Chromogranin A: The Neuroendocrine Tumor Marker
Peptide Biomarkers
439 amino acids
Chromogranin A is a 439-amino-acid glycoprotein stored in the secretory granules of neuroendocrine cells, serving as both the primary blood biomarker for neuroendocrine tumors and a precursor to bioactive peptides.
Angelone et al., Clinica Chimica Acta, 2012
Angelone et al., Clinica Chimica Acta, 2012
If you only read one thing
Chromogranin A is a protein your body's hormone-secreting cells release into the blood. Doctors use it as a tumor marker because neuroendocrine tumors pump out a lot of it. The catch: acid-suppressing drugs like omeprazole push CgA levels sky-high in people who don't have cancer at all — over 80% of PPI users get elevated readings. The most useful thing CgA can do is track how an already-diagnosed tumor is responding to treatment, not screen healthy people.
Chromogranin A (CgA) is a 439-amino-acid glycoprotein found inside the secretory granules of neuroendocrine cells throughout the body. When neuroendocrine cells release their contents, CgA enters the bloodstream alongside hormones, neuropeptides, and catecholamines. That release makes CgA measurable in blood, and elevated levels became the first widely used circulating biomarker for neuroendocrine tumors (NETs). CgA remains the most commonly ordered blood test in NET diagnosis and monitoring, despite well-documented limitations in specificity.
But CgA is more than a tumor marker. The protein is a prohormone, cleaved into at least five bioactive peptide fragments, including catestatin (a catecholamine release inhibitor), vasostatin (a vasorelaxant), and pancreastatin (a regulator of insulin secretion and glucose metabolism). These derived peptides have independent biological functions in cardiovascular regulation, immune modulation, and metabolic homeostasis. CgA sits at an intersection of oncology, cardiology, and peptide biology. Understanding it as both a clinical tool and a biological molecule clarifies why a single blood test carries so much clinical weight, and why its interpretation requires more nuance than most biomarkers. For context on other peptide-based cancer therapies targeting neuroendocrine tumors, see Anticancer Peptides: How They Selectively Kill Tumor Cells.
Key Takeaways
- Chromogranin A is a 439-amino-acid glycoprotein stored in neuroendocrine secretory granules, released alongside hormones and catecholamines[1]
- CgA is the most widely used circulating biomarker for neuroendocrine tumors, with higher sensitivity than neuron-specific enolase or alpha-subunit of glycoprotein hormones
- Proton pump inhibitors cause false-positive CgA elevations in over 80% of PPI users, with levels that can mimic malignancy; CgA drops 82% after PPI discontinuation
- Serial CgA measurements (changes greater than 17%) predict treatment response versus disease progression with 91.2% sensitivity and 82.9% specificity
- CgA is a prohormone that yields at least five bioactive peptides: catestatin, vasostatin-I/II, pancreastatin, chromofungin, and serpinins (Helle et al., Cardiovascular Research, 2010)
- Catestatin, a CgA-derived peptide, has emerged as an independent research target in hypertension, heart failure, and antimicrobial defense[2]
What Chromogranin A Is
Chromogranin A belongs to the granin family of acidic secretory proteins. Granins are stored inside dense-core secretory granules, the specialized vesicles that neuroendocrine cells use to package and release hormones, neuropeptides, and catecholamines. CgA is the most abundant granin and constitutes approximately 40% of the soluble protein content in adrenal medullary chromaffin cells.[1]
The protein was first identified in chromaffin granules of the adrenal medulla (hence "chromogranin"), but it is expressed across a wide range of neuroendocrine tissues: the adrenal medulla, the enterochromaffin-like (ECL) cells of the gastric mucosa, the pancreatic islets, the anterior pituitary, the thyroid C cells, and the parathyroid glands. It is also found in neurons throughout the central and peripheral nervous systems. This broad tissue distribution is why CgA levels can reflect neuroendocrine activity from multiple organ systems, and why interpreting elevated results requires clinical context about potential non-tumor sources.
CgA serves at least three known functions inside secretory granules. First, it participates in granule biogenesis, helping to form and stabilize the dense-core vesicle structure. Second, it binds calcium ions within granules, contributing to the high intragranular calcium concentration needed for hormone packaging. Third, upon release from the cell, CgA is cleaved by proteases into bioactive peptide fragments that exert their own physiological effects on the cardiovascular system, immune cells, and metabolic regulation.
CgA as a Tumor Biomarker
Neuroendocrine tumors arise from neuroendocrine cells and retain their secretory machinery, including CgA-containing granules. As NET cells proliferate and secrete, CgA accumulates in the bloodstream at concentrations proportional to tumor burden. This relationship between tumor mass and circulating CgA is the basis for its use as a biomarker.
Diagnostic Use
CgA is typically measured via immunoassay from a serum or plasma sample. Elevated CgA levels support a diagnosis of neuroendocrine neoplasm, particularly when combined with imaging findings and clinical symptoms. CgA has higher diagnostic sensitivity for NETs than alternative markers including neuron-specific enolase (NSE) and the alpha-subunit of glycoprotein hormones.
Sensitivity varies by tumor type and grade. Well-differentiated, low-grade gastroenteropancreatic NETs (GEP-NETs) produce CgA reliably. Poorly differentiated neuroendocrine carcinomas may not express CgA as consistently. Functioning tumors (those that produce hormones like serotonin, insulin, or gastrin) tend to have higher CgA levels than non-functioning tumors of the same size.
For NETs specifically, CgA elevation correlates with several clinically relevant features: tumor size, degree of differentiation, presence of liver metastases, and overall disease burden. Patients with metastatic disease typically have higher CgA levels than those with localized tumors.
CgA levels differ across NET subtypes. Pheochromocytomas and paragangliomas (tumors of the adrenal medulla and sympathetic ganglia) often produce extremely high CgA levels because chromaffin cells are dense with secretory granules. Midgut carcinoid tumors, the classic "carcinoid" tumors that can cause carcinoid syndrome with flushing and diarrhea, reliably elevate CgA in proportion to disease volume. Pancreatic NETs are more heterogeneous in their CgA secretion: insulinomas (which produce insulin) tend to have lower CgA levels than gastrinomas or non-functioning pancreatic NETs of similar size. Small-cell lung cancer, a high-grade neuroendocrine carcinoma, may elevate CgA but less consistently than well-differentiated tumors, and neuron-specific enolase tends to be a more useful marker in that context.
The WHO classification system grades neuroendocrine neoplasms (NENs) based on proliferation rate (Ki-67 index and mitotic count). Grade 1 (low-grade, Ki-67 less than 3%) and Grade 2 (intermediate, Ki-67 3-20%) tumors tend to express CgA robustly. Grade 3 NETs and poorly differentiated neuroendocrine carcinomas (Ki-67 above 20%) may have reduced CgA expression, making the marker less reliable in aggressive disease precisely when prognostic information is most needed.
Monitoring Treatment Response
Where CgA adds the most value is in serial monitoring during treatment. A single CgA level has limited diagnostic specificity, but the trend over time is clinically actionable. Research has demonstrated that a change in paired CgA measurements (delta CgA) of greater than 17% can predict partial response or stable disease versus progressive disease with 91.2% sensitivity and 82.9% specificity.
This makes CgA most useful not for initial diagnosis (where imaging and pathology are primary tools) but for tracking disease trajectory. Rising CgA during treatment suggests progression. Falling CgA suggests response. A greater than 50% reduction in CgA is commonly used as a biochemical response criterion in clinical trials evaluating NET therapies, including peptide receptor radionuclide therapy (PRRT). For more on PRRT in neuroendocrine tumors, see Alpha-Emitter PRRT: The Next Generation of Radioactive Peptides.
Prognostic Value
Baseline CgA levels at diagnosis carry prognostic information. Multiple studies have demonstrated that higher baseline CgA is associated with shorter overall survival in GEP-NET patients. A 2014 study reported that plasma CgA levels independently predicted survival and tumor response in patients with advanced GEP-NETs, even after adjusting for tumor grade and extent of disease.
The False Positive Problem: Proton Pump Inhibitors
Not Every Elevated CgA Means Cancer
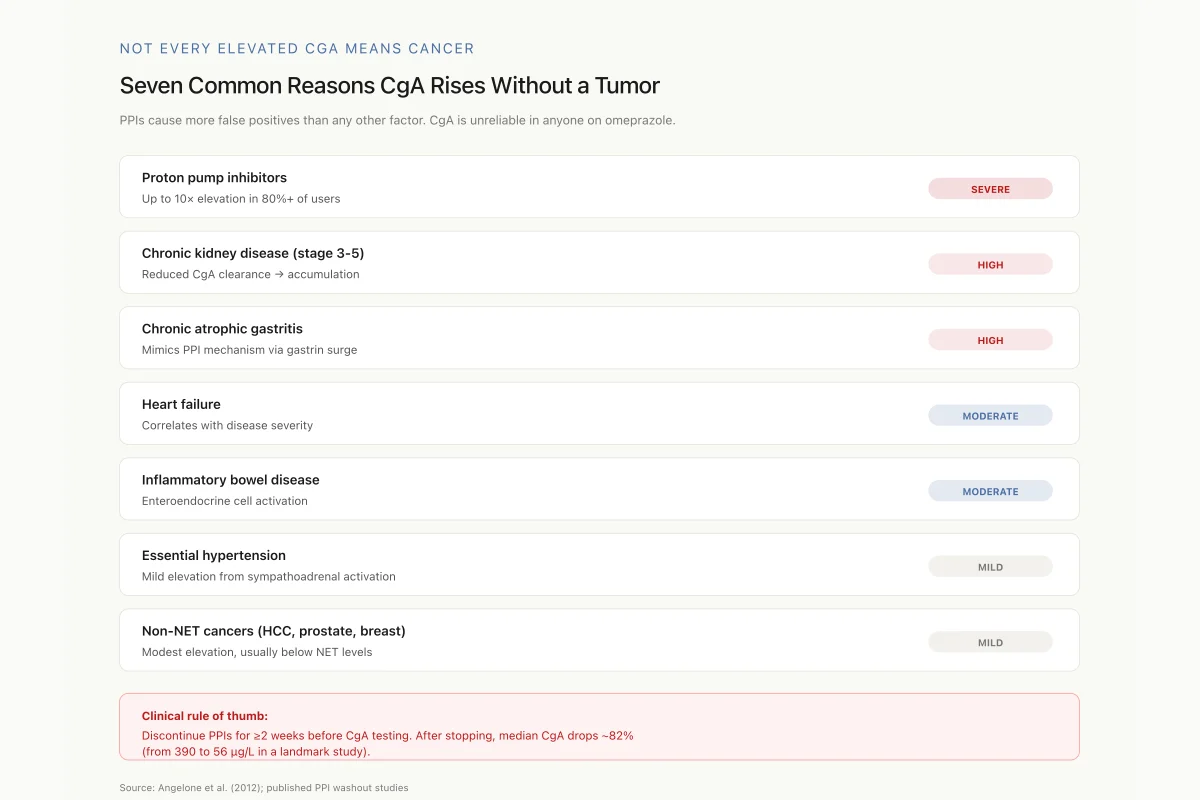
Seven Common Reasons CgA Rises Without a Tumor
PPIs alone cause more false positives than any other factor. CgA is not reliable in someone taking omeprazole, esomeprazole, or lansoprazole.
Proton pump inhibitors
Up to 10× elevation in 80%+ of users
Chronic kidney disease (stage 3-5)
Reduced CgA clearance → accumulation
Chronic atrophic gastritis
Mimics PPI mechanism via gastrin surge
Heart failure
Correlates with disease severity
Inflammatory bowel disease
Enteroendocrine cell activation
Essential hypertension
Mild elevation from sympathoadrenal activation
Non-NET cancers (HCC, prostate, breast)
Modest elevation, usually below NET levels
Clinical rule of thumb: Discontinue PPIs for ≥2 weeks before CgA testing. After stopping PPIs, median CgA drops ~82% (from 390 µg/L to 56 µg/L in a landmark study).
Source: Angelone et al. (2012); published PPI washout studies
 View as image
View as imageThe single most important clinical pitfall with CgA testing is its elevation by proton pump inhibitors (PPIs) such as omeprazole, esomeprazole, and lansoprazole. PPIs are among the most widely prescribed medications globally, and their effect on CgA levels is dramatic.
PPIs suppress gastric acid production by inhibiting the proton pump in parietal cells. The resulting hypochlorhydria (low stomach acid) triggers a compensatory increase in gastrin secretion from G cells, which in turn stimulates proliferation of ECL cells in the gastric mucosa. ECL cells are neuroendocrine cells that store and release CgA. The proliferative stimulus from elevated gastrin increases both ECL cell mass and CgA secretion, sometimes producing CgA levels that mimic or exceed those seen in neuroendocrine tumors.
The numbers are stark. In PPI-treated individuals, CgA is elevated in over 80% of cases. Case reports have documented ten-fold CgA elevations from PPI use alone, in patients with no neuroendocrine pathology. In one study, patients on PPIs with no evidence of neuroendocrine disease had a median CgA of 390 micrograms per liter. After PPI discontinuation, median CgA fell to 56 micrograms per liter, an 82% reduction. In patients who switched from PPIs to H2-receptor antagonists (like ranitidine or famotidine), CgA decreased by 77%.
The clinical consequence: any CgA measurement in a patient taking a PPI is unreliable for neuroendocrine tumor evaluation. Current guidelines recommend discontinuing PPIs for at least two weeks (some experts suggest three or more weeks) before measuring CgA. When PPI discontinuation is not clinically feasible, CgA results must be interpreted with extreme caution, and alternative markers or imaging should guide decision-making.
This is not a minor caveat. PPI use is extraordinarily common. Approximately 15% of adults in the United States take a PPI, many of them long-term and some over-the-counter without physician oversight. The overlap between the PPI-using population and patients undergoing workup for GI symptoms (which could include NET evaluation) is substantial.
Safety
ModerateDon't interpret a CgA result while on a PPI
Concern
In PPI users, median CgA is ~390 µg/L. After stopping the PPI for at least two weeks, that drops to ~56 µg/L — an 82% reduction, in people with no neuroendocrine disease at all. Acting on a CgA taken during PPI use can lead to unnecessary imaging, biopsies, or anxiety.
What the research says
Guidelines recommend stopping PPIs for ≥2 weeks (some experts say 3+) before CgA testing. H2 blockers like famotidine are a short-term alternative if acid control is still needed — they raise CgA far less.
Particularly relevant for: Anyone on omeprazole, esomeprazole, lansoprazole, or related PPIs undergoing NET workup
What to do
Before ordering or interpreting CgA, verify PPI status and washout duration. If PPI can't be stopped, rely on imaging (68Ga-DOTATATE PET) and alternative biomarkers.
Published CgA-PPI pharmacology literature
Other Causes of CgA Elevation
PPIs are the most common cause of false-positive CgA, but not the only one. Other conditions that elevate CgA include:
Renal impairment. CgA is cleared by the kidneys. Reduced glomerular filtration rate leads to CgA accumulation, with levels correlating inversely with kidney function. Chronic kidney disease stages 3-5 can produce clinically significant CgA elevation.
Chronic atrophic gastritis. Autoimmune or Helicobacter pylori-associated destruction of parietal cells mimics the PPI effect: reduced acid production leads to compensatory gastrin elevation, ECL cell hyperplasia, and increased CgA secretion.
Heart failure. CgA is elevated in heart failure and correlates with disease severity. This reflects the neuroendocrine activation that characterizes heart failure pathophysiology. The elevation is clinically relevant because CgA-derived peptides (catestatin, vasostatin) have direct cardiovascular effects.[1] For other peptide biomarkers used in heart failure, see BNP and NT-proBNP: How Heart Failure Is Diagnosed with Peptide Biomarkers.
Inflammatory bowel disease. Both Crohn's disease and ulcerative colitis can elevate CgA through enteroendocrine cell activation.
Essential hypertension. Mild CgA elevation is observed in some hypertensive patients, consistent with sympathoadrenal activation.
Other neoplasms. Certain non-neuroendocrine cancers (hepatocellular carcinoma, prostate cancer, breast cancer) can produce modest CgA elevations, though typically lower than those seen with NETs.
The composite effect of these confounders is that CgA has modest diagnostic specificity when used in isolation. It functions best as one component of a multimodal diagnostic approach and is most reliable for monitoring established disease rather than screening or initial diagnosis.
Chromogranin A as a Prohormone: The Bioactive Peptides
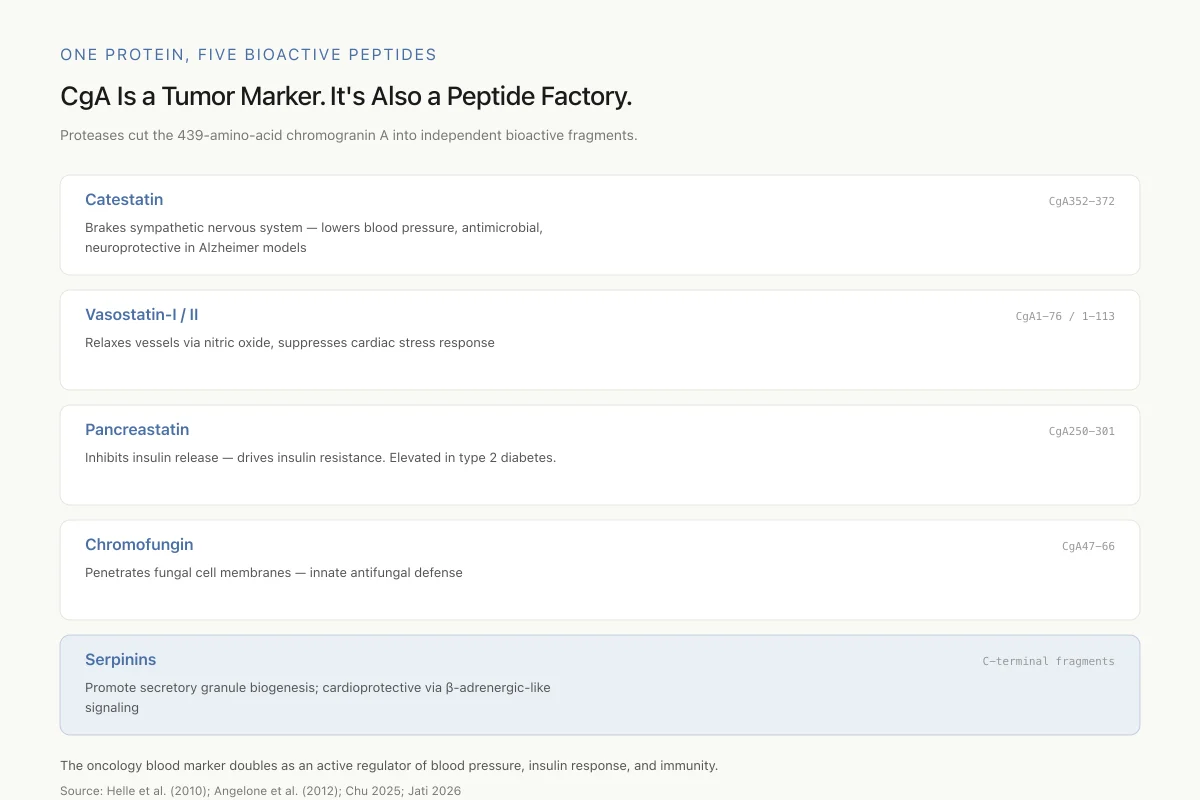
One Protein, Five Bioactive Peptides
CgA Is a Tumor Marker. It’s Also a Peptide Factory.
Proteases cut the 439-amino-acid chromogranin A into independent bioactive fragments. Their effects reach far beyond oncology.
Brakes sympathetic nervous system — lowers blood pressure, antimicrobial, neuroprotective in Alzheimer models
Relaxes vessels via nitric oxide, suppresses cardiac stress response
Inhibits insulin release — drives insulin resistance. Elevated in type 2 diabetes.
Penetrates fungal cell membranes — innate antifungal defense
Promote secretory granule biogenesis; cardioprotective via β-adrenergic-like signaling
The same protein that oncologists use to track neuroendocrine tumors is actively shaping blood pressure, insulin response, immunity, and neurodegeneration — depending on which fragment is released.
Source: Helle et al. (2010); Angelone et al. (2012); Chu 2025; Jati 2026
 View as image
View as imageCgA's role extends well beyond tumor markers. The protein contains multiple cleavage sites recognized by prohormone convertases and other proteases. Depending on the cell type and its enzymatic machinery, CgA is processed into different combinations of bioactive peptides. At least five have been characterized:
Catestatin (CgA352-372)
Catestatin is the most extensively studied CgA-derived peptide. This 21-amino-acid fragment acts as a potent inhibitor of catecholamine release from chromaffin cells. By suppressing norepinephrine and epinephrine secretion, catestatin functions as an endogenous brake on sympathoadrenal activity. In clinical studies, reduced plasma catestatin levels are associated with hypertension, and catestatin supplementation lowers blood pressure in animal models.[2]
Beyond cardiovascular regulation, catestatin has demonstrated antimicrobial activity. A 2026 study showed that catestatin promotes clearance of cutaneous Staphylococcus aureus infections, suggesting an innate immune defense function.[3] Catestatin has also been identified as a predictor of cardiac death in heart failure patients with mildly reduced or preserved ejection fraction, positioning it as both a biomarker and a potential therapeutic target.[2]
A 2026 study further expanded catestatin's known biology by demonstrating that it ameliorates tauopathy and amyloidogenesis (hallmarks of Alzheimer's disease) through adrenergic inhibition, suggesting a neuroprotective role.[4]
Vasostatin-I and Vasostatin-II (CgA1-76 and CgA1-113)
The vasostatin peptides are derived from CgA's N-terminal region. They exert vasorelaxant effects through calcium-independent, PI3-kinase-dependent nitric oxide release from endothelial cells. Vasostatin-I and -II also have cardiosuppressive and antiadrenergic properties, contributing to cardiac stabilization under both normal and stress conditions (Helle et al., Cardiovascular Research, 2010).
A 2025 study demonstrated that vasostatin-2 attenuates injury-induced neointimal hyperplasia (the excessive cell growth that narrows blood vessels after injury) through the ACE2/MasR/PPARgamma/NF-kappaB signaling axis, identifying a new vascular protective mechanism.[5]
Pancreastatin (CgA250-301)
Pancreastatin is a 52-amino-acid peptide that inhibits insulin secretion from pancreatic beta cells and inhibits glucose-stimulated insulin release. It has been described as a "dysglycemic" peptide because its effects oppose glucose homeostasis. Pancreastatin levels are elevated in type 2 diabetes and metabolic syndrome, and it has emerged alongside catestatin as a therapeutic target in diabetes research. The pancreastatin-catestatin axis represents an internal regulatory balance within CgA biology: pancreastatin promotes insulin resistance while catestatin enhances insulin sensitivity. Their relative concentrations may influence metabolic outcomes, though this remains an area of active investigation.
Other Derived Peptides
Chromofungin (CgA47-66) has antifungal activity and can penetrate fungal cell membranes. Serpinins (derived from CgA) promote secretory granule biogenesis and exert cardioprotective effects through beta-adrenergic-like mechanisms. These peptides are less extensively characterized than catestatin and vasostatin but add to the evidence that CgA functions as a multi-peptide prohormone with diverse physiological roles.
CgA in Context: Comparing Peptide Biomarkers
CgA in the Wider Biomarker Landscape
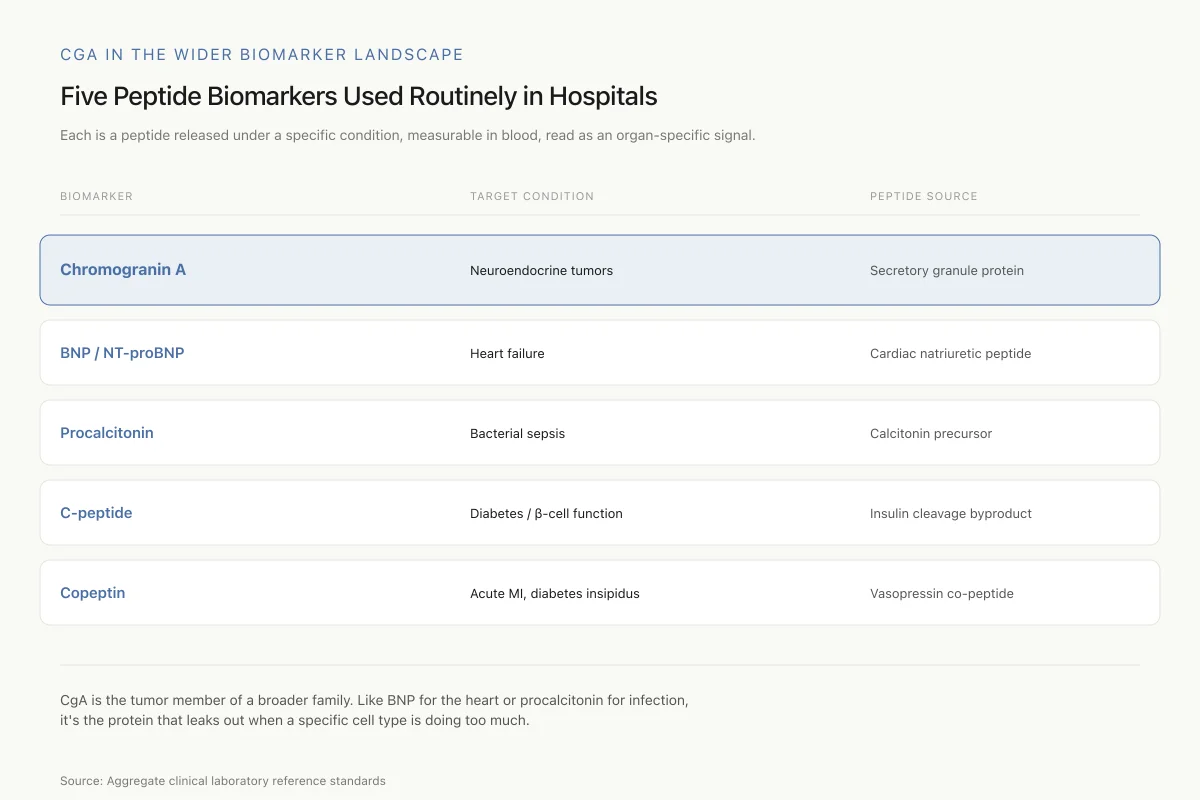
Five Peptide Biomarkers Used Routinely in Hospitals
All share the same principle: a peptide released under a specific condition, measurable in blood, read as a signal of what’s happening in one organ system.
Chromogranin A
Target:Neuroendocrine tumors
Source:Secretory granule protein
BNP / NT-proBNP
Target:Heart failure
Source:Cardiac natriuretic peptide
Procalcitonin
Target:Bacterial sepsis
Source:Calcitonin precursor
C-peptide
Target:Diabetes / β-cell function
Source:Insulin cleavage byproduct
Copeptin
Target:Acute MI, diabetes insipidus
Source:Vasopressin co-peptide
CgA is the tumor member of a broader family. Like BNP for the heart or procalcitonin for infection, it’s the protein that leaks out when a specific cell type is doing too much.
Source: Aggregate clinical laboratory reference standards
 View as image
View as imageCgA is one of several peptide-based biomarkers used in clinical medicine, each tailored to a specific organ system:
| Biomarker | Target Condition | Peptide Type |
|---|---|---|
| Chromogranin A | Neuroendocrine tumors | Secretory granule protein |
| BNP / NT-proBNP | Heart failure | Cardiac natriuretic peptide |
| Procalcitonin | Bacterial sepsis | Calcitonin precursor |
| C-peptide | Diabetes (beta cell function) | Insulin cleavage byproduct |
| Copeptin | Acute MI, diabetes insipidus | Vasopressin co-peptide |
Each of these biomarkers shares a common principle: a peptide or protein released from specific cell types under specific conditions, measurable in blood, providing diagnostic or prognostic information. For details on each, see the cluster articles: BNP and NT-proBNP: How Heart Failure Is Diagnosed with Peptide Biomarkers, Procalcitonin: The Peptide Biomarker That Detects Sepsis, C-Peptide: Why This Insulin Byproduct Is a Crucial Diabetes Marker, Copeptin: The Vasopressin Surrogate Biomarker for Acute Conditions, and Amyloid-Beta as an Alzheimer's Biomarker: The Peptide Plaque Connection.
Limitations and the Future of NET Biomarkers
CgA's limitations are well-documented. Its specificity is compromised by PPI use, renal impairment, and multiple non-neoplastic conditions. Its sensitivity varies by tumor type and grade.
Assay standardization remains a significant practical problem. At least four commercially available immunoassay platforms measure CgA using different antibodies that recognize different epitopes on the protein. The DAKO ELISA targets the C-terminal region. The CIS RIA targets the mid-region. The Thermo Fisher BRAHMS KRYPTOR assay uses yet another epitope. Because CgA undergoes variable proteolytic processing in different tissues and tumors, different fragments predominate in different clinical settings. An assay optimized for full-length CgA may underreport levels in a patient whose tumor primarily releases processed fragments, and vice versa. Results from different platforms can vary by two-fold or more, making it essential that serial monitoring use the same assay throughout. Switching laboratories or assay platforms during disease monitoring invalidates trend data.
These limitations have driven research into alternative and complementary NET biomarkers. NETest, a multi-gene blood transcript analysis measuring 51 NET-associated gene transcripts in peripheral blood, has shown higher sensitivity (93%) and specificity (97%) than CgA in prospective studies, and is not affected by PPI use. However, NETest is more expensive and less widely available. Circulating tumor DNA is emerging as a marker for tumor heterogeneity and treatment resistance in NETs, though its role in routine monitoring remains investigational. Protein biomarker panels combining CgA with neuron-specific enolase, pancreatic polypeptide, and other analytes attempt to improve on single-marker performance.
Functional imaging, particularly gallium-68 DOTATATE PET/CT, has transformed NET diagnosis and staging by directly visualizing somatostatin receptor expression on tumor cells. This imaging modality has higher sensitivity than any circulating biomarker for detecting primary tumors and metastases, and it directly informs decisions about peptide receptor radionuclide therapy (PRRT) eligibility.
CgA is unlikely to be replaced entirely. Its ease of measurement, wide availability, and established prognostic value in serial monitoring ensure continued clinical use. But the era when CgA was the only circulating NET biomarker has passed. Its role is evolving from a stand-alone diagnostic marker to one component of a multi-analyte, multi-modal assessment strategy.
Meanwhile, the CgA-derived peptides continue to generate independent research interest. Catestatin's roles in blood pressure regulation, immune defense, and neurodegeneration position it as a therapeutic candidate, not merely a byproduct of a tumor marker.[2][3][4] The parent protein that clinicians measure as a cancer biomarker turns out to be a peptide factory with functions extending far beyond oncology.
The Bottom Line
Chromogranin A is a 439-amino-acid secretory granule protein that serves dual roles: the most widely used circulating biomarker for neuroendocrine tumors, and a prohormone yielding at least five bioactive peptides with cardiovascular, immune, and metabolic functions. Its clinical utility is strongest for serial monitoring of established NET disease, where changes greater than 17% predict treatment response with over 90% sensitivity. Its biggest limitation is false-positive elevation by proton pump inhibitors, which affect over 80% of PPI users. The CgA-derived peptides catestatin and vasostatin are emerging as independent research targets beyond oncology.
Sources & References
- 1RPEP-01897·Angelone, T et al. (2012). “Chromogranin-A: a multifaceted cardiovascular role in health and disease..” Current medicinal chemistry.Study breakdown →PubMed →↩
- 2RPEP-10493·Chu, Song-Yun et al. (2025). “Catestatin as a predictor for cardiac death in heart failure with mildly reduced and preserved ejection fraction..” ESC heart failure.Study breakdown →PubMed →↩
- 3RPEP-15243·Guth, Colin et al. (2026). “Neuroendocrine Peptide Catestatin Clears MRSA Skin Infections by Activating Mast Cells.” Mucosal immunology.Study breakdown →PubMed →↩
- 4RPEP-15372·Jati, Suborno et al. (2026). “Neuroendocrine Peptide Catestatin Reduces Alzheimer's Tau Pathology and Amyloid Through Adrenergic Inhibition.” bioRxiv : the preprint server for biology.Study breakdown →PubMed →↩
- 5RPEP-10410·Chen, Qiujing et al. (2025). “Natural Peptide Vasostatin-2 Prevents Artery Re-Narrowing After Stent Placement.” Cardiovascular research.Study breakdown →PubMed →↩