Annexin V: The Protein That Images Dying Heart Cells
Peptide-Based Cardiac Imaging
0.036 nM Kd
Annexin V binds phosphatidylserine on dying cells with sub-nanomolar affinity. Labeled with technetium-99m, it produces SPECT images of cell death in the living heart, detecting myocardial infarction, transplant rejection, and unstable atherosclerotic plaques.
Tait et al., Biochemistry, 1992
Tait et al., Biochemistry, 1992
If you only read one thing
Annexin V is a protein that grabs onto dying cells — specifically, a fatty molecule called phosphatidylserine that flips to the outside of cells as they start to die. Attach a tiny bit of radioactive tracer to annexin V, inject it into a patient, and you can take a picture of exactly where cells are dying in the body. It was first used to image dying heart cells after a heart attack in 2000. The science works. The problem is clinical adoption: it's a big, slow-clearing molecule, every study has been small, and smaller peptide alternatives are starting to replace it. Still one of the cleanest examples of molecular imaging turning biology into a picture.
In 1998, Francis Blankenberg injected technetium-99m-labeled annexin V into mice with acute liver failure, rats rejecting transplanted hearts, and mice bearing lymphoma tumors. External radionuclide imaging showed a two- to sixfold increase in tracer uptake at sites of apoptosis across all three models.[1] It was the first demonstration that a protein targeting dying cells could produce images of cell death in a living organism. Two years later, the same approach was used to photograph dying heart muscle in human patients after myocardial infarction.[2] The tool that made both experiments possible was annexin V, a 35.8 kDa protein with extraordinary affinity for phosphatidylserine, the lipid that flips to the outside of cell membranes when cells begin to die.
Annexin V is not technically a peptide; at 320 amino acids, it is a small protein. But its mechanism, its clinical applications, and its position in the broader landscape of molecular imaging probes place it squarely at the intersection of peptide science and cardiovascular medicine. It pioneered the concept of imaging cell death in real time, and it remains the benchmark against which every newer apoptosis-targeting probe is measured. For how annexin V fits into the broader field of peptide-based cardiac imaging, see Peptide-Based Cardiac Imaging: Beyond Traditional Heart Scans.
Key Takeaways
- When cells start dying, they flip a molecular "I'm dying" flag to the outside of their membrane. Annexin V is a protein that grabs those flags.
- Attach a radioactive tag to annexin V, inject it, and a scanner can literally photograph where in your body cells are currently dying.
- The binding is astonishingly strong — tighter than most antibodies bind their targets, which is why it produces crisp, clean images.
- After a heart attack, 6 out of 7 patients showed annexin V lighting up the dying heart muscle on scans taken within hours of treatment.
- In heart transplant patients, every person whose heart glowed on the annexin V scan turned out to have serious rejection on biopsy.
- In mice, giving annexin V as a drug (not just an imaging tracer) cut heart attack damage by 27% and improved pumping function by 29%.
- Despite the science working, adoption has been slow. A smaller 19-amino-acid peptide competitor is starting to replace it in newer research.
What Is Annexin V?
Annexin V (also called annexin A5) was first isolated from human placenta in the early 1980s, when Bohn and colleagues identified it as placental protein 4 (PP4). Independently, Chris Reutelingsperger's group at Maastricht University purified a potent anticoagulant from human umbilical cord arteries in 1985 and named it vascular anticoagulant-alpha (VAC-alpha).[3] The two proteins turned out to be the same molecule. Over the following decade, it acquired at least seven names: placental protein 4, vascular anticoagulant-alpha, lipocortin V, endonexin II, calphobindin I, anchorin CII, and placental anticoagulant protein I. The annexin nomenclature was adopted in 1990 to unify this confusing taxonomy.
Structure and Binding
Annexin V is a 320-amino-acid protein (35.8 kDa) folded into a planar cyclic arrangement of four homologous repeat domains, each composed of five alpha-helical segments. The convex surface of the protein faces the cell membrane and contains calcium-binding sites in three of the four repeat domains. When calcium ions are present, the protein undergoes a conformational change that exposes a concave binding surface with high affinity for negatively charged phospholipids, particularly phosphatidylserine (PS).
The binding affinity is remarkable. Tait et al. measured the equilibrium dissociation constant at 0.036 nM when PS content exceeded 10% of membrane phospholipids, with calcium concentrations above 1 mM.[4] This sub-nanomolar Kd means annexin V binds PS tighter than most antibodies bind their targets. Equally notable, annexin V self-assembles into two-dimensional crystalline lattices on PS-rich membrane surfaces, creating a protein shield that stabilizes the membrane into a gel phase and blocks access of coagulation factors to the underlying phospholipids. This explains its potent anticoagulant activity, the property that led to its original discovery.
How Dying Cells Become Visible
In healthy cells, phosphatidylserine sits exclusively on the inner leaflet of the plasma membrane. This asymmetry is actively maintained by flippase enzymes (ATP-dependent aminophospholipid translocases) that continuously shuttle PS from the outer to the inner leaflet. During apoptosis, this machinery is disrupted through two simultaneous events: caspases inactivate the flippases, and calcium-dependent scramblases are activated, randomizing phospholipid distribution across the bilayer. The result is rapid externalization of PS onto the outer cell surface, where it becomes accessible to extracellular annexin V.
PS externalization occurs early in the apoptotic cascade, typically within hours of the initiating stimulus, and precedes DNA fragmentation and cell shrinkage. This timing is critical for imaging: annexin V can detect cells that are committed to death but have not yet disintegrated. Vermes et al. established the annexin V/propidium iodide dual-staining assay in 1995, demonstrating that fluorescein-labeled annexin V binding preceded membrane permeabilization (marked by propidium iodide uptake).[5] This assay became the most widely used flow cytometry method for detecting apoptosis and forms the conceptual foundation for all subsequent annexin V imaging work.
One important caveat: PS also becomes exposed during necrosis, when the plasma membrane loses integrity entirely. Annexin V alone cannot distinguish apoptosis from necrosis. In flow cytometry, propidium iodide exclusion solves this problem; in whole-body imaging, the distinction is more difficult and remains a source of debate in the literature.
How PS Exposure Works
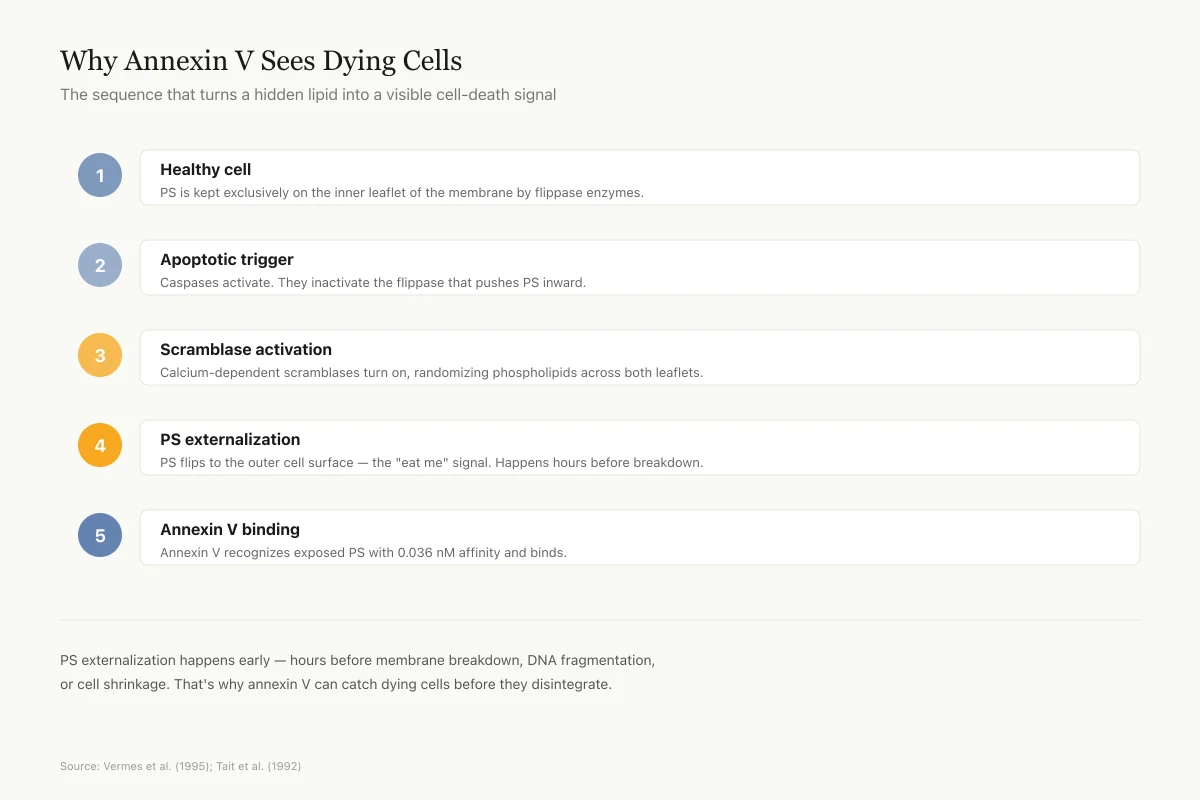
Why Annexin V Sees Dying Cells
The sequence that turns a hidden lipid into a visible cell-death signal
Healthy cell
Phosphatidylserine (PS) is kept exclusively on the inner leaflet of the membrane by flippase enzymes.
Apoptotic trigger
Caspases activate. They inactivate the flippase that pushes PS inward.
Scramblase activation
Calcium-dependent scramblases turn on, randomizing phospholipid distribution across both leaflets.
PS externalization
PS flips to the outer cell surface — the "eat me" signal. This happens hours before membrane breakdown.
Annexin V binding
Extracellular annexin V (natural or injected) recognizes exposed PS with 0.036 nM affinity and binds.
PS externalization happens early in cell death — hours before membrane breakdown, DNA fragmentation, or cell shrinkage. That's why annexin V can catch dying cells before they disintegrate.
Source: Vermes et al. (1995) annexin V/PI assay; Tait et al. (1992)
 View as image
View as imageLabeling Annexin V for Medical Imaging
The transition from laboratory assay to clinical imaging required attaching a detectable label to annexin V without disrupting its PS-binding function. The most successful approach, developed by Blankenberg and colleagues, uses hydrazinonicotinamide (HYNIC) as a bifunctional chelator to bind technetium-99m (99mTc) to annexin V. Technetium-99m is the workhorse radionuclide of nuclear medicine: it emits 140 keV gamma rays ideal for SPECT (single-photon emission computed tomography) cameras, has a 6-hour half-life suited to clinical imaging workflows, and is readily available from molybdenum-99 generators in nuclear medicine departments.
99mTc-HYNIC-annexin V preserves the protein's PS-binding affinity while adding a radioactive signal detectable through tissue. After intravenous injection, the labeled protein distributes through the bloodstream, binds to PS-expressing cells at sites of apoptosis, and clears from non-target tissues over several hours. SPECT imaging is typically performed at both early (2-4 hours) and late (16-24 hours) time points to distinguish specific binding from blood pool activity.
The pharmacokinetics of annexin V imaging are shaped by its protein size. At 35.8 kDa, annexin V is above the renal filtration threshold, which means clearance is slower than for small peptide probes. Kidney uptake is significant, which can interfere with imaging of abdominal structures. These pharmacokinetic limitations ultimately drove the search for smaller peptide alternatives.
Imaging Heart Attacks
The first human cardiac imaging study with annexin V was published in The Lancet in 2000 by Hofstra and colleagues at Maastricht University Medical Center.[2] Seven patients with acute myocardial infarction were treated with percutaneous coronary angioplasty to restore blood flow (achieving TIMI III flow in the infarct-related artery). Two hours after reperfusion, 1 mg of annexin V labeled with 584 MBq of technetium-99m was injected intravenously. SPECT images were acquired at a mean of 3.4 hours (early) and 20.5 hours (late) after injection.
Six of seven patients showed increased uptake of 99mTc-annexin V in the infarct area on both early and late SPECT images. No increased uptake was observed outside the infarct area. All patients with positive annexin V uptake also showed a matching perfusion defect on standard myocardial perfusion imaging. One control patient with no infarction showed no cardiac uptake.
The study demonstrated that cell death in the human heart after myocardial infarction could be visualized noninvasively within hours of the event.
The clinical significance was the potential to quantify the extent of myocardial injury in real time, rather than relying on delayed biomarkers like troponin (which measure leaked intracellular contents) or late gadolinium enhancement on MRI (which visualizes scar tissue days to weeks later). For how other peptide-based imaging agents are being developed to track the repair process after heart attacks, see Integrin Imaging After Heart Attack: Tracking Cardiac Repair.
Detecting Transplant Rejection
Cardiac allograft rejection is a leading cause of death after heart transplantation. Standard monitoring relies on endomyocardial biopsy, an invasive procedure that samples a small piece of heart muscle through a catheter threaded through the jugular vein. Biopsies are performed routinely in the first year after transplant and carry risks of perforation, arrhythmia, and sampling error (rejection can be patchy, and a single biopsy may miss an affected region).
Narula et al. published a landmark study in Nature Medicine in 2001 testing whether 99mTc-annexin V could noninvasively detect transplant rejection.[6] Eighteen heart transplant recipients underwent both annexin V imaging and endomyocardial biopsy within 24 hours. Thirteen patients showed negative myocardial uptake of annexin V, and none of these patients had significant rejection on biopsy. Five patients showed positive myocardial uptake. All five had at least moderate transplant rejection confirmed histologically, and all five showed positive caspase-3 staining in their biopsy specimens, confirming active apoptosis.
The correlation was striking: annexin V imaging had 100% sensitivity and 100% specificity for moderate-to-severe rejection in this small cohort. The clinical implication was clear. If validated in larger studies, annexin V imaging could replace some of the 15-20 endomyocardial biopsies that transplant patients typically undergo in their first year. The study also demonstrated that apoptosis, rather than just necrosis, is a significant mechanism of myocyte loss during rejection, a finding with implications for anti-rejection therapy design.
Unstable Atherosclerotic Plaques
Most heart attacks are caused not by gradual narrowing of arteries but by sudden rupture of atherosclerotic plaques that may not have been causing significant stenosis. Identifying which plaques are vulnerable to rupture before they cause events has been called the "holy grail" of cardiovascular imaging. Plaque instability is driven partly by apoptosis of macrophages and smooth muscle cells within the plaque, which thins the fibrous cap and makes rupture more likely.
Kietselaer et al. published a brief but influential report in the New England Journal of Medicine in 2004 demonstrating that 99mTc-annexin A5 could detect apoptosis in human carotid atherosclerotic plaques in vivo.[7] Four patients scheduled for carotid endarterectomy underwent annexin V imaging before surgery. Plaques with positive annexin V uptake on SPECT imaging showed histological evidence of macrophage infiltration and apoptosis in the resected specimens. The study was small, but it established proof of concept for noninvasive detection of plaque vulnerability using molecular imaging of cell death.
This application connects to a broader theme in cardiovascular medicine: the shift from anatomical imaging (how narrow is the artery?) to molecular imaging (what biological processes are occurring within the plaque?). Annexin V was among the first molecular probes to demonstrate this concept in human patients.
Heart Failure and Ongoing Cell Death
Unlike the acute cell death of a heart attack, heart failure involves chronic, ongoing loss of cardiomyocytes that gradually weakens the heart. Whether annexin V could detect this slower, more diffuse process was tested by Kown et al. in a study published in the Journal of Nuclear Medicine in 2007.[8]
Nine patients with advanced nonischemic cardiomyopathy (eight dilated, one hypertrophic) underwent 99mTc-annexin A5 imaging. Five of nine patients showed focal, multifocal, or global left ventricular uptake. Annexin A5 uptake correlated with subsequent deterioration in left ventricular function, suggesting it identified patients with active, ongoing cell death. Two control relatives of the hypertrophic cardiomyopathy patient (same genetic background, no heart failure) showed no cardiac uptake.
The study was proof of concept rather than a practice-changing trial. It demonstrated that annexin V imaging could detect the chronic apoptotic cell loss that drives heart failure progression.
Clinical Evidence Every Human Cardiac Annexin V Study Proof of concept across four disease contexts — but all small studies Population: 7 acute MI patients 6 of 7 showed cardiac uptake in infarct area Population: 18 heart transplant recipients All 5 with positive uptake had confirmed moderate+ rejection Population: 4 carotid endarterectomy patients Plaque uptake correlated with histological apoptosis Population: 9 nonischemic cardiomyopathy patients 5 of 9 showed cardiac uptake; predicted deterioration Every study was small (4–18 patients) and proof-of-concept. None were large enough for regulatory approval. This is why annexin V, despite 20+ years of positive findings, never became a routine clinical tool. Source: Hofstra (2000); Narula (2001); Kietselaer (2004); Kown (2007)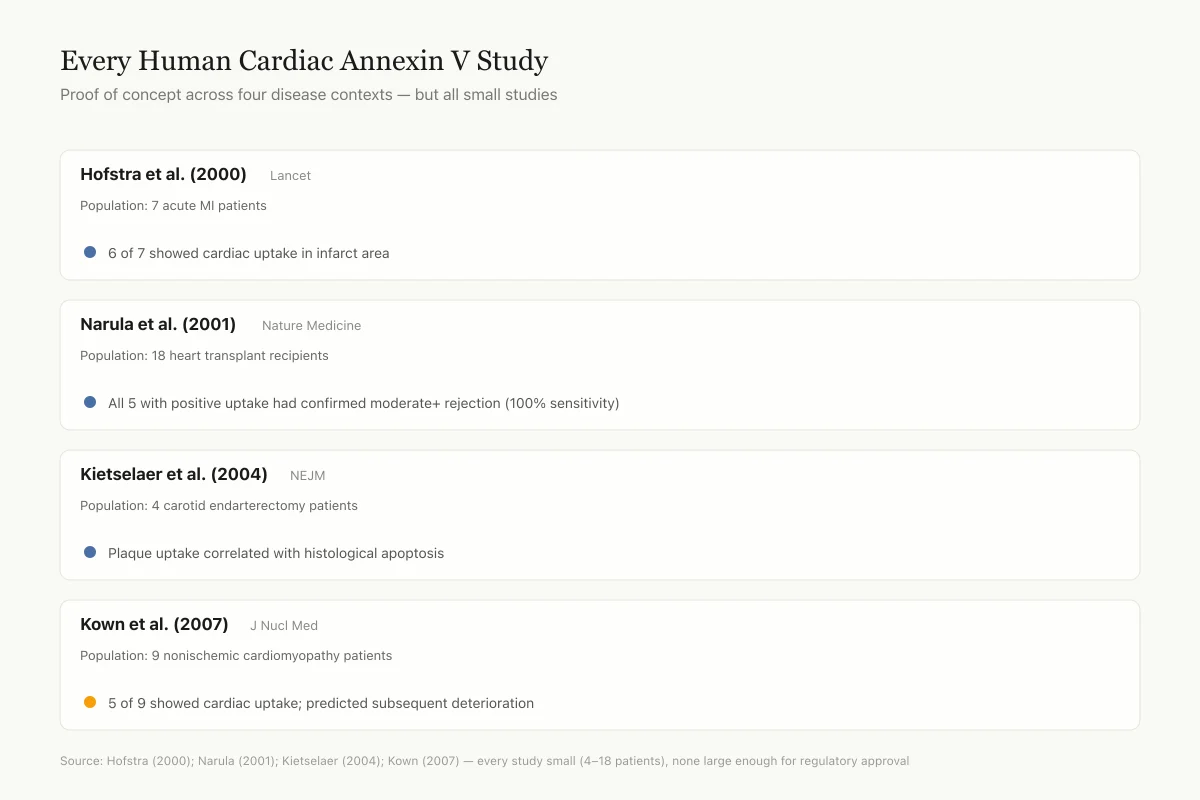 View as image
View as image
The heart also has its own local peptide defense systems. Cardiomyocytes synthesize and secrete ghrelin, a growth hormone-releasing peptide with direct cardioprotective and anti-apoptotic effects, suggesting the heart has intrinsic peptide-based mechanisms for resisting cell death.[9] Understanding these endogenous protective pathways may eventually inform therapeutic strategies that complement diagnostic imaging.
Beyond Imaging: Annexin A5 as a Therapeutic
An unexpected finding emerged from the imaging research: annexin A5 itself has therapeutic potential. Beyond its PS-binding and imaging capabilities, annexin A5 suppresses inflammation, a process that drives tissue damage after ischemia-reperfusion injury.
De Jong et al. demonstrated this in a 2018 study using hypercholesterolemic mice subjected to myocardial ischemia-reperfusion injury.[10] Animals treated with annexin A5 showed a 27% reduction in infarct size compared to vehicle-treated controls. End-diastolic and end-systolic volumes decreased by 22% and 38% respectively. Left ventricular ejection fraction improved by 29%. Fibrous content in the left ventricle was reduced by 42%, accompanied by a 17% increase in wall thickness of the infarcted area.
The mechanism appears to involve annexin A5's ability to shield externalized PS from recognition by inflammatory cells. When macrophages encounter PS on damaged cells, they mount an inflammatory response. Annexin A5 binding masks the PS signal, dampening inflammation and reducing secondary tissue damage. In a separate study, Schutters et al. showed that annexin A5 treatment attenuated vascular inflammation and remodeling in mice, improving endothelial function, with implications for atherosclerotic cardiovascular disease.[11]
These findings recast annexin A5 from a passive diagnostic tool to a potential therapeutic agent. No human therapeutic trials have been completed, but the dual diagnostic-therapeutic profile (theranostic) is attractive for cardiovascular applications where imaging could guide and monitor treatment simultaneously.
Why Annexin V Has Not Reached Routine Clinical Use
Despite two decades of promising clinical studies, 99mTc-annexin V has not been approved as a clinical imaging agent. Several factors explain this gap between research success and clinical adoption.
First, annexin V cannot reliably distinguish apoptosis from necrosis. Both processes expose PS, and in clinical settings like myocardial infarction, both occur simultaneously. This limits the biological specificity of the signal. A positive annexin V scan confirms cell death but cannot determine the mechanism, which matters for therapeutic decision-making.
Second, the pharmacokinetics are suboptimal. At 35.8 kDa, annexin V clears slowly from blood, resulting in high background signal at early time points and requiring delayed imaging. Kidney uptake is substantial, producing high radiation doses to the kidneys and bladder and interfering with imaging of structures near the abdomen. The 6-hour half-life of technetium-99m is well matched to clinical workflows, but the protein's slow tissue clearance means optimal images may require 16-24 hours, stretching the practical imaging window.
Third, the regulatory and commercial pathway has been difficult. Annexin V imaging addresses a genuine unmet need (noninvasive detection of cell death), but the clinical scenarios where it would change management have not been clearly defined in large, randomized trials. For myocardial infarction, troponin testing and cardiac MRI already provide clinically actionable information. For transplant rejection, reducing biopsy frequency requires proving that negative annexin V scans reliably exclude rejection, which demands large multicenter studies that have not been completed.
Fourth, competition from other modalities has intensified. Cardiac MRI with late gadolinium enhancement can assess infarct size without radiation. PET imaging with fluorine-18-labeled probes offers higher spatial resolution than SPECT. And newer peptide probes with better pharmacokinetics are entering preclinical and early clinical testing.
Next-Generation Alternatives
The limitations of annexin V have driven development of smaller, faster-clearing molecular probes that retain apoptosis-targeting specificity. Two approaches are most advanced.
Duramycin
Duramycin is a 19-amino-acid lantibiotic peptide (molecular weight approximately 2 kDa) produced by Streptoverticillium cinnamoneus. Unlike annexin V, which targets phosphatidylserine, duramycin binds phosphatidylethanolamine (PE) with high affinity (Kd approximately 5 nM) and specificity. PE is also externalized during apoptosis and necrosis, making it a valid molecular target for cell death imaging.[12]
99mTc-duramycin offers several pharmacokinetic advantages over 99mTc-annexin V. At 2 kDa versus 35.8 kDa, duramycin clears from blood and non-target tissues far more rapidly, allowing earlier imaging time points and lower radiation doses to non-target organs. In a porcine model of myocardial ischemia-reperfusion injury, 99mTc-duramycin showed avid target uptake with fast background clearance appropriate for cardiac imaging.[13] Direct comparison studies in atherosclerotic plaque imaging showed that 99mTc-duramycin detected apoptosis comparably to 99mTc-annexin V but with reduced non-target organ radiation burden.[14]
Probe Comparison
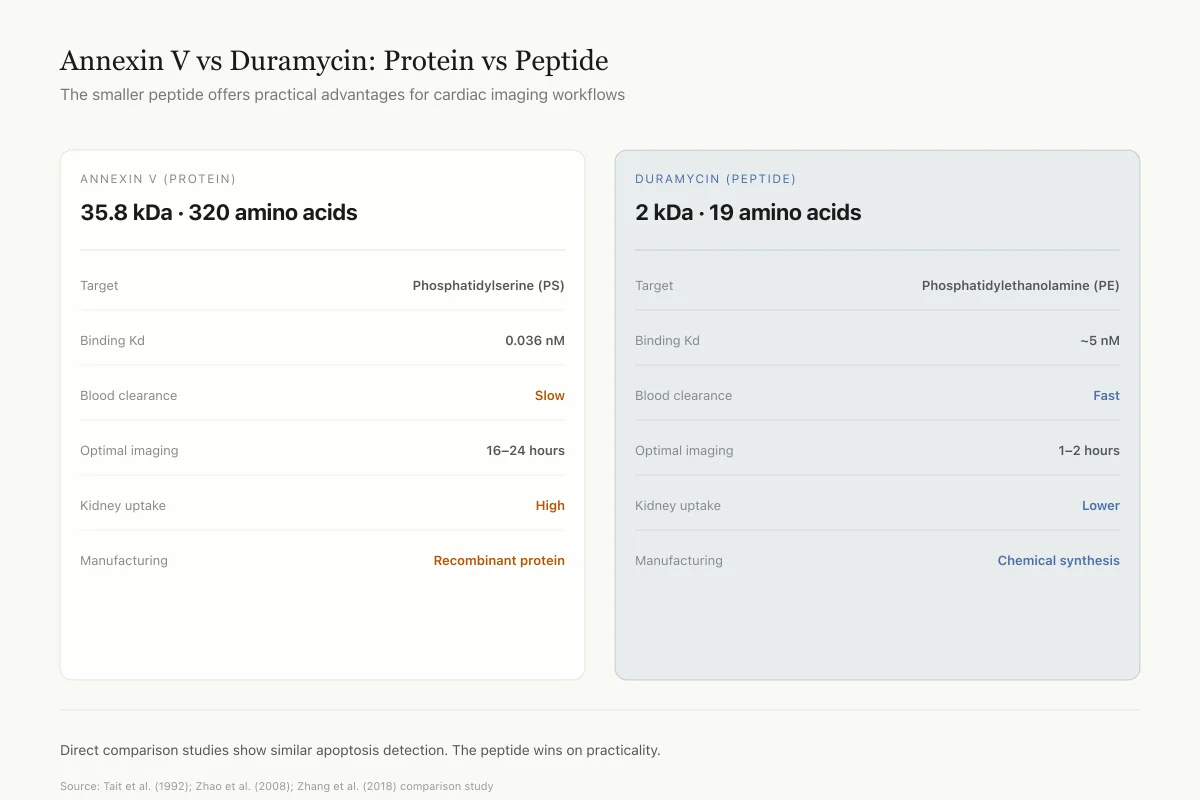
Annexin V vs Duramycin: Protein vs Peptide
The smaller peptide offers practical advantages for cardiac imaging workflows
ANNEXIN V (PROTEIN)
35.8 kDa · 320 amino acids
DURAMYCIN (PEPTIDE)
2 kDa · 19 amino acids
Direct comparison studies in atherosclerotic plaque imaging show similar apoptosis detection between the two probes. The peptide wins on practicality: faster clearance means same-day imaging, lower kidney burden, and much easier chemical synthesis.
Source: Tait et al. (1992); Zhao et al. (2008); Zhang et al. (2018) comparison study
 View as image
View as imagePhage-Display Peptides
Several peptide sequences identified through bacteriophage display technology specifically bind phosphatidylserine with affinities approaching that of annexin V. PSP1 (phosphatidylserine-specific peptide 1) is a 14-amino-acid peptide that showed comparable tumor apoptosis detection to annexin V in mouse models, with the pharmacokinetic advantages of a small peptide: rapid clearance, low immunogenicity, and easier synthesis and labeling.[15] These peptide probes represent the most direct application of peptide science to the problem that annexin V first defined.
The transition from protein-based to peptide-based apoptosis probes follows a pattern seen across molecular imaging: large protein probes establish proof of concept, then smaller peptide analogs optimize pharmacokinetics for clinical translation. The same trajectory is playing out in Integrin Imaging After Heart Attack: Tracking Cardiac Repair, where RGD peptides are replacing larger integrin-binding proteins for tracking angiogenesis and repair. For the full landscape of peptide radiopharmaceuticals being developed for the heart, see Peptide-Based Cardiac Imaging: Beyond Traditional Heart Scans.
Where Annexin V Research Stands
Annexin V established a field. Before Blankenberg's 1998 experiment, imaging cell death in a living organism was theoretical. After it, the question shifted from whether it was possible to how to make it clinically practical. The subsequent human studies in myocardial infarction, transplant rejection, atherosclerosis, and heart failure validated the biological principle: externalized phosphatidylserine is an accessible, abundant, and early marker of cell death that can be detected noninvasively.
The protein has not become a routine clinical tool, and it may never be. The pharmacokinetic limitations of a 36 kDa protein, the inability to distinguish apoptosis from necrosis, and the absence of large randomized trials have stalled the regulatory path. But the concept annexin V proved, that molecular imaging of cell death can reveal cardiovascular disease activity invisible to anatomical imaging, continues to drive the field forward.
The most active research directions now involve smaller peptide probes (duramycin, PSP1, and other phage-display peptides) that retain apoptosis-targeting specificity while offering the faster clearance, lower radiation burden, and easier manufacturing that peptide platforms provide. Simultaneously, the discovery that annexin A5 itself is cardioprotective has opened a separate therapeutic avenue, one where the imaging agent becomes the drug.
The natriuretic peptide system represents another intersection of cardiac biology and peptide science. Just as annexin V revealed that cell death could be imaged, the discovery of ANP and the natriuretic peptide family revealed that the heart communicates with the kidneys through peptide hormones. Both stories illustrate how peptide and protein biology has reshaped cardiovascular medicine over the past four decades.
Boersma et al. captured the trajectory in their 2005 review: annexin A5 moved from protein discovery to diagnostic tool to potential therapeutic in 20 years, a timeline that reflects both the promise and the complexity of translating molecular biology into clinical practice.[3]
The Bottom Line
Annexin V is a 35.8 kDa protein that binds phosphatidylserine on dying cells with 0.036 nM affinity. Labeled with technetium-99m, it pioneered in vivo imaging of apoptosis in 1998 and was subsequently tested in human studies of myocardial infarction (6/7 patients positive), transplant rejection (5/5 with rejection detected), atherosclerotic plaque instability, and heart failure. Clinical adoption has been limited by slow clearance, inability to distinguish apoptosis from necrosis, and lack of large randomized trials. Smaller peptide probes like duramycin (19 amino acids, 2 kDa) are now being developed to overcome these limitations while preserving the apoptosis-targeting principle annexin V established. Separately, therapeutic annexin A5 reduced infarct size by 27% in a mouse model, opening a potential dual diagnostic-therapeutic pathway.
Sources & References
- 9RPEP-00928·Iglesias, María J et al. (2004). “Heart Cells Make Their Own Ghrelin: A Local Cardiac Ghrelin System.” Cardiovascular research.Study breakdown →PubMed →↩