Bacteriocins: Antimicrobial Peptides From Bacteria
Bacteriocins and Microbiome Peptides
410+ Bacterial Bacteriocins Cataloged
The APD3 antimicrobial peptide database catalogs over 410 bacteriocins produced by bacteria. These peptides kill competing bacterial strains through membrane disruption and pore formation, and they have been used in food preservation for decades.
APD3 Database, University of Nebraska Medical Center, 2025
APD3 Database, University of Nebraska Medical Center, 2025
If you only read one thing
Bacteriocins are small antimicrobial peptides that bacteria make to kill their rivals — microbial warfare that's been going on for billions of years. One of them, nisin, has been used as a food preservative since the 1950s and has never had a significant resistance problem. That's remarkable. Bacteria make these by the thousands; a single oral-bacterium strain can carry 14 different bacteriocin genes. They're being studied as antibiotic alternatives for drug-resistant infections like MRSA, but the main barriers are narrow activity ranges, rapid breakdown in the body, and manufacturing cost. Your own gut bacteria make bacteriocins constantly — they're a core part of how the microbiome keeps itself in balance.
Bacteria have been killing each other with antimicrobial peptides for billions of years. Long before humans discovered antibiotics, bacterial species were producing small proteins called bacteriocins to eliminate competing strains in their ecological niche. These peptides are produced by nearly all bacterial species and represent one of the most abundant and diverse classes of antimicrobial compounds in nature. The best-known bacteriocin, nisin, has been used as a food preservative since 1953 and received FDA GRAS (Generally Recognized as Safe) status in 1988. Despite decades of use in food safety, bacteriocins are now attracting renewed interest as potential weapons against antibiotic-resistant pathogens. For the broader landscape of antimicrobial peptides as antibiotic alternatives, see Antimicrobial Peptides vs Antibiotics: The Evidence.
Key Takeaways
- Bacteria have been making tiny peptide weapons to kill their rivals for billions of years. We call them bacteriocins, and there are over 410 cataloged so far.
- Your gut bacteria make these constantly. Part of how probiotics actually work is not crowding — it's chemical warfare against the bad bacteria.
- One bacteriocin, nisin, has preserved cheese and processed food since 1953 — and bacteria still haven't evolved meaningful resistance to it. That's remarkable.
- Some of these peptides kill MRSA and other drug-resistant bugs in the lab. Researchers are testing them as antibiotic alternatives right now.
- A single probiotic bacterium can carry up to 14 different bacteriocin genes, each targeting a different competitor. Your microbiome is far more arms-race than "balanced."
- Despite decades of nisin in food, only a handful of bacteriocins have reached medical use. The hurdles are short half-life, narrow targeting, and manufacturing cost.
- Combining bacteriocins with conventional antibiotics may be a way to slow resistance — a 2025 study showed nisin-plus-azithromycin worked without selecting for resistant strains.
What Are Bacteriocins?
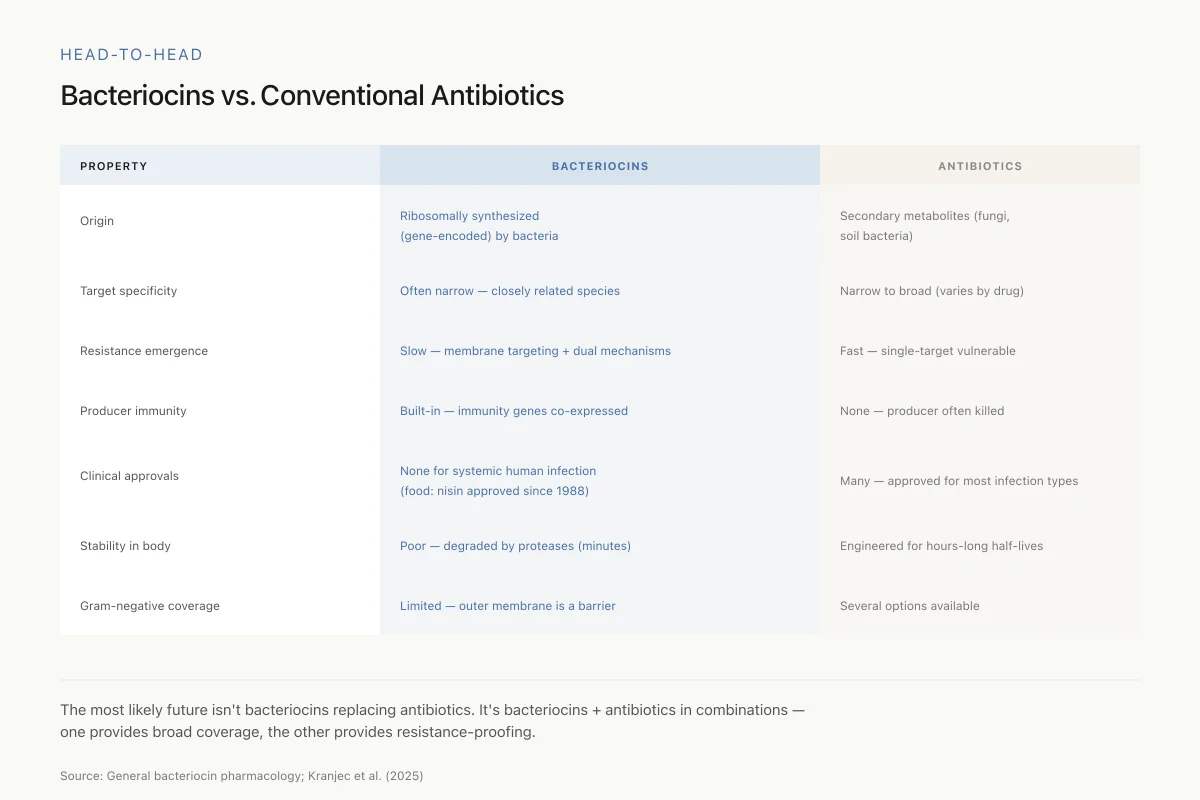
Bacteriocins are antimicrobial peptides or small proteins produced by bacteria through ribosomal synthesis. Unlike conventional antibiotics, which are typically secondary metabolites produced by fungi or soil bacteria, bacteriocins are gene-encoded peptides that bacteria produce specifically to kill closely related competitors. A bacterium that produces a bacteriocin is immune to its own product through co-expressed immunity proteins, giving it a competitive advantage in environments where resources are limited.
The defining characteristics of bacteriocins include:
- Ribosomal synthesis: bacteriocins are encoded in bacterial DNA and produced by ribosomes, unlike many antibiotics that are assembled by non-ribosomal peptide synthetases
- Narrow spectrum: most bacteriocins kill only closely related species or strains, though some (like nisin) have broader activity
- Self-immunity: producing strains carry immunity genes that protect them from their own bacteriocin
- Ecological function: bacteriocins mediate competition between bacterial strains in complex communities including the human gut, soil, and food environments
Lactic acid bacteria (LAB) are the most prolific and best-studied bacteriocin producers. Genera including Lactococcus, Lactobacillus, Pediococcus, Enterococcus, and Streptococcus produce a wide variety of bacteriocins with activity against foodborne pathogens and clinically relevant organisms. The association of LAB with fermented foods and the human gut microbiome has made their bacteriocins particularly attractive for both food safety and therapeutic applications.
Classification: Three Major Classes
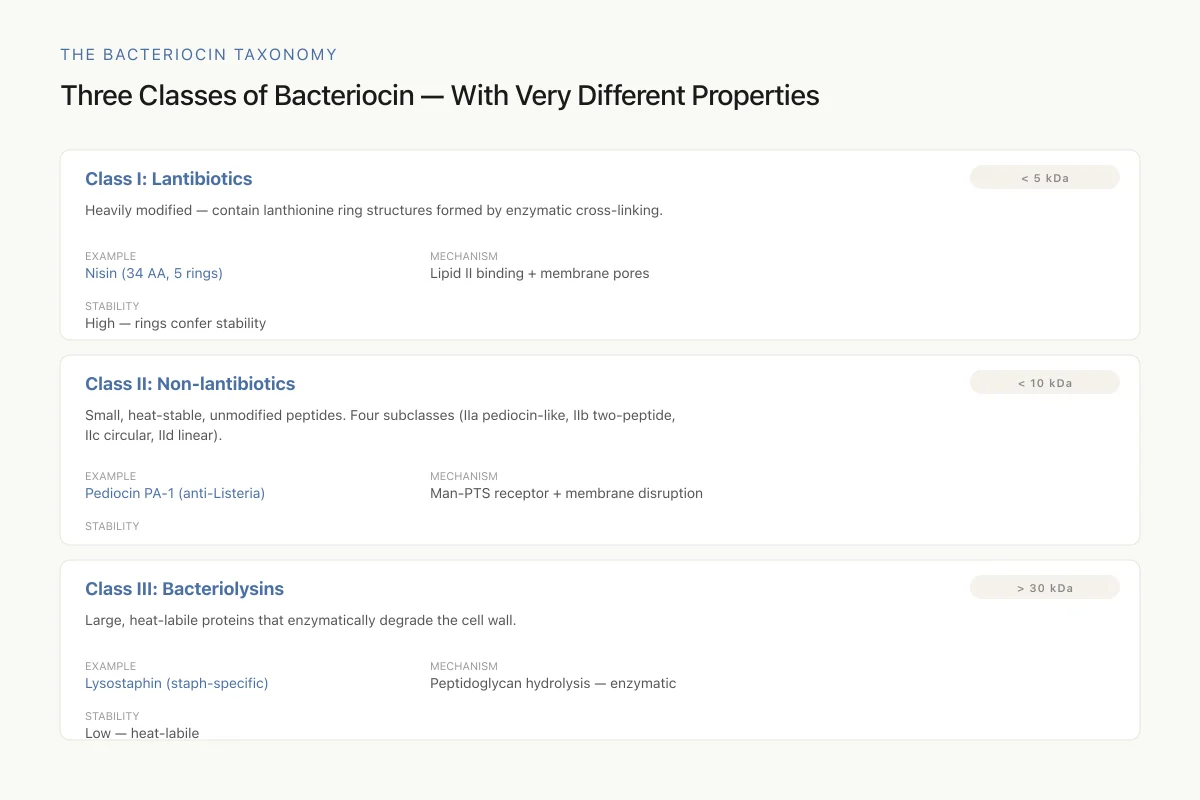
Bacteriocins are classified based on their size, structure, and post-translational modifications. The most widely used system divides them into three classes.
The Bacteriocin Taxonomy
Three Classes of Bacteriocin — With Very Different Properties
All three classes appear in the same gut microbiome at the same time. Bacteria use mixed arsenals — one strain can encode 14+ different bacteriocins across multiple classes, deploying them strategically depending on which competitors are around.
Source: APD3 Database (2025); Hourigan et al. (2025); Revilla-Guarinos et al. (2026)
 View as image
View as imageClass I: Lantibiotics
Lantibiotics are small peptides (typically under 5 kDa) that undergo extensive post-translational modification. The defining feature is the presence of unusual amino acids, lanthionine and methyllanthionine, formed by enzymatic dehydration and thioether cross-linking of serine, threonine, and cysteine residues. These modifications create characteristic ring structures that confer stability and biological activity.
Nisin is the most important lantibiotic. Produced by Lactococcus lactis, nisin is a 34-amino-acid peptide with five intramolecular ring structures. It kills bacteria through a dual mechanism: binding to lipid II (a critical cell wall synthesis precursor) and forming pores in the bacterial membrane. This dual mechanism makes resistance evolution particularly difficult because bacteria would need to simultaneously alter two fundamental cellular processes. Nisin has potent activity against gram-positive bacteria including Listeria monocytogenes, Staphylococcus aureus, Clostridium difficile, and vancomycin-resistant Enterococci (VRE).
A 2025 study developed nisin-loaded niosomes in fast-dissolving oral films for buccal drug delivery, demonstrating activity against MRSA and expanding nisin's potential beyond food preservation into clinical antimicrobial delivery.[1]
Hourigan et al. (2025) conducted a genome-wide analysis of lanthipeptide gene clusters across prokaryote genomes, finding that lanthipeptide production systems frequently co-localize with genetic exchange elements and defense systems, suggesting bacteriocin production is deeply integrated into bacterial competition and horizontal gene transfer strategies.[2]
Class II: Non-Lantibiotic Peptides
Class II bacteriocins are small, heat-stable peptides that lack the extensive post-translational modifications of lantibiotics. They are further divided into subclasses:
Class IIa (pediocin-like): These are the most studied non-lantibiotics. They share a conserved N-terminal YGNGV motif and have strong anti-Listeria activity. Pediocin PA-1, produced by Pediococcus acidilactici, is commercially available as ALTA 2341 for food biopreservation.
Class IIb (two-peptide): These require two complementary peptides for full activity. Neither peptide alone has significant antimicrobial function; synergy between the pair is required for membrane disruption.
Class IIc (circular): These bacteriocins have their N- and C-termini covalently linked to form a circular structure, providing exceptional stability against proteases and extreme pH conditions.
Class IId (unmodified, linear): A catch-all category for non-modified peptides that do not fit other subclasses.
Class III: Bacteriolysins
These are large (>30 kDa) heat-labile proteins that kill bacteria through enzymatic degradation of the cell wall rather than pore formation. Lysostaphin, which specifically cleaves the pentaglycine cross-bridge in staphylococcal cell walls, is the best-known example. Their size and enzymatic mechanism make them functionally distinct from the smaller peptide bacteriocins.
How Bacteriocins Kill
The killing mechanisms of bacteriocins parallel those of other antimicrobial peptides but with additional specificity conferred by receptor recognition. The primary mechanisms include:
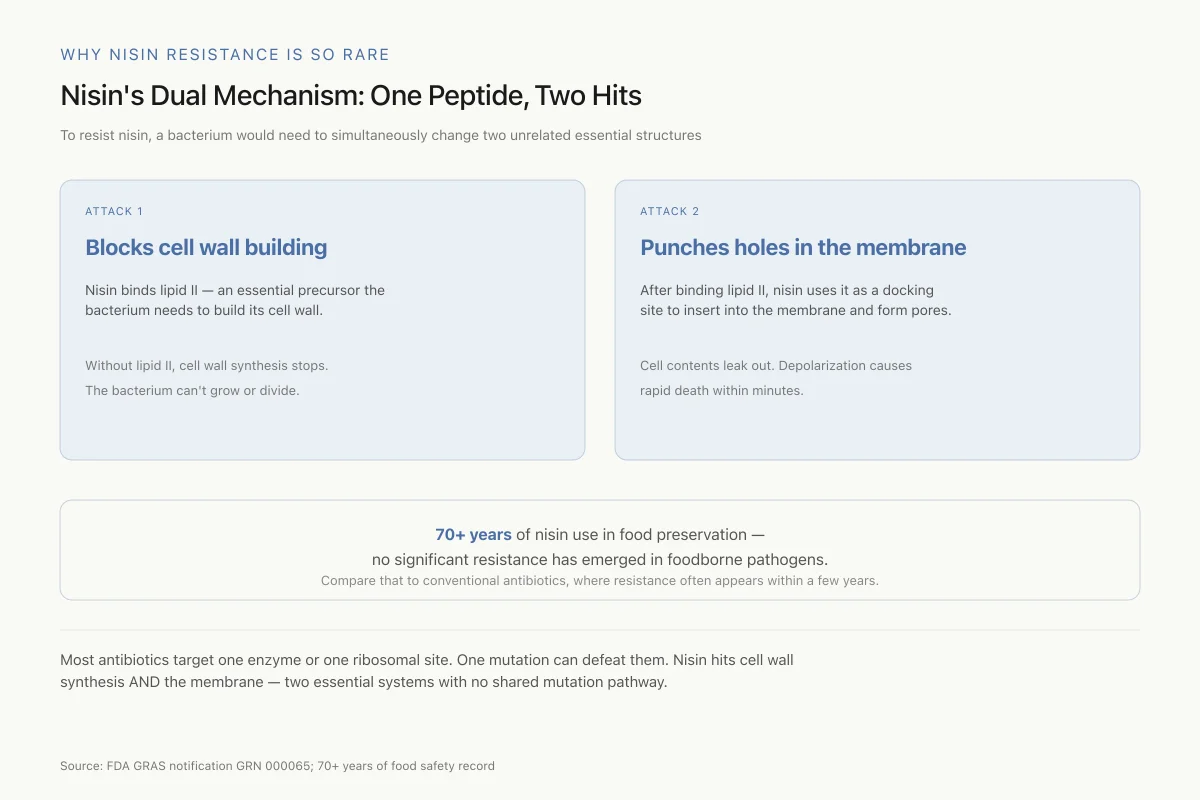
Lipid II binding and pore formation (Class I). Nisin and related lantibiotics bind to lipid II, a universally conserved precursor in bacterial cell wall synthesis. This binding serves two purposes: it blocks cell wall synthesis directly, and it uses lipid II as a docking molecule to insert into the membrane and form large pores. The pores cause rapid depolarization, loss of cytoplasmic contents, and cell death within minutes. This dual mechanism of action is why nisin resistance is rare despite decades of commercial use.
Membrane disruption without receptor binding (Class IIa). Pediocin-like bacteriocins interact with the mannose phosphotransferase system (Man-PTS) on the target cell surface, using it as a receptor to initiate membrane permeabilization. This receptor dependency explains the narrow spectrum of activity: only bacteria expressing compatible Man-PTS variants are susceptible.
Enzymatic cell wall degradation (Class III). Bacteriolysins degrade specific bonds in the peptidoglycan layer. Because the target bond varies between bacterial species (the pentaglycine bridge is specific to staphylococci), these enzymes can be highly selective killers.
Bacteriocins Against Drug-Resistant Pathogens
The antimicrobial resistance crisis has renewed scientific interest in bacteriocins as therapeutic agents. Several recent studies demonstrate their potential against the most problematic resistant organisms.
Ohdan et al. (2026) discovered a Staphylococcus capitis strain (HBC3) that naturally produces two bacteriocins simultaneously: capidermicin and micrococcin P1.[3] This dual-bacteriocin producer showed broad-spectrum antimicrobial activity against MRSA. The finding illustrates a natural strategy that bacteria themselves use: deploying multiple antimicrobial peptides simultaneously to overcome resistance.
Laxmi et al. (2026) reviewed the evidence for lactic acid bacteria-derived bacteriocins against multidrug-resistant neonatal sepsis pathogens.[4] Neonatal sepsis kills an estimated 680,000 newborns annually, and rising multidrug resistance among the causative organisms (E. coli, Klebsiella, Enterobacter, group B Streptococcus) is making standard antibiotic regimens increasingly unreliable. LAB-derived bacteriocins represent a potential alternative with activity against several of these pathogens, though clinical translation remains in early stages.
A 2025 study demonstrated an antibiotic-free antimicrobial combination: bacteriocins paired with a peptidoglycan hydrolase for treating bovine mastitis.[5] The combination showed efficacy both in vitro and in vivo, confirming that bacteriocin-based formulations can work against real infections in animal models without any conventional antibiotic component. This is significant because bovine mastitis treatment is a major contributor to agricultural antibiotic use and resistance selection.
For analysis of how AMPs more broadly are being combined with conventional antibiotics for synergistic effects, see Antimicrobial Peptides vs Antibiotics: The Evidence.
Nisin: The Gold Standard Bacteriocin
Why Nisin Resistance Is So Rare
Nisin's Dual Mechanism: One Peptide, Two Hits
To resist nisin, a bacterium would need to simultaneously change two unrelated essential structures
Most antibiotics target one enzyme or one ribosomal site. One mutation can defeat them. Nisin simultaneously targets cell wall synthesis AND the membrane — two essential systems with no shared mutation pathway. That's why it has aged so well.
Source: FDA GRAS notification GRN 000065; 70+ years of food safety record
 View as image
View as imageNisin deserves special attention as the only bacteriocin with widespread commercial use as an antimicrobial agent. Produced by certain strains of Lactococcus lactis during milk fermentation, nisin was first described in 1928 and has been deliberately used in food preservation since the early 1950s. It received FDA GRAS status in 1988 and is approved for food use in over 50 countries.
The safety record is extensive. Decades of use in dairy products, canned foods, processed meats, and beverages have produced no reports of allergic reactions, toxicity, or resistance emergence in foodborne pathogens. This track record is unusual for any antimicrobial agent and supports the argument that bacteriocin-based approaches may be inherently less prone to resistance than conventional antibiotics.
Recent research is pushing nisin beyond food preservation into clinical antimicrobial applications. Gelen Gungor et al. (2025) developed liposomes co-delivering azithromycin and nisin for skin infections, finding that the combination maintained bactericidal activity while reducing the selection pressure for antimicrobial resistance compared to azithromycin alone.[6] The liposomal delivery format protected nisin from proteolytic degradation, addressing one of the main obstacles to clinical use of peptide antimicrobials.
Amer et al. (2025) took a different delivery approach, formulating nisin-loaded niosomes (non-ionic surfactant vesicles) into fast-dissolving oral films for buccal administration.[1] The platform showed activity against MRSA and other gram-positive pathogens, representing a potential oral healthcare application for a peptide originally developed for food safety.
The Untapped Diversity of Bacteriocin Arsenals
One of the most remarkable findings from recent genomic studies is the sheer scale of bacteriocin diversity that remains unexplored. Revilla-Guarinos et al. (2026) characterized Streptococcus dentisani 7746, an oral commensal bacterium that encodes 14 different bacteriocins regulated by two quorum sensing systems (Com and Blp-like).[7] A single bacterial strain carrying 14 distinct antimicrobial peptides indicates the extent of chemical warfare occurring in complex microbial communities like the oral cavity.
This diversity has practical implications. Each bacteriocin has a distinct spectrum of activity, mechanism of action, and resistance profile. Deploying combinations of bacteriocins, as bacteria themselves do naturally, could provide broader-spectrum coverage and reduce the probability of resistance emergence. The challenge is moving from genomic identification to functional characterization and formulation at scale.
Caly-Simbou et al. (2026) reviewed bacteriocin applications in plant pathology, demonstrating that the potential extends beyond human health into agricultural crop protection.[8] Bacteriocins with activity against phytopathogenic bacteria could reduce agricultural antibiotic use, which is a significant contributor to environmental resistance selection.
For more on how the microbiome's peptide production extends beyond antimicrobial functions to influence brain chemistry and behavior, see How the Microbiome Produces Neuroactive Peptides That Affect Your Brain. For the concept that bacterial metabolites retain biological activity even after the producing bacteria die, see Postbiotics: When Dead Bacteria Still Produce Useful Peptides.
Limitations and Challenges
Despite their promise, bacteriocins face several obstacles on the path from laboratory to clinic. These are genuine barriers that temper the enthusiasm generated by in vitro studies and animal models.
Head-to-Head
Bacteriocins vs. Conventional Antibiotics
Different strengths for different problems — combination approaches are where the field is heading
The most likely future isn't bacteriocins replacing antibiotics. It's bacteriocins + antibiotics in combinations — one providing broad coverage, the other providing resistance-proofing. That's how bacteria themselves fight each other naturally.
Source: General bacteriocin pharmacology; Kranjec et al. (2025); Gelen Gungor et al. (2025)
 View as image
View as imageNarrow spectrum. The specificity that makes bacteriocins ecologically precise also limits their clinical utility. A bacteriocin active against one Listeria species may have no effect on Salmonella. Clinical infections often involve organisms that standard bacteriocin panels do not cover, particularly gram-negative pathogens where the outer membrane provides an additional barrier to peptide access. The nisin-type lantibiotics that target lipid II are primarily effective against gram-positive organisms. Extending bacteriocin activity to gram-negative pathogens, the group that includes many of the most dangerous antibiotic-resistant organisms (carbapenem-resistant Enterobacteriaceae, MDR Pseudomonas, Acinetobacter), remains a significant challenge. Approaches under investigation include combining bacteriocins with outer membrane permeabilizers, engineering chimeric bacteriocins with gram-negative targeting domains, and using bacteriocin-antibiotic combinations where the antibiotic provides gram-negative coverage.
Proteolytic degradation. Like all peptides, bacteriocins are susceptible to degradation by human proteases. Systemic delivery faces the same challenges as other antimicrobial peptides: short half-lives, rapid renal clearance, and limited tissue penetration. Liposomal and nanoparticle delivery systems are being developed to address this, as shown by the nisin formulation studies described above. Circular bacteriocins (Class IIc) offer a natural solution: their covalently closed backbone provides inherent resistance to exopeptidases, though endopeptidases can still cleave them.
Manufacturing scale and cost. Producing bacteriocins at pharmaceutical grade and scale is more expensive than conventional antibiotic manufacturing. Fermentation-based production using the native producing organism is the most practical route, but purification to clinical standards adds cost. Heterologous expression in E. coli or yeast can increase yields for some bacteriocins, but post-translational modifications required by lantibiotics must be replicated by co-expressing the modification enzymes, adding complexity. The cost equation may improve as fermentation technology advances and as the economic burden of antibiotic resistance (estimated at $55 billion annually in the US alone) increases the willingness to invest in alternatives.
Regulatory ambiguity. Bacteriocins for food use have clear regulatory pathways (GRAS for nisin, food additive approvals for pediocin). Bacteriocins for clinical therapeutic use face the full drug approval process, which requires investment that the narrow-spectrum nature of individual bacteriocins makes commercially difficult to justify. A bacteriocin effective against only Listeria or only S. aureus has a smaller potential market than a broad-spectrum antibiotic, making the cost of clinical trials harder to recoup. Combination products (multiple bacteriocins, or bacteriocin-antibiotic pairs) face even more complex regulatory paths because each component requires safety data.
Safety
LowClinical bacteriocin therapy is still experimental
Concern
Nisin's 70-year food safety record is excellent, but clinical use (systemic infection treatment, MRSA therapy) is a different application with different risks. Liposomal nisin and bacteriocin-antibiotic combinations are in early preclinical and animal studies — no bacteriocin has completed a Phase 3 trial for human infection.
What the research says
Current clinical bacteriocin research is largely focused on topical applications (skin infection, oral care) and animal models (mastitis). Systemic human use would require dedicated safety studies. The favorable food-safety track record is encouraging but doesn't automatically transfer to injected or systemic dosing.
Particularly relevant for: Anyone considering bacteriocin-based products for infection treatment
What to do
Current evidence supports nisin as a food preservative. Clinical bacteriocin products should be considered experimental until Phase 2/3 trial data becomes available.
FDA GRAS notification for nisin; current clinical trial registry
Resistance is rare but not impossible. While nisin resistance has not been a practical problem in food applications over 70 years, laboratory studies can select for resistance through sequential passage at sub-inhibitory concentrations. Known resistance mechanisms include modifications to the lipid II target, changes in cell membrane composition, and production of nisin-degrading enzymes (nisinase). Clinical use at higher selective pressures and longer durations could potentially generate resistance that food applications have not. The dual mechanism of nisin (lipid II binding plus pore formation) and the difficulty of simultaneously altering both targets are reasons for cautious optimism, but resistance cannot be assumed impossible.
Bacteriocins and the Gut Microbiome
The human gut contains trillions of bacteria engaged in constant competitive interactions, and bacteriocins are among the primary weapons in this competition. The bacteriocin profile of an individual's gut microbiome influences which bacterial species can colonize and persist. Probiotic bacteria often owe part of their beneficial effects to bacteriocin production: by killing pathogenic competitors, they create ecological space for themselves and other beneficial organisms.
This has implications for understanding how the microbiome is shaped and maintained. Bacteriocin-producing strains can resist colonization by pathogenic invaders not through the host immune system but through direct chemical warfare. The observation that a single oral commensal strain can encode 14 different bacteriocins[7] suggests that the antimicrobial peptide landscape of the human microbiome is far more complex and dynamic than currently appreciated.
The relationship between bacteriocin production and broader microbiome health is an active area of research. Whether supplementing the gut with specific bacteriocin-producing strains could treat dysbiosis, prevent pathogenic colonization, or modulate immune responses remains to be established in controlled human studies.
The concept extends to the oral microbiome as well. The discovery of S. dentisani 7746 with its 14-bacteriocin arsenal[7] came from oral cavity sampling, and the strain has been investigated as a potential probiotic for dental caries prevention. By producing bacteriocins that kill cariogenic Streptococcus mutans while being itself a commensal organism, S. dentisani exemplifies the therapeutic potential of harnessing natural bacteriocin production within microbial communities.
The broader implication is that bacteriocins are not just potential drugs to be manufactured and delivered exogenously. They are endogenous components of the microbiome's self-regulation system. Understanding which bacteriocins are produced, by which organisms, under which conditions, and with what ecological effects is a prerequisite for any attempt to deliberately manipulate microbiome composition for health benefit. The field of postbiotics, which focuses on beneficial compounds produced by microorganisms including dead ones, overlaps significantly with bacteriocin research. For more on this intersection, see Postbiotics: When Dead Bacteria Still Produce Useful Peptides.
The connection between gut microbial peptides and systemic health extends beyond antimicrobial function. Some microbial peptides cross the gut-brain axis and influence neurological function. For that dimension of microbiome peptide biology, see How the Microbiome Produces Neuroactive Peptides That Affect Your Brain. For a broader view of how alpha-defensins produced by Paneth cells interact with bacterial bacteriocins to shape the intestinal microbial ecosystem, see our defensin coverage.
The Bottom Line
Bacteriocins are a diverse and ancient class of antimicrobial peptides produced by bacteria to kill competing strains. Nisin, the most commercially successful, has been used safely in food preservation for over 70 years without significant resistance emergence. Recent research is expanding bacteriocin applications toward treating drug-resistant infections, with studies demonstrating efficacy against MRSA, neonatal sepsis pathogens, and agricultural infections. Genomic surveys reveal enormous untapped diversity, with individual bacterial strains encoding arsenals of 14 or more distinct bacteriocins. The main barriers to clinical translation remain narrow spectrum of activity, proteolytic instability, manufacturing costs, and regulatory pathways. These are real challenges, but the fundamental biology of bacteriocins, their membrane-targeting mechanisms, their natural diversity, and their decades-long safety record in food applications, provides a strong foundation for continued development as antimicrobial resistance narrows the conventional antibiotic arsenal.
Sources & References
- 1RPEP-09924·Amer, Ali A et al. (2025). “Nisin Antimicrobial Peptide in Fast-Dissolving Oral Films: Novel Delivery.” Molecules (Basel.Study breakdown →PubMed →↩
- 2RPEP-11428·Hourigan, David et al. (2025). “Antimicrobial Peptide Genes Sit Next to Defense and Gene-Transfer Systems in Bacterial Genomes.” BMC genomics.Study breakdown →PubMed →↩
- 3RPEP-15822·Ohdan, Keijuro et al. (2026). “Staphylococcus capitis strain producing dual bacteriocins, capidermicin and micrococcin P1, shows broad-spectrum antimicrobial activity..” Scientific reports.Study breakdown →PubMed →↩
- 4RPEP-15486·Laxmi, Vijay et al. (2026). “Antimicrobial Peptides from Probiotic Bacteria Could Fight Drug-Resistant Infections in Newborns.” Probiotics and antimicrobial proteins.Study breakdown →PubMed →↩
- 5RPEP-11922·Kranjec, Christian et al. (2025). “An antibiotic-free antimicrobial combination of bacteriocins and a peptidoglycan hydrolase: in vitro and in vivo assessment of its efficacy..” Applied and environmental microbiology.Study breakdown →PubMed →↩
- 6RPEP-11077·Gelen-Gungor, Dilek et al. (2025). “Co-delivery of azithromycin and nisin through liposomes for skin infection to reduce antimicrobial drug resistance..” International journal of pharmaceutics.Study breakdown →PubMed →↩
- 7RPEP-15990·Revilla-Guarinos, Ainhoa et al. (2026). “Oral Probiotic Bacterium Carries a Record 14 Antimicrobial Peptide Genes — More Than Any Known Bacterial Isolate.” Journal of oral microbiology.Study breakdown →PubMed →↩
- 8RPEP-14928·Caly-Simbou, Eva et al. (2026). “Bacteriocins as Natural Antimicrobial Peptides for Protecting Crops from Disease.” Biochemical and biophysical research communications.Study breakdown →PubMed →↩