Collagen Peptide Markers in Liver Fibrosis
Collagen Peptide Diagnostics
97% sensitivity
In chronic viral liver disease, serum type III procollagen peptide detected cirrhosis in 97% of patients, outperforming three other fibrosis markers.
Murawaki et al., Hepatology, 1994
Murawaki et al., Hepatology, 1994
If you only read one thing
When the liver scars, it leaks tiny protein pieces into the blood. Doctors can measure those pieces with a simple blood draw to tell how much scarring is happening — no needle biopsy required. The best of these tests (PRO-C3 and the ELF score) are accurate enough to guide real decisions, especially for ruling out advanced liver damage. They don't replace a biopsy in every case, but they let millions of people get screened who otherwise never would.
When the liver scars, it produces collagen. When it produces collagen, peptide fragments are cleaved from procollagen molecules and released into the bloodstream. These fragments, measurable through simple blood tests, have become some of the most studied noninvasive biomarkers for detecting and staging liver fibrosis. The concept dates to the early 1980s, when researchers first measured procollagen type III peptide (PIIINP) in serum and bile of patients with alcoholic cirrhosis.[1] Four decades later, collagen-derived peptide markers have evolved from single analytes into composite scoring systems used in clinical hepatology worldwide. This article traces the evidence from foundational studies through modern neo-epitope assays, covering what these markers measure, how well they perform, and where they fall short. For coverage of peptide-based approaches to treating fibrosis rather than diagnosing it, see peptide approaches to liver fibrosis.
Key Takeaways
- Procollagen type III N-terminal peptide (PIIINP) was the first collagen peptide biomarker for liver fibrosis, with serum levels correlating with hepatic collagen synthesis activity (r = 0.76, Torres-Salinas et al., 1986)
- PIIINP was markedly elevated in 90% of patients with alcoholic hepatitis and cirrhosis compared to 60-80% for type I procollagen peptide (Savolainen et al., 1984)
- Type IV collagen 7S domain outperformed PIIINP, type IV collagen triple-helix, and TIMP-1 for detecting cirrhosis in chronic viral liver disease (Murawaki et al., 1994)
- PRO-C3, a modern neo-epitope assay measuring true type III collagen formation, achieves an AUC of 0.81 for detecting significant fibrosis in NAFLD patients
- The Enhanced Liver Fibrosis (ELF) score combines PIIINP, hyaluronic acid, and TIMP-1 into a validated composite that predicts fibrosis progression and hepatic decompensation
- All collagen peptide markers reflect extracellular matrix turnover, not fibrosis directly, meaning elevated levels can indicate either active scarring or active scar remodeling
How Collagen Peptides Become Biomarkers
Liver fibrosis is the progressive accumulation of extracellular matrix (ECM), predominantly collagen, in response to chronic liver injury. Whether the cause is alcohol, hepatitis virus, metabolic dysfunction, or autoimmune disease, the fibrotic process follows a similar pattern: activated hepatic stellate cells transform into myofibroblasts and deposit collagen, primarily types I, III, IV, V, and VI. For deeper coverage of how peptides target this cellular process, see stellate cell targeting: how peptides attack liver fibrosis at the source.
Collagen is synthesized as procollagen, a precursor molecule with extension peptides at both ends (N-terminal and C-terminal propeptides). During collagen maturation, specific enzymes cleave these propeptides. The released fragments enter the circulation, where they can be measured by immunoassays. Because the rate of propeptide release reflects the rate of collagen synthesis (and, in some assays, collagen degradation), these fragments serve as surrogate markers of fibrotic activity.
The critical distinction: collagen peptide markers measure ECM turnover, not the amount of scar tissue already deposited. A patient with active fibrogenesis (new scar being laid down) and a patient with active fibrolysis (old scar being broken down) can both have elevated collagen peptide levels. This is both the strength and limitation of these markers. They detect dynamic change, which liver biopsy, a single snapshot, may miss. But they cannot definitively distinguish between scarring and healing.
The Foundational PIIINP Studies (1983-1987)
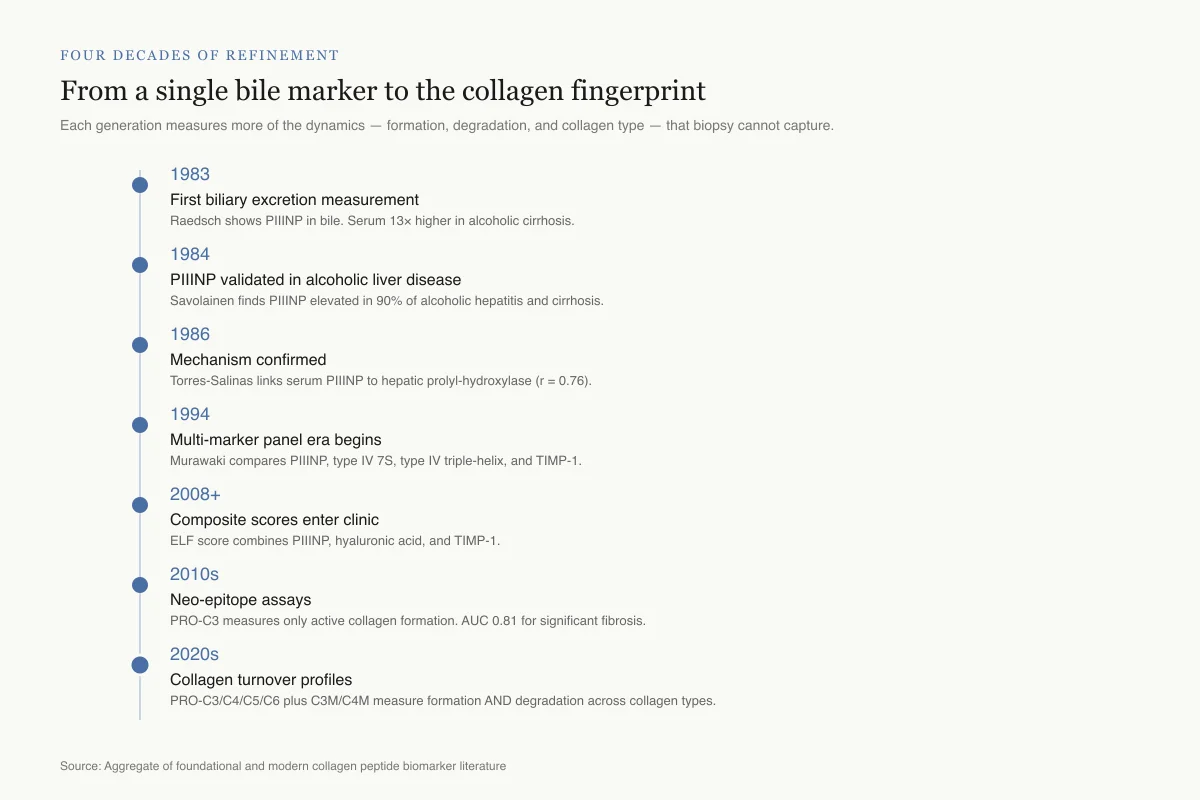
Four decades of refinement
From a single bile marker to the collagen fingerprint
Each generation measures more dynamics — formation, degradation, and specific collagen types — that a snapshot biopsy cannot capture.
1983
First biliary excretion measurement
Raedsch shows PIIINP is excreted in bile. Serum levels 13× higher in alcoholic cirrhosis than controls.
1984
PIIINP validated in alcoholic liver disease
Savolainen finds PIIINP elevated in 90% of alcoholic hepatitis and cirrhosis cases.
1986
Mechanism confirmed
Torres-Salinas links serum PIIINP to hepatic prolyl-hydroxylase activity (r = 0.76).
1994
Multi-marker panel era begins
Murawaki compares PIIINP, type IV 7S, type IV triple-helix, and TIMP-1. Different markers reflect different ECM compartments.
2008+
Composite scores enter clinic
ELF score combines PIIINP, hyaluronic acid, and TIMP-1. Validated for staging and outcome prediction.
2010s
Neo-epitope assays
PRO-C3 developed to measure only active collagen formation. AUC 0.81 for significant fibrosis.
2020s
Collagen turnover profiles
Multi-marker panels (PRO-C3, PRO-C4, PRO-C5, PRO-C6, C3M, C4M) measure formation AND degradation across collagen types.
Source: Aggregate of foundational and modern collagen peptide biomarker literature
 View as image
View as imageFirst Measurements in Bile and Serum
The story begins with Raedsch et al. (1983), who demonstrated that procollagen type III peptide (PIIINP) is excreted in bile, establishing that biliary clearance is a quantitatively important pathway for this peptide fragment.[1] In healthy controls, serum PIIINP was 2.5 ng/ml; in patients with alcoholic cirrhosis, it was 33.6 ng/ml, a 13-fold increase. Biliary excretion rates were also higher in cirrhotics (0.98 nmol/h vs. 0.4 nmol/h in controls), while urinary excretion of a PIIINP fragment (Col 1) was actually lower in cirrhotics (0.11 vs. 0.25 nmol/h). This established that elevated serum PIIINP in liver disease reflects both increased synthesis and altered clearance.
Diagnostic Accuracy in Alcoholic Liver Disease
Savolainen et al. (1984) measured both type I C-terminal and type III N-terminal procollagen peptides in 60 patients with alcoholic and 14 with nonalcoholic liver disease.[2] PIIINP was elevated in 90% of patients with alcoholic hepatitis and cirrhosis. Type I procollagen peptide was elevated in 60-80% of the same patients, making PIIINP the more sensitive marker. Both markers were moderately elevated in fatty liver but could not reliably distinguish simple steatosis from early fibrosis. The highest PIIINP values were observed in patients with numerous Mallory bodies, suggesting the marker also reflects hepatic inflammation intensity.
Correlation with Collagen Synthesis
Torres-Salinas et al. (1986) provided the mechanistic link: serum PIIINP levels correlated closely with hepatic prolyl-hydroxylase activity (r = 0.76, p < 0.001), an enzyme directly involved in collagen synthesis.[3] In 19 patients with alcoholic hepatitis, those with severe fibrosis had PIIINP levels of 49.1 ng/ml compared to 20.4 ng/ml in those with mild fibrosis (p < 0.01). Prolyl-hydroxylase activity showed the same pattern: 1,208 cpm/mg in severe fibrosis versus 734 cpm/mg in mild fibrosis.
This study established a principle that distinguished procollagen peptide markers from other liver tests. Standard liver function tests (ALT, AST, GGT) measure hepatocyte damage, the consequence of disease. Procollagen peptide markers measure the fibrogenic response to that damage, which is the actual process that leads to cirrhosis and liver failure. A patient can have normal transaminases while actively depositing scar tissue, and conversely can have elevated transaminases during acute inflammation without significant fibrosis. PIIINP captures the scarring process itself, not just the insult that triggers it.
Extension Beyond Alcoholic Liver Disease
The clinical utility of PIIINP extended beyond the alcoholic liver disease population where it was first validated. Morelli et al. (1985) showed that type III procollagen peptide levels were elevated in chronic active hepatitis and early stages of liver disease before cirrhosis develops, across multiple etiologies including viral hepatitis.[4] This was important because it suggested the marker was tracking the fibrotic process itself rather than something specific to alcohol-induced liver injury.
El-Mohandes et al. (1987) demonstrated utility in schistosomal liver fibrosis, a major cause of hepatic disease affecting over 200 million people in tropical regions.[5] N-terminal procollagen peptide levels correlated with histological fibrosis grade, showing that the same collagen peptide fragment measured in Finnish alcoholics was equally informative in Egyptian schistosomiasis patients. The universality of the marker across such different disease mechanisms strengthened the case that PIIINP measures a fundamental fibrotic pathway.
Tanaka et al. (1986) explored combining PIIINP with serum proline measurements for improved fibrosis assessment in alcoholic patients, an early example of the multi-analyte approach that would later define the field.[6]
By the late 1980s, the evidence from these foundational studies established a consistent finding across alcoholic hepatitis, chronic viral hepatitis, and parasitic liver disease: serum procollagen peptide levels rise with fibrosis severity and correlate with hepatic collagen synthesis. The marker worked across geographies, etiologies, and clinical settings.
Beyond Type III: Multi-Collagen Panels
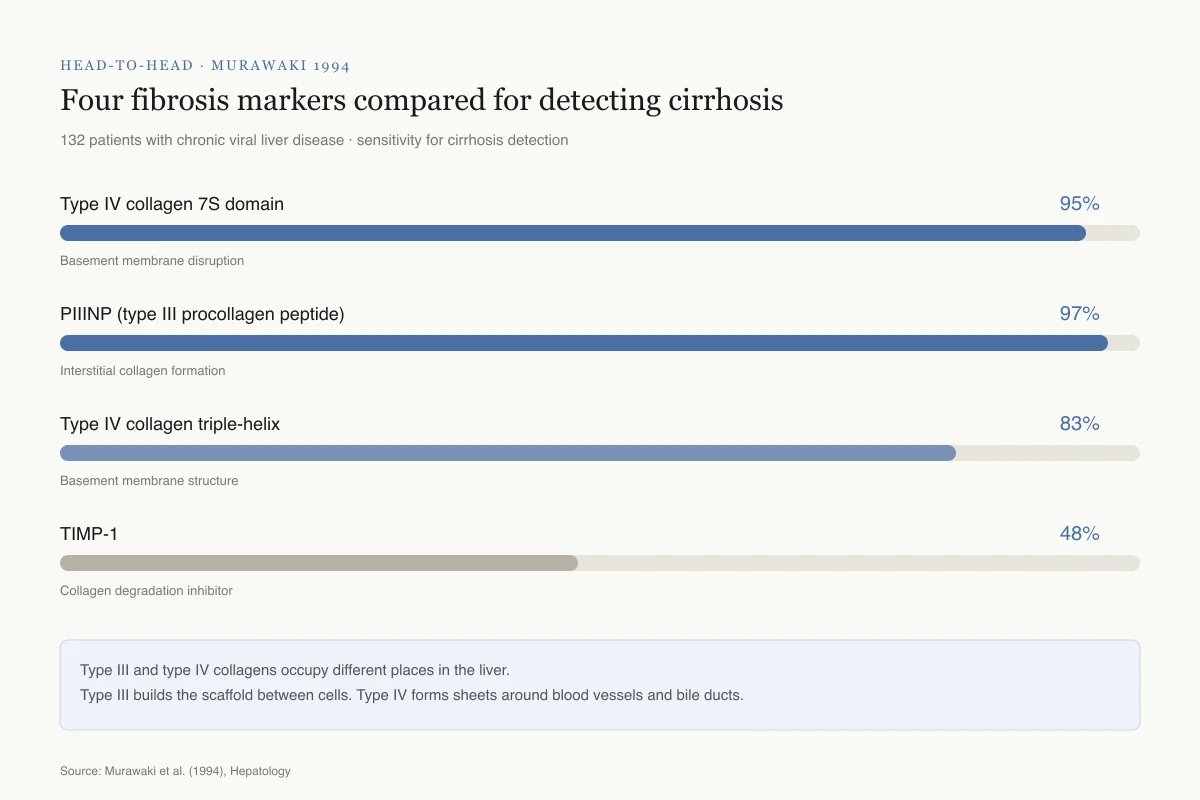
Head-to-Head · Murawaki 1994
Four fibrosis markers compared for detecting cirrhosis
132 patients with chronic viral liver disease · simultaneous serum measurement · sensitivity for cirrhosis detection
Type IV collagen 7S domain
95%
Basement membrane disruption
PIIINP (type III procollagen peptide)
97%
Interstitial collagen formation
Type IV collagen triple-helix
83%
Basement membrane structure
TIMP-1
48%
Collagen degradation inhibitor
Why two winners: Type III and type IV collagens occupy different places in the liver. Type III builds the scaffold between cells; type IV forms the thin sheets around blood vessels and bile ducts. Measuring both catches scarring wherever it starts.
Source: Murawaki et al. (1994), Hepatology
 View as image
View as imageType IV Collagen and Composite Assessment
The liver ECM contains multiple collagen types, and type III procollagen peptide alone does not capture the full picture. Shahin et al. (1992) compared PIIINP with collagen type VI for assessing hepatic fibrosis in both schistosomiasis and alcoholic liver disease, finding that combining markers improved diagnostic accuracy beyond either analyte alone.[7]
The most comprehensive early comparison came from Murawaki et al. (1994), who measured four serum markers simultaneously in 132 patients with chronic viral liver disease: type III procollagen peptide, type IV collagen 7S domain, the central triple-helix of type IV collagen, and tissue inhibitor of metalloproteinases (TIMP-1).[8]
All four markers increased with disease progression, but their sensitivity for detecting cirrhosis differed: type III procollagen peptide detected 97% of cirrhosis cases, type IV collagen 7S domain detected 95%, the type IV triple-helix detected 83%, and TIMP-1 detected only 48%. Using receiver operating curve analysis, type IV collagen 7S domain was the best single test for detecting cirrhosis, with type III procollagen peptide as the second best.
This study revealed an important principle: different collagen markers reflect different aspects of ECM remodeling. Type III collagen is an interstitial collagen that forms the reticular framework of the liver; its propeptide indicates new scar formation in the tissue spaces between hepatocytes. Type IV collagen forms the basement membrane underlying blood vessels and bile ducts; its 7S domain indicates disruption of these structures, which occurs as fibrosis distorts the liver's architecture. TIMP-1 is not a collagen fragment at all but an enzyme inhibitor that prevents matrix metalloproteinases from degrading collagen; elevated TIMP-1 suggests the balance has shifted toward collagen preservation over breakdown.
Together, these markers provide a multi-dimensional view of fibrotic activity that no single analyte can capture. The Murawaki study also demonstrated that these markers correlated more strongly with fibrosis and periportal necrosis than with intralobular inflammation, confirming that collagen peptide markers are primarily fibrosis indicators rather than inflammation indicators.
Modern Neo-Epitope Assays: PRO-C3 and Beyond
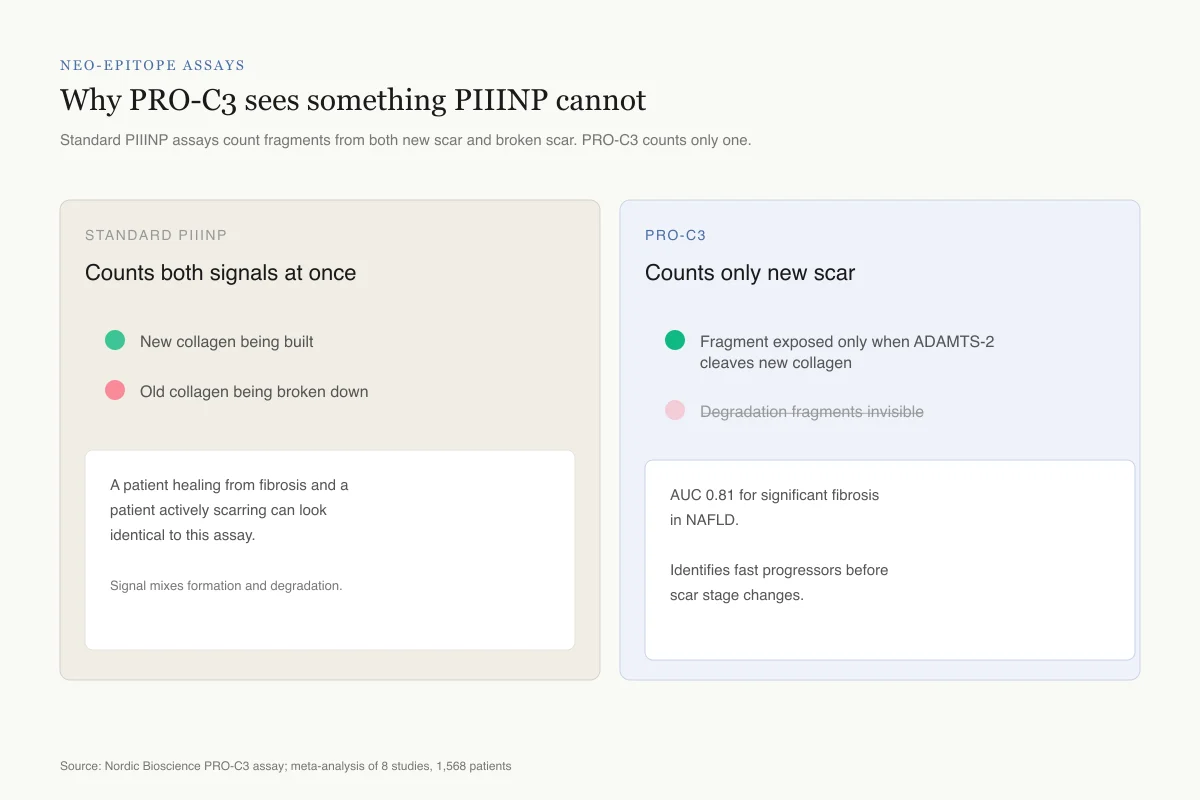
Neo-Epitope Assays
Why PRO-C3 sees something PIIINP cannot
Standard PIIINP assays count fragments from both new scar and broken scar. PRO-C3 counts only one.
Standard PIIINP
Counts both signals at once
A patient healing from fibrosis and a patient actively scarring can look identical.
PRO-C3
Counts only new scar
AUC 0.81 for significant fibrosis in NAFLD. Identifies "fast progressors" before scar stage changes.
The neo-epitope trick: design an antibody that only recognizes the exact molecular surface created by a single, formation-specific cleavage event. Everything else stays invisible.
Source: Nordic Bioscience PRO-C3 assay; meta-analysis of 8 studies, 1,568 patients
 View as image
View as imageWhat PRO-C3 Measures
The limitation of first-generation PIIINP assays became apparent as the field matured: the antibodies used in standard PIIINP immunoassays detect both intact PIIINP (released during collagen formation) and PIIINP fragments generated during collagen degradation. This means a standard PIIINP measurement cannot distinguish between a liver actively laying down new scar tissue and a liver actively breaking down old scar tissue. Both processes release type III collagen fragments that the assay counts.
PRO-C3, developed by Nordic Bioscience in Denmark, addresses this by targeting a specific neo-epitope, a molecular surface that only becomes exposed when the ADAMTS-2 enzyme cleaves the N-terminal propeptide from intact type III collagen during its maturation into functional collagen fibrils. The antibody used in the PRO-C3 assay recognizes only this cleavage-specific epitope. An intact PIIINP that has not yet been cleaved from its collagen molecule will not be detected. A PIIINP fragment generated by degradation of existing collagen will not be detected. Only the specific fragment produced during new collagen deposition registers.
In clinical validation studies across NAFLD, hepatitis C, hepatitis B, and alcoholic liver disease, PRO-C3 achieves an area under the receiver operating curve (AUC) of 0.81 for detecting significant fibrosis (F2 or greater) and 0.79 for advanced fibrosis. In a meta-analysis of eight studies including 1,568 patients, the summary AUC was 0.81 (95% CI 0.77-0.84) for significant fibrosis. Patients with baseline PRO-C3 levels of 20.2 ng/ml or higher showed fibrosis progression over follow-up, while those with lower levels did not progress. This prognostic capability, identifying "fast progressors" before their fibrosis advances, represents a step beyond static staging and could guide decisions about treatment timing and monitoring intensity.
PRO-C3 has also been shown to be heritable, with genetic factors influencing baseline levels. In a genome-wide association study, PRO-C3 correlated with liver fibrosis measured by magnetic resonance elastography (MRE), and the combination of genetic risk score and PRO-C3 improved prediction beyond either alone.
The ELF Score
The Enhanced Liver Fibrosis (ELF) test combines three direct ECM biomarkers into a single composite score: PIIINP, hyaluronic acid (a glycosaminoglycan that accumulates in fibrotic tissue), and TIMP-1. The ELF score has been validated against biopsy-proven fibrosis across multiple chronic liver disease etiologies in both adult and pediatric populations.
The ELF score predicts hepatic decompensation and mortality. In a validation study, patients with higher ELF scores had increased risk of liver-related clinical events including variceal bleeding, ascites, hepatocellular carcinoma, and liver-related death. This prognostic ability is a step beyond simple staging: it suggests that dynamic ECM biomarkers carry information about disease trajectory that static biopsy staging may miss.
Collagen Turnover Profiles
Recent work has moved beyond single markers or simple composites toward "collagen turnover profiles" that measure formation and degradation of multiple collagen types simultaneously. A 2019 study assessed PRO-C3 (type III formation), PRO-C5 (type V formation), PRO-C4 (type IV formation), and C4M (type IV degradation) in liver transplant recipients. PRO-C3, PRO-C4, and C4M were all elevated in patients whose fibrosis recurred after transplantation, with the combination outperforming any individual marker.
Stemann et al. (2025) applied a similar multi-marker approach using PRO-C3, PRO-C6 (type VI collagen formation), and C3M (type III collagen degradation) to monitor kidney function decline in patients undergoing peptide receptor radionuclide therapy.[9] Urinary C3M was the most predictive marker, demonstrating that collagen turnover peptides have diagnostic applications beyond liver disease.
Strengths and Limitations of Collagen Peptide Markers
What These Markers Do Well
Collagen peptide biomarkers are noninvasive, reproducible, and scalable. A single blood draw replaces a liver biopsy, which carries a small but real risk of bleeding (1-3%) and pain (20-30%), samples only 1/50,000th of the liver (introducing substantial sampling error, since fibrosis distribution is often heterogeneous), and requires pathologist interpretation subject to inter-observer variability that can differ by one or two fibrosis stages even between expert hepatopathologists. Serum markers can be repeated over time to track fibrosis dynamics, something biopsy cannot practically achieve. A patient might reasonably undergo blood testing every 6 to 12 months, while serial biopsies at that frequency would be clinically and ethically unjustifiable.
These markers are particularly strong at ruling out advanced fibrosis. In most validation studies, the negative predictive value (the probability that a low score truly means no advanced fibrosis) exceeds 90%. This makes them valuable as screening tools in primary care and hepatology clinics: patients with low collagen peptide levels are unlikely to have advanced disease and can avoid biopsy. Given that millions of people worldwide have undiagnosed NAFLD, a scalable screening blood test is arguably more important than a perfect staging tool.
Where They Fall Short
Collagen peptide markers cannot replace liver biopsy for all purposes. Their ability to distinguish adjacent fibrosis stages (F1 vs. F2, or F2 vs. F3) is limited, with substantial overlap between stages. Biopsy remains necessary for initial diagnosis in ambiguous cases, for assessing features like steatohepatitis grade and ballooning that peptide markers do not capture, and for research protocols requiring histological endpoints.
PIIINP levels are influenced by factors beyond liver fibrosis. Active wound healing anywhere in the body, bone turnover, rheumatic diseases, and even growth in children can elevate PIIINP. This is why composite scores like ELF perform better than single analytes: combining multiple markers with different confounders reduces false positives.
The distinction between fibrogenesis and fibrolysis remains a challenge. PRO-C3 partially addresses this for type III collagen by measuring only formation, but the clinical reality is that formation and degradation often occur simultaneously in a fibrotic liver. A declining PRO-C3 could mean slowing fibrosis (good) or could mean collagen maturation is complete and the scar is stable (neutral). Context matters.
Diagnostic Use Across Liver Disease Etiologies
The performance of collagen peptide markers varies by disease etiology, and understanding these differences matters for clinical interpretation.
In alcoholic liver disease, where the foundational studies were conducted, PIIINP performs well because fibrogenesis is typically active and progressive. The Torres-Salinas correlation of r = 0.76 between PIIINP and prolyl-hydroxylase activity was established in this population.[3] The confounding factor is that alcohol cessation can rapidly reduce PIIINP levels even before fibrosis resolves, because the marker tracks active collagen synthesis rather than accumulated scar.
In NAFLD/NASH, the dominant modern epidemic affecting an estimated 25% of the global population, PRO-C3 has shown consistent diagnostic value across multiple cohorts. This matters because NAFLD fibrosis progresses silently for years, and most patients are diagnosed incidentally or late. A blood test that identifies the subset of NAFLD patients with active fibrogenesis could transform screening strategies.
In hepatitis C, collagen peptide markers have been validated both for staging and for monitoring treatment response. After sustained virologic response to antiviral therapy, PIIINP and PRO-C3 levels decline, providing evidence that fibrogenesis slows when the underlying cause is eliminated. This makes collagen markers valuable for confirming treatment success beyond viral clearance.
In hepatitis B and autoimmune hepatitis, the evidence is more limited but directionally consistent. The geographic variation in validation data reflects where research groups have been active rather than fundamental differences in marker biology.
For related coverage of how peptide therapies might directly address liver fibrosis, see BPC-157 and liver protection.
Where the Field Is Heading
The convergence of collagen peptide biomarkers with imaging, genetics, and artificial intelligence is reshaping fibrosis diagnostics. Combining PRO-C3 with FIB-4 (a simple score using age, AST, ALT, and platelets) improves diagnostic accuracy beyond either alone. MRI-based elastography can measure liver stiffness noninvasively, and combining stiffness measurements with collagen peptide levels further improves staging precision.
Collagen hybridizing peptides (CHPs), synthetic peptides that selectively bind damaged collagen through triple-helix formation, represent a next-generation approach. These peptides can be conjugated to imaging agents for direct visualization of fibrotic tissue, potentially providing both the anatomical information of imaging and the molecular specificity of blood biomarkers.
The integration of collagen peptide panels into clinical practice guidelines is advancing. The American Association for the Study of Liver Diseases (AASLD) and the European Association for the Study of the Liver (EASL) now recommend noninvasive tests as first-line assessment for liver fibrosis in specific populations, with biopsy reserved for cases where noninvasive results are indeterminate. The FDA has accepted collagen peptide markers as exploratory endpoints in clinical trials for antifibrotic drugs, recognizing that dynamic biomarkers may capture treatment effects faster than histological endpoints.
As the peptide biomarker portfolio expands to include formation-specific, degradation-specific, and collagen-type-specific assays, the goal is a blood-based "collagen fingerprint" that captures the full dynamics of ECM remodeling. The trajectory from Raedsch's 1983 biliary excretion study to modern multi-collagen turnover profiles represents four decades of progressive refinement. The fundamental insight has not changed: when the liver scars, it leaves peptide fingerprints in the blood. What has changed is our ability to read those fingerprints with increasing specificity and clinical utility.
The Bottom Line
Collagen peptide biomarkers for liver fibrosis have evolved from single-analyte PIIINP measurements in the 1980s to sophisticated multi-marker panels and composite scores. The evidence base spans alcoholic hepatitis, chronic viral hepatitis, schistosomiasis, and NAFLD/NASH. Modern assays like PRO-C3 achieve AUCs above 0.80 for detecting significant fibrosis, while composite scores like ELF predict clinical outcomes including decompensation and mortality. These markers measure ECM turnover rather than scar volume, which is both their diagnostic strength (capturing dynamic change) and their primary limitation (difficulty distinguishing active scarring from active healing).
Sources & References
- 1RPEP-00019·Raedsch, R et al. (1983). “Biliary excretion of procollagen type III peptide in healthy humans and in patients with alcoholic cirrhosis of the liver..” Gastroenterology.Study breakdown →PubMed →↩
- 2RPEP-00023·Savolainen, E R et al. (1984). “Can Blood Tests for Collagen Fragments Reveal How Badly Alcohol Has Damaged the Liver?.” Alcoholism.Study breakdown →PubMed →↩
- 3RPEP-00039·Torres-Salinas, M et al. (1986). “Serum procollagen type III peptide as a marker of hepatic fibrogenesis in alcoholic hepatitis..” Gastroenterology.Study breakdown →PubMed →↩
- 4RPEP-00030·Morelli, A et al. (1985). “Type III procollagen peptide and PZ-peptidase serum levels in pre-cirrhotic liver diseases..” Clinica chimica acta; international journal of clinical chemistry.Study breakdown →PubMed →↩
- 5RPEP-00042·el-Mohandes, M et al. (1987). “A Blood Test for Liver Scarring: How a Collagen Peptide Tracks Fibrosis Without Biopsy.” Experimental and molecular pathology.Study breakdown →PubMed →↩
- 6RPEP-00038·Tanaka, Y et al. (1986). “A Blood Peptide That Reveals Hidden Liver Scarring in Alcoholic Liver Disease.” Digestive diseases and sciences.Study breakdown →PubMed →↩
- 7RPEP-00248·Shahin, M et al. (1992). “Blood Tests for Collagen Fragments Can Track How Fast Your Liver Is Scarring.” Hepatology (Baltimore.Study breakdown →PubMed →↩
- 8RPEP-00303·Murawaki, Y et al. (1994). “Serum type III procollagen peptide, type IV collagen 7S domain, central triple-helix of type IV collagen and tissue inhibitor of metalloproteinases in patients with chronic viral liver disease: relationship to liver histology..” Hepatology (Baltimore.Study breakdown →PubMed →↩
- 9RPEP-13680·Stemann Lau, Tobias et al. (2025). “Urinary collagen degradation marker may predict kidney function decline during peptide radionuclide therapy.” Frontiers in endocrinology.Study breakdown →PubMed →↩