Alpha-Defensins: Neutrophil Peptides That Kill
Defensins and Innate Immunity
6 Human Alpha-Defensins
Six alpha-defensins have been identified in humans: four in neutrophil granules (HNP-1 through HNP-4) and two in intestinal Paneth cells (HD-5 and HD-6). Together they form one of the oldest and most conserved antimicrobial defense systems in mammalian biology.
Ganz et al., Journal of Clinical Investigation, 1985
Ganz et al., Journal of Clinical Investigation, 1985
If you only read one thing
Alpha-defensins are tiny germ-killing peptides your body makes on its own. You have six of them — four ride inside white blood cells called neutrophils, and two live in your gut lining. When a neutrophil finds a bacteria, it dumps these peptides onto it and they punch holes in its membrane until it dies. The gut versions protect your intestines from the trillions of bacteria living there. They were discovered in 1985 and are still one of our oldest, most reliable immune weapons.
Key Takeaways
- Your body makes six tiny germ-killing peptides called alpha-defensins. Four live in your white blood cells, two live in your gut lining.
- They're not rare. In your neutrophils — a common white blood cell — alpha-defensins make up 5% of all the protein. That's a lot of weaponry.
- When a neutrophil finds bacteria, it dumps these peptides on them. They punch holes in the bacterial outer layer and the bacteria die within minutes.
- One of the gut versions (HD-6) does something surprising. It doesn't kill bacteria directly — it weaves itself into a net that physically traps them.
- Crohn's disease involves a broken version of this system. People with Crohn's often have Paneth cells that produce too few defensins, letting gut bacteria invade.
- Despite 40 years of research, no defensin has become an FDA-approved drug. Short half-lives and manufacturing cost have blocked every attempt.
- Bacteria can't easily evolve resistance to them the way they do to antibiotics — which is why defensin-inspired drugs are still being pursued.
Human Alpha-Defensin Family
All Six, Side by Side
Four in neutrophils, two in the gut — each with a distinct job
Source: Neutrophil
Abundance: ~5% total neutrophil protein
Most abundant, 30aa, alanine at position 1
Source: Neutrophil
Abundance: Proteolytic product
N-terminal cleavage of HNP-1/HNP-3
Source: Neutrophil
Abundance: ~5% total neutrophil protein
Differs from HNP-1 by one residue (aspartate)
Source: Neutrophil
Abundance: ~100× less than HNP-1
Outlier — 22 residues differ from HNP-1
Source: Paneth cell
Abundance: Constitutive + induced
Strongest cationic charge, broad-spectrum killing
Source: Paneth cell
Abundance: Constitutive + induced
Self-assembles into nanonets that trap bacteria
HNP-1/2/3 are nearly identical by sequence but encoded by separate genes. HNP-4 is an outlier with broader activity against certain Gram-negatives. HD-5 kills bacteria directly; HD-6 traps them — a mechanism unique among all known antimicrobial peptides.
Source: Ganz et al. (1985); Hu et al. (2019); Chairatana et al. (2019); Xu et al. (2018)
 View as image
View as imageIn 1985, a team led by Tomas Ganz, Michael Selsted, and Robert Lehrer isolated three small peptides from human neutrophil granules that killed Staphylococcus aureus, Pseudomonas aeruginosa, and Escherichia coli on contact in vitro.[1] They named them defensins. Four decades later, alpha-defensins have been found in neutrophils, intestinal Paneth cells, and the female reproductive tract. They kill bacteria, fungi, and enveloped viruses. They modulate inflammation. They influence blood clotting and tumor biology. And they remain one of the most studied families of antimicrobial peptides in immunology. For the broader defensin family including beta-defensins, see Defensins: Your Body's First Line of Antimicrobial Defense.
The Six Human Alpha-Defensins
Humans express six alpha-defensins, divided into two groups by their source tissue.
Neutrophil Alpha-Defensins (HNP-1 through HNP-4)
Human neutrophil peptides 1, 2, and 3 (HNP-1, HNP-2, HNP-3) are stored in azurophilic (primary) granules of neutrophils. Together they constitute roughly 5% of total neutrophil protein by mass, making them among the most abundant proteins in these white blood cells.[1] When neutrophils encounter pathogens and degranulate, HNP-1 through HNP-3 are released at concentrations high enough to kill bacteria directly.
HNP-1 and HNP-3 are 30 amino acids long, differing by a single residue at position 1: alanine in HNP-1, aspartate in HNP-3. HNP-2 is 29 amino acids, a proteolytic product formed by cleavage of the N-terminal residue from either HNP-1 or HNP-3. Despite these minimal sequence differences, the three peptides are encoded by separate genes (DEFA1 and DEFA3), and the DEFA1/DEFA3 gene cluster shows significant copy number variation between individuals, ranging from 2 to 17 copies per diploid genome. This variation directly affects neutrophil defensin levels and may influence susceptibility to infection.
HNP-4 is the outlier. Encoded by DEFA4, it is 33 amino acids long, with 22 residues differing from HNP-1 through HNP-3. It is a minor component of neutrophil granules, present at roughly 100-fold lower concentrations than HNP-1. A 2019 systematic mutational analysis of HNP-4 found that its unique charge distribution confers broader antimicrobial activity than HNP-1 against certain Gram-negative organisms.[2]
Enteric Alpha-Defensins (HD-5 and HD-6)
Human defensin 5 (HD-5) and human defensin 6 (HD-6) are produced by Paneth cells, specialized secretory cells at the base of small intestinal crypts. When bacteria contact the crypt surface, Paneth cells release HD-5 and HD-6 into the intestinal lumen.
HD-5 has the strongest cationic charge among human alpha-defensins, which correlates with its broad-spectrum bactericidal activity.[3] HD-6 functions differently: rather than killing bacteria through membrane disruption, it self-assembles into ordered structures called nanonets that physically entangle microbes, preventing them from reaching the epithelial surface.[4] This trapping mechanism is unique among known antimicrobial peptides. Butyric acid and leucine, products of microbial fermentation, stimulate Paneth cell alpha-defensin secretion, creating a feedback loop between the microbiome and host defense.[5]
For more on how Paneth cell defensins maintain microbial balance, see Defensins and the Gut: Maintaining Microbial Balance in Your Intestines.
Structure: Three Disulfide Bonds
All alpha-defensins share a conserved structural scaffold: a triple-stranded beta-sheet stabilized by three intramolecular disulfide bonds connecting six conserved cysteine residues. The disulfide pairing pattern (Cys1-Cys6, Cys2-Cys4, Cys3-Cys5) distinguishes alpha-defensins from beta-defensins, which have a different Cys1-Cys5, Cys2-Cys4, Cys3-Cys6 pairing. This structural difference is one of the key distinctions covered in Beta-Defensins: The Epithelial Barrier Peptides in Skin, Gut, and Lungs.
The disulfide bonds were long assumed to be essential for antimicrobial activity. A 2026 study in PLoS Pathogens challenged this assumption: breaking the disulfide bonds of a weakly bactericidal alpha-defensin released a more potent antimicrobial form.[6] The reduced (disulfide-free) peptide adopted a different conformation that enhanced membrane interaction. This finding suggests the disulfide scaffold may serve structural and regulatory roles rather than being directly required for bacterial killing.
A 2025 structural comparison of HNP-1 with the cathelicidin LL-37 and magainin-2 revealed that each antimicrobial peptide uses a distinct membrane-interaction mechanism despite convergent killing outcomes.[7] HNP-1 uses a carpet-like model, coating the bacterial membrane surface until critical concentration is reached, while LL-37 operates through a toroidal pore mechanism. For more on how these peptides distinguish microbial membranes from host cells, see How Defensins Distinguish Bacteria from Your Own Cells.
How Alpha-Defensins Kill Microbes
Bacteria
The original 1985 discovery demonstrated direct killing of S. aureus (Gram-positive), P. aeruginosa and E. coli (Gram-negative), and the fungus Candida albicans.[1] The killing mechanism depends on electrostatic attraction: the cationic (positively charged) defensin peptides are attracted to the anionic (negatively charged) phospholipids that dominate bacterial cell membranes. Mammalian cell membranes have a neutral outer leaflet rich in cholesterol, which defensins do not target efficiently. This charge asymmetry is the basis for selectivity.
Once bound to the bacterial membrane, alpha-defensins disrupt membrane integrity through what structural studies describe as a carpet model: peptides accumulate on the membrane surface until reaching a threshold concentration, at which point they insert into and permeabilize the lipid bilayer.[7] The result is loss of membrane potential, leakage of cellular contents, and death. This mechanism is difficult for bacteria to evolve resistance against because it targets a fundamental physical property of the membrane rather than a specific protein receptor.
Mechanism Comparison
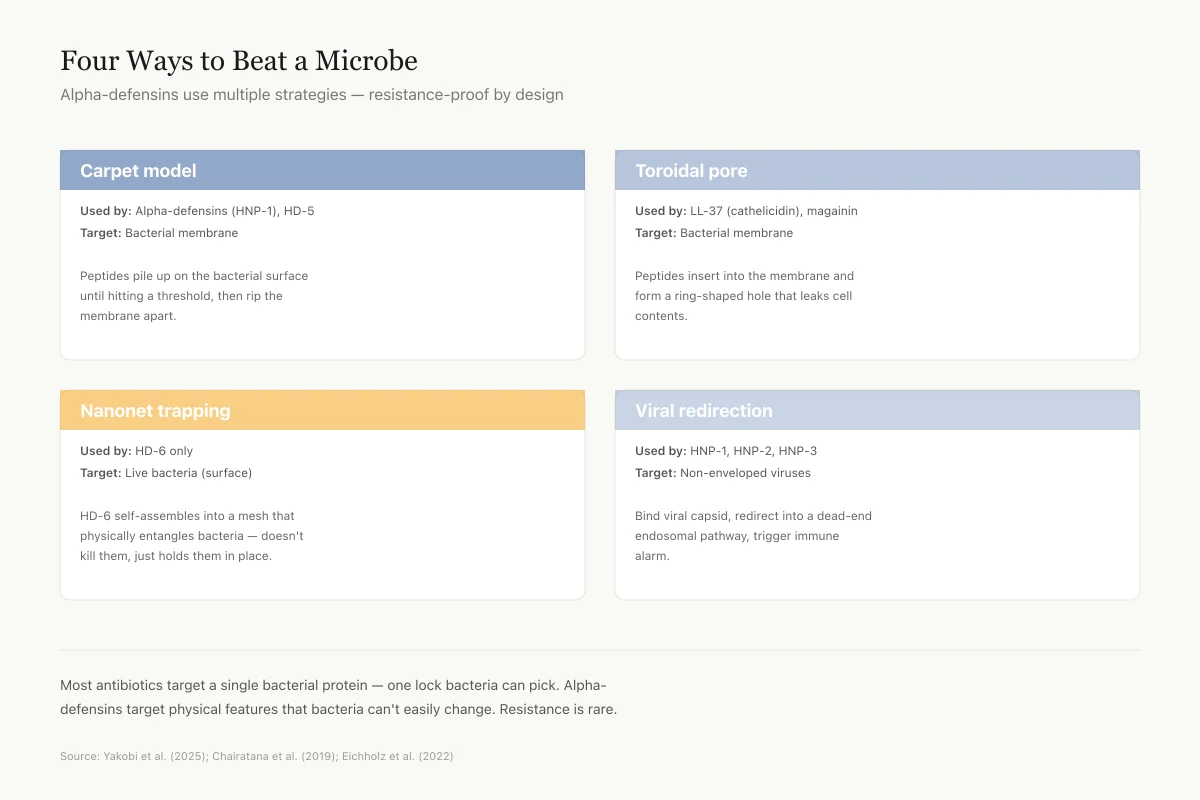
Four Different Ways to Beat a Microbe
Alpha-defensins use multiple strategies — resistance-proof by design
Carpet model
Used by: Alpha-defensins (HNP-1), HD-5
Target: Bacterial membrane
Peptides pile up on the bacterial surface until they hit a threshold, then rip the membrane apart.
Toroidal pore
Used by: LL-37 (cathelicidin), magainin
Target: Bacterial membrane
Peptides insert into the membrane and form a ring-shaped hole that leaks cell contents out.
Nanonet trapping
Used by: HD-6 only
Target: Live bacteria (surface)
HD-6 self-assembles into a mesh-like net that physically entangles bacteria without killing them directly.
Viral redirection
Used by: HNP-1, HNP-2, HNP-3
Target: Non-enveloped viruses (adenovirus)
Bind to viral capsid, redirect the virus into a dead-end endosomal pathway, trigger immune alarm.
Most antibiotics target a single bacterial protein — one lock bacteria can eventually pick. Alpha-defensins target physical features (charge, membrane lipids, viral capsid shape) that bacteria and viruses can't easily change. That's why resistance is rare.
Source: Yakobi et al. (2025); Chairatana et al. (2019); Eichholz et al. (2022)
 View as image
View as imageA 2025 review of HNP-1 described additional intracellular mechanisms: after membrane permeabilization, HNP-1 may inhibit bacterial DNA, RNA, and protein synthesis, suggesting a multi-target killing strategy.[8]
Viruses
Alpha-defensins directly inactivate enveloped viruses including herpes simplex virus type 1 (demonstrated in the original 1985 study), influenza A, and HIV-1. The antiviral mechanism differs from antibacterial killing. For enveloped viruses, defensins bind to viral envelope glycoproteins and block fusion with host cell membranes, preventing the virus from entering cells. The lectins and glycoprotein interactions involved are specific to each virus type.
For non-enveloped viruses, alpha-defensins use a distinct strategy. Eichholz et al. (2022) showed that HNP-1 through HNP-3 bind to adenovirus capsids and redirect the virus into a non-productive endosomal pathway, triggering NLRP3 inflammasome activation and IL-1-beta maturation in macrophages.[9] The virus enters the cell but is diverted into a degradative compartment rather than reaching the nucleus. This represents a fundamentally different antiviral mechanism: instead of destroying the virus directly, defensins exploit it to activate innate immune signaling. A 2025 study confirmed that alpha-defensins also neutralize adeno-associated virus (AAV) vectors through similar capsid-binding mechanisms, which has implications for gene therapy delivery.
Fungi
Alpha-defensins kill Candida albicans and other fungal species through membrane disruption similar to their antibacterial mechanism. The positively charged peptide binds to the negatively charged fungal cell membrane, causing permeabilization and cell death. Theta-defensins, cyclic defensins found in non-human primates that evolved from alpha-defensin precursors, show even stronger antifungal activity against multidrug-resistant Candida auris, killing the fungus at concentrations where conventional antifungals fail.[10] Humans lost the ability to produce theta-defensins due to a premature stop codon in the theta-defensin gene, though the gene remnants persist in the human genome as pseudogenes.
Resistance
One of the most cited advantages of antimicrobial peptides over conventional antibiotics is the difficulty bacteria face in evolving resistance. Because alpha-defensins target the physical properties of the bacterial membrane (charge and lipid composition) rather than a specific protein target, bacteria would need to fundamentally alter their membrane architecture to evade killing. Some bacteria have evolved partial resistance by modifying their surface charge through lipid A modifications or by producing proteases that degrade defensins, but these adaptations typically impose fitness costs. Mycobacterium abscessus, for example, can escape defensin bactericidal activity when opsonized (coated with antibodies), redirecting the immune response in a way that reduces defensin effectiveness.[19]
Beyond Killing: Immune Regulation
Alpha-defensins do more than kill pathogens. Research over the past two decades has revealed roles in inflammation control, adaptive immunity, and tissue repair.
Inflammation Control
Brook and colleagues published a 2016 study in PNAS that identified a specific anti-inflammatory mechanism for HNP-1.[11] HNP-1 enters macrophages, the immune cells that drive inflammatory responses, and directly inhibits mRNA translation without triggering the unfolded protein response or degrading mRNA. By shutting down protein synthesis in activated macrophages, HNP-1 functions as a molecular brake: it allows neutrophils to clear pathogens while preventing macrophages from producing excessive inflammatory cytokines that cause tissue damage.
This dual function (pathogen killing plus inflammation limitation) positions alpha-defensins as coordinators of the acute immune response rather than simple antimicrobial agents.
A 2025 study by Lee and colleagues found that alpha-defensins increase binding of non-typeable Haemophilus influenzae (NTHi) to macrophages without increasing engulfment, a mechanism relevant to chronic airway inflammation in alpha-1 antitrypsin deficiency.[12]
Cell Adhesion and Wound Healing
Howell et al. (2018) demonstrated that human defensins promote cell adhesion, a property relevant to wound closure and tissue repair.[13] Alpha-defensins enhanced the adhesion of epithelial cells and fibroblasts to extracellular matrix components, suggesting a role in tissue repair that extends beyond infection control.
Thrombosis
Abu-Fanne and colleagues published in Blood (2019) that neutrophil alpha-defensins promote thrombosis in vivo by altering fibrin formation, structure, and stability.[14] When neutrophils degranulate at sites of vascular injury, released HNP-1 through HNP-3 incorporate into forming fibrin clots and make them denser and more resistant to fibrinolysis (clot breakdown). This connection between innate immunity and coagulation helps explain why severe infections are often accompanied by thrombotic complications.
Alpha-Defensins and Disease
Crohn's Disease
Wehkamp and colleagues demonstrated in 2005 that patients with ileal Crohn's disease have reduced Paneth cell expression of HD-5 and HD-6.[15] This reduction was specific to ileal (small intestinal) Crohn's, not colonic Crohn's or ulcerative colitis. The finding provided a mechanistic link between defensin deficiency and the microbial dysbiosis observed in Crohn's patients: with fewer defensins protecting the crypt, bacteria gain access to the epithelial surface and trigger the chronic inflammatory cycle characteristic of the disease.
Shimizu et al. (2020) extended this finding, showing that alpha-defensin misfolding, not just reduced expression, correlates with dysbiosis and ileitis in mouse models of Crohn's disease.[16] Misfolded defensins lose antimicrobial function even when present at normal concentrations, suggesting that defensin quality matters as much as quantity. The genetic basis for this deficiency involves reduced Wnt/Tcf-4 signaling in Paneth cells, which controls defensin gene transcription, and NOD2 mutations that independently impair defensin expression. NOD2 mutations are the strongest known genetic risk factor for ileal Crohn's disease. This Crohn's connection is the most clinically advanced area of alpha-defensin research.
Cancer
Alpha-defensins have shown both pro-tumor and anti-tumor effects depending on context. The 2020 review by Xu et al. described this as the "double-edged sword" of defensins in host immunity.[17] HNP-1 through HNP-3 are elevated in the serum of patients with certain cancers. Whether they are fighting the tumor, responding to associated infection, or contributing to tumor-associated inflammation remains unresolved.
Tsiaoussis et al. (2018) measured alpha-defensin expression alongside CD20+ B-lymphocytes and CD3+ T-lymphocytes in colonic neoplasia, finding altered defensin expression patterns in adenoma and carcinoma tissue compared to normal mucosa.[18]
Atherosclerosis and Vascular Disease
Alpha-defensins are increasingly recognized as participants in atherogenesis. Neutrophils that infiltrate atherosclerotic plaques release HNP-1 through HNP-3, which promote smooth muscle cell proliferation, enhance LDL uptake by macrophages, and increase endothelial permeability. A 2025 review in Frontiers in Immunology described alpha-defensins as active contributors to vascular pathology in dialysis patients, where chronically elevated neutrophil activation drives defensin release into the circulation. The dual nature of alpha-defensins, protective against infection but potentially harmful in chronic inflammatory conditions, reflects a recurring theme in innate immunity research.
Periprosthetic Joint Infection
Outside of basic science, alpha-defensin has a clinical diagnostic application. The Synovasure alpha-defensin test detects elevated alpha-defensin levels in synovial fluid as a biomarker for periprosthetic joint infection after hip or knee replacement. When neutrophils respond to infection in the joint space, they release alpha-defensins at concentrations measurably higher than in sterile inflammation. The test has sensitivity and specificity above 90% in validated studies, making it one of the more reliable biomarkers in orthopedic infection diagnosis. This is currently the most direct clinical use of alpha-defensin measurement.
Evolutionary Conservation
Alpha-defensins are ancient. Defensin-like genes have been identified across mammals, birds, reptiles, and even invertebrates, suggesting the family originated hundreds of millions of years ago. In mammals, the alpha-defensin gene cluster has undergone extensive duplication and diversification, particularly in primates and rodents. Mice express over 20 Paneth cell alpha-defensins (called cryptdins), compared to just HD-5 and HD-6 in humans.
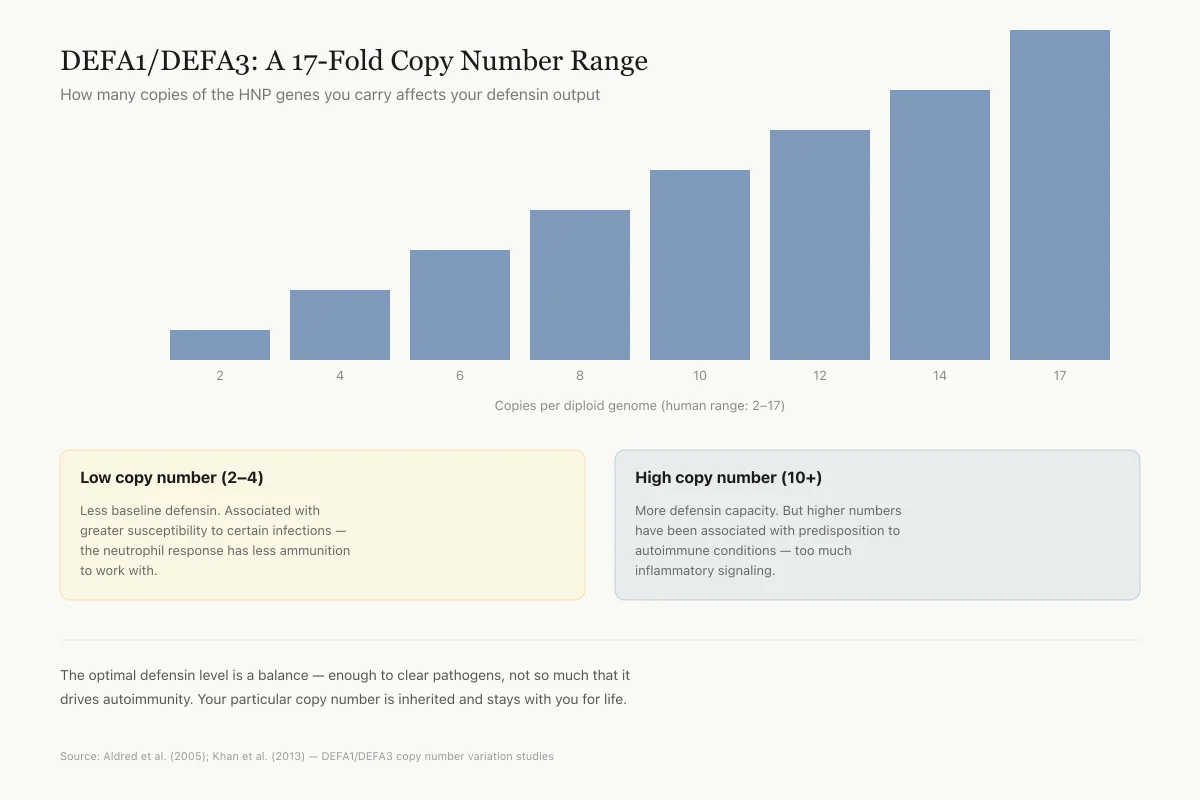
The copy number variation in the human DEFA1/DEFA3 gene cluster is one of the more dramatic examples of structural variation in the human genome. Individuals carry between 2 and 17 copies per diploid genome, and lower copy numbers have been associated with increased susceptibility to certain infections while higher copy numbers may predispose to autoimmune conditions. This variation suggests that the optimal defensin level is a balance: enough to control pathogens, not so much as to drive excessive inflammation.
Genetic Variation
DEFA1/DEFA3: A 17-Fold Copy Number Range
How many copies of the HNP genes you carry affects your defensin output
Copies per diploid genome (human range: 2–17)
Low copy number (2–4)
Less baseline defensin. Associated with greater susceptibility to certain infections — the neutrophil response has less ammunition to work with.
High copy number (10+)
More defensin capacity. But higher numbers have been associated with predisposition to autoimmune conditions — too much inflammatory signaling.
This is one of the most dramatic examples of structural variation in the human genome. The optimal defensin level is a balance — enough to clear pathogens, not so much that it drives autoimmunity. Your particular copy number is inherited and stays with you for life.
Source: Aldred et al. (2005); Khan et al. (2013) — DEFA1/DEFA3 copy number variation studies
 View as image
View as imageNakamura et al. (2020) characterized the expression and localization patterns of Paneth cells and their alpha-defensins across the length of the small intestine, finding highest expression in the ileum, which corresponds to the region most affected in Crohn's disease.[20]
What We Know and What We Do Not
Alpha-defensins are among the best-characterized antimicrobial peptides in human biology. The killing mechanisms are well-documented in vitro. The Paneth cell biology is established. The connection to Crohn's disease is supported by multiple independent groups.
What remains less clear is how alpha-defensin function can be therapeutically modulated. No alpha-defensin-based drug has been approved for any indication. The challenges are familiar to peptide pharmacology: short half-life in circulation, potential for toxicity at high systemic concentrations, and the difficulty of delivering peptides to specific tissue compartments. Recombinant production of correctly folded alpha-defensins with intact disulfide bonds adds manufacturing complexity. The 2025 HNP-1 review by Zhang et al. noted that while structure-to-function relationships are increasingly understood, translation to clinical application remains at early stages.[8]
Several therapeutic directions are being explored. The Crohn's disease connection offers the most direct clinical angle: if reduced Paneth cell defensin expression contributes to disease, could restoring defensin levels prevent or treat ileal Crohn's? That question has been asked since Wehkamp's 2005 publication and remains unanswered by clinical trial data. Approaches under investigation include small molecules that upregulate endogenous defensin production, recombinant defensin peptides for topical application in wound care, and defensin-derived fragments optimized for systemic stability.
The diagnostic application of alpha-defensin testing in periprosthetic joint infection demonstrates that clinical utility does not require therapeutic use. Biomarker applications may expand to other inflammatory conditions where neutrophil-derived defensin levels correlate with disease activity, including atherosclerosis and chronic lung disease.
Alpha-defensins also intersect with the broader antimicrobial peptide field. They work alongside cathelicidins like LL-37, beta-defensins in epithelial tissues, and other innate immune effectors. Understanding how these peptide families coordinate, potentially synergize, and sometimes conflict in vivo is an active area of research that will shape future therapeutic approaches.
The Bottom Line
Alpha-defensins are a family of six antimicrobial peptides: four stored in neutrophil granules (HNP-1 through HNP-4) and two secreted by intestinal Paneth cells (HD-5 and HD-6). They kill bacteria, fungi, and enveloped viruses through membrane disruption, while also regulating inflammation by blocking macrophage protein translation. Reduced Paneth cell defensin expression is linked to Crohn's disease. No alpha-defensin therapeutic has reached clinical approval, but the diagnostic alpha-defensin test for joint infection is in routine use.
Sources & References
- 1RPEP-00025·Ganz, T et al. (1985). “Defensins. Natural peptide antibiotics of human neutrophils..” The Journal of clinical investigation.Study breakdown →PubMed →↩
- 2RPEP-04235·Hu, Han et al. (2019). “Systematic mutational analysis of human neutrophil α-defensin HNP4..” Biochimica et biophysica acta. Biomembranes.Study breakdown →PubMed →↩
- 3RPEP-03995·Xu, Dan et al. (2018). “Human Enteric α-Defensin 5 Promotes Shigella Infection by Enhancing Bacterial Adhesion and Invasion..” Immunity.Study breakdown →PubMed →↩
- 4RPEP-04025·Unknown (2019). “Chairatana 2019 Dynamics Of Human Defensin.” .Study breakdown →↩
- 5RPEP-04503·Takakuwa, Akiko et al. (2019). “Butyric Acid and Leucine Induce α-Defensin Secretion from Small Intestinal Paneth Cells..” Nutrients.Study breakdown →PubMed →↩
- 6RPEP-15614·Luo, Gan et al. (2026). “Breaking disulfide bonds in a weakly bactericidal α-defensin unleashes a potent antimicrobial peptide with an altered conformation..” PLoS pathogens.Study breakdown →PubMed →↩
- 7RPEP-14295·Yakobi, Sinethemba H et al. (2025). “Structural and mechanistic divergence in LL-37, HNP-1, and Magainin-2: An integrated computational and biophysical analysis..” Current research in structural biology.Study breakdown →PubMed →↩
- 8RPEP-14500·Zhang, Jiaqi et al. (2025). “HNP-1: The Human Neutrophil Defensin That Kills Bacteria and Viruses — Structure, Function, and Clinical Potential.” Microorganisms.Study breakdown →PubMed →↩
- 9RPEP-06103·Eichholz, Karsten et al. (2022). “Alpha-Defensin Peptides Redirect Adenovirus Vaccines to Trigger Inflammasome Activation in Immune Cells.” Journal of virology.Study breakdown →PubMed →↩
- 10RPEP-03579·Basso, Virginia et al. (2018). “Monkey-Derived Circular Peptides Killed Drug-Resistant Candida Fungi 200 Times More Effectively Than Human Saliva Peptides.” Antimicrobial agents and chemotherapy.Study breakdown →PubMed →↩
- 11RPEP-02884·Brook, Matthew et al. (2016). “Neutrophil-derived alpha defensins control inflammation by inhibiting macrophage mRNA translation..” Proceedings of the National Academy of Sciences of the United States of America.Study breakdown →PubMed →↩
- 12RPEP-12018·Lee, Jungnam et al. (2025). “Alpha-defensins increase NTHi binding but not engulfment by the macrophages enhancing airway inflammation in Alpha-1 antitrypsin deficiency..” Frontiers in immunology.Study breakdown →PubMed →↩
- 13RPEP-03713·Howell, Katie et al. (2018). “Cell adhesion properties of human defensins..” Biochemical and biophysical research communications.Study breakdown →PubMed →↩
- 14RPEP-04033·Abu-Fanne, Rami et al. (2019). “Neutrophil α-defensins promote thrombosis in vivo by altering fibrin formation, structure, and stability..” Blood.Study breakdown →PubMed →↩
- 15RPEP-01099·Wehkamp, Jan et al. (2005). “Crohn's Disease of the Small Intestine Is Linked to a Specific Deficiency of Antimicrobial Peptides.” Proceedings of the National Academy of Sciences of the United States of America.Study breakdown →PubMed →↩
- 16RPEP-05132·Shimizu, Yu et al. (2020). “Misfolded Alpha-Defensins in Gut Paneth Cells Drive Microbiome Disruption and Crohn's-Like Inflammation.” Life science alliance.Study breakdown →PubMed →↩
- 17RPEP-05209·Xu, Dan et al. (2020). “Defensins Can Both Fight and Fuel Infections — A Review of Their Dual Role in Immunity.” Frontiers in immunology.Study breakdown →PubMed →↩
- 18RPEP-03952·Tsiaoussis, Georgios I et al. (2018). “Gut Antimicrobial Peptides α-Defensins Are Depleted in Liver Cirrhosis, Linked to Bacterial Toxin Leakage.” Digestive diseases and sciences.Study breakdown →PubMed →↩
- 19RPEP-07471·Touré, Hamadoun et al. (2023). “Mycobacterium abscessus Opsonization Allows an Escape from the Defensin Bactericidal Action in Drosophila..” Microbiology spectrum.Study breakdown →PubMed →↩
- 20RPEP-05021·Nakamura, Kiminori et al. (2020). “Paneth Cell Defensins Increase 4-50x from Upper to Lower Small Intestine in Mice.” Frontiers in immunology.Study breakdown →PubMed →↩