BPC-157 and Spinal Cord Injury: Animal Models
BPC-157 and Spinal Cord Injury
1 year sustained recovery
Rats with spinal cord compression injuries showed functional recovery lasting one full year after a single BPC-157 injection given 10 minutes post-injury.
Perovic et al., Journal of Orthopaedic Surgery and Research, 2019
Perovic et al., Journal of Orthopaedic Surgery and Research, 2019
If you only read one thing
BPC-157 reversed tail paralysis in rats after a spinal cord compression injury, and a single injection kept them recovered for a full year. The effect has been seen in five different nervous system injury models — all in rats, all from one lab in Zagreb. No human trial has ever tested BPC-157 for spinal cord injury. Rat tail recovery is not the same thing as human limb recovery.
A single intraperitoneal injection of BPC-157 given 10 minutes after spinal cord compression produced functional recovery in rats that lasted one full year.[1] That finding, published in the Journal of Orthopaedic Surgery and Research in 2019, represents the most detailed study to date on BPC-157's effects in spinal cord injury. A follow-up study in 2022 extended the evidence by demonstrating that even rats with definitive, established spinal cord injuries at day 4 post-injury responded to BPC-157 therapy with rapid functional improvement.[2] These results, combined with BPC-157's documented neuroprotective effects in peripheral nerve transection, traumatic brain injury, and stroke models, position the peptide as one of the more broadly studied neuroprotective candidates in preclinical research.[5] All evidence remains in animal models. No human spinal cord injury trial has been conducted.
Key Takeaways
- A single intraperitoneal BPC-157 injection (200 or 2 mcg/kg) given 10 minutes after spinal cord compression restored tail motor function and resolved spasticity by day 15 in rats (Perovic et al., 2019).[1]
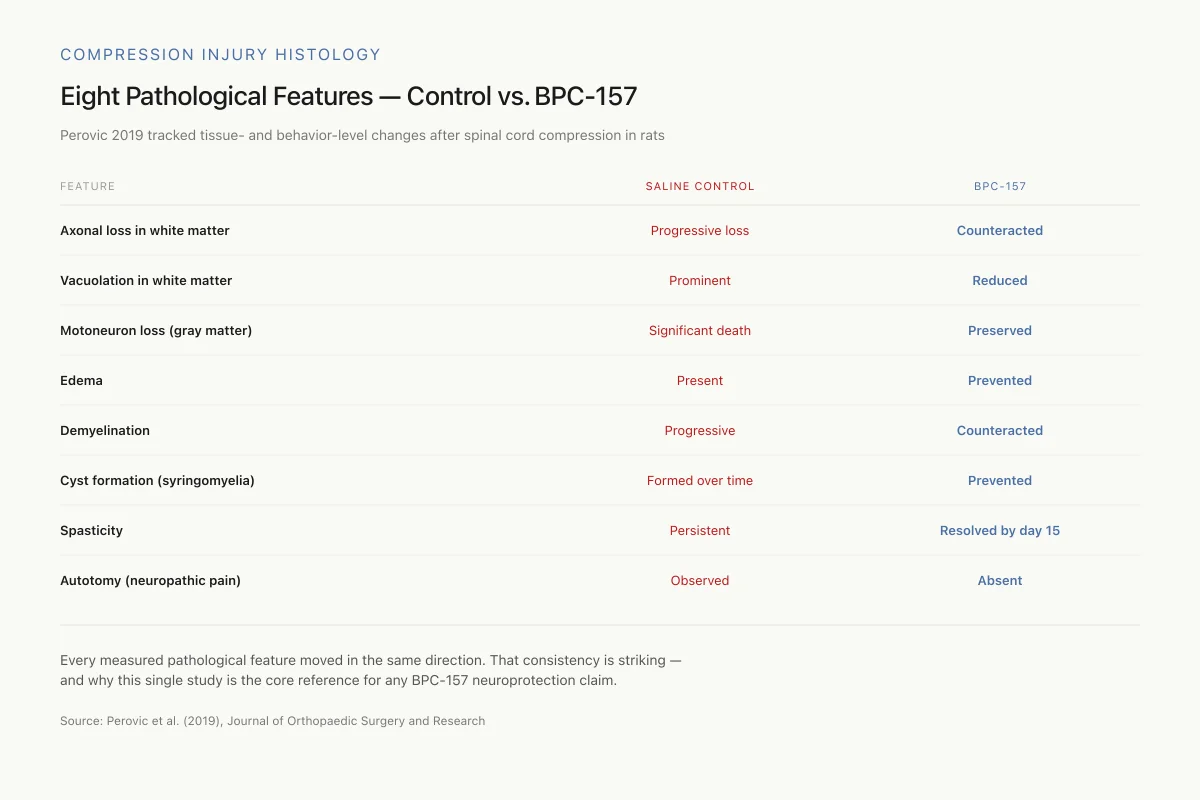
- Histological analysis showed BPC-157 counteracted axonal loss in white matter, motoneuron loss in gray matter, edema, vacuolation, demyelination, and cyst formation (Perovic et al., 2019).[1]
- Even rats with established spinal cord injuries at day 4 responded to BPC-157 oral therapy with rapid tail function recovery and no demyelination over a month-long observation (Perovic et al., 2022).[2]
- The 2022 study showed BPC-157 increased Nos1, Nos2, and Nos3 gene expression at 30 minutes post-injury, implicating the nitric oxide system in its neuroprotective mechanism.[2]
- BPC-157 also improved outcomes in rat models of sciatic nerve transection, traumatic brain injury, hippocampal ischemia, and cuprizone-induced demyelination, suggesting broad CNS and PNS protective effects.[3]
- All evidence comes from a single research group in Zagreb using rat models. No independent replication, no human data, no randomized controlled trials exist for BPC-157 in spinal cord injury.
The 2019 spinal cord compression study
The primary evidence for BPC-157 in spinal cord injury comes from Perovic et al. (2019), published in the Journal of Orthopaedic Surgery and Research. The study used a compression injury model: laminectomy at L2-L3 followed by 60-second compression of the exposed dural sac of the sacrocaudal spinal cord using a neurosurgical piston weighing 60-66 grams. This produced consistent tail paralysis in Wistar rats.[1]
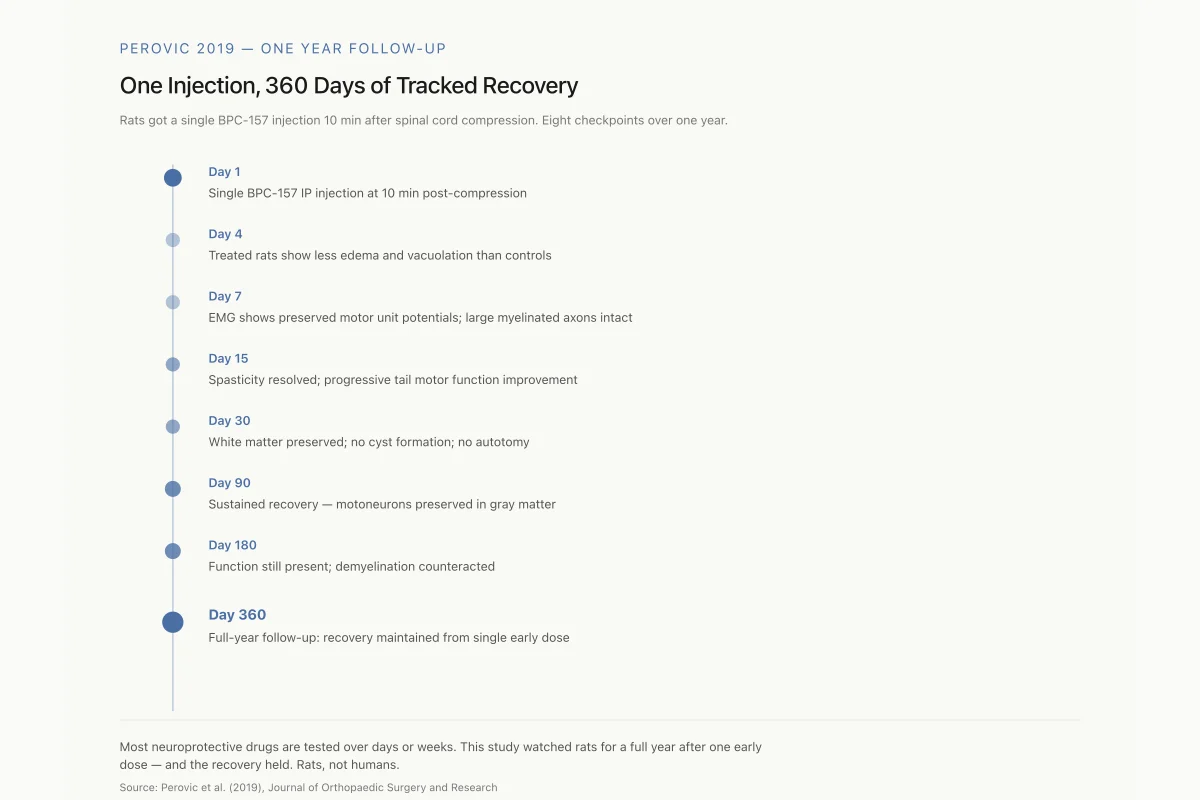
Treatment was a single intraperitoneal injection at 10 minutes post-injury. Two doses were tested: 200 mcg/kg and 2 mcg/kg, against 0.9% saline controls at 5 ml/kg. Assessments were performed at 1, 4, 7, 15, 30, 90, 180, and 360 days after injury, making this one of the longest follow-up periods in BPC-157 research.
Motor function recovery. All BPC-157-treated rats showed consistent clinical improvement with increasingly better motor function of the tail. Autotomy (self-mutilation, a behavioral indicator of neuropathic pain) was absent in treated animals. Spasticity resolved by day 15. Control rats showed persistent tail paralysis and spasticity throughout the observation period.
White matter changes. BPC-157 counteracted the formation of vacuoles and the loss of axons in white matter. In control animals, progressive vacuolation and axonal degeneration were prominent features of the injury. BPC-157-treated animals showed markedly less white matter damage at every time point assessed.
Gray matter changes. BPC-157 prevented edema formation and motoneuron loss in gray matter. The preservation of motoneurons in the ventral horn is particularly significant because motoneuron death is a primary driver of permanent motor deficits after spinal cord injury.
Caudal nerve assessment. Starting from day 7, BPC-157-treated rats showed preserved large myelinated axons in the caudal nerve distal to the injury. EMG recordings confirmed this structural preservation with markedly improved motor unit potentials in tail muscles compared to controls.
Demyelination and cyst formation. BPC-157 counteracted both demyelination and cyst formation, two hallmarks of chronic spinal cord injury that contribute to permanent disability. The prevention of cyst formation (syringomyelia) is notable because post-traumatic cysts can expand over time and worsen neurological deficits months or years after the initial injury.
Both the 200 mcg/kg and 2 mcg/kg doses produced therapeutic effects. The study concluded that BPC-157 therapy "can impact all stages of the secondary injury phase," a claim based on the peptide's effects on inflammation, edema, demyelination, axonal degeneration, and cyst formation.
Perovic 2019 — One Year Follow-up
One Injection, 360 Days of Tracked Recovery
Rats got a single BPC-157 injection 10 minutes after spinal cord compression. Assessors checked them eight times across a full year.
Most neuroprotective drugs are tested over days or weeks. This study watched rats for a full year after one early dose — and the recovery held. Rats, not humans.
Source: Perovic et al. (2019), J Orthop Surg Res
 View as image
View as imageCompression Injury Histology
Eight Pathological Features — Control vs. BPC-157
Perovic 2019 tracked tissue- and behavior-level changes across saline and BPC-157-treated rats after spinal cord compression.
Every measured pathological feature moved in the same direction. That consistency is striking — and is why this study is the core reference for any BPC-157 neuroprotection claim.
Source: Perovic et al. (2019), J Orthop Surg Res
 View as image
View as imageThe 2022 follow-up: definitive spinal cord injury
Perovic et al. (2022) extended these findings with a three-part study that addressed a critical question: can BPC-157 help once a spinal cord injury is established, not just when given immediately after trauma?[2]
Part 1: Immediate effects (10-30 minutes post-injury). Using the same sacrocaudal compression model, BPC-157 was given intraperitoneally at 10 minutes post-injury, and assessments were performed at 10-30 minutes after injury. BPC-157-treated rats showed only discrete edema and minimal hemorrhage compared to controls. Gene expression analysis at 30 minutes revealed increased Nos1, Nos2, and Nos3 expression, the three isoforms of nitric oxide synthase. This is the earliest documented molecular response to BPC-157 in a spinal cord injury model.
Part 2: Definitive injury (day 4 treatment). By day 4 after compression, the spinal cord injury is considered definitive in this model. BPC-157 was administered intragastrically (oral route) at day 4, and assessments were performed 20 minutes after treatment. Even at this delayed timepoint, BPC-157-treated rats showed only mild hemorrhage and discrete tissue vacuolation compared to more severe pathology in controls.
Part 3: Sustained oral therapy (days 4-30). BPC-157 was given continuously in drinking water from day 4 through day 30 post-injury. Rats with definitive tail paralysis rapidly presented tail function recovery. Luxol fast blue staining confirmed no demyelination process in BPC-157-treated animals. This finding is relevant because demyelination is one of the progressive pathological processes that worsens spinal cord injury outcomes over time. Preventing or reversing demyelination in an established injury could be clinically meaningful.
The three-part design demonstrated that BPC-157 had effects at every stage: acute (minutes), subacute (days), and chronic (weeks). The oral route effectiveness in parts 2 and 3 is notable because a therapy that works orally is more practical for clinical translation than one requiring injection.
How BPC-157 might protect the spinal cord
The mechanisms behind BPC-157's spinal cord effects are not fully elucidated, but several pathways are supported by evidence from the SCI studies and related neuroscience research.
Nitric oxide system modulation
The 2022 Perovic study demonstrated that BPC-157 rapidly upregulated all three NOS isoforms (Nos1, Nos2, Nos3) within 30 minutes of administration in spinal cord injury rats.[2] This is consistent with broader BPC-157 research showing that the peptide interacts extensively with the nitric oxide system. Vukojevic et al. (2020) demonstrated in a hippocampal ischemia model that BPC-157 elevated Nos3 and Nos1 gene expression while decreasing Nos2 and Nfkb expression during reperfusion, a pattern suggesting selective modulation rather than blanket upregulation.[7]
Nitric oxide plays complex roles in spinal cord injury. In the acute phase, endothelial NO (from eNOS/Nos3) promotes blood flow and can be protective, while excessive inducible NO (from iNOS/Nos2) contributes to secondary injury through oxidative stress. BPC-157's ability to modulate all three NOS isoforms simultaneously could theoretically address both the beneficial and harmful aspects of NO signaling after spinal cord trauma.
Angiogenic and vascular effects
BPC-157 has established angiogenic properties. Seiwerth et al. (2018) reviewed how BPC-157 interacts with standard angiogenic growth factors (EGF, FGF, VEGF) and concluded that BPC-157 was "consistently effective" in promoting healing across models of acute and chronic tissue injury, implementing "its own angiogenic effect in the healing" process.[9]
In the spinal cord context, angiogenesis matters because secondary injury after spinal cord trauma involves vascular disruption, hemorrhage, and ischemia. The 2019 SCI study documented that BPC-157 counteracted edema and hemorrhage, both vascular phenomena. Vukojevic et al. (2020) showed that BPC-157 elevated Vegfr2 expression in hippocampal tissue after ischemia/reperfusion, providing molecular evidence for its angiogenic action in central nervous system tissue specifically.[7]
Gene expression pathways
The hippocampal ischemia study by Vukojevic et al. (2020) provided the most detailed gene expression data for BPC-157 in CNS injury. They found strongly elevated expression of Egr1, Akt1, Kras, Src, Foxo, Srf, Vegfr2, Nos3, and Nos1, with decreased expression of Nos2, Nfkb, and no activation of Mapk1.[7]
The Egr1 (Early Growth Response 1) gene is relevant to nerve regeneration: it regulates genes involved in cell growth, differentiation, and survival, overlapping functionally with neurotrophic factors like BDNF. The Akt1 activation is associated with anti-apoptotic signaling, which could protect motoneurons and other spinal cord cells from secondary injury-induced cell death. Sikiric et al. (2016) noted that BPC-157's effects on JAK-2 pathways were also implicated in its neuroprotective activity.[6]
These gene expression patterns provide molecular plausibility for BPC-157's spinal cord effects but do not constitute a proven mechanism. The gene studies were performed in hippocampal tissue after ischemia, not in spinal cord tissue after compression. Whether the same expression patterns occur in spinal cord injury specifically remains to be confirmed.
The Proposed Mechanism
How BPC-157 May Protect the Liver: The Src-Caveolin-1-eNOS Pathway
A signaling cascade that ends in more nitric oxide, better hepatic blood flow, and reduced liver damage
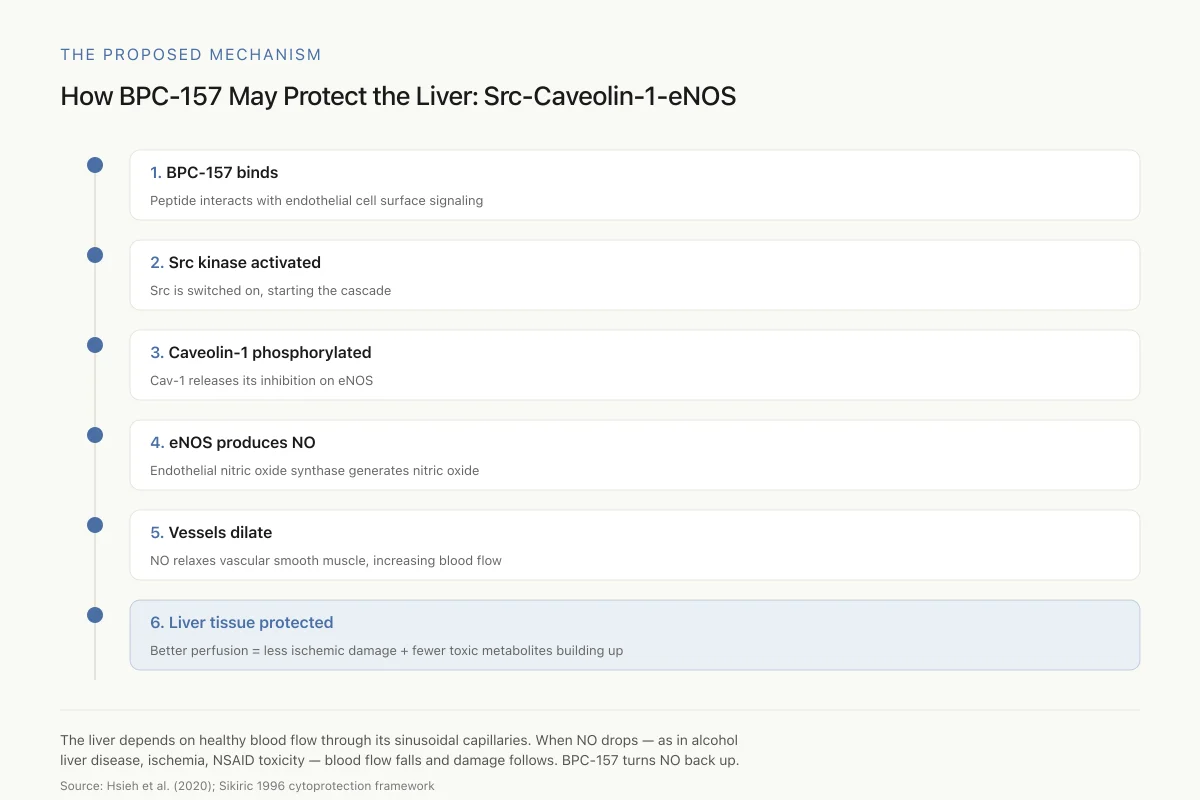
The liver depends on healthy blood flow through its sinusoidal capillaries. When NO production drops — as it does in alcohol-related liver disease, ischemia, and NSAID toxicity — blood flow falls and damage follows. BPC-157 seems to turn the NO signal back up.
Source: Hsieh et al. (2020); Sikiric 1996 cytoprotection framework
 View as image
View as imageBPC-157 in other nervous system injury models
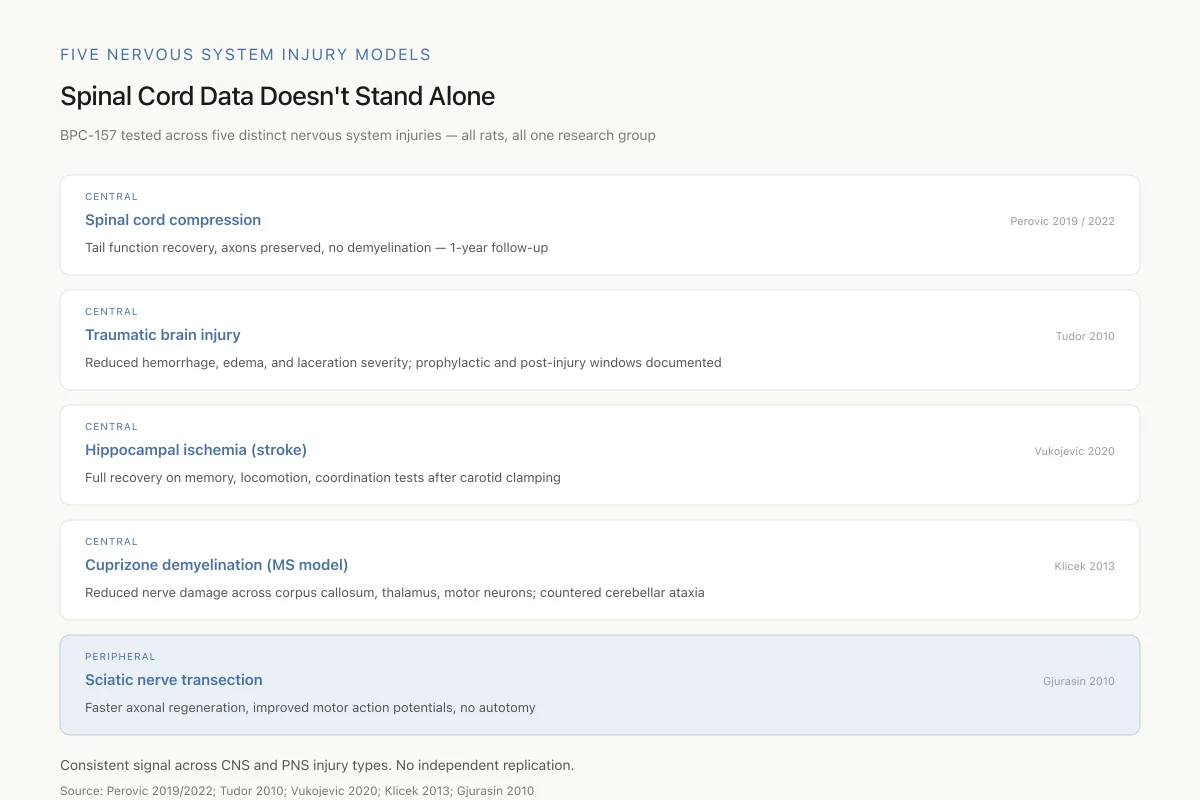
The spinal cord injury data does not exist in isolation. BPC-157 has been tested across several nervous system injury models, creating a pattern of CNS and PNS protective effects.
Peripheral nerve transection
Gjurasin et al. (2010) tested BPC-157 in rats with transected sciatic nerves. Treatment was administered via three routes: intraperitoneally, intragastrically, or locally at the anastomosis site. BPC-157-treated rats showed faster axonal regeneration with improved neural fascicle presentation, homogeneous regeneration patterns, increased density and size of regenerating fibers, and increased blood vessel presence. Electrophysiological testing confirmed increased motor action potentials, and functional testing showed improved sciatic functional index scores. Autotomy was absent in treated animals.[3] For deeper coverage of this evidence, see our dedicated article on BPC-157 and peripheral nerve repair.
Traumatic brain injury
Tudor et al. (2010) demonstrated that BPC-157 attenuated damage from traumatic brain injury in mice using a falling weight model. Both microgram and nanogram doses improved the conscious/unconscious/death ratio across a range of force impulses. BPC-157 reduced subarachnoid and intraventricular hemorrhage, brain laceration severity, and brain edema. When given prophylactically 30 minutes before injury, it was effective against force impulses ranging from 0.068 to 0.159 Ns. Post-injury, the time window for effectiveness varied with injury severity: 5 minutes for moderate (0.130 Ns), 20 minutes for severe (0.145 Ns), and 30 minutes for the most severe impulse tested (0.159 Ns).[4]
Hippocampal ischemia and stroke
Vukojevic et al. (2020) tested BPC-157 in rats after bilateral common carotid artery clamping for 20 minutes, a model of hippocampal ischemia/reperfusion injury (stroke). BPC-157 applied as a 1 ml bath to the operated area during reperfusion counteracted both early (24-hour) and delayed (72-hour) neural hippocampal damage. Full functional recovery was documented via Morris water maze (memory), inclined beam-walking (locomotion), and lateral push (coordination) tests.[7]
Cuprizone-induced demyelination
Klicek et al. (2013) tested BPC-157 in rats exposed to cuprizone, a copper chelator that induces demyelination similar to what occurs in multiple sclerosis. Cuprizone-treated control rats developed nerve damage in multiple brain areas, with the most prominent damage in corpus callosum, laterodorsal thalamus, nucleus reuniens, and anterior horn motor neurons. BPC-157 consistently reduced nerve damage in all affected areas, with the greatest protective effect in the most severely damaged regions. BPC-157 also counteracted cuprizone-induced cerebellar ataxia and impaired forelimb function.[8]
This demyelination data is particularly relevant to spinal cord injury because demyelination is a major pathological process in both acute and chronic SCI. The fact that BPC-157 counteracted both traumatic demyelination (in the SCI models) and toxic demyelination (in the cuprizone model) suggests its anti-demyelination effects may operate through a common protective mechanism. For broader context on neurotrophic peptides and peptide approaches to nerve regeneration, see our companion articles.
Five Nervous System Injury Models
Spinal Cord Data Doesn’t Stand Alone
BPC-157 has been tested across five distinct nervous system injuries — all rats, all one research group.
Consistent signal across CNS and PNS injury types. The breadth is unusual for a single peptide. So is the fact that independent labs haven’t replicated any of it.
Source: Perovic 2019/2022; Tudor 2010; Vukojevic 2020; Klicek 2013; Gjurasin 2010
 View as image
View as imageBPC-157 and neurotransmitter modulation
BPC-157's central nervous system effects extend to neurotransmitter systems, which provides additional context for understanding its spinal cord injury results.
Tohyama et al. (2004) measured the effects of BPC-157 on serotonin synthesis rates across rat brain regions using alpha-methyl-L-tryptophan autoradiography. Acute BPC-157 administration reduced serotonin synthesis in the dorsal thalamus, hippocampus, lateral geniculate body, and hypothalamus, while increasing synthesis in the substantia nigra reticulata and medial anterior olfactory nucleus. After 7 days of treatment, synthesis decreased in the dorsal raphe nucleus and increased in the substantia nigra, lateral caudate, accumbens nucleus, and superior olive.[10]
Boban Blagaic et al. (2005) demonstrated that BPC-157 was effective against serotonin syndrome in rats, counteracting the excessive serotonergic signaling produced by combined SSRI and MAO inhibitor administration.[11]
Sikiric et al. (2016) reviewed the accumulated evidence for BPC-157's effects on dopaminergic and serotonergic systems, concluding that the peptide "modulates serotonergic and dopaminergic systems" and "beneficially affects various behavioral disturbances" resulting from damaged or overstimulated neurotransmitter systems.[6]
The neurotransmitter data is relevant to spinal cord injury because serotonergic and dopaminergic signaling play roles in motor function, pain processing, and autonomic regulation. Disruption of descending serotonergic pathways after SCI contributes to motor deficits and neuropathic pain. A peptide that modulates these systems could theoretically influence SCI recovery through neurotransmitter-mediated mechanisms in addition to its direct tissue-protective effects.
Safety
ModerateNo human spinal cord injury data
Concern
Every rodent neuroprotective drug that worked in SCI models — methylprednisolone, minocycline, riluzole, GM1 ganglioside — has either failed or produced only modest benefit in human trials. BPC-157 has never been tested in human SCI at all.
What the research says
The rat data is consistent and internally coherent. But consistency in rats has not historically translated to efficacy in human SCI. Nothing about this peptide has earned an exception to that pattern yet.
Particularly relevant for: Anyone considering BPC-157 for a spinal cord injury or neurological condition
What to do
Do not self-administer BPC-157 in the context of spinal cord injury. Consult a rehabilitation physician and discuss evidence-based options.
Review of past SCI translation failures; Perovic et al. 2019/2022
Limitations of the evidence
The BPC-157 spinal cord injury evidence has significant constraints that should be understood before drawing conclusions.
Single research group. Virtually all BPC-157 neuroscience research originates from Sikiric's laboratory at the University of Zagreb. The SCI studies, TBI study, nerve transection study, hippocampal ischemia study, and cuprizone study all involve overlapping author lists. Independent replication by separate research groups is absent for the spinal cord injury findings specifically.
Animal models only. No human clinical trial has tested BPC-157 for spinal cord injury or any nervous system condition. BPC-157 has been tested in phase II inflammatory bowel disease trials and more recently in a multiple sclerosis trial, but spinal cord injury has not been targeted clinically.[1]
Compression model limitations. The sacrocaudal compression model used in the SCI studies produces tail paralysis, not the limb paralysis that characterizes most human SCI. The rat sacrocaudal spinal cord differs anatomically and functionally from the cervical and thoracic segments most commonly injured in humans. Tail function recovery, while measurable and reproducible, does not directly predict whether BPC-157 would restore limb function after thoracic or cervical cord injury.
Sample sizes and blinding. The published SCI studies do not report large sample sizes, and the details of randomization and blinding procedures are limited. The 2019 study used random allocation but does not specify whether outcome assessors were blinded to treatment group.
Dose-response questions. Both the 200 mcg/kg and 2 mcg/kg doses produced effects in the 2019 study. This 100-fold dose range producing similar outcomes is unusual in pharmacology and raises questions about dose-response relationships and the potential for a ceiling effect at very low doses.
Mechanism uncertainty. While the gene expression data from the 2022 SCI study and the 2020 hippocampal ischemia study provide molecular correlates, a clear mechanistic pathway from BPC-157 administration to spinal cord protection has not been established. The peptide's receptor has not been identified. How a 15-amino-acid peptide from gastric juice reaches the spinal cord, crosses the blood-spinal cord barrier, and produces local effects remains unexplained.
Translation gap. Numerous neuroprotective agents have shown promise in rodent SCI models but failed in human trials. Methylprednisolone, minocycline, riluzole, and ganglioside GM1 all demonstrated preclinical efficacy that did not translate to consistent clinical benefit. BPC-157 faces the same translational challenge, with the additional obstacle that its safety profile in humans is documented only from short-term IBD trials with small sample sizes.
Context within SCI research
Spinal cord injury affects approximately 250,000-500,000 people globally each year. Despite decades of research, effective pharmacological treatments remain limited. Methylprednisolone, the only drug historically used in acute SCI, has fallen out of favor due to modest benefits and significant side effects.
Current research focuses on several approaches: chondroitinase ABC to dissolve scar tissue, stem cell transplantation, epidural electrical stimulation, and various neuroprotective and neuroregenerative agents. BPC-157 fits within the neuroprotective category, alongside agents like erythropoietin, minocycline, and riluzole.
What distinguishes BPC-157 from other preclinical neuroprotective candidates is the breadth of its documented effects across injury types. Most neuroprotective agents target a single pathway: anti-inflammation, anti-apoptosis, or anti-excitotoxicity. BPC-157 appears to influence multiple pathways simultaneously, including angiogenesis, nitric oxide modulation, anti-demyelination, and neurotransmitter regulation. Whether this multi-target profile is genuinely advantageous or simply reflects insufficient mechanistic understanding is an open question.
The oral bioavailability demonstrated in the 2022 SCI study (effectiveness when given in drinking water) would be a practical advantage for clinical translation if confirmed in humans, as most neuroprotective agents in SCI trials require intravenous or intrathecal administration.
BPC-157 has been described as an antiulcer peptide that was later found to have systemic effects across virtually every tissue type. This pattern of expanding therapeutic claims from a single peptide, without a clearly identified receptor or signaling cascade, warrants scientific caution even as the preclinical results remain internally consistent.
The Bottom Line
BPC-157 produced sustained functional recovery in rats after spinal cord compression injury, counteracting axonal loss, demyelination, edema, and motoneuron death across observation periods up to one year. Two studies from 2019 and 2022 form the core evidence, supported by related findings in peripheral nerve, brain, and demyelination models. All data comes from a single research group using rat models. No human spinal cord injury trial exists, and the translation from rat sacrocaudal compression to human clinical SCI remains entirely theoretical.
Sources & References
- 1RPEP-04418·Perovic, Darko et al. (2019). “Stable gastric pentadecapeptide BPC 157 can improve the healing course of spinal cord injury and lead to functional recovery in rats..” Journal of orthopaedic surgery and research.Study breakdown →PubMed →↩
- 2RPEP-06429·Perovic, Darko et al. (2022). “Novel Therapeutic Effects in Rat Spinal Cord Injuries: Recovery of the Definitive and Early Spinal Cord Injury by the Administration of Pentadecapeptide BPC 157 Therapy..” Current issues in molecular biology.Study breakdown →PubMed →↩
- 3RPEP-01616·Gjurasin, Miroslav et al. (2010). “BPC-157 Heals Traumatic Nerve Injury in Rats.” Regulatory peptides.Study breakdown →PubMed →↩
- 4RPEP-01705·Tudor, Mario et al. (2010). “BPC-157 Protects Against Traumatic Brain Injury in Mice.” Regulatory peptides.Study breakdown →PubMed →↩
- 5RPEP-06570·Vukojevic, Jakša et al. (2022). “Pentadecapeptide BPC 157 and the central nervous system..” Neural regeneration research.Study breakdown →PubMed →↩
- 6RPEP-03116·Sikiric, Predrag et al. (2016). “Brain-gut Axis and Pentadecapeptide BPC 157: Theoretical and Practical Implications..” Current neuropharmacology.Study breakdown →PubMed →↩
- 7RPEP-05186·Vukojević, Jakša et al. (2020). “BPC-157 Protects Against Brain Damage From Blocked Blood Flow in Rats.” Brain and behavior.Study breakdown →PubMed →↩
- 8RPEP-02211·Klicek, R et al. (2013). “Stable gastric pentadecapeptide BPC 157 heals cysteamine-colitis and colon-colon-anastomosis and counteracts cuprizone brain injuries and motor disability..” Journal of physiology and pharmacology : an official journal of the Polish Physiological Society.Study breakdown →PubMed →↩
- 9RPEP-03561·Unknown (2018). “Seiwerth 2018 Bpc 157 And Standard.” .Study breakdown →↩
- 10RPEP-00987·Tohyama, Y et al. (2004). “BPC-157 Changes Serotonin Production Across Multiple Brain Regions.” Life sciences.Study breakdown →PubMed →↩
- 11RPEP-01012·Boban Blagaic, Alenka et al. (2005). “BPC-157 Effectively Treats Serotonin Syndrome in Rats.” European journal of pharmacology.Study breakdown →PubMed →↩