Alpha-PRRT: Next-Gen Radioactive Peptide Therapy
Peptide Receptor Radionuclide Therapy
60% Tumor Shrinkage
In the ALPHAMEDIX-02 Phase 2 trial, 212Pb-DOTAMTATE produced tumor shrinkage in approximately 60% of treatment-naive patients with advanced neuroendocrine tumors, earning FDA Breakthrough Therapy Designation in February 2024.
Sanofi/Orano Med, ESMO 2025
Sanofi/Orano Med, ESMO 2025
If you only read one thing
Alpha-PRRT is a newer, more powerful version of radioactive peptide cancer therapy. Instead of using beta particles that scatter over several millimeters, it uses alpha particles that travel less than a hair's width but hit 100 to 1,000 times harder. In clinical trials, it's shrinking tumors in 60% of patients who haven't had PRRT before — and even working in some patients whose cancer stopped responding to the standard treatment. It's not FDA-approved yet, but it has Breakthrough Therapy status.
Alpha-emitter PRRT represents a fundamental shift in how radioactive peptides kill cancer cells. Standard peptide receptor radionuclide therapy (PRRT) uses beta-emitting isotopes like lutetium-177, which fire electrons that travel several millimeters through tissue. Alpha-emitting PRRT replaces those beta particles with helium nuclei that travel less than 100 micrometers but deliver 100 to 1,000 times more energy per unit distance.[1] The result is a precision weapon: more lethal to tumor cells, less damaging to surrounding tissue, and potentially effective against cancers that have stopped responding to conventional PRRT.
The clinical data is no longer theoretical. Phase 2 trials of lead-212 labeled peptides have produced tumor shrinkage in 60% of PRRT-naive patients. Actinium-225 labeled peptides have achieved complete remissions in patients who progressed after lutetium-177 therapy. And a combined "TANDEM" approach, using both alpha and beta emitters simultaneously, has generated responses in previously refractory disease.[2] This article covers the physics, the clinical evidence, and the open questions for each alpha-emitting isotope under investigation. For a broader introduction to the field, see Peptide Receptor Radionuclide Therapy (PRRT) Explained. For current survival data with conventional beta-PRRT, see PRRT Clinical Outcomes: Survival Data for Neuroendocrine Tumors.
Key Takeaways
- Alpha-PRRT is the next-generation version of radioactive peptide cancer therapy — more powerful, more precise, and in late-stage trials right now.
- A peptide carries the radioactive payload directly to tumor cells, docks onto them, and delivers radiation from inside the cell instead of from outside the body.
- The punch is 100 to 1,000 times harder than standard PRRT, but travels less than a hair's width — which means it kills tumors without torching the tissue around them.
- In phase 2, it shrank tumors in about 60% of patients who hadn't had PRRT before, and 35% of patients whose cancer had stopped responding to the standard beta version.
- One patient was declared cancer-free at autopsy after alpha-PRRT following progression on the standard treatment — the kind of result that's impossible with most cancer drugs.
- Kidneys typically take damage from radiation therapy. Early data shows alpha-PRRT spares them far better than the older version — less than 5% kidney function loss per year.
- Nothing alpha-emitting is FDA-approved yet. The lead candidate has Breakthrough Therapy status, which speeds review but is not the same as approval.
How PRRT Works: Peptides as Radioactive Delivery Vehicles
Peptide receptor radionuclide therapy exploits a biological vulnerability of certain cancers. Neuroendocrine tumors (NETs) overexpress somatostatin receptors on their cell surfaces at densities 10 to 100 times higher than normal tissue. PRRT attaches a radioactive isotope to a somatostatin analogue peptide (such as DOTATATE or DOTATOC), creating a molecule that binds to these receptors, gets internalized into the cell, and delivers lethal radiation from inside the tumor.[3]
The NETTER-1 Phase III trial established lutetium-177 DOTATATE (Lutathera) as a standard treatment, demonstrating prolonged progression-free survival and improved quality of life in patients with midgut NETs. Since its FDA approval in 2018, beta-PRRT has become a cornerstone of NET management.[4] For a detailed explanation of how Lutathera works, see Lutathera (177Lu-DOTATATE): How Radioactive Peptides Treat Cancer.
But beta-PRRT has limitations. Beta particles travel 1 to 12 millimeters through tissue, which means they irradiate healthy cells neighboring the tumor. Some patients develop resistance or fail to respond. And the energy deposited per unit of tissue (linear energy transfer, or LET) is relatively low, meaning that DNA damage from beta particles is often repairable by tumor cells. Real-world outcome data confirms these limitations: Lazarenko and colleagues documented that while Lu-177 DOTATATE produces meaningful responses, a substantial proportion of patients experience disease progression within two years of completing treatment.
Virgolini and colleagues characterized the current state of PRRT in a 2026 review, noting that while NETTER-1 established the clinical utility of beta-PRRT, the field is moving toward combination strategies, dosimetry-guided approaches, and alpha-emitting isotopes to overcome these limitations.[4]
Alpha-emitter PRRT addresses each of these limitations.
Beta vs. Alpha: Why the Particle Type Changes Everything
The physics distinction between alpha and beta particles determines their biological effects.
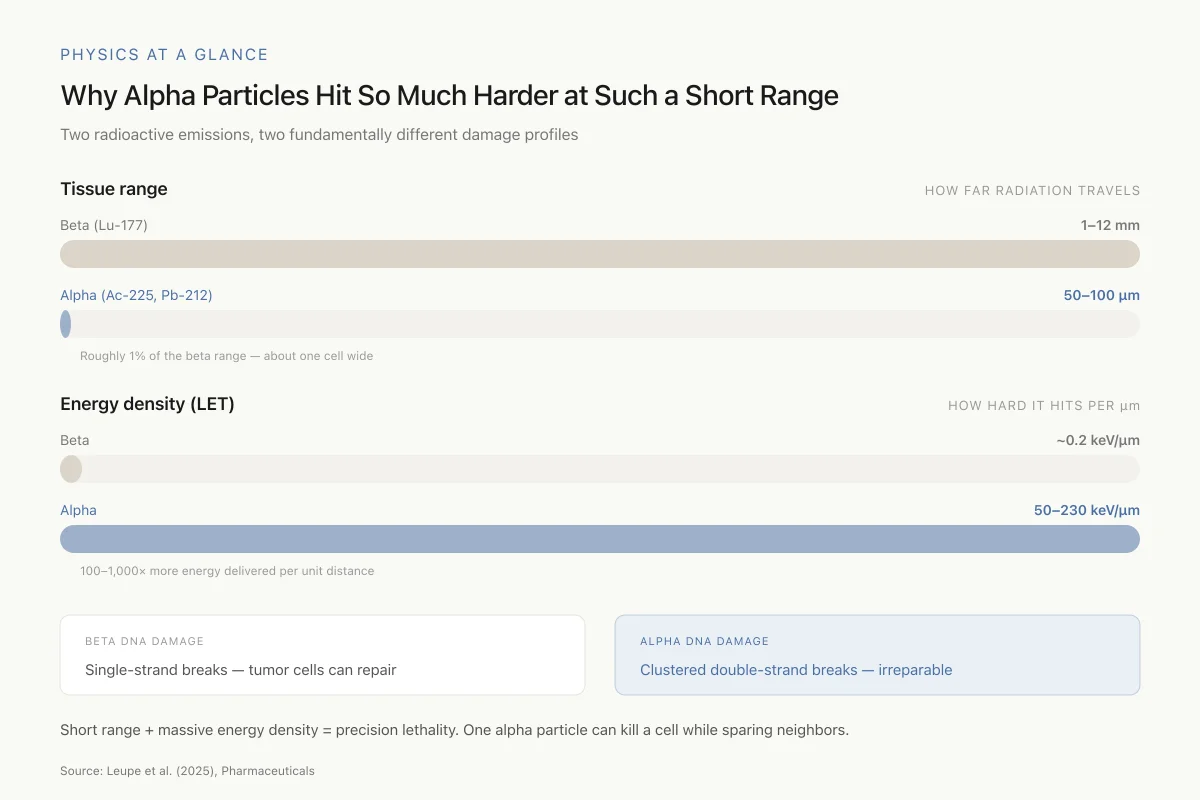
Beta particles (electrons) from lutetium-177 have a tissue range of 1 to 12 mm and a linear energy transfer of approximately 0.2 keV per micrometer. This means they deposit energy gradually over a relatively long path, allowing tumor repair mechanisms time to fix sublethal DNA damage. The longer range can be therapeutically useful for larger tumors (the "crossfire effect"), but it also irradiates healthy tissue beyond the tumor margin.
Alpha particles (helium-4 nuclei) have a tissue range of only 50 to 100 micrometers and a linear energy transfer of 50 to 230 keV per micrometer. This 100- to 1,000-fold increase in energy density produces clustered, irreparable double-strand DNA breaks. A single alpha particle traversing a cell nucleus can kill the cell. Repair mechanisms cannot fix this type of damage.[1]
Physics at a Glance
Why Alpha Particles Hit So Much Harder at Such a Short Range
Two radioactive emissions, two fundamentally different damage profiles
Roughly 1% of the beta range — about one cell wide
100–1,000× more energy delivered per unit distance
Short range + massive energy density = precision lethality. One alpha particle crossing a cell nucleus can kill the cell outright, while leaving neighbors a tenth of a millimeter away untouched.
Source: Leupe et al. (2025), Pharmaceuticals
 View as image
View as imageThe short range of alpha particles creates a therapeutic paradox: they are extraordinarily lethal at close range but essentially harmless beyond 100 micrometers. This means that alpha-PRRT can destroy tumor cells while sparing adjacent normal tissue, but only if the radioactive peptide reaches every tumor cell. Small, well-vascularized tumors with high receptor density are ideal targets. Large, poorly vascularized tumors with heterogeneous receptor expression may be better suited to beta-PRRT or a combination approach.
For a comparison of the two beta-emitting isotopes used in conventional PRRT, see Yttrium-90 vs Lutetium-177 PRRT: Two Isotopes, Two Approaches.
Actinium-225: Four Alpha Particles Per Decay
Actinium-225 is the most potent alpha emitter under clinical investigation for PRRT. With a half-life of 9.9 days, it undergoes a cascade decay producing four alpha particles before reaching stable bismuth-209. This quadruple alpha emission makes each Ac-225 decay extraordinarily cytotoxic.[1]
Clinical evidence: Ac-225 PRRT
Perrone and colleagues reported what may be the most striking result in alpha-PRRT to date: the first autopsy-confirmed complete remission of a metastatic neuroendocrine neoplasm after a single cycle of Ac-225 labeled somatostatin receptor antagonist (DOTA-LM3). A 78-year-old man with recurrent NEN of tailgut cyst origin, previously treated with surgery, lanreotide, and Lu-177 DOTATATE, received one cycle of [225Ac]Ac-DOTA-LM3. At autopsy (performed after death from unrelated causes), no viable tumor was found at any metastatic site.[5]
In their larger retrospective series, Perrone and colleagues reported on 35 patients who received a total of 57 [225Ac]Ac-DOTA-LM3 cycles between March 2022 and September 2024. The cohort included both monotherapy (24 cycles) and TANDEM therapy with simultaneous Lu-177 (33 cycles). The treatment showed a manageable safety profile with encouraging efficacy, including responses in patients who had progressed after standard Lu-177 PRRT.[2]
The daughter isotope problem
Ac-225's quadruple decay chain creates a unique challenge: the daughter isotopes (francium-221, astatine-217, bismuth-213) are themselves radioactive and can detach from the targeting peptide. If free daughter isotopes redistribute through the body, they deliver untargeted radiation to healthy organs, particularly the kidneys. This is one of the central safety questions for Ac-225 PRRT, and the clinical data so far suggests the nephrotoxicity may be lower than anticipated, with annual eGFR loss below 5%.[6]
Agonists vs. antagonists
A notable development in Ac-225 PRRT is the shift from somatostatin receptor agonists (like DOTATATE) to antagonists (like DOTA-LM3). Receptor antagonists bind more receptor sites on the tumor surface without requiring internalization, potentially increasing the amount of radioactivity delivered to the tumor. Speicher and colleagues documented a case where switching from agonist-based to antagonist-based somatostatin receptor theranostics enabled successful PRRT in a patient with insufficient receptor uptake on conventional imaging.[7]
Lead-212 (AlphaMedix): The Clinical Frontrunner
Lead-212 has emerged as the most clinically advanced alpha emitter for PRRT, primarily through the AlphaMedix program (212Pb-DOTAMTATE), now jointly developed by Orano Med and Sanofi.
ALPHAMEDIX-02 Phase 2 results
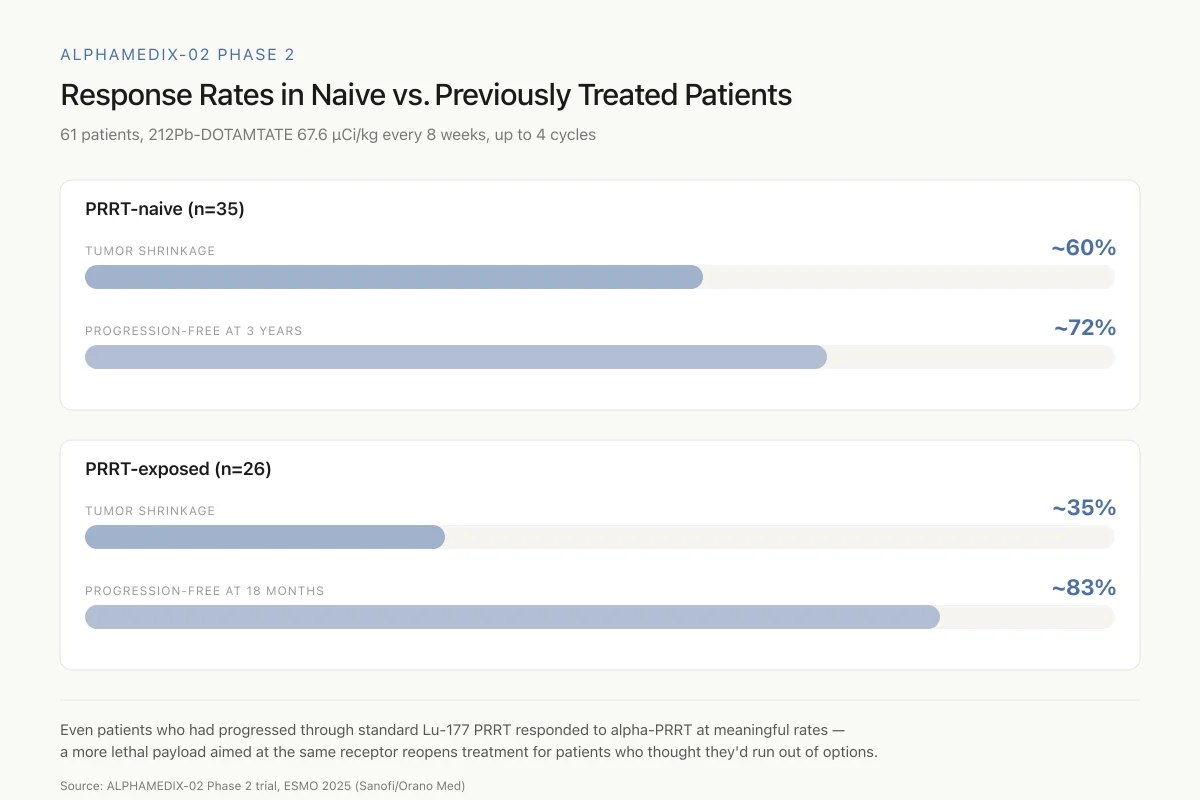
The ALPHAMEDIX-02 trial, presented at ESMO 2025, enrolled 61 patients across two cohorts: 35 PRRT-naive and 26 PRRT-exposed patients with unresectable or metastatic somatostatin receptor-positive gastroenteropancreatic NETs. AlphaMedix was administered at 67.6 microcuries per kilogram every eight weeks for up to four cycles.
The results met all primary efficacy endpoints. In PRRT-naive patients, approximately 60% achieved tumor shrinkage, with roughly 70 to 75% remaining progression-free at three years. In PRRT-exposed patients, approximately 35% achieved tumor shrinkage, with about 83% progression-free at 18 months.
ALPHAMEDIX-02 Phase 2
Response Rates in Naive vs. Previously Treated Patients
61 patients, 212Pb-DOTAMTATE 67.6 µCi/kg every 8 weeks, up to 4 cycles
The most striking finding: even patients who had already progressed through standard Lu-177 PRRT responded to alpha-PRRT at meaningful rates. Targeting the same receptor with a more lethal payload reopens treatment for patients who thought they'd run out of options.
Source: ALPHAMEDIX-02 Phase 2 trial, ESMO 2025 (Sanofi/Orano Med)
 View as image
View as imageWhy lead-212 may have advantages
Lead-212 has a half-life of 10.6 hours and produces one alpha particle (from its daughter bismuth-212) before reaching stable lead-208 or thallium-208. The shorter decay chain compared to Ac-225 means fewer daughter isotopes that can redistribute to healthy tissue.[8]
Lee and colleagues at the University of Alabama published structural modifications to improve lead-212 peptide radiopharmaceuticals, demonstrating that chelator design and peptide linker chemistry can enhance tumor uptake and reduce kidney retention. Their work showed that Pb-212 labeled peptides targeting SSTR2 may provide improved effectiveness compared to existing beta-emitting alternatives.[9]
Saidi and colleagues conducted a direct side-by-side comparison of [212Pb]Pb-DOTAMTATE against other SSTR2-targeting compounds in vivo. Their 2025 study in the Journal of Nuclear Medicine compared pharmacokinetics, tumor uptake, and organ dosimetry across multiple peptide variants, providing the first head-to-head data on how 212Pb-labeled peptides perform relative to established alternatives.[8]
Safety in the Phase 2 trial
The ALPHAMEDIX-02 safety profile was manageable and consistent across both cohorts. The most common adverse events were fatigue, nausea, and alopecia, all predominantly Grade 1 or 2. Hematological toxicity was present but generally mild. An unexpected finding was achalasia (difficulty swallowing) in a small number of patients, a toxicity not previously associated with PRRT. The mechanism is under investigation but may relate to alpha particle effects on the esophageal myenteric plexus, which expresses low levels of somatostatin receptors.
Kidney function monitoring showed preserved renal function in most patients, consistent with the theoretical advantage of alpha particles' short tissue range limiting renal parenchymal exposure. However, the follow-up duration in Phase 2 is insufficient to characterize late renal toxicity, which can manifest years after treatment in beta-PRRT.
Regulatory status
The FDA granted Breakthrough Therapy Designation to AlphaMedix in February 2024 for treatment of PRRT-naive patients with progressive SSTR-expressing GEP-NETs. This was the first targeted alpha therapy to receive this designation in any indication, signaling regulatory recognition of the clinical potential. Phase 3 planning is underway, with the pivotal trial expected to compare AlphaMedix against standard of care in SSTR-positive GEP-NET patients.
TANDEM Therapy: Combining Alpha and Beta
Rather than choosing between alpha and beta emitters, TANDEM therapy administers both simultaneously. The rationale: beta particles provide the crossfire effect needed for larger tumor deposits, while alpha particles deliver lethal doses to individual cells, including those at tumor margins or in micrometastases that beta radiation alone cannot eradicate.
Perrone and colleagues' experience with 177Lu/225Ac-DOTA-LM3 TANDEM therapy represents the largest published dataset. Their retrospective analysis of 35 patients demonstrated that the combination was feasible, with a safety profile comparable to monotherapy. The approach produced responses in patients who had exhausted other options, including prior beta-PRRT.[2]
A 2024 case report from the same group described an "impressive response" in a 58-year-old woman with rapidly progressing pancreatic NET (initially Grade 2) with extensive liver, bone, and lymph node metastases. After progressing through chemotherapy and standard Lu-177 PRRT, she received TANDEM therapy with 177Lu/225Ac-DOTA-LM3 and achieved a marked partial response.[10]
The TANDEM concept also addresses a practical consideration: isotope availability. Because Ac-225 is scarce, using small amounts of Ac-225 in combination with readily available Lu-177 allows more patients to receive alpha radiation than a pure Ac-225 monotherapy approach would permit. The beta component provides bulk tumor debulking while the alpha component targets resistant cells and micrometastases. This combined approach may prove to be the most clinically practical path to incorporating alpha emitters into standard PRRT protocols, particularly while Ac-225 production capacity remains limited.
The dosimetry of TANDEM therapy is complex. Each isotope has a different energy profile, tissue range, and decay timeline. Calculating the combined radiation dose to the tumor and to dose-limiting organs (kidneys, bone marrow) requires sophisticated modeling that accounts for both alpha and beta contributions. Standardized dosimetry protocols for TANDEM therapy do not yet exist, and current practice relies on institutional experience and conservative dose limits.
Beyond Neuroendocrine Tumors
While NETs remain the primary target, alpha-PRRT is being explored for other receptor-expressing cancers.
Krolicki and colleagues reported on the safety and efficacy of bismuth-213 labeled DOTA-Substance P for recurrent glioblastoma, targeting the neurokinin type 1 receptor (NK-1R) rather than somatostatin receptors. Their results demonstrated that targeted alpha therapy is feasible in brain tumors when a suitable peptide-receptor pair exists.[11]
Research is also expanding to cholecystokinin-2 receptor (CCK2R) targeting for medullary thyroid carcinoma and other CCK2R-expressing tumors, and to prostate-specific membrane antigen (PSMA) targeting for prostate cancer (though PSMA agents are typically non-peptide small molecules).
The principle is consistent: if a cancer overexpresses a receptor for which a peptide ligand exists, that peptide can deliver alpha particles to the tumor. The limiting factors are receptor density, tumor accessibility, and isotope availability.
The systematic review by Zampella and colleagues examined PRRT outcomes in high-grade (Grade 3) neuroendocrine neoplasms, a population that historically responds poorly to standard treatments. Their meta-analysis of studies published through March 2025 found that even in these aggressive tumors, PRRT with radiolabeled somatostatin analogues produced disease control, suggesting that alpha-PRRT, with its greater cytotoxic potential, could further improve outcomes in this difficult-to-treat population. High-grade NETs with adequate somatostatin receptor expression may represent an ideal application for alpha particles because the greater DNA damage per particle could overcome the faster proliferation rate that makes these tumors resistant to beta radiation.
High-grade neuroendocrine neoplasms
The distinction between low-grade and high-grade NETs is clinically important for alpha-PRRT. Low-grade (G1/G2) NETs are slow-growing, well-differentiated tumors that respond well to beta-PRRT. High-grade (G3) neuroendocrine neoplasms are aggressive, rapidly proliferating tumors with a poorer prognosis. Conventional beta-PRRT has shown modest benefit in G3 disease, partly because beta radiation produces reparable DNA damage that fast-dividing cells can overcome between mitoses. Alpha particles, which produce irreparable clustered double-strand breaks, may be more effective against rapidly proliferating cells because the damage cannot be repaired regardless of cell cycle timing.
Safety: Nephrotoxicity and Hematological Effects
Kidney damage is the primary long-term concern for any form of PRRT. Radioactive peptides are filtered and partially reabsorbed by renal tubular cells, exposing the kidneys to radiation during every treatment cycle.
Das and colleagues published a comprehensive review of PRRT-related nephropathy in 2026, characterizing it as "a new era of radiation nephropathy" given the expanding use of radiopeptide therapies. They documented that the risk varies based on the isotope used, the peptide carrier, cumulative dose, and patient-specific factors including baseline renal function and concurrent nephrotoxic medications.[6]
Critically for alpha-PRRT, early data suggest that the short range of alpha particles may actually reduce renal toxicity compared to beta-PRRT. Ac-225 DOTATOC produced less than 5% annual eGFR decline in five-year follow-up data, compared to the more variable renal effects seen with Lu-177. The short 50 to 100 micrometer range of alpha particles means that radiation is confined to the cells that directly interact with the radioactive peptide, rather than spreading to adjacent nephron structures as beta radiation does.
Mohindroo and Ramirez reviewed the full spectrum of PRRT toxicities in 2026, noting that hematological toxicity (cytopenias) remains the most common acute adverse effect across all PRRT modalities, while renal toxicity and myelodysplastic syndrome represent the primary long-term concerns. For alpha-PRRT specifically, the limited sample sizes in current studies mean that rare toxicities may not yet be apparent.[12]
Safety
ModerateLong-term kidney and blood effects still unknown
Concern
All PRRT carries a risk of kidney damage and blood cell suppression. Alpha-PRRT is so new that long-term data beyond 5 years doesn't exist. The Ac-225 daughter isotope redistribution adds an additional layer of uncertainty about off-target radiation exposure.
What the research says
Early data is actually encouraging — alpha particles' short range may cause less kidney damage than beta-PRRT. Ac-225 showed less than 5% annual kidney function decline over 5 years. But 'less than beta-PRRT' is not the same as 'safe,' and larger, longer studies are needed.
Particularly relevant for: Patients considering alpha-PRRT or enrolled in clinical trials
What to do
Regular kidney function monitoring during and after treatment is essential. Discuss the known unknowns with your oncology team — this is cutting-edge therapy with promising but limited safety data.
Das et al. (2026), Nephrology Dialysis Transplantation; Mohindroo & Ramirez (2026)
The AlphaMedix Phase 2 trial reported a manageable safety profile with mostly mild adverse events (fatigue, nausea, hair thinning). An unexpected finding was achalasia (difficulty swallowing) in some patients, a toxicity not previously associated with PRRT and under investigation.
The Supply Problem
Alpha-emitting isotopes are far more difficult to produce than beta emitters. Lutetium-177 is reactor-produced in quantities sufficient for widespread clinical use. The alpha emitters face different production challenges.
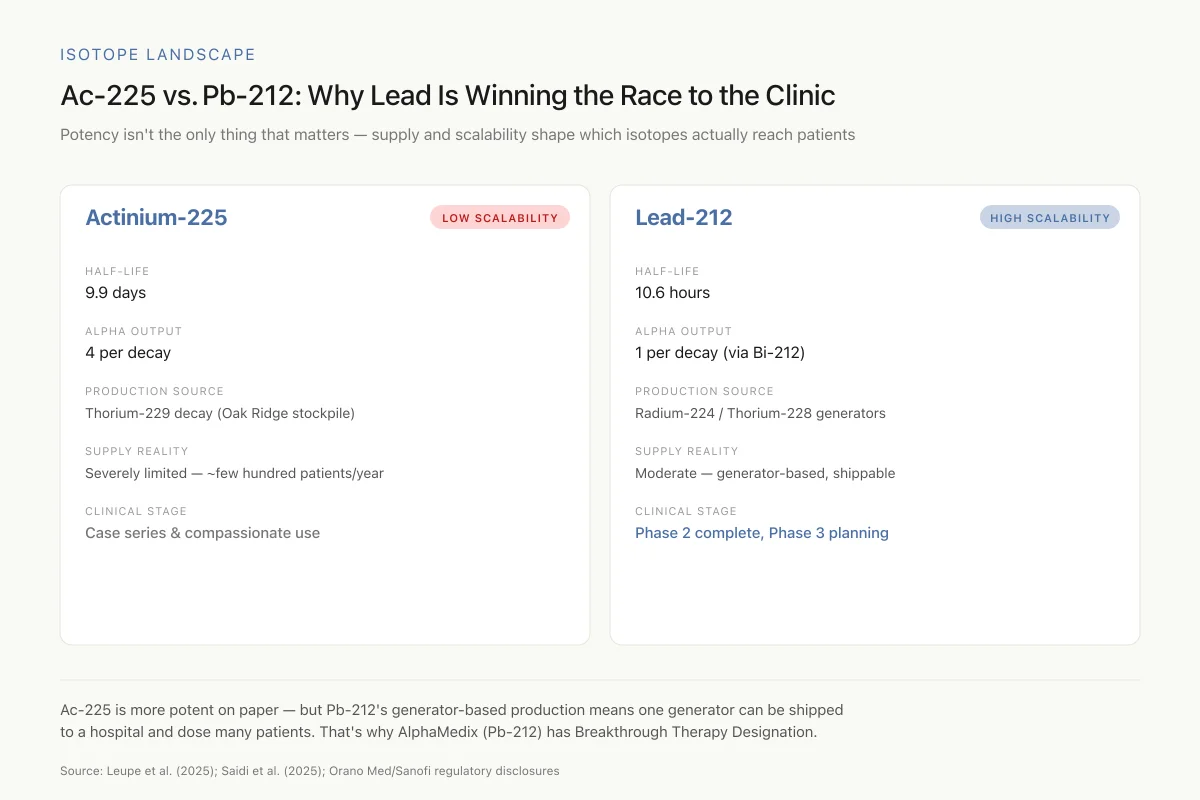
Actinium-225 is primarily produced from the decay of thorium-229 in limited government stockpiles (Oak Ridge National Laboratory, Institute for Transuranium Elements). Global production capacity is estimated at enough for only a few hundred patients per year. Accelerator-based and reactor-based production methods are under development, but scaling remains a major barrier to widespread clinical use.
Lead-212 is produced from radium-224 generators, which themselves derive from thorium-228. The generator-based approach is more scalable than Ac-225 production, which is one reason the AlphaMedix program has advanced faster in clinical development. Orano Med has invested in production infrastructure to support potential commercialization.
This supply constraint shapes the entire field. Even if alpha-PRRT proves superior to beta-PRRT in clinical trials, widespread adoption requires solving the isotope production problem. Current estimates suggest that meeting projected demand would require a 10- to 100-fold increase in production capacity for alpha-emitting isotopes.
Multiple approaches are under development. The Department of Energy has invested in accelerator-based Ac-225 production at several national laboratories. Isotope harvesting from existing medical cyclotrons is being explored. And the thorium-229 stockpile at Oak Ridge, while limited, continues to serve as the primary source for clinical trials. The timeline from production scaling to clinical availability is measured in years, not months, and the supply problem remains one of the largest obstacles to alpha-PRRT becoming a standard treatment option.
Isotope Landscape
Ac-225 vs. Pb-212: Why Lead Is Winning the Race to the Clinic
Potency isn't the only thing that matters — supply and scalability shape which isotopes actually reach patients
Ac-225 is the more potent isotope on paper (4 alpha particles per decay) — but it's produced from a tiny government stockpile. Pb-212's generator-based production means one radium-224 generator can be shipped to a hospital and dose many patients. That's why AlphaMedix (Pb-212) has Breakthrough Therapy Designation while Ac-225 is still mostly in case series.
Source: Leupe et al. (2025); Saidi et al. (2025); Orano Med/Sanofi regulatory disclosures
 View as image
View as imageFor Pb-212, the generator-based approach offers a structural advantage. A single radium-224/lead-212 generator can produce multiple patient doses over its useful lifetime, and generators can be shipped to clinical sites rather than requiring the isotope to be produced on-site. Orano Med has built dedicated production facilities to support the AlphaMedix clinical program and potential commercialization, positioning Pb-212 as the more supply-ready alpha emitter for near-term clinical use.
What Remains Unknown
Alpha-PRRT is moving rapidly from preclinical concept to clinical reality, but several critical questions remain unanswered.
Long-term safety data beyond five years does not exist for any alpha-PRRT approach. The Ac-225 daughter redistribution problem requires larger patient cohorts and longer follow-up to fully characterize. Optimal dosing, fractionation schedules, and patient selection criteria have not been established through randomized controlled trials.
The comparison between Ac-225 and Pb-212 has not been made in a head-to-head clinical trial. Each isotope has theoretical advantages, and the choice may ultimately depend on tumor biology, prior treatment history, and practical availability.
Whether alpha-PRRT should be used as first-line therapy, as salvage after beta-PRRT failure, or in combination (TANDEM) is an active area of investigation. The NETTER-1 trial established beta-PRRT as a standard, and alpha-PRRT trials are building from that foundation rather than replacing it.
The Kobayashi 2026 long-term data from Japan confirmed that conventional Lu-177 PRRT produces durable responses with acceptable toxicity, reinforcing that beta-PRRT remains the established standard against which alpha-PRRT must demonstrate superiority or complementary benefit.[13]
The regulatory pathway will also shape adoption. AlphaMedix's Breakthrough Therapy Designation provides an accelerated review mechanism, but Phase 3 data will still be required for full FDA approval. For Ac-225 based approaches, the path from compassionate use and small retrospective series to randomized controlled trials requires the isotope supply problem to be solved first. The field is in a transitional period: the preclinical and early clinical evidence is compelling, but the infrastructure needed to deliver alpha-PRRT at scale does not yet exist.
The Bottom Line
Alpha-emitter PRRT uses peptides to deliver alpha particles directly to cancer cells, producing irreparable DNA damage within a range of less than 100 micrometers. Phase 2 data for lead-212 (AlphaMedix) and clinical series for actinium-225 demonstrate responses in both treatment-naive and treatment-refractory neuroendocrine tumor patients. Key unresolved questions include long-term toxicity, optimal sequencing relative to beta-PRRT, and isotope supply scalability.
Sources & References
- 1RPEP-12048·Leupe, Hannes et al. (2025). “Clinical Experience with Targeted Alpha-Emitter Peptide Receptor Radionuclide Therapy (α-PRRT) for Somatostatin Receptor-Positive Neuroendocrine Tumors..” Pharmaceuticals (Basel.Study breakdown →PubMed →↩
- 2RPEP-13027·Perrone, Elisabetta et al. (2025). “Alpha Radiation Therapy for Resistant Neuroendocrine Cancers: Safety and Survival in 35 Patients.” Cancers.Study breakdown →PubMed →↩
- 3RPEP-07871·Bodei, Lisa et al. (2024). “How Radioactive Peptides Are Used to Treat Neuroendocrine Tumors.” PET clinics.Study breakdown →PubMed →↩
- 4RPEP-16322·Virgolini, Irene J et al. (2026). “Peptide receptor radionuclide therapy in neuroendocrine tumours: advances, combination strategies, and future directions..” European journal of nuclear medicine and molecular imaging.Study breakdown →PubMed →↩
- 5RPEP-13026·Perrone, Elisabetta et al. (2025). “First Autopsy-Confirmed Complete Cancer Remission After Single Cycle of Alpha Radiation Therapy.” Clinical nuclear medicine.Study breakdown →PubMed →↩
- 6RPEP-15077·Das, Abhirami et al. (2026). “Managing Kidney Damage from Peptide-Targeted Radiation Cancer Therapy.” Nephrology.Study breakdown →PubMed →↩
- 7RPEP-16159·Speicher, Tilman et al. (2026). “Switching From Agonist to Antagonist Peptide Enabled Successful Targeted Radiation Therapy for a Previously Untreatable Neuroendocrine Tumor.” Clinical nuclear medicine.Study breakdown →PubMed →↩
- 8RPEP-13368·Saidi, Amal et al. (2025). “Side-by-Side Comparison of the In Vivo Performance of [212Pb]Pb-DOTAMTATE and Other SSTR2-Targeting Compounds..” Journal of nuclear medicine : official publication.Study breakdown →PubMed →↩
- 9RPEP-08642·Lee, Dongyoul et al. (2024). “Improved Octreotide-Based Radiotherapy Achieves 80% Survival in Neuroendocrine Tumor Mouse Model.” European journal of nuclear medicine and molecular imaging.Study breakdown →PubMed →↩
- 10RPEP-09062·Perrone, Elisabetta et al. (2024). “Dramatic Tumor Response to Dual-Radionuclide Peptide Therapy in a Patient with Treatment-Resistant Pancreatic Neuroendocrine Cancer.” Diagnostics (Basel.Study breakdown →PubMed →↩
- 11RPEP-04296·Królicki, Leszek et al. (2019). “Safety and efficacy of targeted alpha therapy with 213Bi-DOTA-substance P in recurrent glioblastoma..” European journal of nuclear medicine and molecular imaging.Study breakdown →PubMed →↩
- 12RPEP-15731·Mohindroo, Chirayu et al. (2026). “How to Manage Side Effects of Radioactive Peptide Therapy for Neuroendocrine Tumors.” Current treatment options in oncology.Study breakdown →PubMed →↩
- 13RPEP-15446·Kobayashi, Noritoshi et al. (2026). “Long-Term PRRT for Neuroendocrine Tumors in Japan: Efficacy and Safety Confirmed.” Annals of nuclear medicine.Study breakdown →PubMed →↩