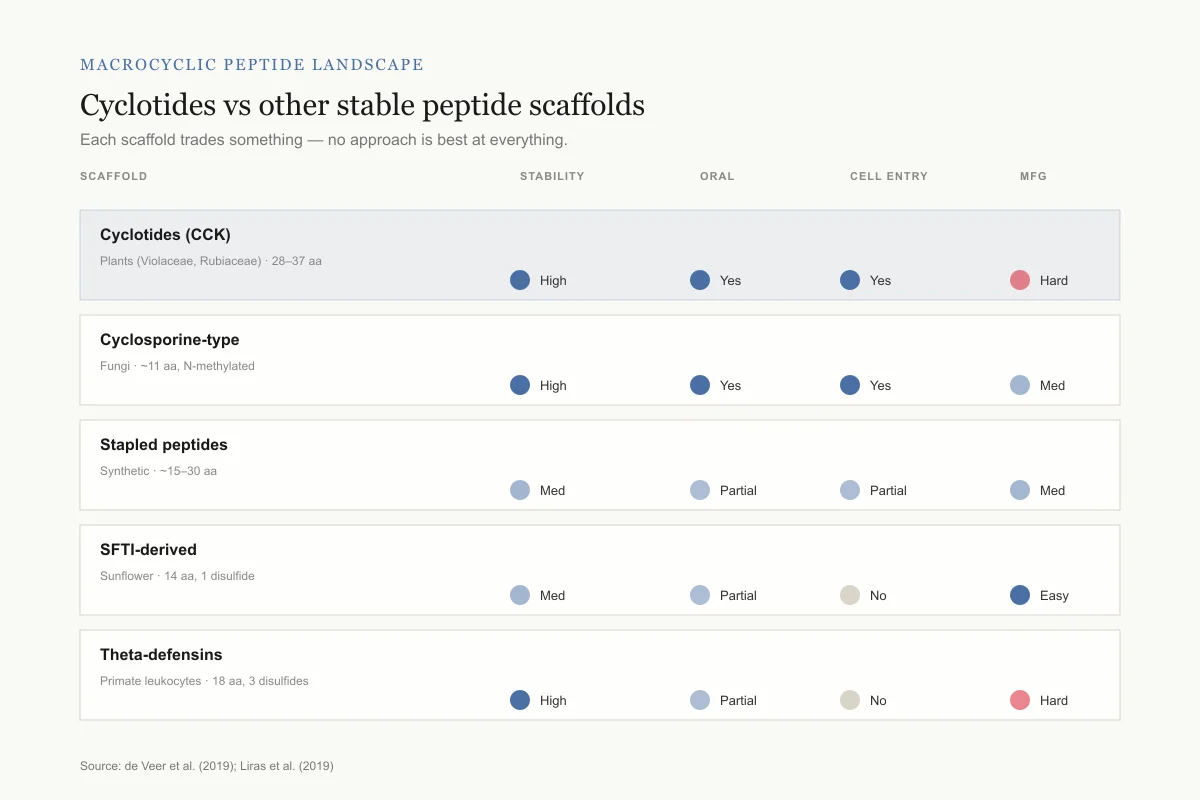
Macrocyclic peptide landscape
Cyclotides vs the other ways to make a stable peptide drug
Each scaffold trades something — no approach is best at everything.
Cyclotides (CCK)
Plants (Violaceae, Rubiaceae) · 28–37 aaCyclosporine-type
Fungi · ~11 aa, N-methylatedStapled peptides
Synthetic · ~15–30 aaSFTI-derived
Sunflower · 14 aa, 1 disulfideTheta-defensins
Primate leukocytes · 18 aa, 3 disulfidesThe cyclotide bet: best all-around profile for stability, oral dosing, and getting inside cells — but the hardest to manufacture. That trade-off is what the field is trying to solve.
Source: de Veer et al. (2019); Liras et al. (2019)
 View as image
View as imageCyclotides in Context: The Macrocyclic Peptide Landscape
Cyclotides represent one approach within the broader field of macrocyclic peptide therapeutics. For how cyclotides compare to other macrocyclic peptide strategies, see Macrocyclic Peptides: The Sweet Spot Between Small Molecules and Biologics.
The macrocyclic peptide space includes:
- Cyclotides: plant-derived, CCK motif, 28-37 amino acids, naturally orally bioavailable
- Cyclosporine-type macrocycles: fungal-derived, N-methylated backbone, oral bioavailability through passive permeability
- Stapled peptides: synthetic, hydrocarbon staple reinforces alpha-helical structure
- SFTI-derived scaffolds: sunflower-derived, smaller (14 amino acids), single disulfide
- Theta-defensins: mammalian, cyclic backbone, three disulfides (structurally similar to cyclotides)
Cyclotides' competitive advantage lies in their combination of stability, cell penetration, oral bioavailability, and tolerance to sequence grafting. Their primary limitation is manufacturing cost: chemical synthesis of cyclic, disulfide-rich peptides is more complex and expensive than linear peptide synthesis. The requirement for correct formation of three specific disulfide bonds and the cyclization step adds complexity compared to linear peptide manufacturing, where solid-phase synthesis is well-established at commercial scale.
Each macrocyclic approach has trade-offs. Cyclosporine-type macrocycles achieve oral bioavailability through N-methylation and backbone modifications but are limited to specific structural classes. Stapled peptides maintain alpha-helical structure but have more limited stability than cyclotides. SFTI-derived scaffolds are simpler to synthesize but accommodate smaller payload sequences. Cyclotides offer the broadest combination of favorable properties but at the highest manufacturing complexity. The optimal scaffold depends on the specific therapeutic application, target location (extracellular vs. intracellular), required stability, and commercial feasibility.
Safety
ModerateCyclotides can damage membranes too
Concern
The same membrane-insertion property that lets cyclotides kill insects and some microbes also makes native cyclotides cytotoxic to human cells at micromolar concentrations. A grafted drug candidate can inherit some of this activity.
What the research says
Drug designers carefully select parent scaffolds with low membrane activity and engineer grafts to minimize off-target membrane effects. This is a real but manageable engineering problem.
Particularly relevant for: Relevant to cyclotide drug developers, not to current patients (no cyclotide drugs are on the market)
What to do
Watch this space — the first human safety trials will be the critical test of whether the therapeutic window is usable.
de Veer et al. (2019), Chemical Reviews
Limitations and Open Questions
No cyclotide-based drug has reached human clinical trials. T20K, the most advanced candidate, has been tested in mouse models but not in humans. The gap between preclinical proof of concept and clinical development is substantial.
Manufacturing scalability is unresolved. Chemical synthesis of cyclotides is feasible at research scale but has not been demonstrated at commercial pharmaceutical manufacturing scale. Recombinant production in plants or microorganisms is being explored but faces its own challenges.
Toxicity from membrane activity. The same membrane-disrupting activity that gives cyclotides their natural insecticidal and antimicrobial functions raises safety questions for human therapeutics. Native cyclotides are cytotoxic at micromolar concentrations. Grafted cyclotides designed for non-membrane targets may retain some of this membrane activity, requiring careful therapeutic index optimization.
Immunogenicity is uncharacterized in humans. Whether cyclotides trigger immune responses after repeated administration is unknown. Their small size argues against strong immunogenicity, but their unique circular structure is foreign to the human immune system.
Oral bioavailability, while demonstrated, remains limited. Absolute oral bioavailability of cyclotide scaffolds is in the low single-digit percentage range. For potent molecules where the target is engaged at nanomolar concentrations, this may be sufficient. For less potent candidates, oral dosing may require impractically high doses. Formulation strategies including enteric coatings, permeation enhancers, and nanoparticle encapsulation are being explored to improve oral delivery.
Structure-activity relationships are incompletely mapped. While the CCK framework is well-characterized structurally, predicting how a specific grafted sequence will affect the overall fold, stability, and biological activity of the resulting chimeric cyclotide remains partially empirical. High-throughput screening methods help, but rational design of grafted cyclotides still requires iterative optimization.
The Bottom Line
Cyclotides are circular plant peptides of approximately 30 amino acids stabilized by a cyclic cystine knot motif that makes them resistant to heat, acid, and enzymatic degradation. Over 280 sequences have been identified across multiple plant families. Their exceptional stability and tolerance to sequence substitution have made them attractive drug design scaffolds, with engineered cyclotides targeting GPCRs, intracellular protein-protein interactions, and cancer pathways. Kalata B1-based grafted cyclotides have demonstrated oral bioavailability in preclinical studies, but no cyclotide-based drug has yet entered human clinical trials.
Sources & References
- 1RPEP-04145·de Veer, Simon J et al. (2019). “Cyclotides: From Structure to Function..” Chemical reviews.Study breakdown →PubMed →↩
- 2RPEP-11493·Hyun, Youbong (2025). “Cyclotides as novel plant-derived scaffolds for orally active cyclic peptide therapeutics..” Molecules and cells.Study breakdown →PubMed →↩
- 3RPEP-05017·Muratspahić, Edin et al. (2020). “Cyclotide Scaffolds Enable Orally Active Peptide Drugs Targeting GPCRs.” RSC chemical biology.Study breakdown →PubMed →↩
- 4RPEP-04342·Liras, Spiros et al. (2019). “Permeability of Cyclic Peptide Macrocycles and Cyclotides and Their Potential as Therapeutics..” ACS medicinal chemistry letters.Study breakdown →PubMed →↩
- 5RPEP-14717·Akinleye, Toluwanimi E et al. (2026). “Discovery, Isolation, and Bactericidal Activity of a Cyclotide from Spigelia anthelmia L. (Loganiaceae)..” Journal of natural products.Study breakdown →PubMed →↩
- 6RPEP-01922·Craik, David J et al. (2012). “Cyclotides as a basis for drug design..” Expert opinion on drug discovery.Study breakdown →PubMed →↩
- 7RPEP-09070·Philippe, Grégoire Jean-Baptiste et al. (2024). “Delivery to, and Reactivation of, the p53 Pathway in Cancer Cells Using a Grafted Cyclotide Conjugated with a Cell-Penetrating Peptide..” Journal of medicinal chemistry.Study breakdown →PubMed →↩
- 8RPEP-08573·Koehbach, Johannes et al. (2024). “A New 'Plug and Play' Method for Building Cyclotide-Based Peptide Drugs.” RSC chemical biology.Study breakdown →PubMed →↩
- 9RPEP-03153·Wang, Conan K et al. (2016). “Biodistribution of the cyclotide MCoTI-II, a cyclic disulfide-rich peptide drug scaffold..” Journal of peptide science : an official publication of the European Peptide Society.Study breakdown →PubMed →↩
- 10RPEP-03263·de Veer, Simon J et al. (2017). “Cyclotides as Tools in Chemical Biology..” Accounts of chemical research.Study breakdown →PubMed →↩