Adrenomedullin: The Vasodilator Peptide
Cardiovascular Peptides
52 amino acids
Discovered in 1993 from a rare adrenal tumor, adrenomedullin turned out to be one of the body's most important vascular regulators, produced by nearly every tissue.
Kitamura et al., Biochem Biophys Res Commun, 1993
Kitamura et al., Biochem Biophys Res Commun, 1993
If you only read one thing
Adrenomedullin is a natural peptide your body makes to relax blood vessels and protect organs. Almost every tissue produces it. Doctors now measure a piece of it in your blood (called MR-proADM) to predict how sick someone with sepsis really is — it's better at predicting outcomes than standard tests. It's not something you can take as a supplement. Researchers are working on an antibody drug that redirects your own adrenomedullin to damaged tissues, and it's in Phase 2 clinical trials for septic shock.
In 1993, a team of Japanese researchers was screening extracts from a rare adrenal gland tumor called pheochromocytoma. They found a 52-amino-acid peptide that caused a potent and long-lasting drop in blood pressure when injected into rats.[1] Because the peptide was abundant in the adrenal medulla, they named it adrenomedullin. What seemed at first like a curiosity from tumor tissue turned out to be one of the body's most widely produced and versatile regulatory peptides. Today, adrenomedullin research spans cardiovascular disease, sepsis, cancer, inflammatory bowel disease, and pulmonary hypertension. This article covers the biology, the clinical evidence, and the emerging therapeutic applications of this peptide. For related cardiovascular peptides, see our articles on endothelin, angiotensin II, bradykinin, apelin, and urotensin II.
Key Takeaways
- Adrenomedullin is one of your body's most powerful natural blood pressure regulators — it can drop blood pressure within seconds of release.
- Almost every tissue in your body makes it. It was discovered by accident in 1993 from an adrenal tumor, but it's in your lungs, kidneys, and heart too.
- Doctors now use a stable piece of it (MR-proADM) to predict who will survive sepsis. It beats the standard tests used today.
- In sepsis, adrenomedullin also holds your blood vessels together. Lose that signal and your vessels start leaking — which is part of how sepsis kills.
- You can't take it as a supplement. It's destroyed within 22 minutes in the bloodstream, so anything you swallow or inject would be gone almost immediately.
- A new antibody drug redirects your own adrenomedullin from blood to damaged tissue. It's in Phase 2 trials for septic shock right now.
- This is the same peptide family as migraine drugs — CGRP peptides. Understanding adrenomedullin helps explain why those drugs work.
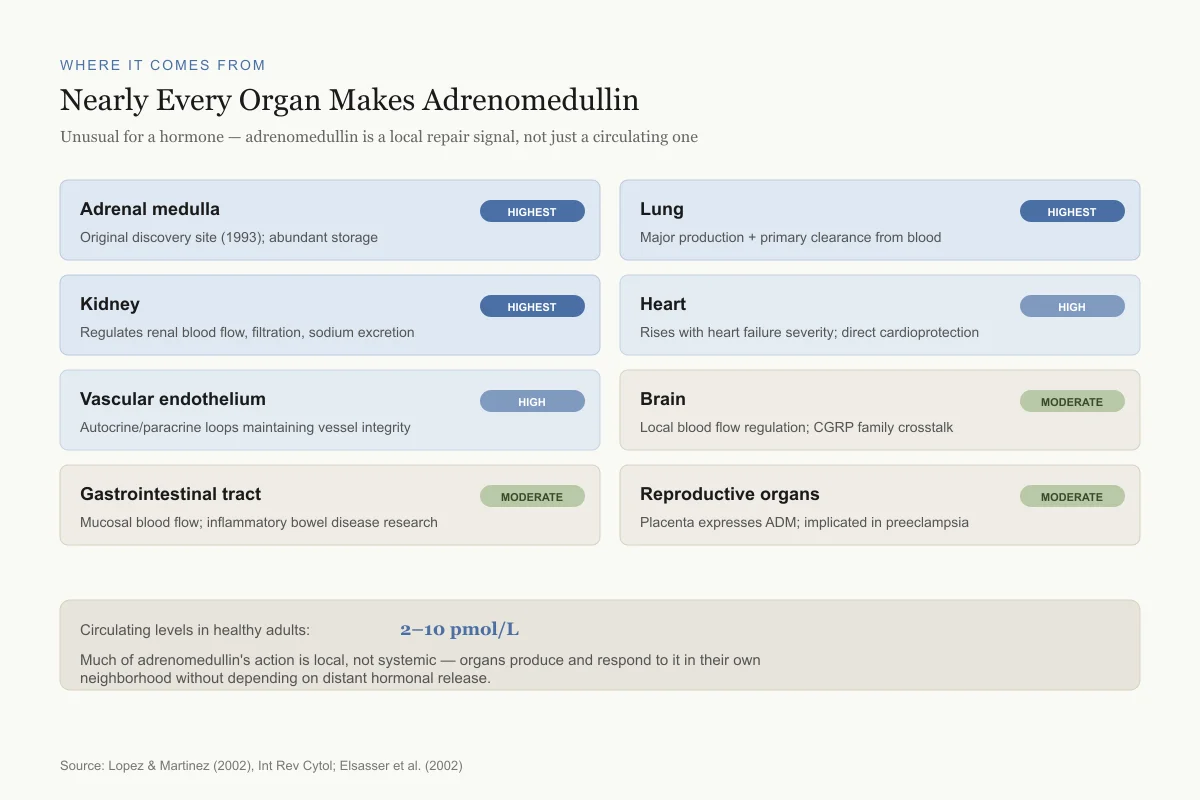
Where It Comes From
Nearly Every Organ Makes Adrenomedullin
Unusually distributed for a hormone — this is what makes it a local repair signal, not just a circulating one
Adrenal medulla
HighestOriginal discovery site (1993); abundant storage
Lung
HighestMajor production + primary clearance site from circulation
Kidney
HighestRegulates renal blood flow, filtration, and sodium excretion
Heart
HighRises with heart failure severity; direct cardioprotection
Vascular endothelium
HighAutocrine/paracrine loops maintaining vessel integrity
Brain
ModerateLocal blood flow regulation; CGRP family crosstalk
Gastrointestinal tract
ModerateMucosal blood flow; inflammatory bowel disease research
Reproductive organs
ModeratePlacenta expresses adrenomedullin; implicated in preeclampsia
Source: Lopez & Martinez (2002), Int Rev Cytol; Elsasser et al. (2002)
 View as image
View as imageDiscovery and Structure
Kitamura and colleagues isolated adrenomedullin from human pheochromocytoma tissue in 1993 by screening for peptides that elevated cyclic AMP (cAMP) in rat platelets.[1] The peptide has 52 amino acids and contains a single internal disulfide bond that forms a six-residue ring structure. This ring is essential for biological activity; breaking it eliminates vasodilatory function.
Adrenomedullin belongs to the calcitonin gene-related peptide (CGRP) superfamily, which also includes CGRP itself, amylin, calcitonin, and intermedin (adrenomedullin 2). These peptides share structural features, particularly the intramolecular ring, and signal through overlapping receptor systems.[2]
Elsasser et al. (2002) documented that adrenomedullin plays protective roles during multiple forms of physiological stress, including hypoxia (low oxygen), inflammation, and hormonal imbalance. The peptide is upregulated in cardiovascular tissues during early hypertension, suggesting the body produces more of it as a defense against rising blood pressure.[3]
The adrenomedullin gene also encodes a second bioactive peptide from the same precursor protein: proadrenomedullin N-terminal 20 peptide (PAMP). PAMP has distinct biological activities, including its own hypotensive effects and antimicrobial properties, though it acts through different receptors than adrenomedullin itself.[3]
Where the Body Makes Adrenomedullin
One of the most surprising findings about adrenomedullin is how widely it is produced. Lopez and Martinez (2002) published a comprehensive review documenting adrenomedullin expression across virtually every organ system: the adrenal medulla, heart, lungs, kidneys, brain, gastrointestinal tract, reproductive organs, and vascular endothelium.[4]
The richest sources are the adrenal medulla (where it was discovered), the lung, and the kidney. The lung is a major site of adrenomedullin clearance from the bloodstream, with specific binding sites on pulmonary endothelial cells removing circulating adrenomedullin and thereby regulating its systemic levels.[4]
Adrenomedullin circulates in the blood at measurable concentrations (2-10 picomoles per liter in healthy adults), but much of its action is local. Endothelial cells and vascular smooth muscle cells both produce and respond to adrenomedullin, creating autocrine and paracrine signaling loops that regulate vascular tone without depending on distant hormonal release.[5]
How Adrenomedullin Signals
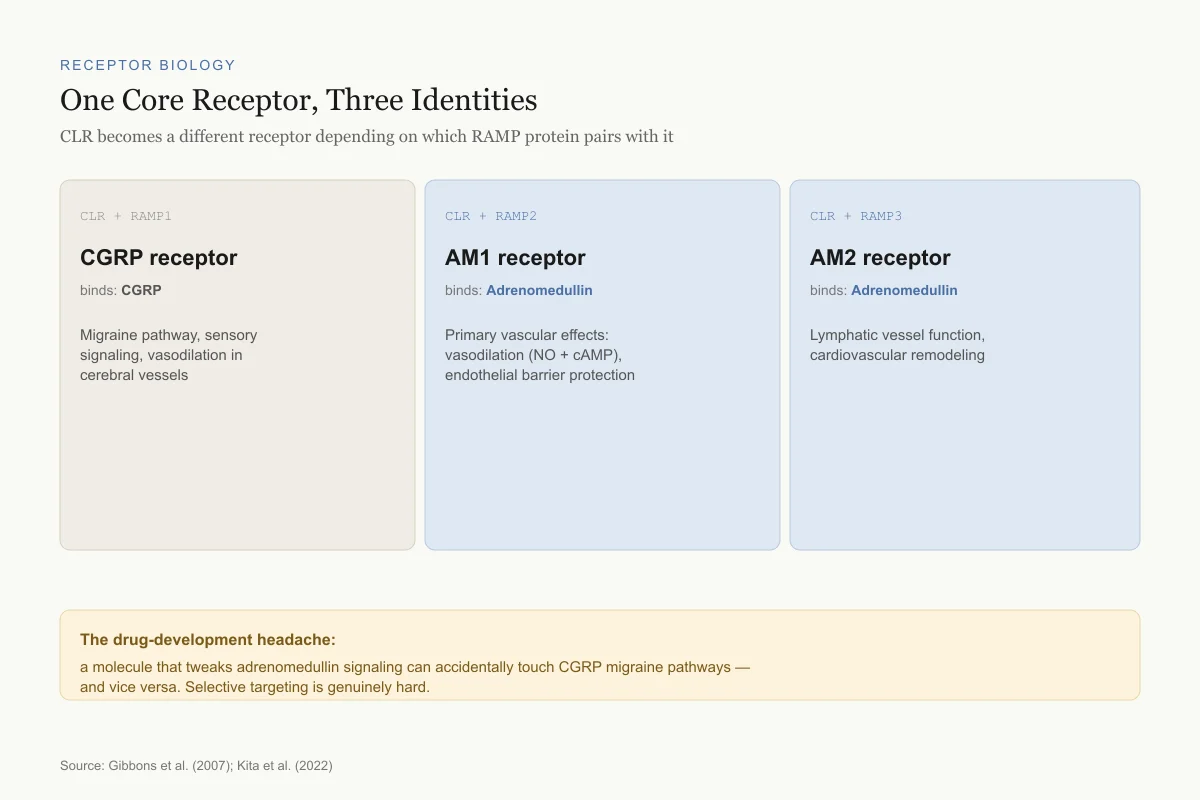
Adrenomedullin acts through a receptor system that is unusual in peptide biology. The core receptor is the calcitonin receptor-like receptor (CLR, also called CRLR), a G protein-coupled receptor that cannot function on its own. It requires a partner protein from the receptor activity-modifying protein (RAMP) family to reach the cell surface and determine ligand specificity.[6]
When CLR pairs with RAMP1, the complex becomes a CGRP receptor. When CLR pairs with RAMP2 or RAMP3, it becomes an adrenomedullin receptor (AM1 or AM2, respectively). This means the same core receptor protein creates different functional receptors depending on which RAMP is available. Gibbons et al. (2007) described how RAMPs act as "molecular switches" that determine the pharmacological identity of CLR.[6]
The downstream effects depend on which receptor complex is activated. AM1 (CLR/RAMP2) mediates most of adrenomedullin's vascular effects: vasodilation through nitric oxide and cAMP production, endothelial barrier protection, and anti-inflammatory signaling. AM2 (CLR/RAMP3) contributes to lymphatic vessel function and may play a role in cardiovascular remodeling.
This receptor system creates a challenge for drug development. Targeting adrenomedullin signaling means working with a receptor that changes identity based on which accessory protein is present, making selective modulation difficult.
Receptor Biology
One Core Receptor, Three Identities
CLR becomes a different receptor depending on which RAMP protein sits with it
CGRP receptor
binds: CGRP
Migraine pathway, sensory signaling, vasodilation in cerebral vessels
AM1 receptor
binds: Adrenomedullin
Primary vascular effects: vasodilation (NO + cAMP), endothelial barrier protection
AM2 receptor
binds: Adrenomedullin
Lymphatic vessel function, cardiovascular remodeling
The drug-development headache: a molecule that tweaks adrenomedullin signaling can accidentally touch CGRP migraine pathways, and vice versa. Selective targeting is genuinely hard.
Source: Gibbons et al. (2007); Kita et al. (2022)
 View as image
View as imageCardiovascular Effects
Blood pressure regulation
Adrenomedullin is among the most potent endogenous vasodilators identified. When infused intravenously in humans, it produces dose-dependent reductions in blood pressure while increasing cardiac output and heart rate.[5] The vasodilation occurs through two mechanisms: an endothelium-dependent pathway via nitric oxide (NO) release, and a direct endothelium-independent pathway through cAMP elevation in vascular smooth muscle cells.
Cheung and Bhatt (2004) reviewed adrenomedullin's role in the cardiovascular system, noting that plasma adrenomedullin levels rise in proportion to disease severity in heart failure, hypertension, and sepsis.[5] Whether these elevated levels represent a protective compensatory response or simply a marker of disease severity remained a key question in the field.
Heart protection
Tsuruda et al. (2019) reviewed the evidence for adrenomedullin as a cardioprotective agent. In animal models of myocardial infarction, adrenomedullin reduced infarct size, decreased cardiomyocyte apoptosis, and improved cardiac function.[7] The mechanisms include direct anti-apoptotic effects on heart muscle cells, reduction of oxidative stress, and suppression of inflammatory cytokines in the infarcted tissue.
Small-scale clinical trials have tested intravenous adrenomedullin infusion in patients with heart failure. The peptide improved hemodynamics (reduced vascular resistance, increased cardiac output) without significant adverse effects in these short-term studies.[7] However, adrenomedullin's half-life in human blood is less than 22 minutes, making continuous infusion impractical for chronic therapy.
The half-life problem
The short half-life is the central obstacle to adrenomedullin-based therapeutics. Kato et al. (2015) published a "bench-to-bedside" pharmacology review that summarized efforts to overcome this limitation.[8] Approaches include:
- Modified adrenomedullin derivatives with polyethylene glycol (PEG) conjugation to extend half-life
- Gene delivery strategies that cause tissues to produce adrenomedullin continuously
- Adrenomedullin analogs with amino acid substitutions that resist enzymatic degradation
None of these has reached large-scale clinical use. The most clinically advanced approach is not administering adrenomedullin directly, but rather targeting it with an antibody (see the adrecizumab section below).
Endothelial Barrier Protection
One of adrenomedullin's most clinically relevant functions is maintaining the endothelial barrier, the layer of cells lining blood vessels that controls what passes between the bloodstream and surrounding tissues. Temmesfeld-Wollbruck et al. (2007) demonstrated that adrenomedullin strengthens endothelial cell-to-cell junctions, reduces vascular permeability, and stabilizes the actin cytoskeleton that gives endothelial cells their structural integrity.[9]
When the endothelial barrier breaks down, fluid leaks from blood vessels into tissues, causing edema and organ dysfunction. This vascular leak is a hallmark of sepsis, acute respiratory distress syndrome (ARDS), and other critical illnesses. The finding that adrenomedullin actively maintains barrier integrity suggested a direct link between adrenomedullin biology and the organ failure seen in severe infections.
This is where adrenomedullin research shifted from basic science curiosity to potential clinical tool. If adrenomedullin protects the vascular barrier, and if sepsis involves barrier breakdown, then measuring or manipulating adrenomedullin levels could have practical value in critical care medicine.
Adrenomedullin as a Sepsis Biomarker
MR-proADM: measuring the unmeasurable
Adrenomedullin itself is difficult to measure in clinical settings because of its short half-life and binding to plasma proteins. The solution came from measuring mid-regional pro-adrenomedullin (MR-proADM), a stable fragment of the adrenomedullin precursor protein that is released in equimolar amounts. MR-proADM has a half-life of several hours and correlates directly with adrenomedullin production.[10]
Valenzuela-Sanchez et al. (2016) reviewed MR-proADM as a biomarker of organ failure, concluding that it outperformed traditional markers like procalcitonin (PCT) and C-reactive protein (CRP) for predicting adverse outcomes in sepsis.[10]
Predicting mortality and organ failure
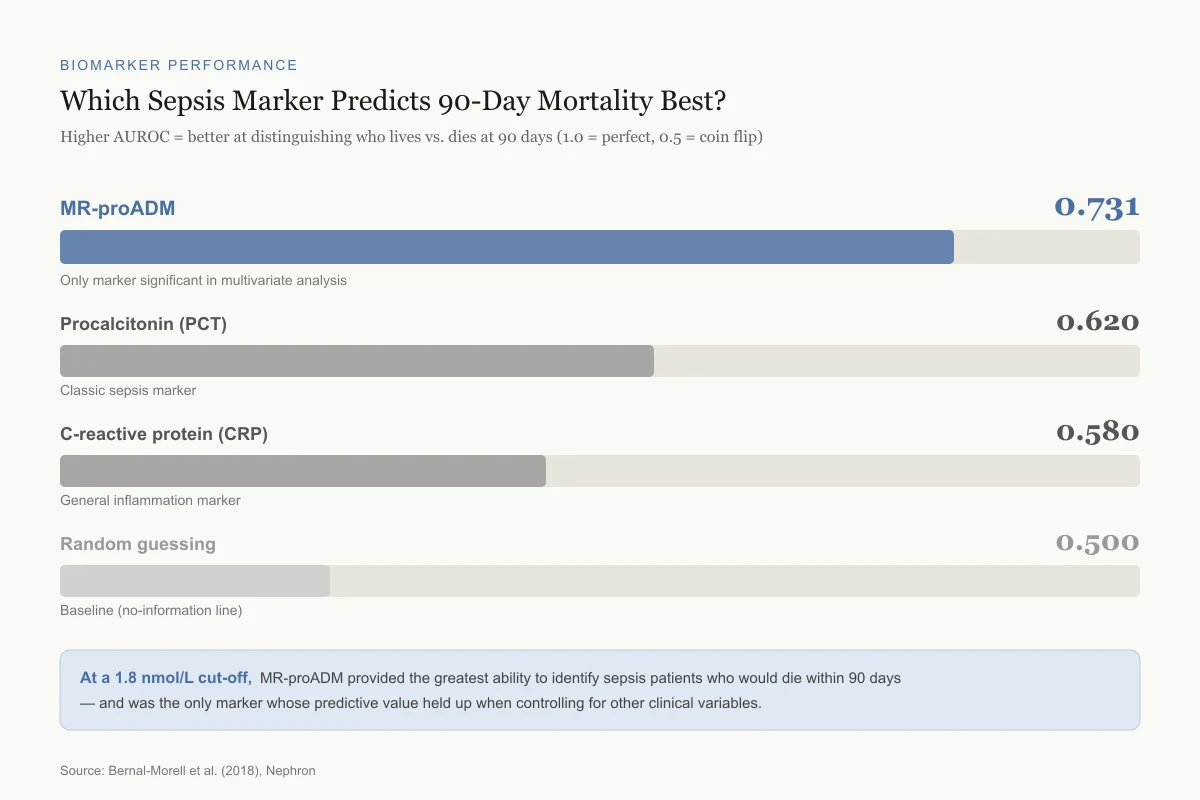
Bernal-Morell et al. (2018) measured MR-proADM in sepsis patients and found it was the best single biomarker for predicting 90-day mortality, with an area under the receiver operating characteristic curve (AUROC) of 0.731 (95% CI 0.612-0.850, p<0.001). It was the only marker that maintained independent predictive value in multivariate analysis. A cut-off of 1.8 nmol/L had the greatest discriminative capacity for 90-day mortality prediction.[11]
Biomarker Performance
Which Sepsis Marker Predicts 90-Day Mortality Best?
Higher AUROC = better at distinguishing who lives vs. dies at 90 days (1.0 = perfect, 0.5 = coin flip)
MR-proADM
0.731
Only marker significant in multivariate analysis
Procalcitonin (PCT)
0.620
Classic sepsis marker
C-reactive protein (CRP)
0.580
General inflammation marker
Random guessing
0.500
Baseline (no-information line)
At a 1.8 nmol/L cut-off, MR-proADM provided the greatest ability to identify sepsis patients who would die within 90 days — and was the only marker whose predictive value held up when controlling for other clinical variables.
Source: Bernal-Morell et al. (2018), Nephron
 View as image
View as imageOnal et al. (2018) published a narrative review synthesizing the MR-proADM sepsis literature. They found consistent evidence across multiple studies that MR-proADM levels correlate with sepsis severity, organ dysfunction scores, and mortality risk. The review noted that MR-proADM performs especially well for identifying patients who appear stable but are deteriorating, a clinical scenario where traditional markers like CRP and white blood cell count often lag behind actual disease progression.[12] The biological logic is straightforward: when organs are failing and the vascular barrier is leaking, the body produces more adrenomedullin in an attempt to stabilize the endothelium. Higher MR-proADM levels reflect greater vascular distress.
A limitation of the biomarker data: MR-proADM is not specific to sepsis. Elevated levels also occur in heart failure, chronic kidney disease, and other conditions involving endothelial stress. It works best as a prognostic marker (how sick is this patient?) rather than a diagnostic one (does this patient have sepsis?).
Adrenomedullin in Disease
Heart failure
Plasma adrenomedullin levels are elevated in heart failure patients in proportion to disease severity. Hirata et al. (1996) were among the first to document this, finding elevated circulating adrenomedullin in patients with hypertension and heart failure.[13] The elevation appears to be a compensatory response: the failing heart and stressed vasculature produce more adrenomedullin to maintain vasodilation and cardiac output. This makes adrenomedullin both a marker of heart failure severity and a potential therapeutic target.
Pulmonary hypertension
Pulmonary hypertension is a condition where blood pressure in the lung arteries is abnormally high, straining the right side of the heart. Because adrenomedullin dilates blood vessels and is produced abundantly in lung tissue, it has been studied as both a biomarker and potential therapy. Intravenous adrenomedullin infusion in patients with pulmonary hypertension reduced pulmonary vascular resistance and improved cardiac output in short-term clinical studies.[8] Inhaled adrenomedullin has also been tested as a way to deliver the peptide directly to the lungs while avoiding systemic hypotension. Early-phase results showed selective pulmonary vasodilation without dangerous drops in systemic blood pressure.
The lung connection runs deeper than therapy. The lung is the primary site for adrenomedullin clearance from the bloodstream; specific binding sites on pulmonary endothelial cells remove circulating adrenomedullin.[4] This clearance mechanism means the lung is simultaneously a major target organ and a major regulator of adrenomedullin levels. In pulmonary disease, disruption of this clearance may contribute to altered adrenomedullin signaling throughout the body.
Kidney protection
The kidney is another organ where adrenomedullin has demonstrated protective effects. Adrenomedullin dilates renal blood vessels, increases glomerular filtration rate, and promotes sodium excretion (natriuresis).[3] In animal models of acute kidney injury, adrenomedullin administration reduced tubular damage, decreased inflammation, and improved renal function recovery. Elsasser et al. (2002) noted that adrenomedullin's multiple roles in kidney biology include regulation of renin secretion, mesangial cell function, and renal blood flow distribution.[3]
This kidney protection has particular relevance to sepsis, where acute kidney injury is one of the most common and dangerous organ failures. The finding that adrenomedullin protects both the vascular endothelium and the kidney directly helps explain why MR-proADM levels predict organ failure outcomes so effectively.
Cancer
Zudaire et al. (2003) reviewed the relationship between adrenomedullin and cancer. Multiple tumor types overexpress adrenomedullin, and the peptide promotes angiogenesis (new blood vessel growth) and tumor cell survival.[14] This is the dark side of adrenomedullin's biological profile: the same pro-survival, pro-angiogenic properties that protect organs in acute illness may help tumors grow and resist treatment. Blocking adrenomedullin signaling in tumors is an active area of preclinical research, though no anti-adrenomedullin cancer therapy has reached clinical trials.
Immunity and tissue protection
A 2025 study by Uddin and colleagues in Nature Immunology identified adrenomedullin 2 (also called intermedin), a closely related peptide in the same family, as a key mediator of tissue-protective immunity. Working through CGRP-family receptors, adrenomedullin 2 promoted tissue repair and limited inflammatory damage during immune responses.[15] This suggests the adrenomedullin peptide family may have broader roles in balancing immune defense with tissue preservation than previously recognized.
Therapeutic Approaches Under Development
Adrecizumab: the antibody strategy
The most advanced clinical approach does not administer adrenomedullin directly. Instead, it uses adrecizumab, a humanized monoclonal antibody that binds the N-terminal portion of adrenomedullin. The antibody does not neutralize adrenomedullin. Rather, it redistributes it from the bloodstream to the tissues where the endothelial barrier needs repair.
Kita et al. (2022) reviewed the translational pipeline for adrenomedullin-based therapies. In animal models of septic shock, adrecizumab improved hemodynamics, renal function, and systemic inflammation while reducing inducible nitric oxide synthase expression. A phase 2 clinical trial (NCT03085758) tested adrecizumab in human septic shock, and a second phase 2 study in acute decompensated heart failure has been planned.[16]
This antibody strategy is clever because it sidesteps the half-life problem entirely. Instead of trying to deliver exogenous adrenomedullin, it optimizes the distribution of the adrenomedullin the body is already producing.
Long-acting derivatives
Researchers have also developed modified adrenomedullin peptides with extended half-lives that can be administered by subcutaneous injection at longer intervals. These derivatives typically reduce the potent vasodilatory activity (which causes problematic hypotension) while preserving the organ-protective and barrier-stabilizing functions.[16] None has reached phase 3 clinical trials.
RNA vaccine approach
A novel strategy published in 2025 used a stable RNA vaccine to generate antibodies against adrenomedullin in mouse models of cardiovascular disease. Tadic and colleagues designed the vaccine to reduce circulating adrenomedullin levels in conditions where excess adrenomedullin may contribute to pathology. The approach reduced tumor-associated angiogenesis in preclinical testing.[17] This is very early-stage work, tested only in mice, and the long-term consequences of sustained anti-adrenomedullin immunity are unknown. Given adrenomedullin's protective roles in the cardiovascular system, reducing it systemically carries real risks. But the approach represents a fundamentally different therapeutic concept: instead of adding adrenomedullin or redirecting it, this strategy reduces it, potentially useful in conditions where adrenomedullin drives pathology rather than protection.
Limitations of the Evidence
Mostly preclinical: Despite 30 years of research, the evidence for adrenomedullin's therapeutic potential remains largely preclinical. Animal models consistently show organ protection, but translating these findings to human therapy has proven difficult.
Short half-life: The sub-22-minute half-life makes direct peptide therapy impractical without continuous infusion or structural modification, and both approaches introduce their own challenges.
Small clinical trials: Human studies of intravenous adrenomedullin infusion have been small and short-term. Long-term safety and efficacy data in humans do not exist for any adrenomedullin-based therapeutic approach.
Safety
ModerateAdrenomedullin's double-edged biology: organ protection vs. tumor growth
Concern
The same properties that make adrenomedullin protective in sepsis and heart failure — promoting blood vessel growth and preventing cell death — can also help tumors survive and grow. Multiple tumor types overproduce adrenomedullin.
What the research says
No adrenomedullin-based therapy exists yet for chronic use. Any future therapy would need careful cancer screening and monitoring. Researchers are actually exploring blocking adrenomedullin in cancer as a separate strategy.
Particularly relevant for: Relevant to future clinical development, not current patients
What to do
This is a theoretical concern for drug development, not a risk for patients today. No adrenomedullin supplement or drug is commercially available.
Zudaire et al. (2003), Regulatory Peptides; Tadic et al. (2025)
Cancer concern: Adrenomedullin's pro-angiogenic and anti-apoptotic properties raise theoretical concerns about promoting tumor growth. Any chronic adrenomedullin-based therapy would need careful cancer safety monitoring.
Biomarker limitations: MR-proADM is a prognostic tool, not a diagnostic one. It does not distinguish between sepsis and other causes of endothelial stress, and its clinical adoption has been slow outside of European ICUs.
Receptor complexity: The CLR/RAMP receptor system makes selective pharmacological targeting difficult. Drugs that activate adrenomedullin receptors may also affect CGRP signaling, and vice versa.
The Bottom Line
Adrenomedullin is a 52-amino-acid vasodilator peptide produced by nearly every tissue in the body. Discovered in 1993, it has proven roles in blood pressure regulation, endothelial barrier protection, and cardioprotection. Its stable precursor fragment MR-proADM serves as a prognostic biomarker in sepsis, outperforming procalcitonin for mortality prediction. Therapeutic development is ongoing, with adrecizumab (an antibody that redistributes adrenomedullin to damaged tissues) in phase 2 clinical trials for septic shock. The peptide's short half-life and complex receptor system remain the primary obstacles to direct clinical application.
Sources & References
- 1RPEP-00266·Kitamura, K et al. (1993). “Adrenomedullin: a novel hypotensive peptide isolated from human pheochromocytoma..” Biochemical and biophysical research communications.Study breakdown →PubMed →↩
- 2RPEP-00605·Minamino, N et al. (2000). “Adrenomedullin: A Blood Vessel Peptide That Dilates From Both Sides.” Clinical hemorheology and microcirculation.Study breakdown →PubMed →↩
- 3RPEP-00726·Elsasser, Ted H et al. (2002). “Adrenomedullin's Complex Role in Inflammation: Both Pro-Inflammatory Responder and Anti-Inflammatory Protector.” Microscopy research and technique.Study breakdown →PubMed →↩
- 4RPEP-00749·López, José et al. (2002). “Adrenomedullin: A Multifunctional Peptide Involved in Blood Pressure, Growth, Hormones, and Disease.” International review of cytology.Study breakdown →PubMed →↩
- 5RPEP-00895·Cheung, Bernard M Y et al. (2004). “Adrenomedullin in the Cardiovascular System: Vasodilation, Cardiac Protection, and Heart Failure.” Seminars in vascular medicine.Study breakdown →PubMed →↩
- 6RPEP-01232·Gibbons, Carrie et al. (2007). “How RAMP Proteins Control Adrenomedullin Receptor Signaling.” Molecular endocrinology (Baltimore.Study breakdown →PubMed →↩
- 7RPEP-04529·Tsuruda, Toshihiro et al. (2019). “Adrenomedullin: Continuing to explore cardioprotection..” Peptides.Study breakdown →PubMed →↩
- 8RPEP-02682·Kato, Johji et al. (2015). “Bench-to-bedside pharmacology of adrenomedullin..” European journal of pharmacology.Study breakdown →PubMed →↩
- 9RPEP-01294·Temmesfeld-Wollbrück, Bettina et al. (2007). “Adrenomedullin Maintains Blood Vessel Barrier Integrity: Why It Matters for Sepsis and Inflammation.” Thrombosis and haemostasis.Study breakdown →PubMed →↩
- 10RPEP-03142·Valenzuela-Sánchez, Francisco et al. (2016). “MR-proADM: A Peptide Biomarker That Predicts Organ Failure Better Than Standard Tests.” Annals of translational medicine.Study breakdown →PubMed →↩
- 11RPEP-03585·Bernal-Morell, Enrique et al. (2018). “Usefulness of midregional pro-adrenomedullin as a marker of organ damage and predictor of mortality in patients with sepsis..” The Journal of infection.Study breakdown →PubMed →↩
- 12RPEP-04021·Önal, Uğur et al. (2018). “A Peptide Fragment Called MR-proADM Shows Promise for Diagnosing and Predicting Sepsis Outcomes.” Healthcare (Basel.Study breakdown →PubMed →↩
- 13RPEP-00364·Hirata, Y et al. (1996). “Adrenomedullin Peptide Levels Surge During Sepsis and Track Disease Severity.” The Journal of clinical endocrinology and metabolism.Study breakdown →PubMed →↩
- 14RPEP-00876·Zudaire, E et al. (2003). “Adrenomedullin in Cancer: Tumor Growth Promoter, Angiogenesis Driver, and Potential Drug Target.” Regulatory peptides.Study breakdown →PubMed →↩
- 15RPEP-13850·Uddin, Jazib et al. (2025). “CGRP-related peptide adrenomedullin 2 from gut neurons promotes protective ILC2 responses and limits intestinal inflammation.” Nature immunology.Study breakdown →PubMed →↩
- 16RPEP-06256·Kita, Toshihiro et al. (2022). “Translational studies of adrenomedullin and related peptides regarding cardiovascular diseases..” Hypertension research : official journal of the Japanese Society of Hypertension.Study breakdown →PubMed →↩
- 17RPEP-13743·Tadic, Srdan et al. (2025). “mRNA vaccine targeting the regulatory peptide adrenomedullin reduces tumor blood vessels and melanoma growth.” International journal of molecular sciences.Study breakdown →PubMed →↩